Abstract
The co-occurrence of diabetes mellitus and eating disorders represents a complex and high-risk combination of health conditions that significantly increases the risk of morbidity and mortality. While the interplay between diabetes mellitus and eating disorders is well-documented, consistent screening and management practices remain underutilized. This article explores the etiology of eating disorders, their manifestations in individuals with diabetes mellitus, and offers clinical insights into recognizing potential eating disorders. Additionally, we highlight the importance of the interdisciplinary care team in the effective prevention, screening, diagnosis, and treatment of patients with the dual diagnosis of diabetes mellitus and eating disorders.
Keywords
“The co-occurrence of DM and EDs presents a high-risk combination of health conditions that significantly increases morbidity and mortality.”
Introduction
Diabetes mellitus (DM) and eating disorders (EDs) represent a complex and dangerous intersection of health conditions that significantly increases morbidity and mortality risk.1-3 Individuals managing both conditions face the dual challenge of achieving a healthy weight while maintaining tight blood glucose control, and are at an increased risk of developing complications such as improper wound healing, increased risk of infection, muscle atrophy, menstrual disruption, diabetic ketoacidosis (DKA), electrolyte imbalances, cardiovascular disease, nerve damage, kidney failure, and death.1,2,4
Individuals with DM share common risk factors for developing EDs with the general population, including a complex interplay of genetic, environmental, and psychological factors; however, their risk is heightened by unique aspects of DM management.5,6 While factors such as high body mass index (BMI), body dissatisfaction, poor coping strategies, family dysfunction, and depression are associated with EDs in both type 1 diabetes mellitus (DM1) and type 2 diabetes mellitus (DM2), the underlying causes, predictive factors, and onset of ED symptoms differ between the 2 conditions.6,7
Effective DM care necessitates a focus on food composition, portion control, physical activity, blood glucose monitoring, and weight management, all of which can foster unhealthy attitudes toward food and body image. 7 Furthermore, the psychological burden of managing a chronic condition often introduces stress, anxiety, and depression, exacerbating difficulties in both DM management and ED behaviors.6,8,9 Behaviorally, patients with EDs may exhibit neglect of DM management, avoiding DM-related appointments, extreme anxiety about body image, preoccupation with food, weight, and calorie consumption, withdrawal from loved ones, and increased incidence of major depressive disorder. 5
Due to the complexity of managing disordered eating and EDs in patients with DM, a collaborative, multidisciplinary care team is crucial for effective prevention, screening, diagnosis and treatment. Additionally, nutritional management must be tailored to each individual and provided by professionals with expertise in both DM and EDs.
Here, we explore the manifestations of EDs in patients with DM, focusing on different types of EDs, clinical presentations, unique challenges of diagnosis, treatment considerations, and future directions of care within the context of both DM1 and DM2.
Methods
A systematic literature review was conducted using the PubMed database, with search period from 2010 to present. The search included combinations of the keywords “diabetes,” “eating disorders,” and “nutrition.” No population restrictions were utilized, and only articles published in English were considered. Retrieved articles were imported into Covidence for further screening for relevance. To supplement the initial search, additional literature searches were performed separately.
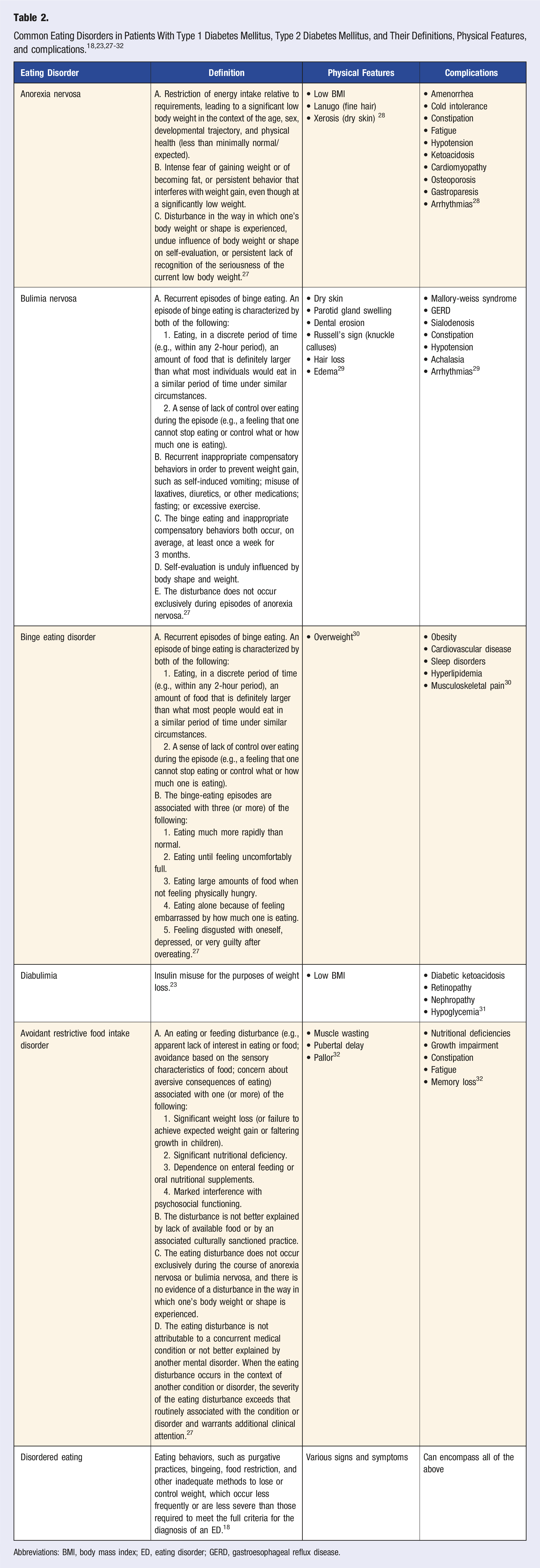
Overview of Eating Disorders
EDs are complex, multifactorial conditions that disrupt eating behaviors and lead to significant health and social impairment. In the United States, the lifetime prevalence of EDs is estimated to be 9% in females and 4% in males. 10 EDs can manifest as early as age five and may persist into adulthood if left untreated. 11 In recent decades, there has been a marked increase in the incidence of ED diagnoses among younger populations, with females disproportionately affected. 11 While each ED has its own unique risk factors, common factors shared across all include female sex, younger age, depression, and body dissatisfaction.12-15 In addition to medical complications, EDs can affect an individual’s psychosocial function, academic and professional performance, and increase susceptibility to other mental health disorders, including anxiety and depression.16,17 Of note, the term “disordered eating” encompasses a broader spectrum of maladaptive eating behaviors that do not meet criteria for formal ED diagnosis. 18
Prevalence of Common Eating Disorders in Patients With Type 1 Diabetes Mellitus, Type 2 Diabetes Mellitus, and the General Population.8,12,19–22

Abbreviations: DM, diabetes mellitus; DM1, type 1 diabetes mellitus; DM2, type 2 diabetes mellitus.
*Not applicable, or no available literature available.
8 Mannucci et al. 2005 provides prevalence from a meta-analysis of female subjects.
19 Hudson et al. 2007 provides data on lifetime prevalence.
Bulimia Nervosa (BN) is characterized by recurrent binge eating episodes, followed by compensatory behaviors such as self-induced vomiting, laxative misuse, fasting, or excessive exercise. 27 In the United States, the lifetime prevalence of BN in the general population is estimated to be 1.0%, with higher rates observed in patients with DM.8,19 BN is particularly prevalent among those with DM1, where it is often associated with insulin misuse, also known as diabulimia.23,36 Rates of insulin misuse have been reported as high as 76% in various cohorts.21,23,36 Although most of the current literature focuses on diabulimia within the DM1 population, studies have shown that individuals with DM2 may also engage in similar insulin misuse in pursuit of weight loss. 37 Common symptoms of BN include abdominal pain, bloating, and fatigue. Physical examination findings include dry skin, parotid gland swelling, dental erosion, Russell’s sign with calluses on knuckles, and hypotension. Medical complications, such as elevated hemoglobin a1c (HbA1c), increased risk of microvascular complications such as retinopathy and nephropathy, as well as Mallory-Weiss syndrome, gastroesophageal reflux disease, constipation, achalasia, and cardiac arrhythmias are also associated with BN in patients with DM.29,38
Binge Eating Disorder (BED) is defined by recurrent binge eating episodes accompanied by a sense of loss of control, without compensatory behaviors. 27 The lifetime prevalence of BED in the general population is estimated to be 2.8%, with rates in patients with DM2 reaching as high as 25%.19,20,39 As BED disrupts glucose metabolism, individuals are at an elevated risk of developing DM2 as a consequence. 40 In addition to DM2, medical complications of BED are linked to obesity, including cardiovascular disease, sleep disruption disorders, and hyperlipidemia. 30
Avoidance/restrictive food intake disorder (ARFID) is characterized by persistent disturbances in eating or feeding, such as an apparent lack of interest in food, avoidance based on sensory characteristics, or concerns about adverse eating consequences, resulting in significant weight loss, dependency on enteral feeding or supplements, or interference with psychosocial functioning. 27 The prevalence of ARFID in the general population is estimated to be 0.5%–5%, though its prevalence in patients with DM is unknown. 22 ARFID is most commonly diagnosed in younger males, and is often associated with neuropsychological disorders, such as autism spectrum disorder. 22 Medical complications include nutritional deficiencies, growth impairment, constipation, fatigue, and memory loss. 32
Interplay Between Diabetes Mellitus and Eating Disorders
Type 1 Diabetes Mellitus
DM1 is an autoimmune disorder marked by the destruction of insulin-producing pancreatic beta cells, thus requiring administration of exogenous insulin. Insulin promotes weight gain by stimulating lipogenesis, inhibiting protein breakdown, and slowing basal metabolism. 41 In individuals with DM1, exogenous insulin therapy is often suboptimal, as both the disease pathophysiology and its treatment can induce insulin resistance, thus necessitating escalating insulin doses to maintain proper glycemic control. Additionally, the perceived and actual risk of hypoglycemia can lead to increased food intake. These factors collectively contribute to peripheral hyperinsulinemia and associated weight gain.41-43
The Diabetes Control and Complications Trial demonstrated the substantial benefits of intensive insulin therapy, which involves increased insulin dosing to achieve tight glucose control, and is now recognized as the cornerstone of managing DM1. 43 As a consequence of greater insulin exposure, the historically normal or underweight phenotype seen in individuals with DM1 has shifted with a rising prevalence of obesity. 42 This shift presents a unique challenge for individuals reliant on insulin therapy to obtain the “ideal” slim body physique that society has deemed as the standard of health and beauty. Weight gain is particularly pronounced following the initiation of insulin therapy and during the transition from adolescence to early adulthood, an age span also associated with an increased risk of ED development. 19 In attempts to counteract weight gain, as the desire to lose weight becomes prioritized over the potential risk of DM complications, vulnerable individuals may develop EDs, which manifest in distinct detrimental patterns and present unique clinical challenges. 44
Anorexia Nervosa
When restrictive EDs, such as AN, occur within the context of DM1, it can be challenging to detect. In addition to the frequent concealment and denial of the presence of an ED, the food restriction associated with AN may lead to recurrent episodes of hypoglycemia and/or HbA1c that is at or below the target range. 6 These in-range or low HbA1c levels are often interpreted by healthcare providers as signs of well managed DM1, leading to positive reinforcement and praise. This feedback can inadvertently reinforce disordered eating behaviors as patients perceive their restrictive actions as successful DM management. Thus, the ED may remain undetected until more severe warning signs appear, such as dangerously low BMI, severe malnutrition, fragility fractures, or seizures. 6
Diabulimia
Although not formally recognized in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, the deliberate omission of insulin doses to induce weight loss, commonly referred to as “diabulimia,” is a well-documented, serious, and life-threatening ED that primarily affects individuals with DM1. This behavior was first described in the 1980’s, when early studies observed that persons with DM1 were intentionally omitting insulin as a weight loss strategy.45-48 The phenomenon was more formally addressed in the 1990’s, when research demonstrated that the intentional underdosing or omission of insulin to control weight was associated with an increased risk of DM-related complications. 1
Insulin facilitates the uptake of glucose from the bloodstream into cells for use and storage. When insulin doses are omitted, the body is unable to access glucose for energy, leading to hyperglycemia. The body attempts to eliminate excess glucose through the urine, resulting in glycosuria and loss of calories. To compensate for the lack of glucose as a form of energy, the body begins breaking down muscle and fat, further contributing to weight loss. 49 This metabolic shift also leads to the production of ketone bodies, which can disrupt the body’s pH balance and potentially result in DKA. 26
Due to the central role of insulin underdosing and omission in diabulimia, individuals with this condition often present with significantly higher HbA1c levels, unexplained weight loss, electrolyte disturbances (including low sodium and potassium), frequent bladder and/or yeast infections, and increased DM-related emergency department visits or hospitalizations, including DKA.5,23 It holds a 3-fold increase in mortality compared with people with DM1 who have never insulin restricted. 50
Distinguishing insulin omission as a typical aspect of pubertal behavior and development of autonomy during the adolescent period or as a deliberate strategy for weight loss can be challenging. It is common for adolescents to occasionally miss insulin doses. One study reported that over a third of adolescents missed more than 15% of their bolus doses the day prior to their DM checkup appointment. 51 Other factors contributing to insulin omission include poor adherence, which often emerges during middle adolescence, denial of DM1 diagnosis in social settings, and fear of hypoglycemia. 52
Of note, as diabulimia is not included in the DSM-5, its definition varies across research studies. For instance, some studies define diabulimia as BN in individuals with insulin dependent DM. 31 Others describe it as the purging component of BN. 53 Some researchers differentiate diabulimia from BN but do not clarify whether it is considered a subset of BN or a distinct condition within the context of their study. 7 Additionally, certain studies focus exclusively on one type of ED, excluding the other from their analysis.5,23 As a result, there is ambiguity in both the prevalence and the distinction between diabulimia and BN in the literature.
Type 2 Diabetes Mellitus
DM2 is a chronic condition characterized by impaired glucose metabolism due to deficient insulin secretion and insulin resistance, which, over time, leads to the inability to maintain glucose homeostasis and results in hyperglycemia. While the development of DM2 is influenced by a complex interplay of genetic, metabolic, and environmental factors, obesity remains the strongest risk factor for the onset of insulin resistance and DM2. 54
Conversely to DM1 and the subsequent development of EDs, the presence of an ED often precedes the diagnosis of DM2, as disordered eating behaviors can contribute to metabolic dysregulation that may increase the risk of developing insulin resistance over time.7,20,55 For instance, individuals with conditions such as BED experience repeated cycles of hyperglycemia due to irregular eating patterns and excessive caloric intake.20,56,57 Such behaviors can lead to metabolic stress, further exacerbating insulin resistance and promoting weight gain, which in turn increases the likelihood of the development of DM2. 58 Research indicates that approximately one third of individuals with BED have co-existing DM2. 40
Similar to DM1, psychosocial factors also play a crucial role in the interplay between DM2 and EDs such as chronic stress, low self-esteem, and societal pressures, leading to a vicious cycle where EDs contribute to the development of insulin resistance and obesity, while the metabolic complications of DM2 may aggravate symptoms of EDs. 59 As such, effective management of both conditions often requires an integrated approach that addresses both the physical and psychological aspects of health.
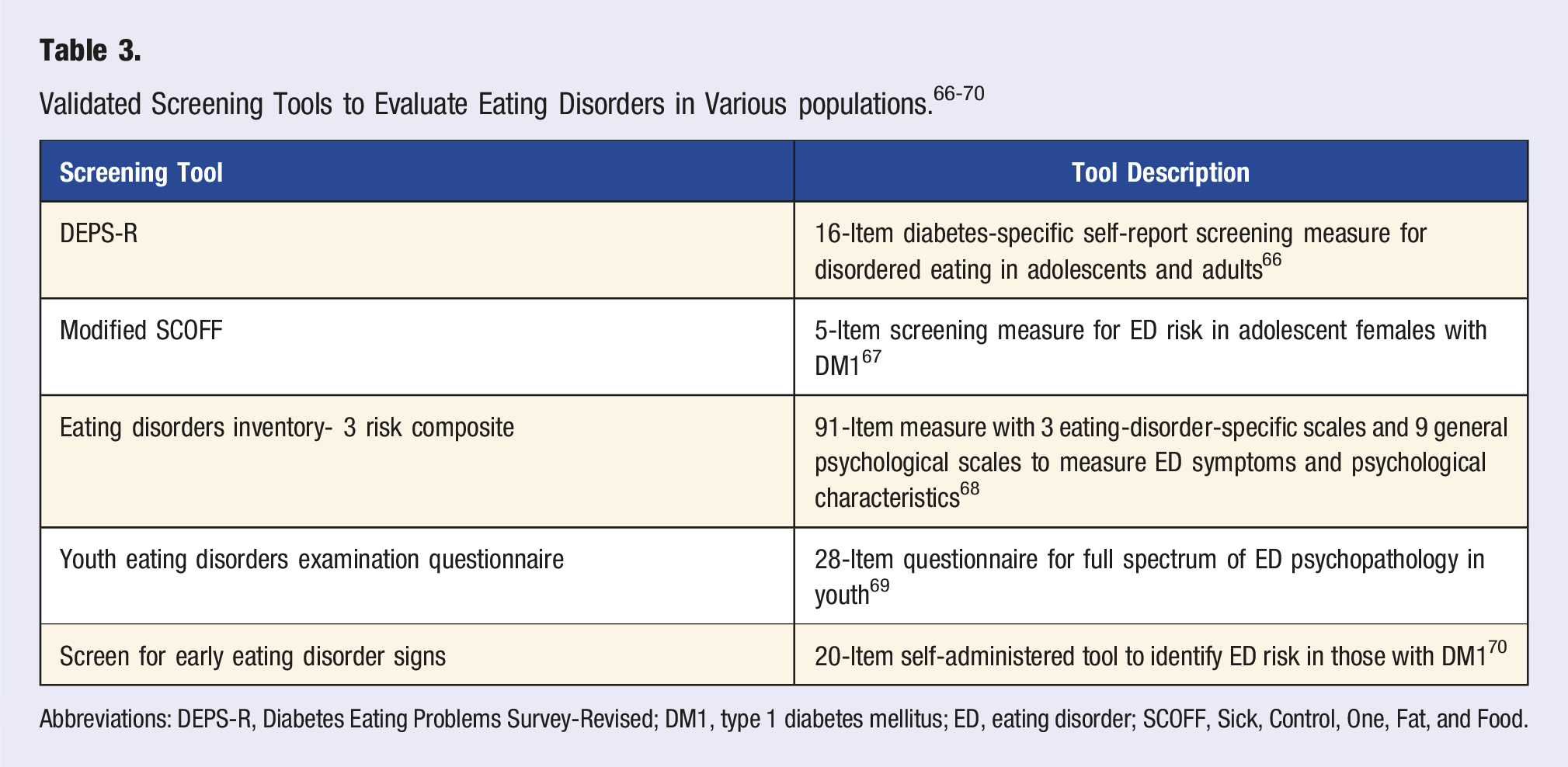
Screening Tools for Eating Disorders
The co-occurrence of DM and EDs is linked to poorer glycemic control, increased risk of complications, and worse treatment outcomes, making early detection, intervention, and prevention crucial to reducing morbidity and mortality. 60 Despite the increasing awareness of EDs, over 60% of DM healthcare professionals report low confidence in identifying comorbid DM1 and EDs, with 78% acknowledging that they do not use screening tools to evaluate disordered eating within this population. 61 Given the higher prevalence of EDs in individuals with DM compared to the general population, the American Diabetes Association recommends ED screening as part of routine care.62,63 However, detecting EDs in patients with DM is complex, as many individuals conceal their relationship with food, with only a small percentage seeking help. Furthermore, there is a spectrum of dysfunctional eating behaviors, with 7% of adolescents with DM1 diagnosed with an ED and up to 40% exhibiting subclinical behaviors. 64 Screening tools designed for the general population may either underestimate or overestimate ED risk in patients with DM, as DM management necessitates careful monitoring of food intake and its impact on blood glucose levels. 65 Therefore, effective care requires a multifaceted approach, including screening tools, DM technology data, and clinical interviews.
To accurately screen and diagnose EDs in individuals with DM, a comprehensive approach is required. This should include diagnostic interviews conducted by clinicians with expertise in both EDs and DM, the use of validated screening tools, interpretation of clinical indicators such as blood glucose levels, HbA1c, BMI, and an understanding of long-term trends derived from DM technology.
Diabetes Mellitus Technology and Eating Disorders
Advancements in DM technology, including CSII (continuous subcutaneous insulin infusion), CGM (continuous glucose monitoring), and their integration into automated insulin delivery systems, have significantly improved DM management by increasing flexibility and optimizing treatment outcomes.73,74 These technologies have demonstrated positive effects on key metrics such as HbA1c, time in range, reduced hypoglycemic episodes, and overall patient satisfaction, leading to widespread adoption as the standard of care for individuals with DM1. 73 However, despite their evident benefits, the impact of these technologies on comorbid conditions, particularly EDs and disordered eating behaviors, remains an area of limited research.
Technological advancements in DM management have significantly alleviated the mental burden associated with the condition, particularly the need for rigid control over food intake. CSII systems deliver a continuous supply of rapid acting insulin (basal) throughout the day, with additional insulin doses administered based on the amount of food consumed (bolus) and blood glucose levels (correction). When combined with CGM data, these systems enable individuals to make timely insulin micro-adjustments guided by real-time glucose data. This integration provides greater flexibility in DM management, reducing the need for strict dietary control and potentially fostering more relaxed eating behaviors. It may also decrease cognitive dietary restraint and reduce preoccupation with food intake.75,76 Furthermore, the ease of blood sugar monitoring and insulin dosing provided by these technologies helps mitigate episodes of disinhibited eating, which often arise from hypoglycemic events triggered by mismatched blood sugar level and insulin doses. These events can lead to rebound hyperglycemia, followed by further insulin correction, creating a vicious cycle that disrupts hunger and satiety signals. By reducing these fluctuations, DM technology offers a more stable physiological environment, which may, in turn, reduce the occurrence of disordered eating behaviors. 77 In theory, this shift toward more relaxed eating behaviors, along with restoring the hypothalamic-gut axis, could contribute to a decrease in ED within this population.75,77
Conflicting data exists regarding the impact of CSII on EDs. Some studies have shown that initiation of CSII is associated with significant reduction in DEPS-R scores, while others have found no significant relationship between insulin delivery type and ED diagnoses, except in the case of AN, where CSII was linked to a nearly 50% reduction in the likelihood of diagnosis.71,78 The use of DM technology in patients with comorbid EDs presents additional challenges, with evidence suggesting that individuals with both conditions are less likely to adopt and continue CSII.79,80 Additionally, although CSII users have better glycemic control and lower daily insulin requirements than those on multiple daily injections, both groups experience similar trends in weight gain over time. 81
Clinical observation has led to speculation that CSII, which allows for precise titration of insulin doses, combined with CGM, which provides real- time glucose data, may enable patients to manipulate insulin delivery in a way that “safely” promotes passive hyperglycemia with a reduced risk of DKA.6,74,82 While this idea seems plausible in theory, it lacks sufficient empirical support. Furthermore, several studies challenge this speculation. Studies have found that individuals with DM1 who engaged in insulin omission or restrictive or binge eating behaviors had significantly lower HbA1c levels when using CSII compared to those on multiple daily injections. 83 Other studies reported that female adolescents using CSII exhibited better glycemic control, with none reporting insulin omission, while 15% of those on multiple daily injections admitted to skipping or reducing insulin doses. 84 Additionally, it was observed that individuals on multiple daily injections were more likely to omit insulin doses than those using CSII. 85
Data from CSII and CGM can be used to detect patterns indicative of underlying ED. Chronic hyperglycemia has been identified as a key CGM marker of dysfunctional eating behaviors.
86
Insulin omission has been found to be less common in the morning and more frequent in the afternoon, peaking between 3:00 and 3:59
The potential benefits of DM technology in addressing EDs remain underexplored. It is hypothesized that such technology may help decrease disordered eating behaviors by providing increased flexibility and reducing mental strain. Although there are concerns of ease of manipulating insulin delivery with DM technology, there is no objective evidence to support this claim. Additionally, DM technology, particularly CGM data, can be used to identify patterns in blood glucose levels and insulin usage data indicative of EDs. Further investigation through larger and more comprehensive studies is necessary to better understand these dynamics.
Multidisciplinary Approach to Treatment
Given the complexity of managing disordered eating and EDs in patients with DM, a collaborative, interprofessional care team is essential for prevention, screening, diagnosis, and treatment. This team should ideally include a practitioner for medical DM oversight, a registered nutritionist for dietary counseling, and a psychotherapist to provide behavioral health support.6,73 Additionally, a strong support system can be transformative for a patient’s recovery. 73
Building rapport between the healthcare team and the patient is critical for fostering positive health behaviors and supporting psychological well-being.88,89 A patient-centered, collaborative approach in a non-judgmental environment not only helps prevent EDs, but also supports treatment if an ED develops.90,91 Language choice is crucial, as judgmental terms can foster shame and impede the healing process. 91 Discussions about weight management should start with motivational interviewing, inquiring whether a patient is willing to engage in the conversation. 73
Treatment goals should be individualized based on the diagnosis, severity, and duration of symptoms. Initial care should prioritize medical safety by addressing behaviors that can lead to acute complications. 6 Clinical interviews should assess the use of maladaptive products such as laxatives, diuretics, and diet pills, and examine insulin restriction practices to counsel patients on the risks of DKA and other health consequences. Once medical safety is established, treatment should focus on small, achievable goals. As patients progress, adjustments to medical nutrition plans, DM management, and self-care goals are necessary. 6
While a multidisciplinary team of healthcare professionals is ideal for managing EDs in patients with DM, resource limitations may prevent its feasibility in all settings. Practitioners must understand their own limitations and abilities to treat, and their scope of practice. By fostering an environment and rapport with patients that facilitates the identification of disordered eating patterns, they can assess the need for additional support and form a patient-centered team tailored to the individual’s needs.
Psychotherapeutic Management
The American Psychiatric Association identified cognitive behavioral therapy, interpersonal psychotherapy, and dialectical behavioral therapy as effective psychotherapeutic interventions for the treatment of EDs. 92 Cognitive behavioral therapy targets maladaptive thoughts and behaviors that contribute to the development and maintenance of EDs and have been adapted for various ED subtypes. 93 For example, enhanced cognitive behavioral therapy was developed specifically for AN, with a focus on normalizing eating patterns, eliminating compensatory behaviors, and addressing distorted cognitions related to body shape and weight. 94 Dialectical behavioral therapy emphasizes emotional regulation, distress tolerance, and mindfulness, aiming to help patients manage rigid, polarized thinking and behavioral patterns. 93 Interpersonal psychotherapy addresses interpersonal difficulties and role transitions to strengthen emotional coping skills and reduce reliance on disordered eating behaviors to manage stress. 93 Among adolescents, family-based treatment, also known as the Maudsley Method, is a first-line behavioral intervention that empowers parents to take primary responsibility for their child’s recovery. 95 Adaptations have been made to create the Maudsley approach for the treatment of AN in adults. 96 These psychotherapeutic approaches, particularly when tailored to the unique needs of individuals with co-occurring DM and EDs, offer a foundation for integrated treatment that addresses both psychological and medical complexes.
Medical Management
Structured psychological therapies remain the first- line treatment for EDs, with limited medications approved for adjuvant therapy. Currently, there are no pharmacological medications approved by the Food and Drug Administration (FDA) for the management of AN. 97 In addition to nutritional rehabilitation and psychotherapy, it is essential to closely monitor patients for medical complications, including refeeding syndrome. Refeeding syndrome can result from reinstating nutrition after fasting, causing electrolyte imbalances that lead to cardiopulmonary, hematological, and neurological dysfunction. 98
Fluoxetine is the only FDA-approved medication for BN, which has been shown to reduce binge and purging episodes and the risk of relapse.99-101 Close monitoring is required during treatment, as the abrupt cessation of purging behaviors can lead to pseudo-Bartter’s syndrome. This condition results from volume depletion caused by purging, which activates the renin-angiotensin-aldosterone system, leading to increased aldosterone secretion and subsequent peripheral edema after cessation of purging behaviors. 102
Lisdexamfetamine is the only FDA-approved medication for BED. 103 Although several medications, such as topiramate, demonstrated efficacy in clinical trials and are frequently prescribed for off-label use, many are associated with unfavorable side effects. 104 Glucagon-like peptide-1 receptor agonists may offer a novel approach for reducing binge eating episodes by modulating appetite and promoting satiety.105,106 However, large-scale randomized controlled trials are needed to confirm their effectiveness for this specific population.
Pharmacological interventions, although limited, offer valuable adjuncts in managing specific EDs. Continued research into novel medications, particularly those targeting appetite regulation, holds promise for advancing pharmacological treatment options.
Nutritional Management
Nutritional rehabilitation for EDs may take place in a variety of locales, including the outpatient setting, inpatient/hospital setting or a residential treatment center. The setting and duration of nutritional management for EDs varies depending on the degree of malnutrition and medical compromise, as well as the patient’s degree of psychological distress. These levels of care may change throughout the course of treatment, as need arises, and as the individual progresses through the phases of ED recovery. 107
A comprehensive nutrition assessment begins with gathering information on the patient’s current and past eating patterns. This includes details on the types and amounts of foods and beverages consumed, timing of consumption, eating environment, food preparation methods, and meal origins such as specific brands, restaurants, or personal cooking. Eating-related behaviors are also evaluated, such as food exclusions, binging, purging, fasting, calorie counting, food measuring, chewing and spitting, excessive water intake and overuse of non-nutritive items such as condiments, salt, pepper, or spices. The assessment further explores the use of diet pills, diuretics, laxatives, and any rituals affecting intake. Additionally, exercise and non-exercise movements, such as refusal to sit or excessive pacing, are examined. 107
The objective of nutritional rehabilitation for all EDs is to restore a balanced and flexible eating pattern with adequate caloric intake and to obtain and maintain an appropriate body weight. Nutritional management varies by ED type. For restriction-based ED such as AN, atypical AN, and ARFID, the primary focus of treatment is to consume sufficient caloric intake to correct the underlying malnutrition.108,109 In contrast, for BN and BED, the emphasis is on correcting eating patterns and behaviors, including eliminating purging, binging, or fasting. 110
A balanced eating pattern should neither be restrictive nor influenced by body image concerns. Balanced eating involves obtaining nutrition from all food groups, with no foods labeled as healthy/unhealthy or good/bad, as all foods contribute to nutrition. The recommended nutritional plan is personalized, based on the type of ED and the patient’s eating pattern prior to developing the disorder. For AN/atypical AN, increasing caloric intake is the first step and requires the patient to not only fight against their ED, but also their own physiology which has adapted to a starved state. For a patient that is malnourished, the initial stages of eating may be extremely uncomfortable and accompanied by gastrointestinal symptoms including abdominal pain, early satiety, bloating and nausea. 108 A patient with BN/BED may also struggle with gastrointestinal symptoms, which are both a trigger for and effect of binging and/or purging. This creates a cycle which must be broken for recovery to progress. 110
Nutritional care for ED varies based on the level of acuity and treatment setting. In outpatient care, a nutrition plan is established to stabilize or gradually increase nutritional intake to restore weight, if required, and address food-related fears and behaviors. 107 Family members often aid, particularly if the patient is an adolescent or young adult; however, as the patient becomes more independent and requires less support, they take greater ownership of their decisions. Some modalities of outpatient treatment, such as Family-Based Treatment, involve family members in preparing, providing, and monitoring meals. 111 Patients are seen regularly by their care team, typically starting with weekly or biweekly visits, then transitioning to monthly check-ins.
In a residential treatment center, day program, or intensive outpatient program, the eating environment is more structured, as the patient is required to adhere to specific meal planning guidelines and schedules. All meals are provided and supervised by staff. Foods previously eliminated or restricted by the patient are reintroduced, often more quickly than in an outpatient care setting. This environment allows challenges to be addressed more efficiently, as patients are immersed in therapy with immediate support.
In the inpatient setting, nutritional care is tightly regulated as patients are monitored for medical stability. In a severely malnourished patient, the total calories needed to achieve weight restoration will most assuredly be above a maintenance level, often reaching 3000 kcals/day or more. Nutritional supplements may be used to achieve the high level of calories required, and in the case of food refusal, a nasogastric tube may be necessary. Although high calorie needs are not specific to an inpatient setting, total caloric intake may be advanced more quickly in a hospital. 112
Medical safety remains a central concern for the treatment of EDs in patients with DM and requires close coordination among members of the multidisciplinary care team. As nutritional rehabilitation typically involves a gradual increase in caloric intake, adjustments to the patient’s DM management plan may be necessary. For instance, as caloric intake increases and weight is restored, insulin regimens may need to be modified accordingly. Importantly, these adjustments should be individualized rather than standardized; insulin changes must be tailored to each patient’s unique physiological and clinical needs.
Sample Meal Plans for Patients With an Eating Disorder (Person A) and for Patients With Comorbid Eating Disorder and Diabetes Mellitus (Person B) With Modifications for Glycemic Control.
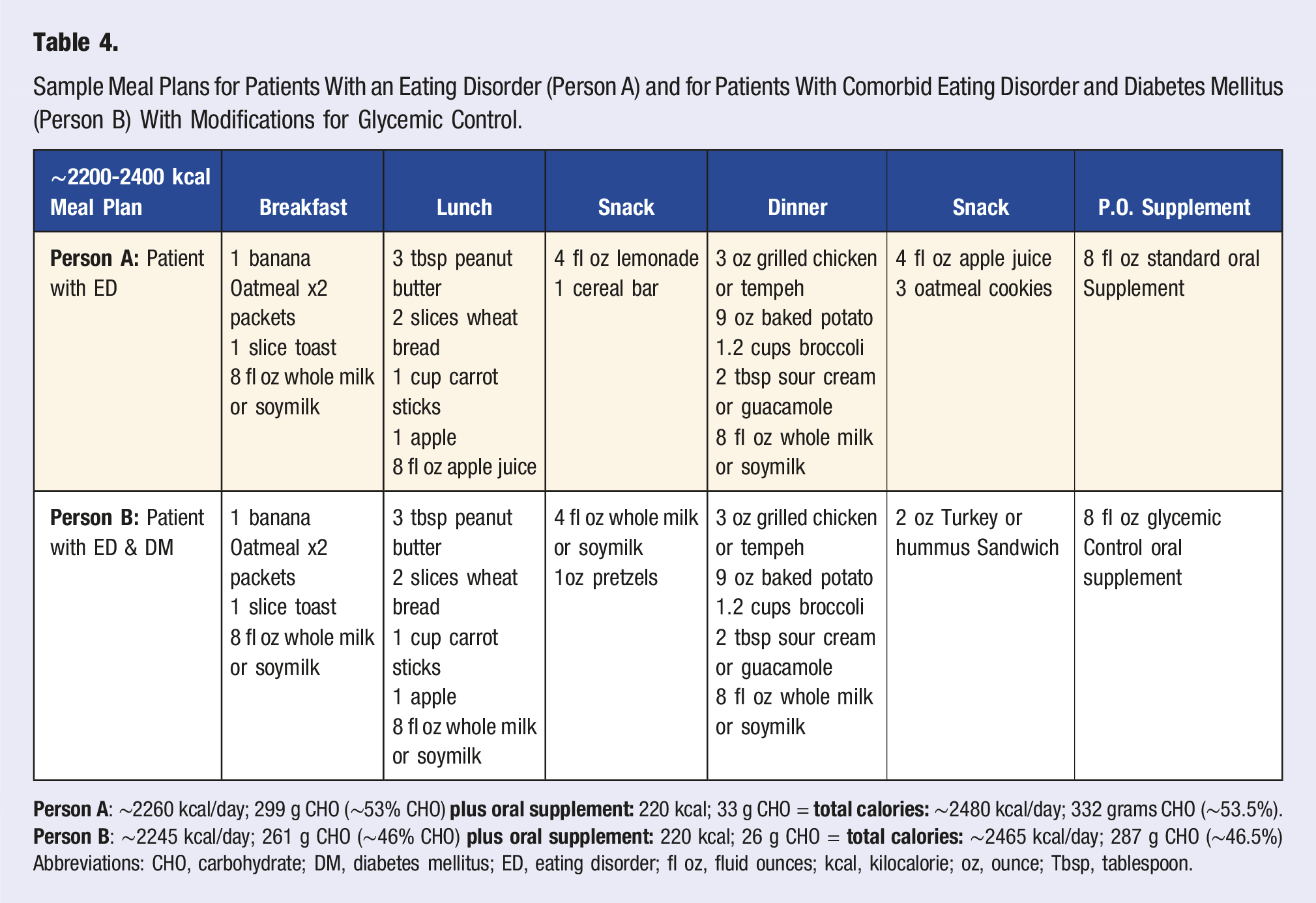
Abbreviations: CHO, carbohydrate; DM, diabetes mellitus; ED, eating disorder; fl oz, fluid ounces; kcal, kilocalorie; oz, ounce; Tbsp, tablespoon.
Discussion
Despite increasing awareness of EDs among patients with DM, challenges persist, including low screening rates, limited confidence among healthcare professionals in making diagnoses, and insufficient multidisciplinary collaboration to address these issues. While a comprehensive, multidisciplinary approach is essential to effectively manage EDs in individuals with DM, several obstacles exist in the prevention, screening, diagnosis, and treatment of EDs and disordered eating.
Although the American Diabetes Association recommends the use of validated screening tools for disordered eating, there are no established guidelines for their implementation.
62
Screening tools have been developed specifically for individuals with DM, yet these tools have notable limitations, including gender and selection biases. Additionally, they often fail to account for essential psychosocial factors, such as concurrent psychiatric conditions, parent-child conflicts related to DM management, and parental distress over DM care. Moreover, many screening tools do not capture the full spectrum of dysfunctional behaviors, therefore many concerning eating patterns may remain undetected. Additionally, screening tools should be used in conjunction with other data sources, including insights from motivational interviewing, objective clinical indicators, and data from DM technology. Clear guidelines for incorporating these various data points into a diagnostic workflow are currently lacking. Figure 1 presents a comprehensive workflow that outlines the processes of prevention, screening, diagnosis, treatment, and ongoing support. Future research should aim to develop more effective screening and diagnostic strategies that incorporate the multifaceted approach needed to accurately care for these individuals. Larger, more inclusive studies are needed to explore the potential advantages of integrating DM technology in the diagnosis and management of ED and disordered eating. Proposed comprehensive workflow to outline the processes of prevention, screening, diagnosis, treatment, and ongoing support of ED in patients with DM. Abbreviations: DM, Diabetes mellitus; ED, eating disorder.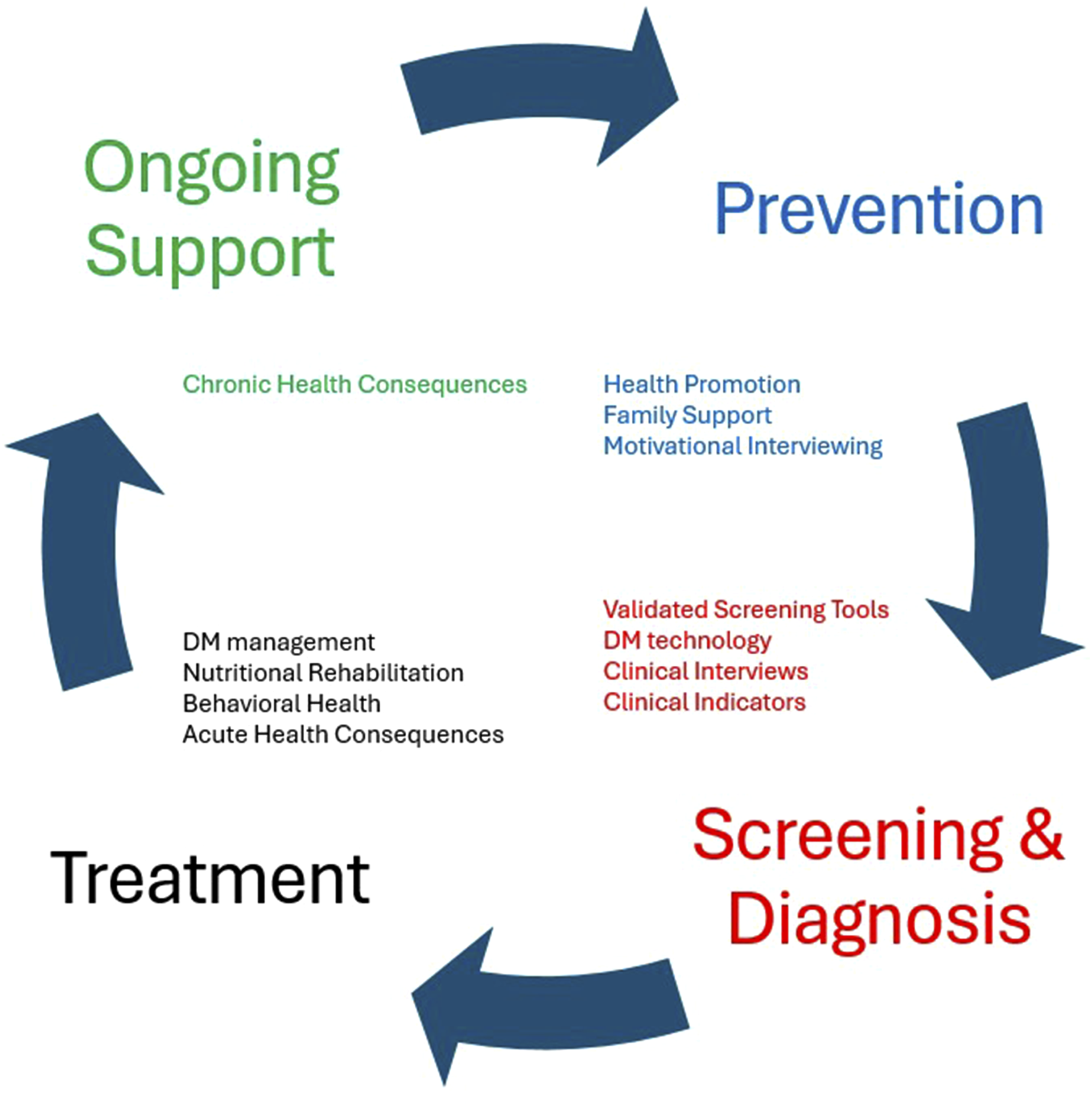
The American Diabetes Association also emphasizes that when evaluating disordered eating symptoms, the underlying etiology and motivation for the behavior should be assessed by qualified professionals with expertise in both DM and ED management. 62 While a patient-centered, collaborative approach is ideal, resource limitations may hinder its implementation in some settings. Future studies should focus on developing strategies to overcome these resource-related barriers and ensure that medical management, nutritional counseling, and behavioral health are adequately supported within multidisciplinary teams, even in resource limited environments. Additionally, healthcare practices should prioritize staff training to ensure that personnel feel confident in diagnosing and managing these patients.
Conclusion
The co-occurrence of DM and EDs presents a high-risk combination of health conditions that significantly increases morbidity and mortality. While individuals with DM share common risk factors for developing EDs with the general population, their risk is exacerbated by the unique challenges of DM management. Prevention, screening, diagnosis, and treatment are key components of managing the dual diagnoses of EDs and DM. Given the complexities involved, a multidisciplinary and holistic approach is paramount for providing effective care.
The process of identification, management, and team assembly in the care of individuals with DM and EDs begins with recognizing potential warning signs of disordered eating. Once concerns are raised, the practitioner should initiate an open and nonjudgemental dialogue with the patient and/or utilize a validated screening tool to further explore the possibility of an ED. If disordered eating or an ED is identified, the practitioner must conduct a comprehensive assessment to evaluate the severity of the condition and the associated medical and psychological risks. The patient’s level of risk will guide clinical decision-making regarding the appropriate level of care- ranging from outpatient support to more intensive inpatient interventions. Following the determination of the appropriate level of care, a multidisciplinary treatment team should be assembled to address the patient’s unique needs.
Footnotes
Author Contributions
H.F. contributed to the drafting, writing, and editing of the final revision of the submission. S.K.K. contributed to the drafting and writing of the manuscript. J.A. and J.Z. contributed to the writing of the manuscript. R.S.R. contributed to the drafting and editing of the final revision of the submission.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RSR received royalties from Springer Nature, publication support and Advisory Board for Sanofi and serves on the board of directors for the American Association of Clinical Endocrinology. HF, SKK, JA, and JZ report no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.