Abstract
Exercise is vitally important in the prevention of weight gain or maintaining weight status, as well as weight loss. High-intensity exercise causes a short-term suppression of hunger of approximately 15 to 60 minutes. Although there is evidence for compensatory food consumption, it usually does not make up for the energy deficit created by exercise. The exception occurs when individuals consume or reward themselves with energy-dense foods or drink. Because people tend to eat the same volume of food each day, on days when they exercise, they will remain in an energy deficit. However, on sedentary days, a positive energy balance is likely if caloric restriction is not imposed, which could result in weight gain. Caloric restriction alone leads to loss of lean body mass, while the inclusion of exercise with an energy deficit helps conserve lean tissue. There are a myriad physiological factors such as the concentration of hormones (GLP-1, PYY3-36, leptin, and ghrelin) and metabolites (free fatty acids and glucose) that either stimulate or inhibit signals for hunger and/or energy intake, but the effect of exercise on these circulating factors is complex and not completely understood.
‘It matters whether an energy deficit and body fat loss is induced through exercise and/or caloric restriction.’
Introduction
Physical activity, whether it is planned, such as exercise, or unplanned, such as nonexercise activity thermogenesis, has been shown to affect both sides of the energy balance equation through daily energy expenditure and by stimulating energy intake. Unfortunately, few Americans engage in regular physical activity and many have adopted a very sedentary lifestyle.
Exercise is vitally important in the prevention of weight gain or maintaining weight status, as well as weight loss. There are a myriad physiological factors that are affected by exercise, such as the concentration of hormones and metabolites that either stimulate or inhibit signals for hunger and/or energy intake.
Exercise, Hunger, and Energy Intake
Hunger is the drive to seek nutrients as negative energy balance or body fat losses increase. Early in the 20th century, Cannon and Washburn described hunger as periodic eruptions of gastric contractions when the stomach was empty. 1 Later investigators described hunger as being sensitive to activation or suppression by internal metabolic events as well as by nonhomeostatic environmental stimuli, which was integrated by the central nervous system. 2 The current view of hunger places an emphasis on central neural mechanisms responding to hormonal and metabolic signaling of energy deficit or body fat loss.
It matters whether an energy deficit and body fat loss is induced through exercise and/or caloric restriction. When a deficit is induced by exercise, there is no conscious detection of caloric withdrawal; however, if the deficit is a result of caloric restriction, hunger increases as a result. 3 Unfortunately, the suppression of hunger following exercise may not always translate to a decrease in food consumption throughout the course of the day, but the imposition of exercise to create a deficit can help keep one in a negative energy balance.
The intensity of the exercise performed can influence the perception of hunger. In acute laboratory studies, it has been well documented that high-intensity exercise decreases hunger during, immediately, and for 15 to 60 minutes following exercise.3-6 What is unclear is how long the diminished hunger due to exercise lasts as most reports indicate a transient suppression3-6 and few reports measure the hunger response to exercise beyond 60 minutes. Likewise, different types of exercise can influence hunger: swimming 7 and cycling submerged in cold water 8 have been associated with increased hunger following the exercise bout and a lack of weight loss following exercise as compared with walking, running, and cycling programs.
When differing energy expenditures have been produced through exercise, compensation for the deficit can be attributed to the portion size and the energy density of the food consumed following the activity. 9 Lean, young, healthy males who have undergone high-intensity exercise had suppressed hunger and a delay until food was requested, but when they consumed a diet that was energy dense, the energy deficit imposed by the exercise regimen was negated. 9 In lean, young women, there was no marked hunger suppression following exercise but again, the energy deficit created by exercise was wiped out by an energy-dense selection of food. 10 Furthermore, when reducing exercise energy expenditure from a moderate-intensity exercise regimen (1.8× resting metabolic rate) to near sedentary levels (1.4× resting metabolic rate) while residing in a whole-body calorimeter with ad libitum access to food for 1 week each, men did not reduce their energy intake in response to the decrease in energy expenditure. This imbalance due to decreased energy expenditure led to a positive energy balance over the 7-day period. 11
People tend to eat for a sense of fullness, and postexercise dietary choices will play a role on whether they remain in a deficit or not. The role of age and exercise intensity on postexercise dietary intake was examined in a study by Wuorinen et al (unpublished data). Three trials of either sedentary, 60 minutes of walking at 40% of V
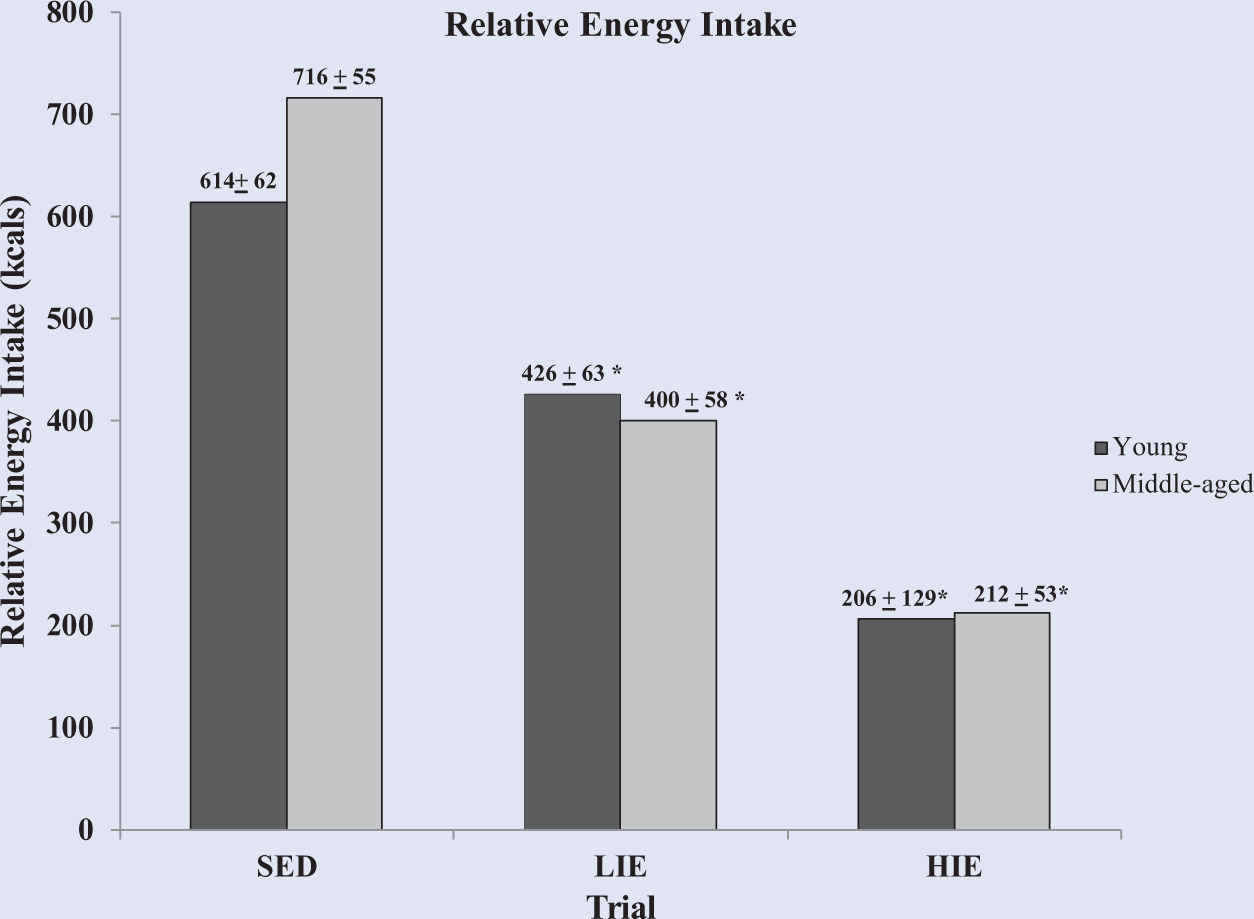
Relative energy intake 30 minutes after sedentary (SED), low-intensity exercise (LIE), and high-intensity exercise (HIE) trials in young and middle-aged overweight women (mean ± standard error). Relative energy intake is calculated as follows: postexercise energy intake − [total energy expenditure of exercise − (exercise time × resting energy expenditure in kcal/min)]. *P < .05 compared with same age group, SED trial. Both young and middle-aged women had significantly lower relative energy intake after the LIE and HIE trials compared with the SED.
Although there is an overwhelming amount of evidence of transient hunger suppression and decreased energy intake following exercise, the inhibition is not complete. Additionally, the expectation that a food reward has been earned by exercising 13 leads some individuals to eat or drink foods that negate the exercise energy deficit. Thus, it is much more difficult to maintain or lose weight if food rewards are consumed on days where exercise takes place.
Hormones and Metabolites
Hunger and subsequent energy intake are physiologically regulated by the neuroendocrine system, specifically gut hormones, that signal for hunger. 14 The urge to eat has been studied for years in animals and humans, but a unifying physiological explanation of hunger and initiation of eating is still not available. 15 The prevailing view is that there is an interaction of the metabolic and endocrine systems with regulatory hypothalamic and ventral tegmental brain areas to provide the information for the initiation of food consumption and the regulation of energy balance. The available evidence is stronger for the termination of meals through the intervention of gastrointestinal hormones such as glucagon-like peptide-1 (GLP-1), and peptide YY (PYY), 16 or the suppression of feeding by adipose tissue hormone leptin, than for the stimulation of the drive to eat thought to be initiated by the gastric hormone ghrelin. 17
Glucagon-like Peptide-1
Glucagon-like peptide-1 (GLP-1) is released by the ileum and colon and has been shown to decrease gastric emptying. 18 GLP-1 is released in proportion to the amount of energy intake and has been shown to inhibit energy intake 19 by altering hypothalamic circuitry. 20 Recent evidence indicates that GLP-1 levels increased following exercise and were associated with a significant decrease in food consumption.5,19 There have also been reports of no effect of exercise on GLP-1 concentrations, 21 indicating that it could simply be the difference between exercise used in the study protocols or the population observed. If the enhanced concentrations of GLP-1 and PYY3-36 are able to aid in decreasing energy intake following exercise, it would be beneficial to determine what types of exercise stimulate GLP-1 secretion.
Peptide Tyrosine Tyrosine
Peptide tyrosine tyrosine (PYY) appears in 2 forms, PYY1-36 and PYY3-36; however, PYY3-36 tends to be the more potent form to inhibit gastric emptying. 22 PYY3-36 is released from the L-cells of the intestine in proportion to the amount of calories consumed at a meal with the resulting effect of signaling satiety, suppression of hunger, and inhibition of energy intake. 23 Evidence is limited on the effects of exercise on PYY3-36 secretion. In some cases, exercise has had a stimulatory effect on total or PYY3-36, which could partially explain the decreased hunger and energy intake following exercise,5,6,21,24 and is decreased in response to caloric restriction. 24 The increased concentrations of PYY3-36 following exercise could partially explain why there is a transient decrease in hunger sensations and diminished energy intake. There is also evidence that exercise had no effect on either total or PYY3-36, 25 again, this could simply be a difference in the duration, intensity, frequency or type of exercise used in various studies, as well as the population examined. Further examination of the effects of exercise duration and intensity on the secretion PYY3-36 is needed.
Leptin
Leptin is released from adipose tissue in proportion to the amount of stored triglycerides. Leptin relays information to the brain areas responsible for hunger and is secreted in response to short-term changes in energy availability. 26 Thus, when there is an energy surplus, plasma leptin levels increase, and hunger should be suppressed. Leptin secretion is reduced in response to exercise energy deficit, 27 which should stimulate hunger and an increased drive for energy intake, thereby attenuating the amount of body mass lost through exercise-induced energy expenditure. However, in the short-term, this is not reflected in hunger or energy intake. It is more likely that leptin acts as a long-term signal as body fat loss occurs to stimulate the drive for energy intake. The signals produced by leptin concentrations in relation to exercise, hunger, and energy intake are varied and further research is needed to provide evidence for leptin’s role.
Ghrelin
Ghrelin is produced primarily by the stomach, but also secreted by other areas such as the hypothalamus.28,29 Circulating ghrelin is influenced by acute and chronic changes in nutritional state. 28 Shortly before a meal, ghrelin is 2-fold that of immediate postprandial levels, and falls to low levels within 1 hour postprandially. 17 There are 2 major forms of endogenous ghrelin, acylated and des-acylated, which together are considered total ghrelin; however, the focus of recent research is on acylated ghrelin, which is the physiological form that stimulates hunger and energy intake. 30 The effects of exercise on concentrations of ghrelin have been mixed. Some investigators have reported a decrease in plasma ghrelin in response to acute exercise,21,31 an increase, 32 or no effect. 19 The varied results of exercise on ghrelin concentrations most likely are because of the varying exercise regimens imposed. In the cases where ghrelin concentrations are decreased in relation to exercise, if hunger was measured, transient hunger suppression was also noted. 21 This relationship between decreased ghrelin and hunger following exercise, provides further evidence in the hunger suppression induced by exercise and the ability to create an energy deficit on exercise days, provided that an overload in energy-dense foods is avoided. When caloric restriction that produces the same energy deficit as exercise is imposed, caloric restriction increases both hunger and acylated ghrelin concentrations. 24
Metabolic Fuels
Removal of metabolic fuels such as free fatty acids and glucose from circulation by exercise energy expenditure neither appears to affect hunger nor produces caloric compensation in meals. This line of evidence justifies the global hypothesis that short-term withdrawal of metabolic fuels through exercise-induced energy expenditure is not consciously perceived as changes in hunger. 33 Instead, they trigger endocrine reflexes such as release of ghrelin during energy deficit and secretion of leptin during energy surplus. These hormones penetrate the median eminence and adjust the availability of metabolic fuels without an effect on the conscious sensing of energy state. 33
The Complex Connection
The full effects of exercise-induced energy expenditure on hunger are yet to be elucidated. The effect of exercise on the milieu of the gastrointestinal hormones and metabolites, known to have a hunger and energy intake suppressing effect, requires further investigation. It is clear that exercise can affect the hormonal and metabolic environment that signal hunger and energy intake, but to what degree in the various populations—men versus women, lean versus obese, and young versus old—is unknown. Furthermore, there is a clear difference between the energy deficit induced through exercise compared with caloric restriction; the physiological changes that occur are different, as are the responses of hunger and energy intake. There are compensatory mechanisms that work elegantly when energy restriction occurs; however, this is bypassed as a result of exercise. Additional evidence is needed to compare the physiological environment that occurs during energy deficit imposed by these 2 different ways to create an energy deficit to produce weight loss and/or maintenance.
What is the most successful way to lose or maintain weight: diet or exercise? It has been shown that when people undergo a weight loss program that involves caloric restriction and moderate aerobic exercise, 75% of the weight loss is from fat and 25% is from lean tissue. 34 However, when people engage in caloric restriction, aerobic and resistance exercise, the amount of lean tissue loss is significantly less, 35 which can mean greater success because of the increase in metabolically active muscle tissue. Therefore, maintaining muscle mass via exercise is vital to weight maintenance, along with regular energy intake as it takes food to stimulate and sustain metabolism. For an individual trying to lose fat and overall mass, an adequate amount of food can complement exercise, whereas restriction of food can antagonize the exercise program. 36
Conclusion
If people engaged in activity to meet the minimum standard set by the American College of Sports Medicine, exercise would contribute significantly to the energy deficit needed to stimulate weight loss or maintenance. Unfortunately, by remaining sedentary, not only are people not expending energy, they are also not compensating by eating less, and thus the obesity epidemic continues on.
Footnotes
Acknowledgements
![]() is from a project supported by the Vermont Genetics Network through Grant Number P20 RR16462 from the INBRE Program of the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of NCRR or NIH. The author would like to thank the subjects who have participated in the studies describe, as well as contributions of the many students who have worked on various projects in the authors laboratory. Additional thanks are given to Katarina Borer, The University of Michigan, for her many years of mentoring.
is from a project supported by the Vermont Genetics Network through Grant Number P20 RR16462 from the INBRE Program of the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of NCRR or NIH. The author would like to thank the subjects who have participated in the studies describe, as well as contributions of the many students who have worked on various projects in the authors laboratory. Additional thanks are given to Katarina Borer, The University of Michigan, for her many years of mentoring.
