Abstract
Keywords
“Cardiometabolic factors displayed robust associations with oxidative stress biomarkers.”
Introduction
The term “oxidative stress” was first coined by Helmut Sies 1 to describe an imbalance between the production of reactive oxygen species (ROS) and the body’s antioxidant defenses that detoxify or neutralize these reactive compounds. 2 ROS—including free radicals and other reactive oxygen-containing molecules—are byproducts of normal cellular metabolism. 3 However if not properly regulated, they can cause damage to cells, proteins, lipids and DNA, 4 This pathophysiological state is increasingly recognized as a key contributor to the development and progression of numerous non-communicable diseases (NCDs), including obesity, type 2 diabetes, cardiovascular disease, neurodegenerative conditions, and biological aging. 5
A growing body of literature has elucidated the key role of oxidative stress in both the pathogenesis and progression these chronic conditions.6,7 In parallel systematic reviews and meta-analyses have consistently emphasized that structured lifestyle interventions—encompassing diet, physical activity, and stress management—play a pivotal role in both prevention and management of NCDs.8-11 Several ecological, case-control and cohort studies indicate that diets rich in whole grain plant/based foods provide protection against NCDs.12-14 Furthermore, numerous studies have established a consensus regarding the pivotal role of dietary antioxidants in reducing the risk of lifestyle-related illnesses.7,15,16 Additionally, Several recent reviews also highlight the potential of broader lifestyle modifications, beyond nutrition alone, in targeting oxidative stress and improving long-term health outcomes.17,18
Acknowledging the significant influence of lifestyle interventions on improving chronic disease management, as evidenced by a several studies 17 and our own Healthy Lifestyle Community Program (HLCP-1, 2, and 3)19-23 which adopts a personalized tailor-made approach and, comprehensive strategic targeting key pillars of health like, nutrition, physical activity, mental wellness, and social engagement. This program has yielded notable impact on health markers, like healthcare expenditures, quality of life and cardiovascular health. Building upon these findings, we aim to delve deeper into the impact of our HLCP-2on various biomarkers of oxidative stress.
Despite the growing recognition of lifestyle interventions as effective tools for targeting oxidative stress and reducing chronic disease risk, there is a lack of studies evaluating the impact of community-based lifestyle interventions on specific oxidative stress biomarkers in the general adult’s population. A major barrier in this area is the challenge of directly measuring free radicals in vivo, given their highly reactive and transient nature. Consequently, researchers often rely on indirect biomarkers—reflecting the body’s antioxidant defenses and oxidative damage—to assess redox balance
To bridge this gap, our study focuses on five key oxidative stress biomarkers: Total Antioxidant Capacity (TAC), Superoxide Dismutase (SOD), Catalase (CAT), Glutathione (GSH), and Lipid Peroxidation (LPO). Together, these biomarkers provide a comprehensive picture of the oxidative stress profile, encompassing both enzymatic and non-enzymatic antioxidant systems, as well as lipid oxidative injury. 24 Analyzing changes in these markers within the context of a community-based lifestyle intervention provides valuable insight into how lifestyle choices influence oxidative stress, and by extension, the aging process and chronic disease development.
In response to this important research gap and acknowledging the significant influence of lifestyle interventions on improving chronic disease management as evidenced by several studies, we built upon our existing Healthy Lifestyle Community Program (HLCP-1, 2, and 3).19-23 HLCP is a multi-component, community-driven intervention targeting key health pillars including nutrition, physical activity, mental well-being, and social connectedness. The program adopts a personalized, tailor-made approach and has demonstrated notable improvements in cardiovascular health, quality of life, and healthcare cost reduction. Building on this foundation, the current study investigates the longitudinal effects of HLCP-2 on oxidative stress biomarkers, offering new insights into the biological pathways through which lifestyle interventions exert their health benefits.
Successfully bridging this research gap may pave the way for more effective and sustainable interventions to combat non-communicable diseases (NCDs). By addressing oxidative stress as an established contributor to chronic disease development through targeted lifestyle modification, we aim to support better health outcomes in a scalable and cost-effective manner. Such community-based strategies hold promise in improving the global NCD burden and constitute a crucial component of modern public health frameworks. Therefore, we hypothesized that the HLCP-2 intervention would lead to significant improvements in systemic oxidative stress biomarkers (TAC, SOD, CAT, GSH, and LPO) following the 10-week intensive phase, with sustained benefits observed at 6- and 12-month follow-ups. We also investigated whether gender-specific responses to the intervention, particularly in catalase activity, would emerge, building on prior evidence of sex-based differences in antioxidant regulation.
Materials and Methods
Study Design
The research was designed as a non-randomized controlled longitudinal intervention study due to its utilization of a living lab approach.
25
The intervention was carried out over a 12-month period (2018-2019) to explore the effects of lifestyle modification on community health in a rural adult population. Data was collected at four intervals: baseline (t0), post-intervention at 10 weeks (t1), 6 months (t2), and 12 months (t3) Figure 1, The current paper presents findings from all four timepoints (T0–T3) to capture both short- and long-term effects of the intervention. Course of the study HLCP cohort 2. Intervention group (IG); Control group (CG); data collection at time t0 (baseline), t1 (after 10 weeks), t2 (follow-up after 6 months), t3 (follow-up after 12 months).
Participants underwent a comprehensive health assessment at each timepoint, at HLCP-2 health checks which included fasting blood sample collection for the analysis of oxidative stress biomarkers—Total Antioxidant Capacity (TAC), Superoxide Dismutase (SOD), Catalase (CAT), Glutathione (GSH), and Lipid Peroxidation (LPO)—as well as key metabolic parameters such as lipid profile, HbA1c, and fasting glucose. In addition, anthropometric data (weight, height, and waist circumference) and vital signs (systolic and diastolic blood pressure) were recorded. Blood samples were collected under standardized fasting conditions, stored in accordance with study protocol, and subsequently transferred to the University Hospital of Münster for laboratory analysis. To gain deeper insight into participant health profiles, validated questionnaires were administered at each timepoint, assessing sociodemographic characteristics, health behaviors, healthcare utilization, quality of life, physical activity, and general well-being. Dietary intake was evaluated using 3-day food diaries, collected during each health assessment and analyzed in accordance with protocols established in previous HLCP studies (Koeder et al). To ensure ethical transparency, participants in the control group were informed of their individual health assessment results.
Due to the nature of the living lab approach employed in HLCP-2, blinding of participants and facilitators was not feasible as previously outlined by Koeder et al, 2021a). 26 However, to minimize bias, all laboratory personnel remained blinded to group allocation throughout the biochemical analyses. Although randomization was not possible within this real-world, community-based framework, the living lab model offered a robust translational platform—bridging scientific evidence with practical implementation—as previously demonstrated in similar interventions.26-28
Registration and Scientific Endeavors The study was registered in the German Clinical Trial Register (DRKS; reference: DRKS00018775; https://www.drks.de/).
Participants
Using a living lab approach,28,29 participants were recruited from two demographically and socioeconomically comparable rural municipalities in the north-western region of Germany. One municipality served as the intervention site, while the other served as the control site. This geographic allocation was chosen to enable contextual relevance and logistical feasibility in real-world settings.
Eligibility criteria included participants aged between 18 to 75 years with the physical and mental capacity to participate in the study. This age demographic was selected to capture variability across life stages and associate health risks. Initially, a total of n = 201 participants enrolled in the study. Of these 22 individuals were excluded prior to the baseline assessment (T0) due to not meeting inclusion criteria (n = 8) or voluntary withdrawal (n = 14). At baseline, 103 participants were allocated to the intervention group (IG) and 72 participants to the control group (CG), based on municipality of residence rather than randomization, in accordance with the principles of the living lab design.
To maximize community engagement a multifaceted recruitment strategy was implemented, including the distribution of posters, flyers, and publication in local newspapers across both municipalities. Additionally, an interactive health market was organized the intervention municipality in collaboration with local health stakeholders to raise awareness and promote study participation. This approach aims to engage participants and raise awareness about the study and its objectives. Participant retention was tracked across four measurement timepoints: baseline (T0), 10 weeks (T1), 6 months (T2), and 12 months (T3). In the IG, retention rates were relatively high: 99 participants completed both T0 and T1, 93 participants completed T2, and 89 participants remained at T3. In the CG, 60 participants completed T1, 60 completed T2, and 54 completed T3. The overall attrition rate was 13.6% in the IG and 25.0% in the CG. This reflects robust participant retention, particularly within the IG, and supports the feasibility of conducting longitudinal lifestyle interventions in real-world community settings. A CONSORT-compliant participant flow diagram illustrating enrollment, allocation, follow-up, and analysis is presented in Figure 2. CONSORT flow diagram illustrating participant progression through the study phases: enrollment, group allocation, follow-up, and analysis. Of the 201 individuals assessed for eligibility, 22 were excluded (15 declined to participate; 7 did not meet inclusion criteria). A total of 103 participants were allocated to the intervention group and 72 to the control group. At the 10-week follow-up, complete data were available for 99 participants in the intervention group and 60 in the control group. At 6 months, 93 participants remained in the intervention group and 60 in the control group; at 12 months, 89 and 54 participants remained, respectively. The diagram presents detailed information on attrition and exclusions at each phase of the study.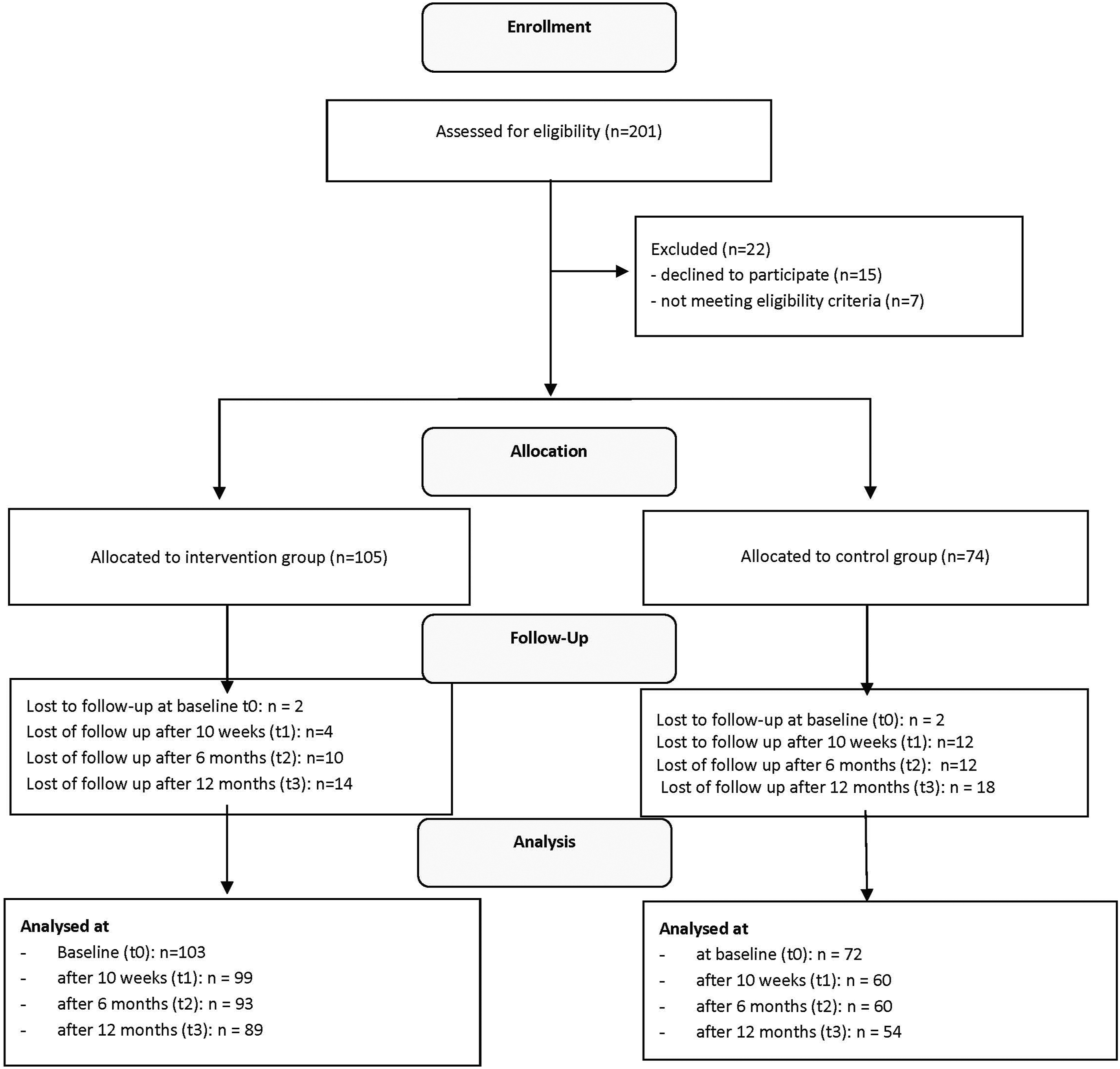
Ethics
The study was conducted in accordance with the Declaration of Helsinki 30 and was approved by the ethics committee of the Westphalia-Lippe Medical Association and the Muenster University (Muenster, Germany; reference: 2018-171-f-S; approved 4 April 2018). All participants provided written informed consent. Before formal inclusion in the study, participants provided their informed written consent, underscoring their voluntary participation.
Lifestyle Intervention
In summary, A comprehensive intervention approach was implemented, and the detailed description of the intervention has been previously published.21,26 In summary, the HLCP-2 intervention centered on addressing the four foundational pillars of health: nutrition : high plant-based whole-food diet, 31 physical activity,32,33 social engagement, 34 and mental well-being : stress management.35,36 The program spanned a 10-week intensive phase, consisting of 14 biweekly interactive seminars, each extending over 2 hours. Rooted in the philosophy of empowering participants with evidence-based knowledge, the intervention aimed to foster personal enrichment and community motivation.
Various workshops were incorporated into the program, including cooking, stress reduction, community-building, nature walk and mindful shopping. 37 These workshops provided practical skills and strategies to facilitate lifestyle changes. Additionally, personalized one-on-one lifestyle coaching sessions were conducted for participants of the IG at baseline (t0) and post-intervention (t1), tailored to individual needs and goals.
Regarding dietary guidance, participants were encouraged to adopt a balanced, whole-food, plant-based diet, with an emphasis on locally sourced, seasonal produce.38,39 Recommendations included reducing intake of meat, high-fat dairy, processed foods, sugar, and salt. 40 Participants also received comprehensive HLCP kits at the conclusion of the program, containing a healthy lifestyle manual, recipe booklet, and summarized lifestyle recommendations.
After the initial 10-week phase, a 12-month alumni program began. Its goal was to maintain progress by offering monthly seminars and digital newsletters. This was instrumental in reinforcing the acquired knowledge and facilitating long-term behavioral changes among participants.
For ethical considerations, both intervention and control groups were informed about their health check results, although only the intervention group received program participation.
Data Collection
Oxidative Stress Biomarkers
It is difficult to determine the levels of free radicals in vivo because of their short half-life, 41 but oxidative stress can be investigated indirectly by measuring the antioxidant defense system.42,43 Taken into consideration the unstable nature of free radicals we analyzed multiple OS biomarkers that will best reflect our defenses system. We aimed to develop an OS profile which gives us a better picture of our body immune response.
Blood samples were collected at baseline and at the end of the 10-week for all the study biomarkers including oxidative stress (TAC, SOD, CAT, GSH activities, and LPO). Fasting blood samples were collected in the morning after an overnight fast of at least 8 hours. Samples were collected in EDTA-coated tubes and were immediately centrifuged at 3000 r/min for 10 minutes at 4°C. 44 The obtained plasma for oxidative stress biomarkers was aliquoted into 4 Eppendorf tubes per biomarker and kept in dry ice for transferring after which they were, frozen at (−80°C), and stored at the Munster hospital laboratory for further examinations. TAC, SOD, CAT, GSH activities and LPO level were detected using commercially available kits from Cayman Chemicals, Ann Arbor, MI, USA according to the manufacturer’s instructions. Spectrophotometry was performed using a microplate reader from TECAN model spark. For each participant, 2 independent assays were performed in duplicates at the laboratory. 42
In summary, data collection for this study included measurements of oxidative stress biomarkers, demographic and anthropometric measurements, vital parameters, blood parameters, questionnaires on physical activity and stress management, nutritional protocol, and quality of life.
Study Hypothesis
The primary outcome parameter of the HLCP-2 study was body weight, and the primary hypothesis was that the participants of the IG would reduce weight significantly during the study period compared to baseline and significantly more participants of the CG. 26 Oxidative stress was defined as a secondary outcome parameter. We hypothesized that multidimensional lifestyle intervention would lead to significant improvements in oxidative stress biomarkers Total Antioxidant Capacity (TAC), Superoxide Dismutase (SOD), Catalase (CAT), Glutathione (GSH), and Lipid Peroxidation (LPO) within the IG following the 10-week intensive intervention phase (t1), relative to baseline (t0). Additionally, it was anticipated that the magnitude of improvement would be significantly greater in the IG than in the CG over the same time period.
Furthermore, we hypothesized that improvements in oxidative stress biomarkers would be sustained at the 6-month (t2) and 12-month (t3) follow-ups, reflecting long-term retention of antioxidant benefits. Lastly, we explored whether these responses differed by gender, given the known sex-based variations in redox biology and antioxidant capacity.
Statistical Analysis
The sample size for this study was determined based on the primary outcome parameter, which was the change in body weight as described previously, 23 and eligible participants were included into the present secondary analyses, accordingly. A predefined analysis plan was used, and all analyses were performed on an available-case analysis (ACA). Descriptive statistics were reported using absolute numbers and percentages (%) for categorical data and means with standard deviation (SD) for quantitative variables. The normality of the data was assessed using the Shapiro-Wilk test, with P < 0.05 indicating a nonnormal distribution supported by visual inspection through histograms and Q-Q plots.
Between-group differences were analyzed by using independent t test for normally distributed and Mann–Whitney-U test for non-normally distributed continuous variables. Fisher’s exact test was used for between-group comparisons of categorical variables. To evaluate within-group changes, paired t test was used for normally and Wilcoxon signed-rank test was used for non-normally distributed variables. All tests were two-sided, and results were considered significant at P < 0.05.
To evaluate the intervention’s effect across all four timepoints, repeated measures analysis of variance (RM-ANOVA) was performed for each oxidative stress biomarker (TAC, SOD, CAT, GSH, LPO), with time as the within-subject factor and group (IG vs CG) as the between-subject factor. Post hoc pairwise comparisons were Bonferroni-adjusted.
Gender-based subgroup analyses were performed within the intervention group using independent samples t-tests or Mann–Whitney U tests, depending on data distribution, to assess differences in biomarker changes between male and female participants. Gender was also included as a covariate in the regression models to explore its predictive value across all oxidative stress biomarkers.
Baseline Characteristics of the Intervention and Control Groups.
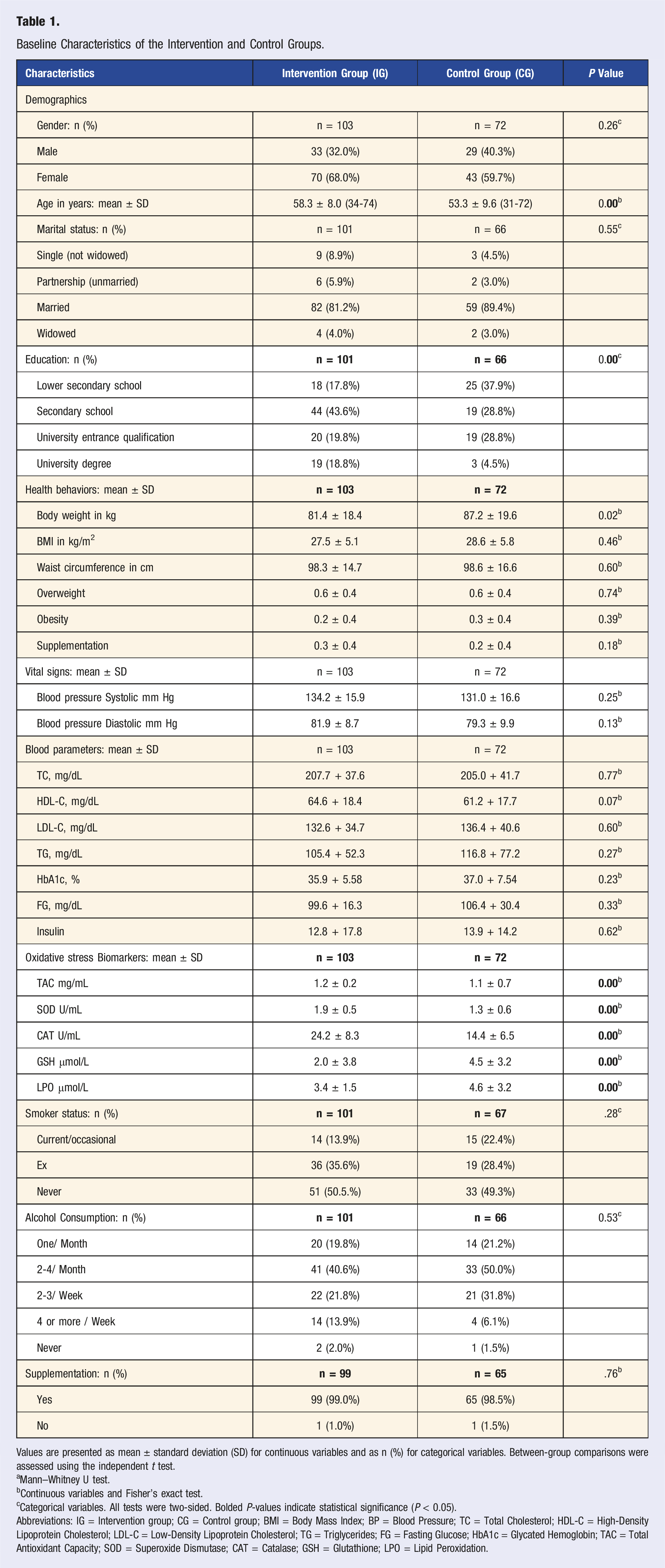
Values are presented as mean ± standard deviation (SD) for continuous variables and as n (%) for categorical variables. Between-group comparisons were assessed using the independent t test.
aMann–Whitney U test.
bContinuous variables and Fisher’s exact test.
cCategorical variables. All tests were two-sided. Bolded P-values indicate statistical significance (P < 0.05).
Abbreviations: IG = Intervention group; CG = Control group; BMI = Body Mass Index; BP = Blood Pressure; TC = Total Cholesterol; HDL-C = High-Density Lipoprotein Cholesterol; LDL-C = Low-Density Lipoprotein Cholesterol; TG = Triglycerides; FG = Fasting Glucose; HbA1c = Glycated Hemoglobin; TAC = Total Antioxidant Capacity; SOD = Superoxide Dismutase; CAT = Catalase; GSH = Glutathione; LPO = Lipid Peroxidation.
Results
This table summarizes the demographic, behavioral, and biochemical characteristics at baseline. Participants in the intervention group were slightly older (58.3 ± 8.0 years) and more educated compared to controls (53.4 ± 9.6 years), with both differences reaching statistical significance (P < 0.01). The majority were female (IG: 68.0%; CG: 59.7%) and overweight (BMI: IG: 27.5 ± 5.1; CG: 28.6 ± 5.8 kg/m2). Cardiometabolic markers, including HbA1c, lipid profile, fasting glucose, and blood pressure—did not significantly differ between groups. Likewise, smoking status, supplementation, and alcohol intake showed no significant differences. At baseline, oxidative stress biomarkers showed some variation between groups. The intervention group demonstrated higher antioxidant levels in total antioxidant capacity (TAC: 1.27 ± 0.21 vs 1.16 ± 0.08 mg/mL) and superoxide dismutase (SOD: 1.90 ± 0.56 vs 1.38 ± 0.62 U/mL), as well as higher catalase activity (CAT: 24.28 ± 8.36 vs 14.41 ± 6.58 U/mL). In contrast, the control group had higher glutathione (GSH: 4.54 ± 3.25 vs 2.02 ± 3.88 μmol/L) and lipid peroxidation levels (LPO: 4.63 ± 3.30 vs 3.47 ± 1.55 μmol/L). Overall, both groups were sufficiently comparable in their baseline oxidative and metabolic profiles to allow for a meaningful evaluation of the intervention’s effects. Due to the use of available-case analysis (ACA), the number of participants (n) varies slightly across some variables depending on data completeness.
Within-Group Pre–Post Changes in Oxidative Stress Biomarkers Following the 10-Week HLCP-2 Intervention.
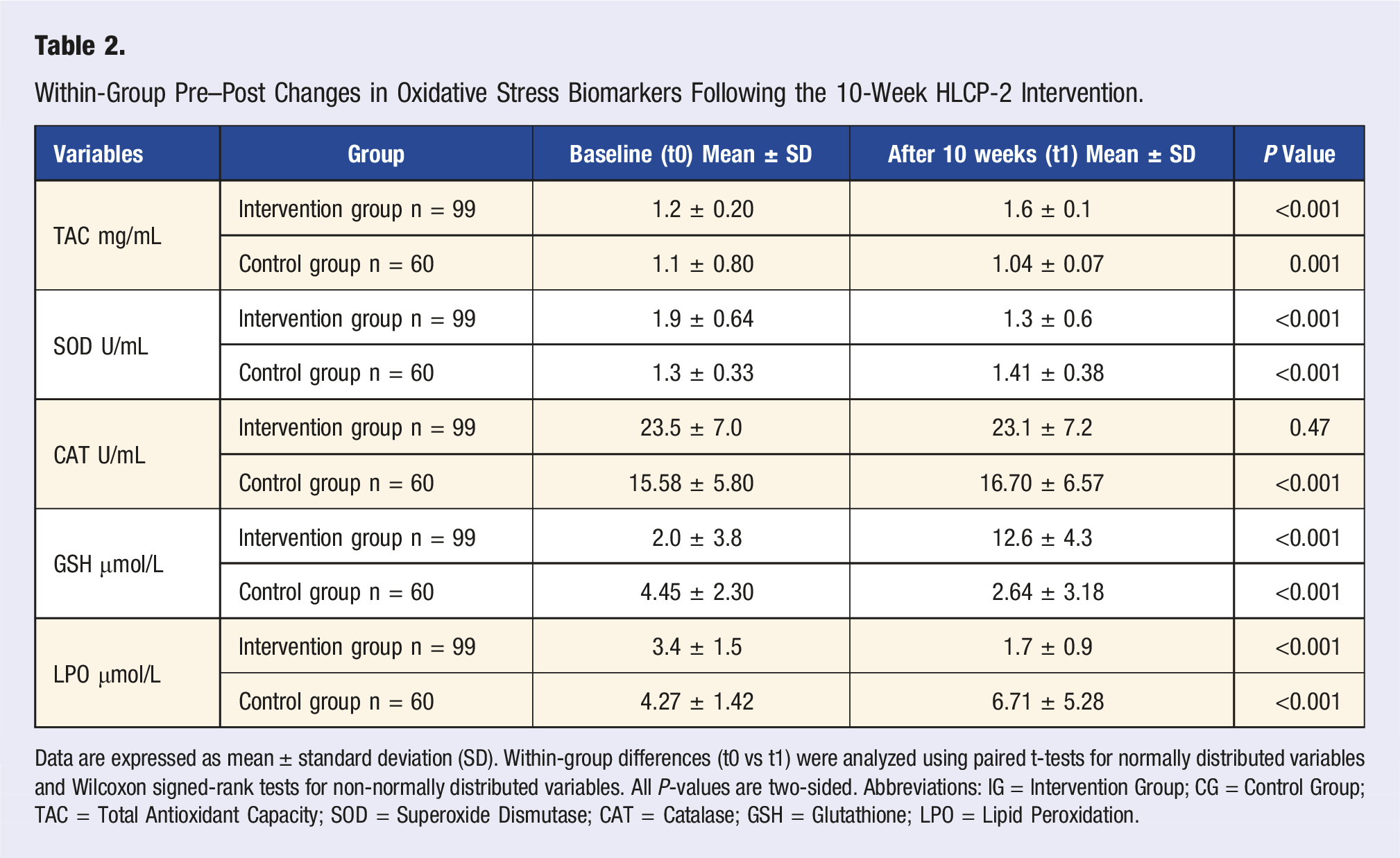
Data are expressed as mean ± standard deviation (SD). Within-group differences (t0 vs t1) were analyzed using paired t-tests for normally distributed variables and Wilcoxon signed-rank tests for non-normally distributed variables. All P-values are two-sided. Abbreviations: IG = Intervention Group; CG = Control Group; TAC = Total Antioxidant Capacity; SOD = Superoxide Dismutase; CAT = Catalase; GSH = Glutathione; LPO = Lipid Peroxidation.
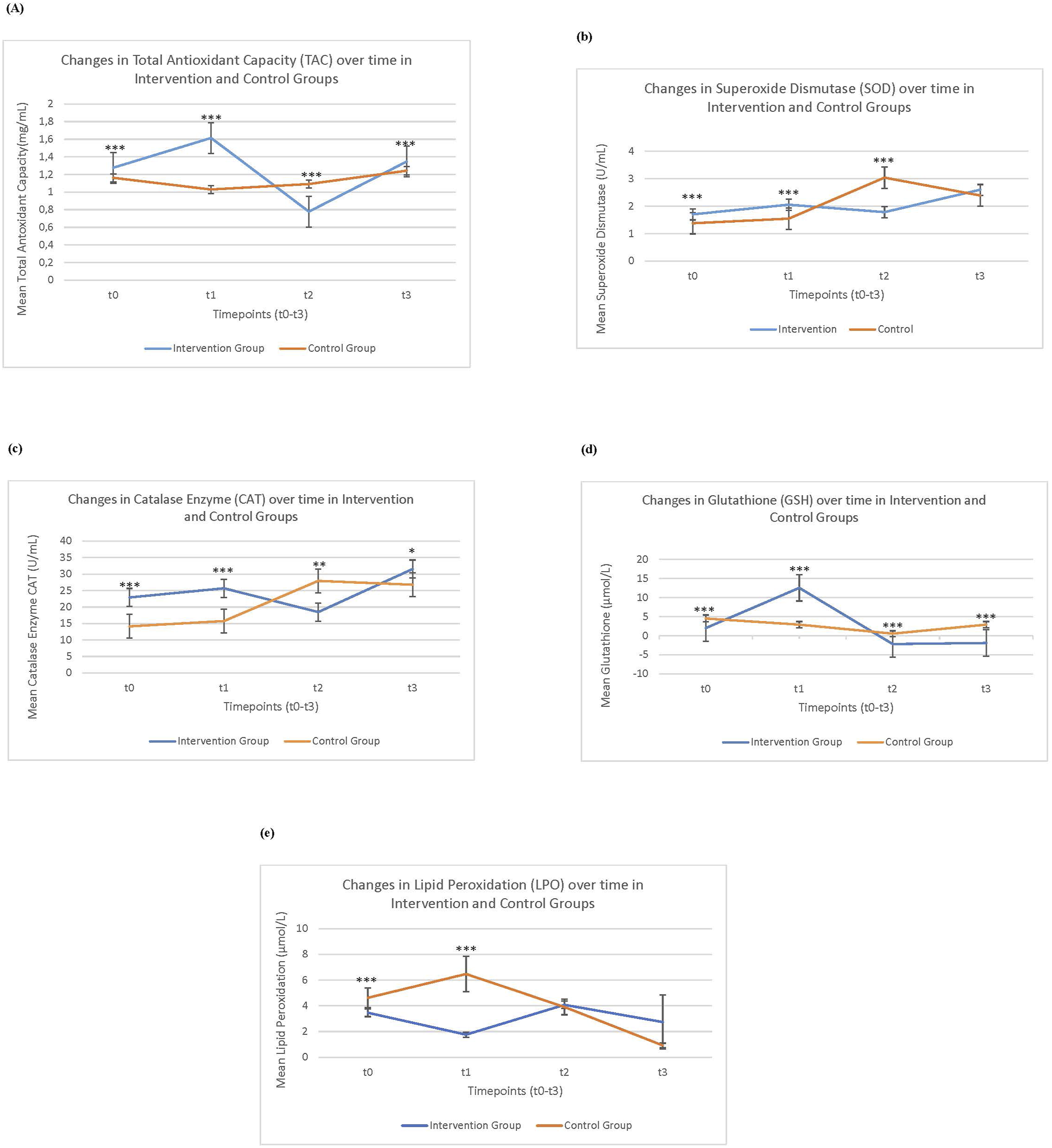
Longitudinal changes in oxidative stress biomarkers (TAC, SOD, CAT, GSH, LPO) across four timepoints in the intervention and control groups. Changes in oxidative stress biomarkers between the intervention group (IG) and control group (CG) over the study period (t0 to t3): (A) Plasma Total Antioxidant Capacity (TAC), (B) Superoxide Dismutase (SOD), (C) Catalase (CAT), (D) Glutathione (GSH), and (E) Lipid Peroxidation (LPO). Data are expressed as mean ±2 standard error (SE). Statistical analysis was conducted using two-way repeated measures ANOVA with Bonferroni-adjusted post hoc comparisons. Asterisks indicate significant between-group differences at individual timepoints (*P < 0.05, **P < 0.01, ***P < 0.001). (A) Total Antioxidant Capacity (TAC) (B) Superoxide Dismutase (SOD) (C) Catalase Enzyme (CAT) (D) Glutathione (GSH) (E) Lipid peroxidation (LPO).
Significant time- and group-dependent effects were observed across all oxidative stress biomarkers. In the intervention group, Total Antioxidant Capacity (TAC) increased significantly from baseline (t0) to post-intervention (t1), with sustained improvement through t3 (P < 0.001), whereas the control group showed minimal change. Superoxide Dismutase (SOD) levels rose moderately in both groups; however, the intervention group demonstrated significantly higher levels at all measured timepoints (P < 0.001), indicating enhanced enzymatic antioxidant defense. Catalase (CAT) activity was consistently elevated in the intervention group compared with controls, showing significant differences at all timepoints (P < 0.05 to P < 0.001), with a marked increase at t3. Glutathione (GSH) levels peaked sharply at t1 in the intervention group and remained significantly higher than in controls at each timepoint (P < 0.001), reflecting a robust intracellular antioxidant response. In contrast, Lipid Peroxidation (LPO) levels—a marker of oxidative damage—were significantly higher in the control group at t0 and t1 (P < 0.001), while remaining low and stable in the intervention group, suggesting a protective effect against oxidative membrane damage. Together, these findings demonstrate that lifestyle-based intervention elicited a comprehensive improvement in systemic antioxidant status and significantly reduced oxidative stress over time (Figure 4). Gender-based analysis of oxidative stress biomarkers pre- and post-intervention. (A) Total antioxidant capacity (TAC mg/mL). (B) Superoxide dismutase (SOD U/mL). (C) Catalase enzyme activity (CAT U/mL). (D) Glutathione (GSH μmol/L). (E) Lipid peroxidation (LPO μmol/L). Illustrates the gender-specific impact of the HPCL-2 on oxidative stress biomarkers after 10 weeks within the intervention group. The analysis was conducted within the intervention group, evaluating biomarkers including: (a) total antioxidant capacity (TAC); (b) superoxide dismutase (SOD); (c) catalase enzyme (CAT); (d) glutathione (GSH), and (e) lipid peroxidation (LPO). The comparison was made between male (n = 32) and female (n = 67) participants to discern variations in biomarker levels after the intervention. Each bar on the graph represents the mean change in biomarker levels, with error bars indicating standard deviations and significance levels set at P < 0.05. Notably, a significant gender-specific difference was observed solely in the catalase enzyme (P = 0.009), while TAC (P = 0.97), SOD (P = 0.23), GSH (P = 0.75), and LPO (P = 0.74) did not exhibit statistically significant gender differences (P > 0.05) after the 10-week intervention within the intervention group.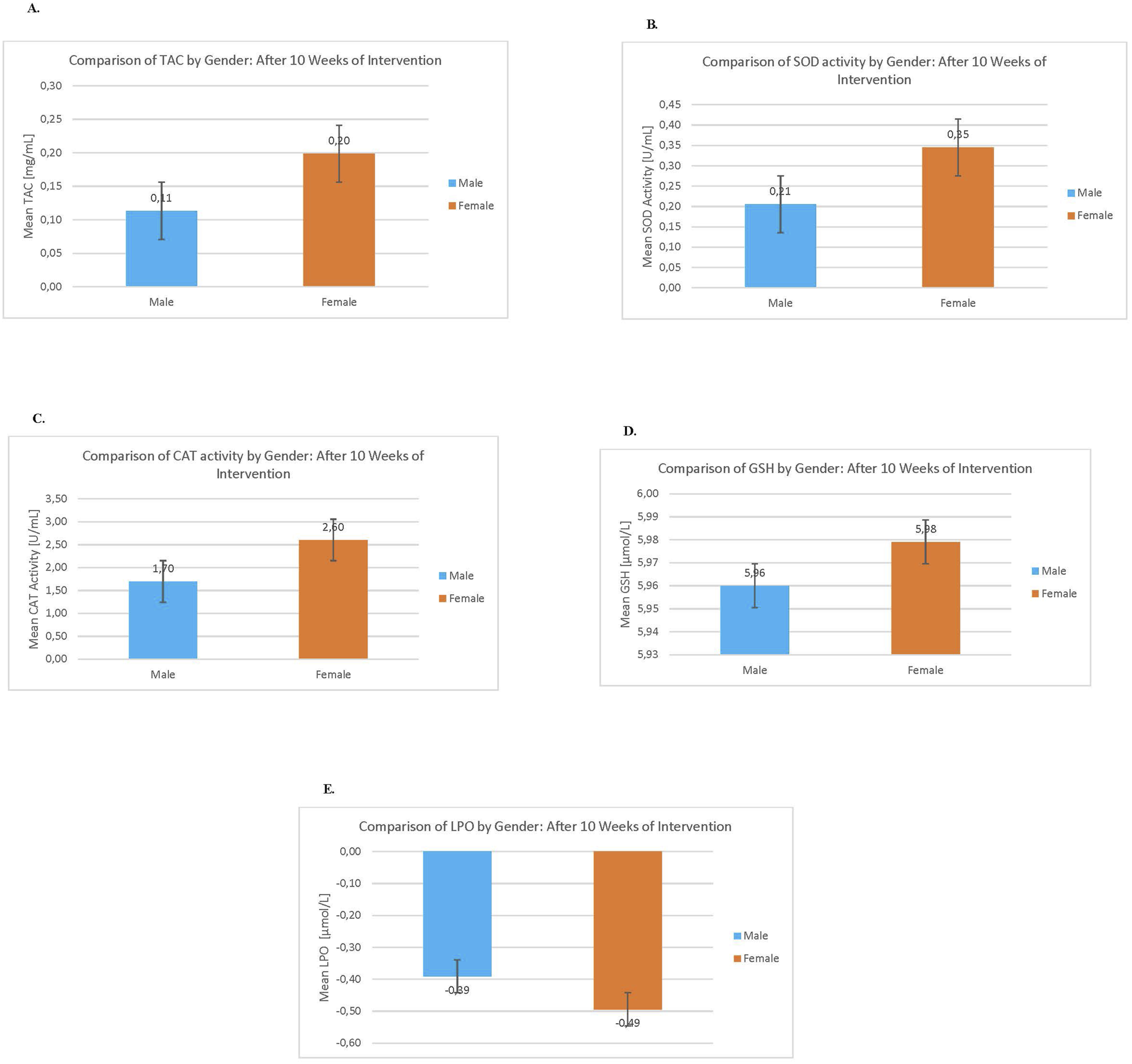
Univariate and Multivariate Linear Regression Analysis
Table 3 shows associations between changes in oxidative stress biomarkers; TAC, SOD, CAT, GSH, LPO) and key predictors ; group, age, sex, BMI, lifestyle, and metabolic factors using multivariate regression analysis.
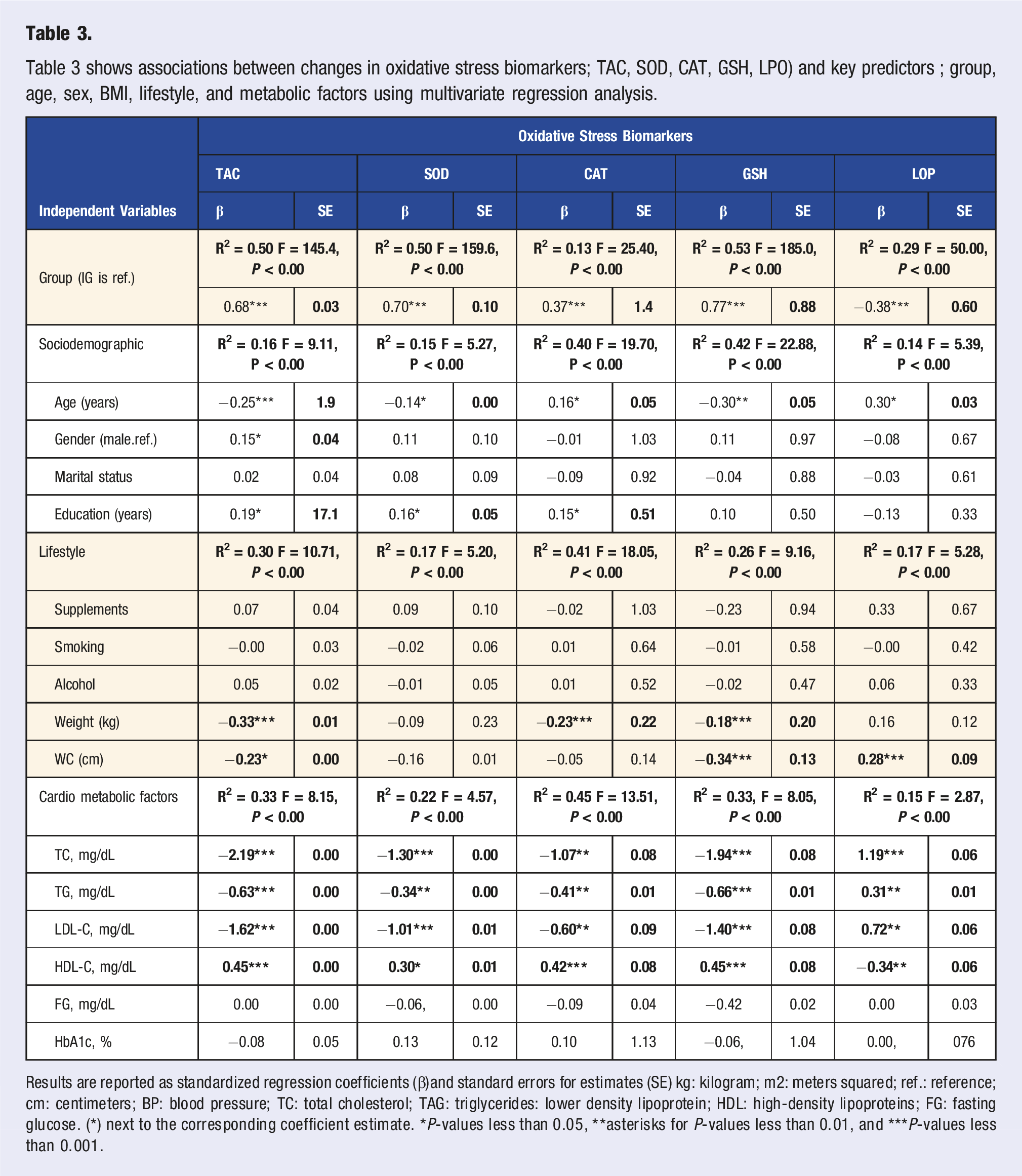
Results are reported as standardized regression coefficients (β)and standard errors for estimates (SE) kg: kilogram; m2: meters squared; ref.: reference; cm: centimeters; BP: blood pressure; TC: total cholesterol; TAG: triglycerides: lower density lipoprotein; HDL: high-density lipoproteins; FG: fasting glucose. (*) next to the corresponding coefficient estimate. *P-values less than 0.05, **asterisks for P-values less than 0.01, and ***P-values less than 0.001.
To evaluate the influence of multiple determinants on oxidative stress biomarkers, univariate regression was conducted using group status (Intervention vs Control), and multivariate regression analyses were performed for sociodemographic, lifestyle, and cardiometabolic parameters. All models were adjusted for baseline BMI, age, sex, physical activity, smoking status, lipid profile, fasting glucose, and HbA1c. Standardized beta coefficients (β) and standard errors (SE) were reported.
The 10-week lifestyle intervention was significantly associated with improved oxidative stress biomarkers, as shown by strong positive associations for TAC (β = 0.68, SE = 0.03, R2 = 0.50, P < 0.001), SOD (β = 0.70, SE = 0.10, R2 = 0.50, P < 0.001), CAT (β = 0.37, SE = 1.40, R2 = 0.13, P < 0.001), and GSH (β = 0.77, SE = 0.88, R2 = 0.53, P < 0.001). LPO was negatively associated with group status (β = −0.38, SE = 0.60, R2 = 0.29, P < 0.001), suggesting reduced lipid peroxidation following intervention.
Among sociodemographic variables, age was inversely associated with TAC (β = −0.25, SE = 1.90), SOD (β = −0.14, SE = 0.00), and GSH (β = −0.30, SE = 0.05), and positively with CAT (β = 0.16, SE = 0.05) and LPO (β = 0.30, SE = 0.03). Education was positively associated with TAC (β = 0.19, SE = 17.1), SOD (β = 0.16, SE = 0.05), and CAT (β = 0.15, SE = 0.51). Gender showed a modest positive association with TAC (β = 0.15, SE = 0.04), whereas marital status did not show significant effects.
Within lifestyle predictors, body weight was negatively associated with TAC (β = −0.33, SE = 0.01), CAT (β = −0.23, SE = 0.22), and GSH (β = −0.18, SE = 0.20). Waist circumference showed a negative relationship with TAC (β = −0.23, SE = 0.00) and GSH (β = −0.34, SE = 0.13), but a positive relationship with LPO (β = 0.28, SE = 0.09). No significant associations were observed for smoking, alcohol use, or supplement intake.
Cardiometabolic factors demonstrate consistent and significant associations. Total cholesterol (TC) had strong inverse relationships with TAC (β = −2.19, SE = 0.00), SOD (β = −1.30, SE = 0.00), CAT (β = −1.07, SE = 0.08), and GSH (β = −1.94, SE = 0.08), and was positively associated with LPO (β = 1.19, SE = 0.06). Similar negative patterns were observed for triglycerides (TG) and LDL-C, while HDL-C was positively associated with all antioxidant biomarkers (TAC, SOD, CAT, GSH) and inversely associated with LPO. Fasting glucose and HbA1c did not show statistically significant associations with any oxidative stress markers.
Discussion
This study is among the first to systematically evaluate the longitudinal impact of a multidimensional lifestyle intervention on five key oxidative stress biomarkers—Total Antioxidant Capacity (TAC), Superoxide Dismutase (SOD), Catalase (CAT), Glutathione (GSH), and Lipid Peroxidation (LPO)—across four timepoints in a rural adult German population. Our findings provide compelling evidence that structured lifestyle modification, delivered through the HLCP-2 program, significantly enhances antioxidant defense systems while reducing oxidative stress load, independent of age, gender, and metabolic risk factors. These results contribute to the growing body of literature supporting the utility of lifestyle medicine interventions in modulating redox balance and promoting healthy aging.2,17,18,45-47
Participants in the intervention group demonstrated significantly improved antioxidant profiles over time, most notably in TAC (β = 0.68, SE = 0.03, P < 0.001), SOD (β = 0.70, SE = 0.10, P < 0.001), and GSH (β = 0.77, SE = 0.88, P < 0.001). These biomarkers represent enhanced activity across both enzymatic (e.g., SOD, CAT) and non-enzymatic (e.g., TAC, GSH) components of the endogenous antioxidant defense system, likely driven by increased intake of antioxidant-rich foods, enhanced metabolic function, and physical activity.48-50 The intervention group also demonstrated a significant reduction in LPO (β = −0.38, SE = 0.60, P < 0.001), indicating reduced oxidative injury to cellular membranes.51,52 Although CAT showed statistically significant between-group differences at all timepoints (P < 0.05 to P < 0.001), within-group improvements were not significant, suggesting that catalase activity may be more stable or require long-term modulation.8,17,53
Gender-stratified analysis of oxidative stress biomarkers following the 10-week HLCP-2 intervention revealed statistically significant differences in catalase activity between males and females within the intervention group (P = 0.009), while other biomarkers—TAC (P = 0.97), SOD (P = 0.23), GSH (P = 0.75), and LPO (P = 0.74)—did not show significant gender-based variation. These findings are consistent with prior research indicating that estrogen enhances catalase expression and activity, potentially providing females with greater resilience to oxidative stress through this pathway.54,55 Studies suggest that sex hormones, particularly estrogen, upregulate antioxidant enzymes and improve redox homeostasis, whereas testosterone may exert pro-oxidant effects.56,57 Other oxidative stress biomarkers did not display gender-specific changes, suggesting that HLCP-2 elicited broad improvements irrespective of sex. This supports emerging evidence that structured lifestyle interventions may mitigate biologically driven sex disparities in oxidative balance.58,59
Multivariate linear regression demonstrated that group allocation was the strongest predictor of improved antioxidant status (TAC R2 = 0.50, SOD R2 = 0.50, GSH R2 = 0.53, CAT R2 = 0.13, LPO R2 = 0.29; all P < 0.001). Age was significantly associated with lower levels of TAC (β = −0.25), SOD (β = −0.14), and GSH (β = −0.30), suggesting age-related declines in the antioxidant defense system. These findings are consistent with previous research demonstrating that aging is accompanied by mitochondrial dysfunction, increased ROS production, and diminished DNA repair capacity—factors that collectively exacerbate oxidative stress.2,60-62 Notably, a 2022 review by Amorim et al and Sinclair in Nature Reviews Endocrinology highlights mitochondrial redox imbalance as a key hallmark of aging and age-related diseases. 18 Gender was positively associated with TAC, with female participants exhibiting higher antioxidant capacity, possibly due to the protective effects of estrogen on redox regulation. Estrogen has been shown to enhance antioxidant enzyme activity and reduce ROS, whereas testosterone may exert pro-oxidant effects, contributing to observed sex differences in oxidative stress.57,63 Educational attainment was also significantly associated with TAC, SOD, and CAT. This may reflect the impact of health literacy and socioeconomic status on dietary quality and physical activity—both known modulators of antioxidant capacity. 64
Body weight and waist circumference emerged as strong negative predictors of antioxidant biomarkers (TAC, CAT, and GSH) and positive predictors of LPO, underscoring the contribution of adiposity to oxidative stress. These associations are consistent with evidence linking visceral fat accumulation to chronic low-grade inflammation, elevated ROS, and impaired antioxidant enzyme expression.65,66 In contrast, the use of supplements, smoking, and alcohol did not show significant associations, indicating that targeted lifestyle modifications—as delivered by HLCP-2—may exert a stronger cumulative impact on redox balance than isolated behaviors. 67
Finally, cardiometabolic factors displayed robust associations with oxidative stress biomarkers. Elevated levels of total cholesterol (TC), triglycerides (TG), and LDL-C were significantly associated with lower antioxidant capacity (TAC, SOD, CAT, and GSH) and higher lipid peroxidation (LPO). In contrast, HDL-C showed protective associations, being positively related to antioxidant biomarkers and inversely associated with LPO. These findings reinforce the tightly coupled relationship between lipid metabolism and oxidative balance and suggest that redox biomarkers may serve as integrative indicators of cardiometabolic health.68,69
While our study demonstrates clear improvements in biomarkers of oxidative stress, discrepancies in existing research must be noted. For instance, Wang et al (2021) reported no change in systemic OS markers after a 6-month intervention in women with obesity, despite weight loss. These inconsistencies may reflect differences in population characteristics, intervention content, or biomarker sensitivity. 70
Nonetheless, a growing body of evidence supports the role of lifestyle modification in reducing chronic disease risk and enhancing antioxidant capacity. For example, a 2023 study in The Lancet Healthy Longevity reported a 44% reduction in mortality risk associated with healthy lifestyle practices in older adults, with significant gains in life expectancy among high-risk populations. 47
The significant enhancements observed across oxidative stress biomarkers suggest that HLCP-2 likely modulates oxidative stress through multiple convergent pathways. Dietary intake of fruits, vegetables, and whole grains may supply exogenous antioxidants; physical activity may enhance the activity of endogenous antioxidant enzymes; and stress-reduction components of the program may reduce ROS generation. Importantly, improvements in weight and metabolic profile are likely to further reduce systemic inflammation and oxidative burden. Taken together, these findings highlight the potential of comprehensive, tailored lifestyle interventions to exert multi-systemic benefits.35,71
Strengths and Limitations
A key strength of our study lies in the comprehensive evaluation of five oxidative stress biomarkers, providing a robust and multidimensional understanding of redox balance within our population. This biomarker-based approach strengthens the scientific rigor and interpretability of our findings regarding the effectiveness of the HLCP-2 intervention. The inclusion of a well-defined control group and implementation in a real-world, community-based setting further enhance external validity—features often lacking in lifestyle medicine research. Despite common challenges in behavioral interventions, including variable adherence and engagement, the study achieved high participation during the intensive phase, suggesting strong feasibility and acceptability of the HLCP-2 model. However, several limitations warrant consideration. The relatively short intervention period may limit the ability to detect long-term changes in certain biomarkers, particularly those with slower physiological turnover, such as CAT. Furthermore, sample size variation—originally based on power calculations for body weight as the primary endpoint—may reduce statistical power for secondary biomarker outcomes. The lack of standardized reference methods for measuring oxidative stress adds complexity to data interpretation and limits comparability across studies. Finally, the resource-intensive nature of biomarker analysis—both in cost and time—presents challenges for scalability and implementation in larger public health contexts. These limitations highlight the importance of future studies employing longer follow-up durations, larger and more diverse cohorts, and standardized biomarker protocols to advance the field of oxidative stress research in lifestyle medicine.
Conclusion
In summary, our findings demonstrate that the HLCP-2 lifestyle intervention leads to meaningful improvements in multiple oxidative stress biomarkers, reinforcing the role of lifestyle medicine in promoting redox balance and healthy aging. By targeting dietary quality, physical activity, stress management, and social connection in a community setting, HLCP-2 offers a feasible and scalable model for oxidative stress modulation and chronic disease prevention. This study adds to the growing evidence base suggesting that multifactorial, tailored interventions can influence biological aging markers and improve long-term health trajectories. Importantly, our data supports the integration of structured lifestyle programs into broader public health strategies aimed at extending health span and reducing disease burden. Future research should aim to elucidate the mechanistic pathways underlying these effects—particularly in aging populations—and evaluate long-term sustainability of biomarker changes. Standardization of oxidative stress measurement and implementation in diverse cultural and geographic settings will be essential for translating these findings into global health policy and practice.
Footnotes
Acknowledgments
I would like to express my sincere gratitude to all those who contributed to the completion of this scientific paper. I would like to acknowledge the participants who generously dedicated their time and efforts to this study. The authors would like to thank the study participants, the local stakeholders involved. Additionally, I extend my appreciation to Dr Matthias Borowski for his statistical advice, Alwine Kraatz and Jennifer Musiol who took part in the investigation, Dr Reinhold Gellner for his valuable contributions in the application process for ethical approvement and his medical advice during the studies, our student helpers and entire research group for their assistance and. This work would not have been possible without their contributions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was conducted as a part of the project “münster.land.leben” and was supported by the German Federal Ministry of Education and Research (BMBF) and the Joint Science Conference (GWK) within the Programme “Innovative Hochschule” (Grant Number: 03IHS062A).
