Abstract
Because higher body fat and lower lean mass is associated with excess morbidity and mortality, health care teams need specific science-based recommendations to advise clients on lifestyle approaches to alter body composition. Combining exercise with modest energy restriction is recommended to cause up to 20% more weight and fat loss, improved function, and reduced lean mass loss than modification of diet alone. The optimal diet for developing a leaner body composition is one that is modestly reduced in energy and contains lower fat and higher protein than average. Aerobic exercise alone may cause minimal change in body weight (1%-3%) and fat if greater than 250 min/wk. Higher intensity or duration of exercise can cause more substantial fat loss but may have poor long-term compliance. Resistance exercise boosts lean body mass and improves some health indicators but generally does not cause fat loss. In summary, consumption of a low-fat (20%-25% of energy), moderately high-protein (>1.2 g/kg and up to 20%-25% of energy) diet with modestly reduced energy (500-1000 kcal/d) combined with aerobic (3-5 d/wk, at least 250 min/wk) and resistance exercise (2 d/wk) can improve body composition to reduce clinically relevant risks associated with obesity.
‘Health care teams need specific science-based recommendations to give appropriate advice about lifestyle approaches for improving body composition.’
Introduction
Reduction in body fat is a common personal and clinical objective, but substantial confusion exists regarding effective and reasonable strategies to achieve this goal. More than 60% of the over 4000 obese adults studied from 2001 to 2006 in the National Health Nutrition Examination Survey (NHANES), reported that they were trying to lose weight. 1 Some claimed success; 40% had lost 5% or more of their body weight, and 20%, at least 10%. It is noteworthy that use of popular diets, liquid diets, nonprescription weight loss pills, and diet products were not associated with reported successful weight loss. Although specific details were not provided, lifestyle changes like eating less fat and exercising more were related to weight loss success in these adults. Physicians and other members of the health care team play key roles in influencing motivation for body fat reduction. Data from the NHANES 2005-2008 showed that being told by a health professional that they were overweight or obese increased the likelihood that people would attempt to lose weight by 5- to 8-fold. 2 Health care teams need specific science-based recommendations to give appropriate advice about lifestyle approaches for improving body composition.
This review will discuss the value of modifying either or both sides of the energy balance equation—energy intake and expenditure—for improvement in body composition in over-fat adults. The most effective practical strategies for reducing body fat while maintaining or increasing lean mass will be summarized while providing the scientific rationale that supports these recommendations. For purposes of focus, inclusion of studies of clinical populations, including those with diabetes, was avoided.
Connection Between Body Composition and Health
Numerous studies connect high body fat, especially located in the visceral region, with higher morbidity and mortality for middle-aged3,4 and older adults. 5 The American Heart Association identifies obesity as a primary risk factor for cardiovascular disease and recommends that weight loss be a primary therapy for obese patients with heart-related disease because this improves most of the risk factors for heart disease—for example, insulin resistance, dyslipidemia, and hypertension. 6 Many of the studies that identify health benefits of weight loss do not measure body composition but use indirect methods such as BMI or waist circumference to estimate degree of excess body fat. The assumption is that most of the benefits of weight loss are secondary to lower body fat, but studies that measure body composition provide more powerful evidence for this connection.
The Health ABC study, a prospective cohort study that measured the body composition of more than 3000 older men and women using dual-energy X-ray absorptiometry (DXA), provides good direct evidence for a link between body fat and health. Analyses of this cohort demonstrate an association between high body fat and conditions such as sarcopenia and cognitive impairment. These associations may be mediated by the connection between high body fat and elevated circulating inflammatory factors. 7 For example, individuals in the Health ABC cohort with higher circulating inflammatory markers had more reduction in thigh muscle mass and grip strength over 5 years than those with lower inflammation. 8 In another analysis, members of the Health ABC cohort who maintained cognitive function over an 8-year period were of lower BMI than those who had minor or major declines in cognitive function. 9 Higher body fat could be linked to cognitive impairment through a greater inflammatory state. 10
Telomere length, an indicator of cellular aging, was negatively associated with percentage body fat in the Health ABC cohort 11 connecting body composition to longevity. Other studies have similarly observed an association between body fat and lower telomere length in young women. 12 Impaired longevity for obese women was supported by the finding that high BMI (>30 kg/m2) in midlife for women was associated with 79% lower odds of healthy survival to 70 years when compared with survival for women with the recommended BMI. 13
As well as total body fat, estimates of body fat distribution (visceral and subcutaneous) were obtained in the Health ABC study using CT scan. The importance of fat distribution on inflammation was demonstrated by substantially higher adipokines and inflammatory markers in individuals with elevated visceral compared with subcutaneous fat. 14 The higher inflammatory status could connect higher visceral fat to the observations of increased risk of comorbidities with this body fat distribution pattern.
In addition to being coupled with poor physical function, low muscle mass, especially when associated with high body fat, is associated with elevated disease risk. Analysis of more than 3000 individuals in the Cardiovascular Health Study 15 demonstrated a 23% increase in cardiovascular disease risk in sarcopenic obese individuals, whereas risk was not elevated in those who were obese or sarcopenic alone. Similarly, sarcopenic obesity alone, more than obesity or sarcopenia, was strongly associated with insulin resistance and dysglycemia in the NHANES III cohort. 16
Thus, data from multiple studies provide strong evidence for a negative effect of high body fat and low lean mass on health and longevity. Distribution of body fat in the visceral region increases risk compared with a similar amount of fat stored subcutaneously.
Dietary Interventions
Modification of diet can clearly influence body composition, but details regarding the quantity and quality of the optimal diet have been controversial.
Energy
Most professional societies and government bodies recommend a traditional goal of reducing energy intake by 500 to 1000 kcal/d (Table 1). This rate of weight loss is selected to minimize a reduction in lean mass and maximize the ability to comply over the long term. A more rapid weight loss is possible with drastic energy restriction such as very-low-calorie diets (VLCDs) of 6 to 10 kcal/kg/d. These diets are often in liquid form, using a high-quality protein source with added vitamins and minerals, because it is difficult to obtain the RDA for micronutrients on such a low-energy diet. The bulk of the evidence shows that VLCDs cause more weight loss initially but are not more effective in the long term. For example, a meta-analysis of 6 studies that compared VLCDs with moderately restricted energy diets (Table 2) revealed that the average weight loss after the intervention period (average of 12.7 weeks) was greater for those on VLCDs (16%) when compared with those on low-energy diets (~10%). 17 However, the difference in weight loss disappeared 1 year after the intervention; each strategy resulted in an average weight 5% to 6% lower than the initial. This lack of difference is likely a result of the difficulty in long-term compliance with VLCDs.
Comparisons of Selected Diet and PA Recommendations for WL and Body Composition Improvement.
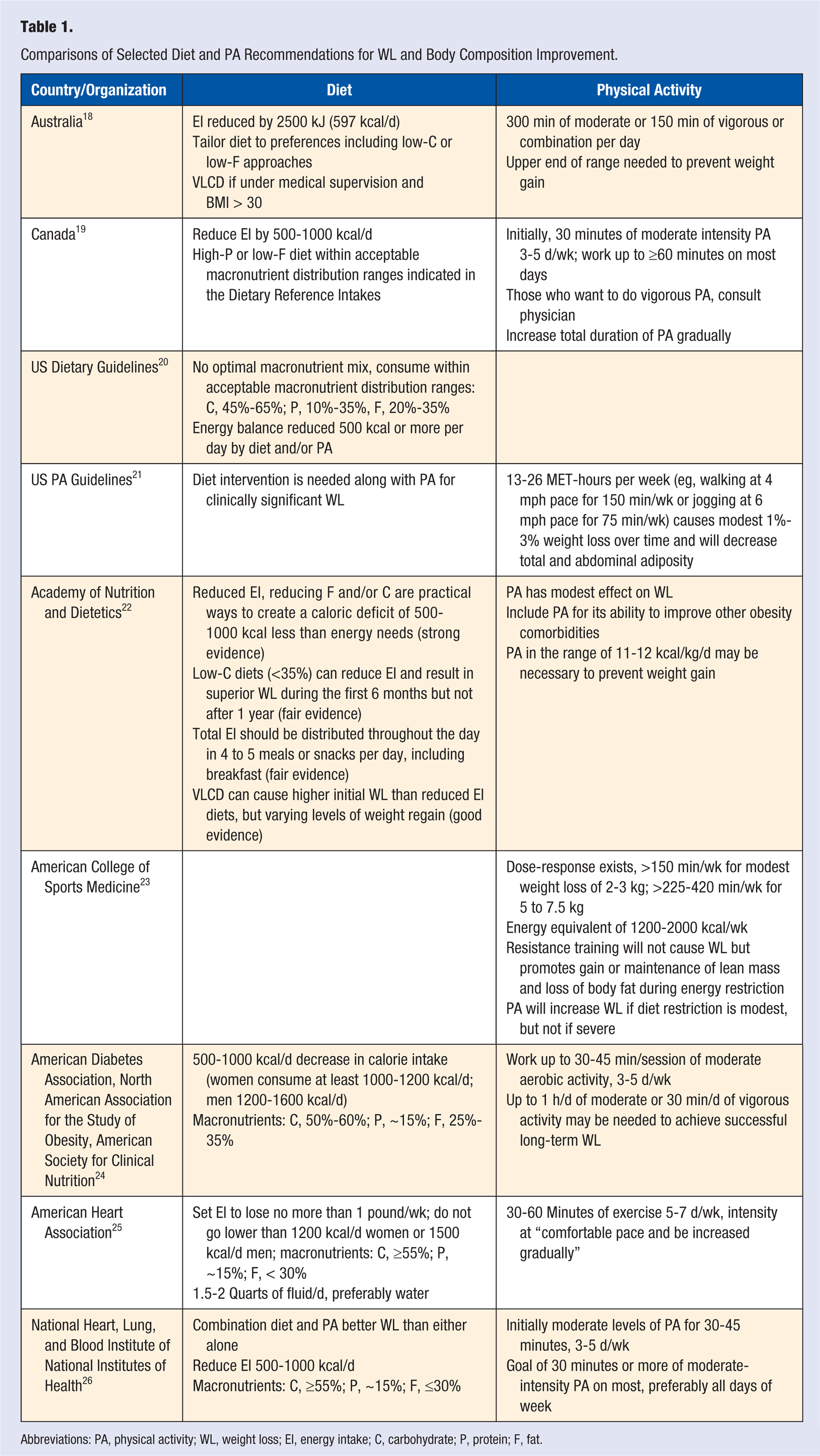
Abbreviations: PA, physical activity; WL, weight loss; EI, energy intake; C, carbohydrate; P, protein; F, fat.
Summary of Dietary Composition Intervention Studies (in Order of Appearance).
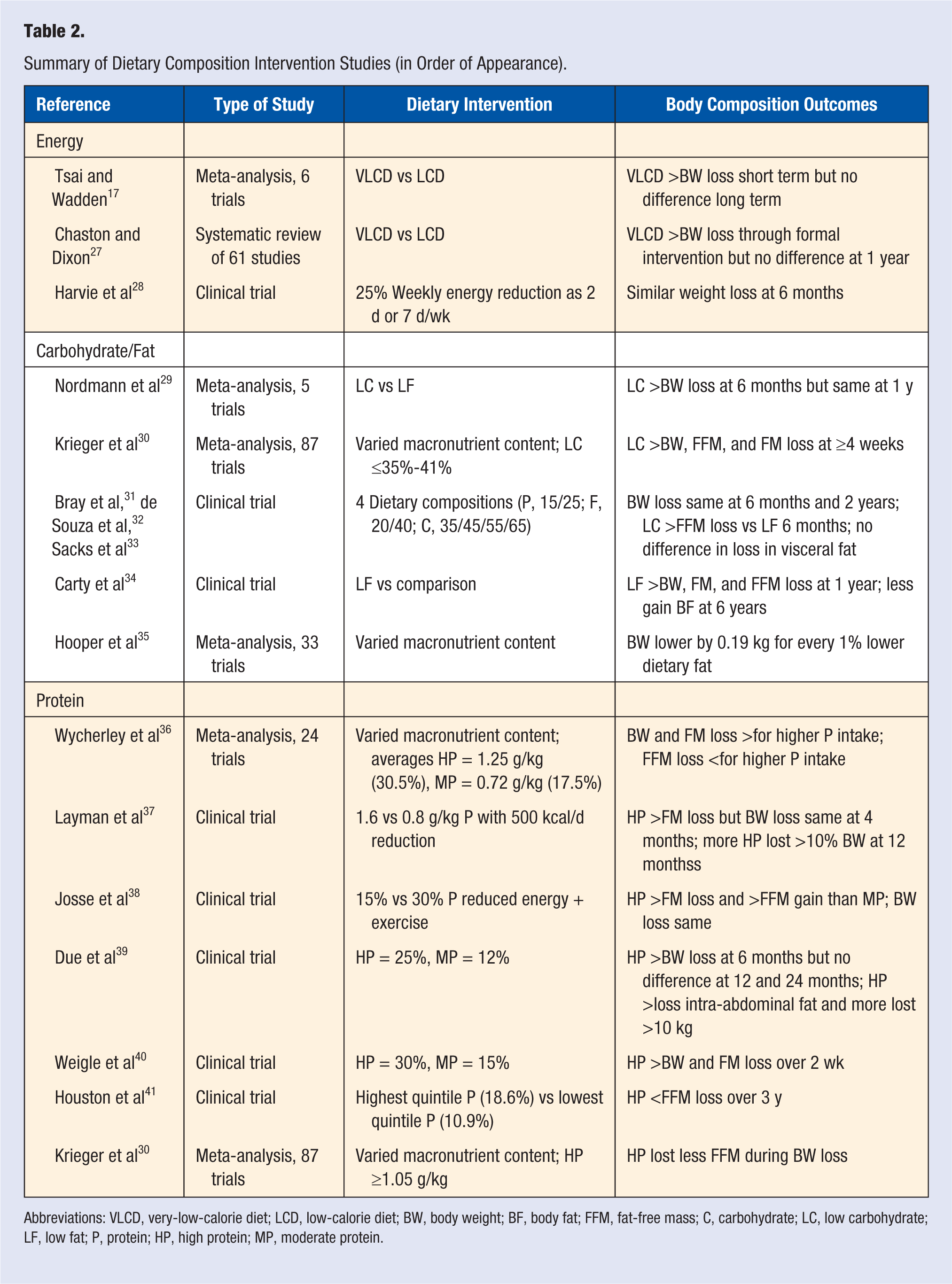
Abbreviations: VLCD, very-low-calorie diet; LCD, low-calorie diet; BW, body weight; BF, body fat; FFM, fat-free mass; C, carbohydrate; LC, low carbohydrate; LF, low fat; P, protein; HP, high protein; MP, moderate protein.
Chaston and Dixon 27 examined whether the magnitude of energy restriction influenced reduction in visceral fat. They did a systematic review of 61 weight loss studies that reported change in total as well as specific depot fat in participants asked to reduce energy intake from 1 to 56 weeks using a low-calorie diet or a VLCD. Initial body fat loss for all interventions was greatest from the visceral depot, with a shift to subcutaneous stores as energy restriction and weight loss progressed. The VLCD interventions caused greater initial loss of visceral fat than moderate-energy restriction up to 4 weeks but became similar to modest energy restriction as the weight loss continued. In other words, loss of visceral fat was secondary to the magnitude of weight loss rather than a particular diet.
Thus, VLCD can cause rapid reduction in visceral fat that coincides with quick improvement of comorbidities over a few weeks. However, this superior benefit is not maintained over time, such that a moderate-energy-restriction diet (500-1000 kcal/d reduction in energy) is recommended for longer-term compliance and adoption of healthful behavior. Similarly, the National Heart, Lung, and Blood Institute of the National Institutes of Health recommends a total energy intake of 1000-1200 kcal/d for most women and 1200-1600 kcal/d for most men. 26 This goal can be personalized based on hunger and weight loss response. Visceral fat will be preferentially reduced in the initial stage of any weight loss regime and contribute less to fat loss as the intervention continues.
A recent clinical trial comparing weight loss (body composition not reported) achieved by reducing the weekly energy intake by 25% with a consistent daily energy-restricted diet or by more dramatic restriction on just 2 days of the week demonstrated that both approaches were similarly effective in reducing body weight by 5 to 6 kg over 6 months and improving a variety of health indicators (eg, serum C-reactive protein and triglycerides). 28 It is interesting to note that fasting insulin and insulin resistance were more reduced for the obese individuals who followed the highly reduced diet for 2 days per week. Thus, novel methods to achieve a modest reduction in weekly energy intake may be useful to reduce body weight.
Carbohydrate and Fat
The quality as well as the quantity of energy may influence weight loss and body composition. Low-carbohydrate (LC) diets have been a consistently popular approach. 42 Although the definition varies from one study to the next, many LC diets prescribe a goal of 10 to 20 g of carbohydrate or <10% of energy per day. However, few individuals can maintain this level of carbohydrate restriction over a prolonged period and may chronically consume diets with ~30% to 40% carbohydrates. These diets have been compared with low-fat (LF) diets, with 20% to 30% of energy from fat and 55% to 60% from carbohydrates. Proponents of the LC approach point out the high satiety value of these diets and the ease with which some people can follow the ad libitum approach. Clinical concerns include the potential for a very high saturated fat intake as well as low fiber and micronutrient intake because of the low intake of grains and fruits. 42 Several studies report a higher serum C-reactive protein, suggesting higher inflammation, with LC diets.43,44 Those who favor a LF approach emphasize the lower energy density of the diet as well as higher fiber and micronutrients.
A meta-analysis that included 5 trials of 447 individuals demonstrated that after 6 months, those following a LC diet lost more weight, had more reduction in blood triglycerides, and better maintenance of high-density lipoprotein compared with LF interventions. 29 However, these differences in weight loss were gone after 1 year. The LF strategy caused more initial reduction in total and low-density lipoprotein cholesterol, which are risk factors for cardiovascular disease.
Although that analysis by Nordmann et al 29 did not consider body composition, Krieger et al 30 examined the effect of dietary composition on body fat and fat-free mass (FFM) change during energy restriction in 87 studies. They concluded that lower carbohydrate diets, defined as ≤35% to 41% carbohydrate, continuing for 12 weeks or more resulted in more loss of FFM (1.74 kg) and fat mass (5.57 kg) compared with those with higher carbohydrate. They did not examine these relationships at time points beyond 12 weeks. This negative effect of a high-fat, LC diet on body composition was confirmed in a subset of 99 individuals from the POUNDS LOST study that compared the effectiveness of 4 dietary compositions. 31 The individuals on the LC diet lost more FFM (1.5 kg) at 6 months compared with those on a LF diet who experienced little change. This was in spite of a similar body weight loss for both dietary groups at this time point. These studies suggest that there can be a negative effect of LC, high-fat diets on lean mass during weight loss.
The Women’s Health Initiative Dietary Modification Trial was an opportunity to examine the effect of a LF diet on body composition without an attempt to alter energy intake. Although close to 49 000 women were included in this trial, only a subset of 4311 women had DXA scans done at baseline and during follow-up at 1, 3, and 6 years. 34 The diet intervention group of this subset (n = 1580) was encouraged to achieve a diet with ≤20% fat along with high fruit, vegetable, and whole grain foods. They participated in 18 intensive behavioral modification sessions to enhance compliance during the first year of the study with quarterly maintenance meetings thereafter. Body fat mass declined modestly, but significantly more (~1.7 kg) for the LF group with little change for the comparison group during the first year of the study. By year 3, the group encouraged to eat less fat had a reduced percentage body fat, whereas the comparison group increased their percentage body fat over baseline. At the last time point (6 years), both groups had an increase in proportional fat mass compared with baseline, but the gain was less for those in the group prescribed a lower-fat diet.
Although the reported dietary intake data for this subset from the Women’s Health Initiative was not presented in that article, dietary data of a larger set of women in this trial 45 showed that those in the LF intervention group reported significantly less total energy intake at 1 year (93 kcal difference from baseline) and at 6 years (114 kcal difference). Although these women did not achieve the goal of 20% of dietary energy as fat, they reduced their dietary fat intake to an average of 24.3% after the first year compared with 36.1% for the comparison group.
A recent systematic review of 33 randomized clinical trials and 10 cohort studies that manipulated dietary fat without attempting to change body weight and lasted at least 6 months showed that body weight, BMI, and waist circumference were lower in those who consumed lower fat. 35 Specifically, they found that body weight was 0.19 kg lower for every 1% reduction in dietary fat. A dose-response was demonstrated, in that magnitude of body weight loss was predicted by a greater reduction in dietary fat.
As a whole, these analyses suggest that a recommendation to follow a LF diet without an intentional change in energy intake can result in small reductions in energy intake and a leaner body composition. More substantial change in body composition would require concurrent focus on deliberate, further reduction in energy intake.
Some research does not support an influence of dietary macronutrient composition on body weight or fat loss. For example, Sacks et al 33 compared weight loss over 2 years in 811 overweight adults asked to consume 4 diets that varied in macronutrient proportion in the POUNDS LOST trial. They were offered a combination of group and individual sessions over the 2 years to help them attain the goals of dietary fat that varied between 20% and 40%, carbohydrate between 35% and 65%, and protein 15% or 25% in the 4 dietary approaches. Weight loss at 6 months was 6 kg regardless of group assignment, with subsequent weight gain for all groups, such that participants weighed 4 kg less than baseline at 2 years. Body composition change, as assessed by DXA scan, in a subset of those women (n = 165) 32 mirrored this result, in that the average fat loss (4.2 kg) and lean loss (2.1 kg) was not different when dietary macronutrient prescription varied. Furthermore, reductions in fat in the abdominal, subcutaneous, and hepatic depots were consistent among diets. There was no difference in reported hunger, satisfaction, or attendance to group instructional sessions among the diet groups. Notably, for all groups, superior attendance predicted greater weight loss success.
A LC diet can reduce muscle and liver glycogen stores and, thus, affect endurance exercise capacity. 46 Evaluation of the physical activity of a subset of individuals in the POUNDS LOST study 31 showed that women on a 20% fat diet had significantly higher spontaneous physical activity than those consuming 40% fat. This suggests that the lower dietary carbohydrate (35%-45%) in the higher-fat diet was a deterrent for voluntary physical activity. Thus, LC diets may interfere with the ability to increase physical activity as part of a weight loss strategy.
Protein
Most studies support the benefit of a higher-than-average protein intake during weight loss to minimize loss of lean tissue and to enhance compliance with a reduced energy diet. A recent meta-analysis of studies that compared a “standard” (0.72 g/kg/d mean with range from 0.5 to 1.24 g/kg, 17.5% of energy) with a “higher” protein intake (1.25 g/kg/d mean and range from 1.05 to 1.83 g/kg, 30.5% of energy) reported that the higher-protein diets outperformed the standard protein diets for body weight loss and improved composition. 36 Specifically, in both short- and longer-term studies, more total body and fat mass and less lean mass was lost when the protein intake was higher. To provide perspective, data from the NHANES 2003-2004 cohort shows that the average adult protein intake in the United States is about 1.0 to 1.3 g/kg and 16% of energy. 47 The highest “acceptable” intake of protein is considered 35% of energy, 20 so these higher protein intakes are within the acceptable range.
Several longer-term studies support the value of higher-protein diets on weight loss. For example, Layman et al 37 demonstrated that a protein intake of twice the RDA (1.6 g/kg) concurrent with reduction in energy (~500 kcal/d) caused 22% more loss of fat mass compared with a diet with the RDA and ~15% of energy as protein over 4 months, even though body weight loss was similar. Additionally, the higher protein intake was associated with superior improvement of blood lipid profiles. Josse et al 38 compared reduced energy diets containing 30% to 15% protein concurrent with an exercise program that included both aerobic and resistance training over 16 weeks. The higher-protein intervention group demonstrated similar total weight loss (~4.3 kg) but more fat loss, with gain of lean tissue (0.7 kg), than the normal protein intake group.
The superiority of higher protein, reduced- energy diet interventions has been shown in studies for up to 12 months. Due et al 39 and Layman et al 37 observed the same average body weight loss in groups that consumed average or higher dietary protein (about twice average) at 12 months, but they found that drop-outs were higher in the lower-protein group. Body composition changes were more desirable for higher-protein groups in both studies, with greater loss of either visceral fat 39 or total body fat 37 than the lower-protein intervention. Both studies also consistently showed that a greater percentage of individuals lost at least 10% of their body weight in the higher-than-average dietary protein groups.
Most short-term studies demonstrate that higher-protein meals increase the magnitude and longevity of satiety. For example, Leidy et al 48 observed about half the daily fullness rating in overweight individuals fed an energy-restricted diet (reduction of 750 kcal/d) with 14% of energy as protein as compared with one with 25% protein, for 12 weeks.
The study by Weigle et al 40 also provides evidence that this benefit of higher dietary protein on long-term weight loss is at least partly related to satiety. They observed a dramatic reduction in “hunger” and increase in “fullness” rating when individuals were changed from a diet of 15% protein to 30% protein fed at isoenergetic levels. When the dietary intake became ad libitum, the daily energy consumed on the 30% protein diet fell immediately by about 20% and was maintained at this lower level through 84 days. Body weight and fat decreased by 4.9 and 3.7 kg, respectively, in spite of a lack of directive to reduce calories or weight.
A favoring of body fat loss by higher-protein diets may be not only secondary to the higher satiety but also the higher energy expenditure associated with digesting and assimilating protein, with fewer of the calories available for storage. One study assessing energy expenditure with a room calorimeter measured a 3% higher 24-hour energy expenditure in individuals consuming a 30% protein diet compared with a 10% protein diet. 49 This metabolic rate boost is modest on a daily basis but could have an impact over the long term. For example, an individual expending 2500 kcal/d would utilize an extra 27 375 kcal over a year if 24-hour energy expenditure increased by 3%.
Dietary protein content logically could affect maintenance of lean tissue during weight loss. Houston et al 41 studied the influence of dietary protein on change in lean body mass in older individuals from the Health ABC study. Those consuming the highest dietary protein (average of 18.6% of energy and 91 g/d) lost 40% less lean mass over 3 years than those in the lowest quintile of protein intake (average of 10.9% of energy and 56.9 g/d). Krieger et al 30 analyzed the effect of dietary protein on lean mass by comparing 87 studies that included protein intake >1.05 g/kg with those with less than this amount. The individuals who followed a higher-protein diet retained more FFM (1.21 kg) during interventions greater than 12 weeks.
In summary, most of the results in the literature appear to provide compelling and consistent evidence that a higher-protein diet (20%-30% of energy or 1.2-1.6 g/kg/d) will generally increase satiety, reduce energy intake, enhance body fat loss, and better maintain lean tissue during energy restriction than protein consumption less than ~1 g/kg/d or 10% to 15% of energy.
Eating Frequency
A popular assumption is that eating many small meals is superior to a few large ones for weight loss. McCrory et al 50 suggest that underreporting artifacts as well as the brief duration of many of the studies examining this claim limit conclusions. They posit that most well-designed experimental interventions do not support an association of meal frequency with weight loss. A randomized controlled trial evaluated the difference in outcomes when 51 overweight men and women were asked to eat the same diet as 3 meals per day or “grazing” with 100 kcal every 2 to 3 hours. Both groups were equally successful in lowering weight over the 6-month trial. 51
Leidy et al 48 studied the interaction of dietary protein (14% vs 25%) with meal frequency (3 vs 6 meals/d) in 27 overweight men on the effect on satiety, blood glucose, and hormones related to appetite. Higher protein, regardless of frequency of ingestion increased fullness. This research group subsequently reviewed the literature and concluded that consumption of 3 meals per day, each with substantial protein, is the best strategy for weight control. 52 Although they acknowledge that the evidence is limited, reducing the meal frequency below 3 can be detrimental for hunger and food intake; increasing to more than 3 is unlikely to have further benefit on energy intake.
Snacks have alternatively been recommended or vilified for attempts to lose body fat. Research shows that individuals rarely compensate for the calories in a snack at the subsequent eating occasion, resulting in a higher total energy intake. 53 Compatible with the recommendations above for protein, snacks with higher protein were more likely to increase fullness and delay the upcoming meal than low protein snacks. 54
Summary of Dietary Interventions
The consensus from the scientific literature supports a modest daily reduction in energy intake in the range of 500 to 1000 kcal/d for body fat loss. More drastic energy restriction may be attempted, but focus on adequate micronutrient intake and fiber as well as medical supervision is suggested for a VLCD approach. In general, initial loss of body fat during any weight loss comes primarily from the visceral depots, with a switch to more reduction from the subcutaneous depots over time. Loss of lean tissue will be greater as the magnitude and rate of weight loss rises. A dietary fat goal of 20% to 25% of energy along with dietary protein up to 1.6 g/kg and 30% of energy is likely to result in more successful weight and fat loss compared with higher-fat and lower-protein diets. A drastic reduction in dietary carbohydrate is likely to cause more rapid initial fat and lean tissue loss, and these diets are generally not more effective in the long term. These LC diets also may be less compatible with an increase in physical activity than higher-carbohydrate diets. A pattern of 3 regularly spaced meals, each containing protein, should be recommended, with snacks as needed, with most snacks containing protein as well.
Physical Activity
Optimal Exercise Dose
Interventions focused only on increasing physical activity of overweight individuals typically result in minimal (0%-3%) weight loss (Table 1.). The US Physical Activity Guidelines committee summarized the evidence from randomized clinical trials demonstrating that moderate- to vigorous-intensity physical activity from 180 to 360 minutes per week over 8 to 16 months was required to cause weight loss in the range of 1 to 3 kg and up to 3% of body weight. 21 More recently, Thorogood et al 55 did a systematic review and meta-analysis of randomized controlled trials comparing exercise-only intervention groups with inactive control groups (Table 3). Both 6-month and year-long programs resulted in weight loss differences in the 1.6- to 1.7-kg range when compared with sedentary controls. Thus, the magnitude of weight loss resulting from an increase in aerobic physical activity as the sole strategy would not be considered by most clinicians to be clinically meaningful.
Summary of Exercise Intervention Studies (in Order of Appearance).
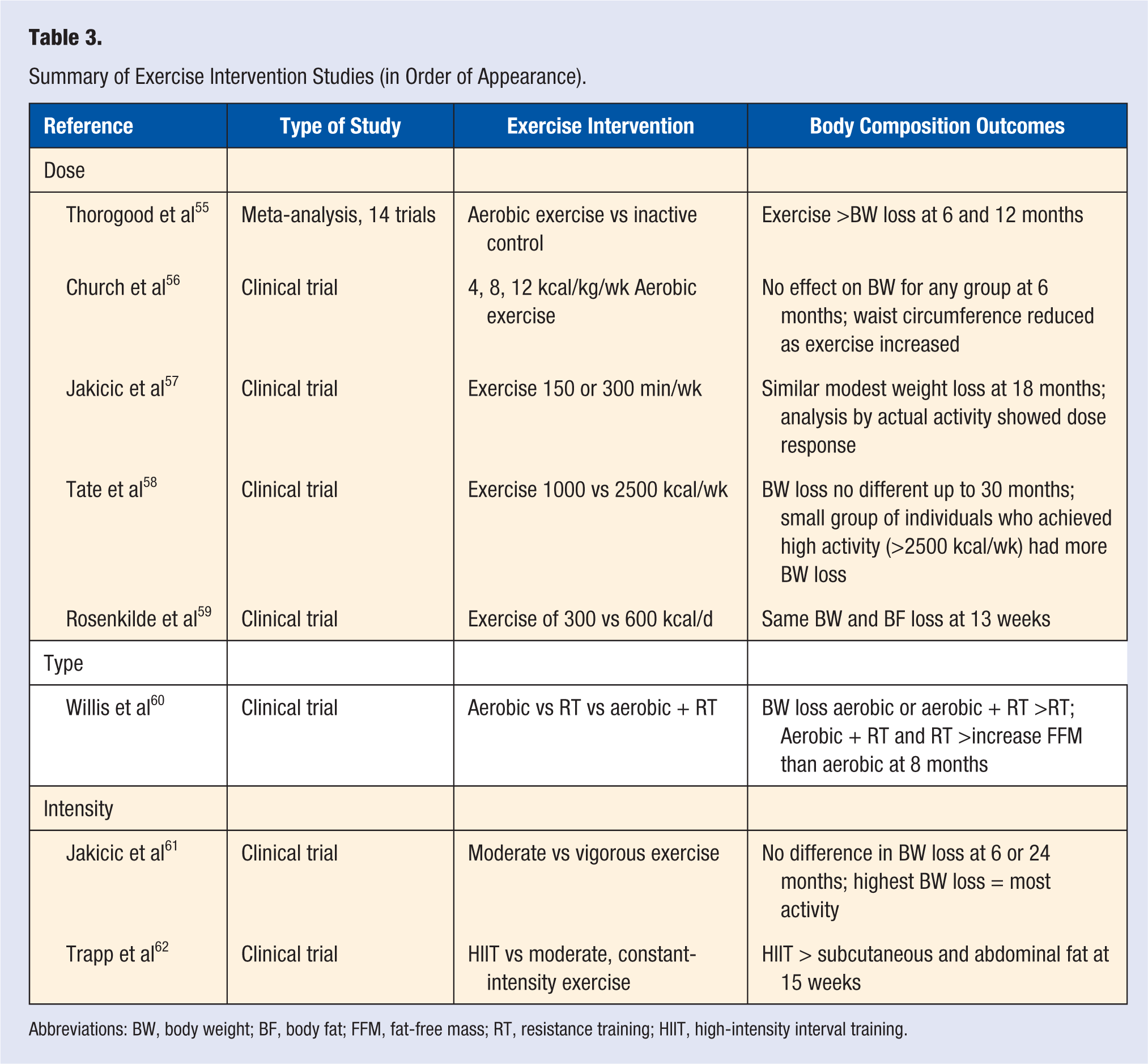
Abbreviations: BW, body weight; BF, body fat; FFM, fat-free mass; RT, resistance training; HIIT, high-intensity interval training.
As an example, Church et al 56 did not observe an effect of physical activity prescription dose (4, 8, or 12 kcal/kg/wk) on body weight in overweight postmenopausal women over 6 months. However, it should be noted that even the highest dose of physical activity in that study, an average of 192 min/wk, is at the lower end of the range discussed as effective for weight loss in the US Physical Activity Guidelines. Body composition was not assessed, but waist circumference was modestly reduced by the rise in physical activity, suggesting a reduction in body fat.
Although it is logical to suspect that there would be a dose-response effect of physical activity on body weight loss, this has not been consistently observed. Several studies that were designed to evaluate substantially different physical activity energy expenditure prescriptions did not find a significant effect of amount of energy expended on weight loss. For example, Jakicic et al 57 observed a similar modest (<2%) body weight loss over 18 months when 278 overweight adults were asked to exercise 150 or 300 minutes per week at moderate to vigorous intensity. Although this suggests that there is no dose-response effect, a different conclusion emerged when the authors retrospectively grouped individuals by reported rather than prescribed exercise. There was progressively greater weight loss when participation was categorized by exercise duration less than 150 min/wk, 150 to <225 min/wk, 225 to 300 min/wk, and more than 300 min/wk at 6 and 12 months of the intervention. This is supported by the fact that those who were most successful in weight loss reported the most activity. These results also demonstrate the hazard in assuming that people perform exercise as prescribed.
Similarly, Tate et al 58 asked more than 200 overweight adults to exercise enough to expend either 1000 or 2500 kcal/wk. The weight lost at 30 months after the start of the treatment was not significantly different—about 1% for the 1000 kcal/wk and 3% for the 2500 kcal/wk. This is likely a result of the fact that the amount of actual activity done by the 2 exercise groups converged and was no longer different at the final measurement period (1390 and 1696 kcal/wk). The small group of individuals who achieved high activity levels (>2500 kcal/wk through 30 months) were successful in losing more weight and maintaining more of this weight loss than those with less activity.
Together these studies support the value of a very high dose of exercise on body weight loss, but few people achieve this high level of activity. The ACSM position stand summarizes that doing physical activity at high volume (eg, up to 420 min/wk) results in more substantial weight loss in the range of 5 to 7 kg 23 and more than 3% of initial body weight. This amount of weight loss is more in line with what is expected to cause clinical improvements. However, the proportion of study participants able to achieve this level of physical activity on a regular basis has been low, and thus, this recommendation does not appear to be feasible for most people without substantial support.
Either dietary compensation or lack of compliance with high volume or intensity of activity could explain the lack of association between added physical activity and body weight and fat loss. Support for energy intake compensation with high-volume physical activity is provided by a study in moderately overweight men, which compared exercise of 300 or 600 kcal/d for 13 weeks. 59 In spite of expending twice as many calories in physical activity, the higher-volume group lost similar amounts of body weight (−3.6 kg and −2.7 kg for moderate and high volume, respectively) and fat (−4.0 kg and −3.8 kg for moderate and high volume, respectively) as the moderate-exercise group. The assumption was that there was more energy intake compensation in the high-volume group. Although not verified by the collected food records, the low precision and underreporting with this method may have made this difficult to verify. This study suggests that a recommendation for 300 kcal/d of physical activity each day could cause modest weight loss. This could translate to as little as ~30 minutes or as much as ~60 minutes of physical activity, depending on the intensity.
This study and others suggest that there may be a threshold of physical activity most likely to moderately reduce body fat without stimulating food intake compensation. However, the state of our knowledge is not precise enough to define this threshold for each population. It may be a different threshold, depending on gender, magnitude of overweight, type of physical activity, fitness, and other factors.
Type of Physical Activity
Some evidence that type of physical activity influences the effect on energy intake is provided by the STRRIDE-AT/RT study comparing aerobic exercise, resistance exercise, and the combination of both in overweight or obese men and women. 63 After 8 months of training, body mass was modestly reduced only for the 2 groups that included aerobic training (−1.3 kg for aerobic and −1.5 kg for combination) but unchanged for those doing only resistance training. 60 These changes were reflected in spontaneous similar reductions in energy intake (~200 kcal/d) for both groups who did aerobic exercise but not the resistance training group. 63 Thus, this study does not support the common notion that being physically active is ineffective for weight loss because it will overstimulate appetite. It also suggests that aerobic exercise is more valuable than resistance training in causing body weight change.
Data from this study also supported the value of aerobic activity on the distribution of body fat. 64 The participants in the aerobic exercise program lost visceral and subcutaneous body fat as well as liver fat, whereas there was no change in body weight for the resistance trainers and only a reduction in the subcutaneous fat depot.
Although unlikely to cause weight and fat loss, resistance training clearly has beneficial effects on lean mass. Resistance exercise is a powerful stimulus for muscle protein synthesis and hypertrophy, even during energy restriction, but most studies do not detect a greater total weight loss when resistance training is added to energy restriction. Individuals typically achieve a leaner body composition because of gains in lean tissue mass and sometimes modest reductions in body fat. 23 For example, individuals from the STRRIDE-AT/RT study who did resistance training 3 days per week or resistance training plus aerobic exercise increased lean body mass by about 1 kg, whereas there was no change for the individuals who only did aerobic exercise. 60
The optimal resistance exercise prescription for a leaner body composition in obese individuals is not known, but most studies begin with the general recommendations of the US Physical Activity Guidelines, 21 2 to 3 days per week using one set of 8 to 12 repetitions per lift for all major muscle groups. Intensity and volume should increase as individuals get stronger because the critical stimulus for strength and muscle mass gain appears to be substantial fatigue. Appropriate training to maintain safety (eg, avoiding breath holding) is critical for those naïve to resistance training.
Intensity of Physical Activity
Limited research has been done to define the optimal exercise intensity for weight loss. The US Physical Activity Guidelines offers equal choice of 150 min/wk if moderate (eg, walking 4 mph) or a shorter, 75 min/wk if more-intense activity is performed (eg, jogging at 6 mph). As discussed earlier, the study by Jakicic et al 61 did not identify a benefit of a higher-exercise intensity prescription for overweight women in a weight loss program. Most recommendations leave the decision of intensity up to the individual, with moderate intensity most often used for the exercise prescription. Vigorous is defined by the Physical Activity Guidelines as a rating of 7 to 8 on a 10-point scale, with jogging, biking uphill, and singles tennis provided as examples. Jakicic et al 61 defined vigorous exercise as 70% to 85% of maximal heart rate, with a rate of perceived exertion of 13 to 15 on a 20-point scale.
In general, high-intensity sprint exercise has not been recommended for body weight or fat loss because the primary fuel for muscles during high-intensity effort is carbohydrate rather than fat. This tradition has been challenged by some provocative studies performed using interval training, repeated interspersed high-intensity with lower-intensity bouts.65,66 These studies provide evidence that interval training, sometimes termed high-intensity interval training (HIIT), may be at least as effective at reducing subcutaneous and abdominal body fat as traditional moderate, constant-intensity exercise. 65 Trapp et al 62 had young overweight women do a HIIT protocol consisting of repeated 8-s cycle sprints followed by 12 s of low-intensity cycling for a period of 20 minutes, 3 times per week and compared this with steady-state cycling at 60% VO2max for 40 minutes. More subcutaneous and abdominal fat was lost by the HIIT group after 15 weeks when compared with the continuous-intensity exercisers. The suggested mechanism for a superior effect of HIIT on body fat loss is the stimulation of catecholamine release during high-intensity activity and subsequent higher fat utilization during recovery. It is also possible that a higher lactate or corticotrophin-releasing factor accumulation could reduce appetite and food intake after HIIT. 65 These studies are provocative but require additional research to verify the value of HIIT for overweight individuals.
Although safety of and compliance with interval training might be questioned, Gremeaux et al 67 demonstrated that 62 overweight and obese people with an average age of 53 years could comply with an exercise plan that included walking, resistance training, and cycle interval training (2 sets that included 15-30 s at 80% of maximal aerobic power followed by the same time as passive recovery repeated for 10 minutes). Overall compliance with the supervised exercise program over 9 months was an amazing 97%. All participants were also counseled to follow a modestly reduced energy diet. The average body mass fell 5.3 kg along with body fat and waist circumference. Although this study did not compare this lifestyle intervention with any other, it demonstrates that overweight individuals can tolerate and respond well to an intervention that includes interval training. It is important to note that those with a history of cardiovascular disease were excluded from this study. Clinical judgment and compliance with recommended risk stratification 68 should be used when deciding on use of high-intensity exercise as a treatment for obesity.
Health Benefits of Exercise Regardless of Weight Change
Although it is beyond the scope of this review to thoroughly examine the many health benefits of physical activity, 69 it is acknowledged that physical activity independent of weight change is highly beneficial. Lack of weight or fat change should not discourage individuals from adopting and maintaining an active lifestyle. Research from the Cooper Clinic database 70 clearly and consistently shows that fitness and physical activity reduce morbidity and mortality, regardless of other risk factors, such as body fat, diet, and blood pressure.
Some studies demonstrate that physical activity interventions may cause redistribution of total body fat to a healthier pattern. Reduction in abdominal and visceral fat 71 has been noted with physical activity interventions, even when body weight did not change.
Healthful behaviors such as physical activity and wholesome diet may overcome at least part of the negative effects of high body fat. For example, Lee et al 70 followed more than 3000 healthy adults for 6 years and monitored who developed hypertension, metabolic syndrome, and hypercholesterolemia. Increased body fatness over the period increased the risk of all these conditions, whereas increasing fitness reduced the risk. In examining the interaction between fitness and fatness, they determined that improved fitness ameliorated but did not completely eliminate the negative effects of high body fat on the development of these cardiovascular risk factors. Another prospective analysis of more than 13 000 women in the Nurse’s Health study illustrated that higher physical activity during midlife predicted up to double the odds of successful survival at age 70. 72 Thus, all overweight individuals should be encouraged to improve their quality of diet and physical activity. Encouraging an improved body composition through a reduction in body fat and maintained or increased lean mass is also prudent to maximize health and longevity.
Summary Physical Activity
In summary, aerobic physical activity less than 150 min/wk is unlikely to result in substantial body weight or fat loss. Moderate-intensity aerobic physical activity greater than 250 min/wk or more will more likely result in modest weight and fat loss. HIIT may be an option if the individual is at low risk of cardiovascular disease. Resistance training is an excellent adjunct to the aerobic exercise prescription to aid in maintenance or increases in lean mass during weight loss but is not expected to appreciably reduce body weight or fat. However, physical activity should be encouraged even if body weight does not change. Reducing the more dangerous visceral fat depot through physical activity and modest weight loss can improve other health-related factors, such as insulin resistance, blood pressure, and blood lipids.
Physical Activity Combined With Dietary Restriction
All professional society and government guidelines for weight loss recommend that diet be modified along with physical activity to have optimal outcomes (Table 1). Most evidence supports that the combination of modest dietary energy restriction with aerobic and resistance exercise will cause more substantial weight loss and favorably influence body composition and health than one strategy alone. For example, Curioni and Lourenco 73 did a meta-analysis of 6 randomized clinical trials that compared diet with diet plus exercise interventions and included a follow-up at 1 year. They reported that the latter caused a 20% greater body weight loss than dietary strategies alone at the end of the intervention period. This differential was maintained when body weight was examined at the 1-year follow-up. In another meta-analysis, Wu et al 74 analyzed 10 studies that compared the body mass response of a diet with a diet plus exercise intervention. The pooled weight loss at the end of the diet plus exercise intervention (3.34 kg) was significantly greater than the 1.38 kg for diet-alone interventions. The differential weight loss continued through 1 year when the difference was 2.29 kg between the 2 types of interventions.
Only 6 of the studies in the meta-analysis 74 reported body composition. A secondary analysis of these studies demonstrated a similar advantage of the combination of diet with exercise on percentage body fat, with a 2% pooled differential. Janssen and Ross 75 assessed changes in body fat distribution using MRI in 60 obese men and women following 16 weeks of a reduced-energy diet or diet with exercise that caused a ~10% weight loss. They found that for all treatments, the visceral depot is preferentially reduced over the subcutaneous in both interventions but that the combination of diet with aerobic exercise targeted more reduction in abdominal subcutaneous adipose tissue than diet alone.
Because exercise stress on the muscle stimulates protein synthesis, one advantage of physical activity during weight loss is a better preservation of lean tissue. Villareal et al 76 followed more than 100 older adults (at least 65 years of age) over a year of either a diet (500-750 kcal/d below energy requirement), exercise (3 d/wk of combination of flexibility, balance, aerobic, and resistance exercises), diet with exercise, or no treatment program. The weight loss was similar: 9% to 10%, for the diet alone and the combination of strategies, but the addition of exercise to the dietary intervention better preserved lean tissue mass (3% loss) over the year compared with the dietary treatment (5% loss). Similarly, Janssen and Ross 75 observed that body fat loss was similar when comparing a diet-alone with a diet plus aerobic exercise intervention in 60 obese men and women. Skeletal muscle mass was maintained in those who exercised while dieting in spite of an average body weight loss of ~11 kg; lean mass was reduced when dietary restriction was the sole intervention.
Although it is beyond the scope of this review to thoroughly explore issues, it is acknowledged that inclusion of increased physical activity in a weight loss intervention has multiple additional health benefits over diet restriction alone. As an example, the study by Villareal et al 76 comparing diet alone with diet plus exercise confirmed that the combination treatment caused superior improvement in physical performance and functional status than the diet-only intervention. Together, these studies support the value of combining dietary and physical activity strategies to most effectively improve body composition, health, and function.
Development of the appropriate nutrition and physical activity prescription is, of course, only the beginning of the process of behavior change. An entire body of research has been devoted to approaches to enhance compliance with lifestyle recommendations. Readers are encouraged to explore reviews77,78 that cover research on techniques such as self-monitoring, goal setting, and social support as well as reviewing recommendations from professional organizations on specific strategies to improve dietary18,22 and physical activity 79 behavior. In 2011, the Centers for Medicare and Medicaid Services decided, 80 based on systematic review by the US Preventive Services, 81 that there was sufficient evidence that intensive behavioral therapy reduces adiposity in obesity and recommended a specific 5-part framework for applying this approach to eligible patients (Table 4).
Centers for Medicare and Medicaid Services 5: A framework for Intensive Behavioral Counseling. 80

Conclusions and Recommendations
Because high body fat and low lean mass are associated with higher morbidity and mortality, clinical teams are often asked about the safest and most-effective methods to lose weight and improve body composition. A combination of dietary modification with an increase in physical activity is recommended for greater weight loss, superior body composition change, and health benefits. Although many diets can be effective in causing weight loss, most of the research supports a modestly reduced energy intake (500-1000 kcal/d reduction) that is LF (20%-25%), at least 50% carbohydrate, and ~1.2 g/kg or 20% to 25% protein. Each meal and snack should contain protein to assist satiety. A LC diet can be effective in the short term but is unlikely to be more effective for weight loss in the long term and may reduce lean tissue and exercise capacity more than a higher-carbohydrate approach.
An increase in physical activity without a change in diet is likely to have only a modest effect on body weight but can improve body composition, function, and health. Performing moderate exercise on most days of the week for a total of 250 minutes per week for at least 9 months may cause a reduction of ~3% of weight, whereas increasing to ~60 minutes on most days can result in up to twice this body mass loss. However, it is difficult for most individuals to maintain that level of physical activity, so achievement of substantial negative energy balance typically also requires diet modification. Aerobic exercise is the most critical type of activity for reduction of body fat, whereas resistance training is valuable to reduce loss of lean tissue during weight loss. Adding energy intake restriction to physical activity is substantially more effective than exercise alone.
Although additional research can be performed to more carefully clarify the optimal exercise and diet prescription, the most pressing need appears to be for strategies to improve motivation for and compliance with any strategy. Researchers and clinicians note a dramatic individual difference in response to interventions. Additional research focused on understanding individual differences related to genetics, response to environment, or effective motivation could assist in the development of individualized approaches to weight loss.
