Abstract
Objectives
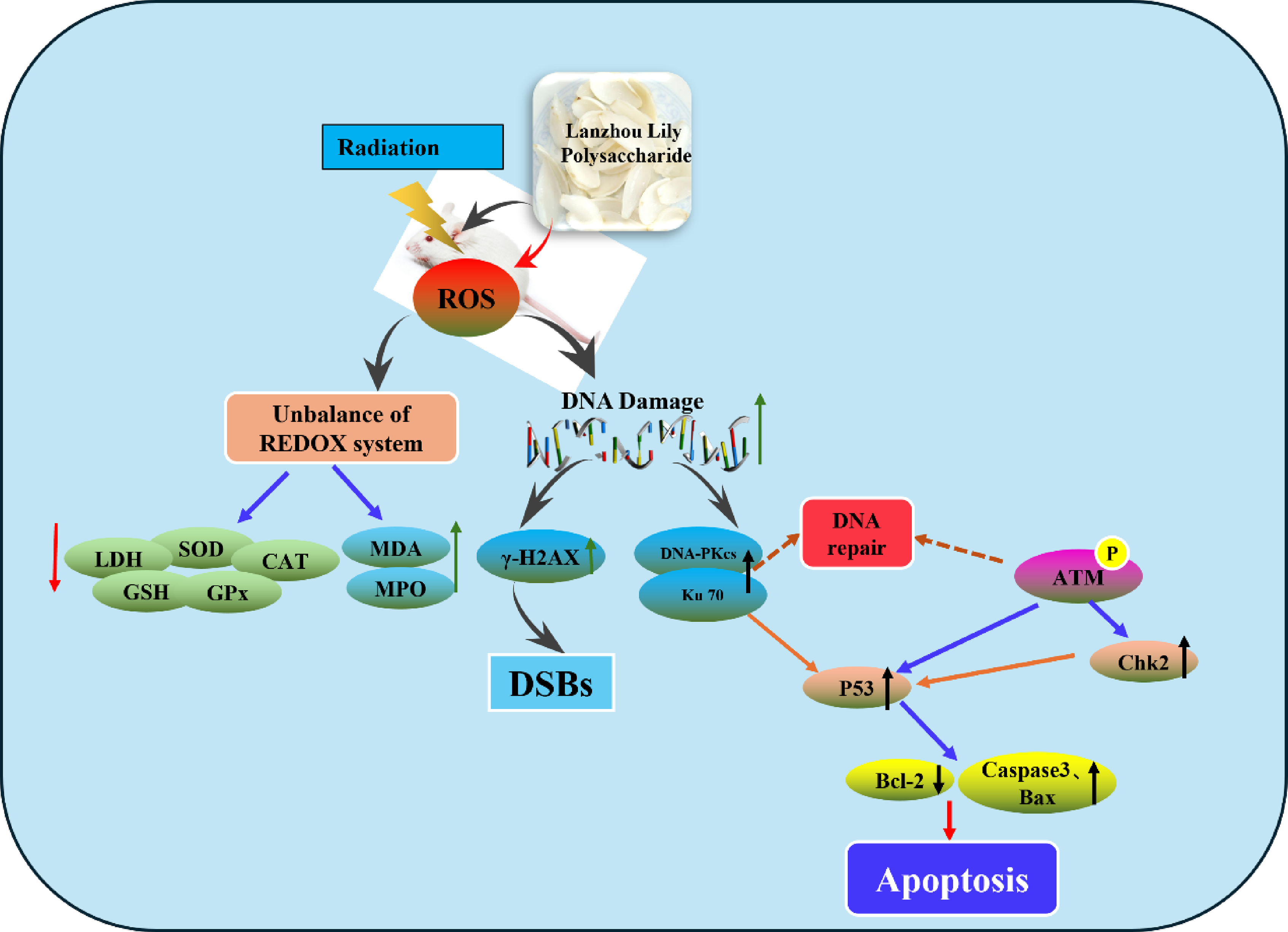
To investigate the protective effect and underlying mechanism of Lanzhou Lily Polysaccharide (LP) against radiation-induced splenic damage in mice.
Methods
Mice were pretreated with LP before X-ray irradiation. (1) Antioxidant enzyme activities (superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx)), glutathione (GSH) content, and levels of malondialdehyde (MDA), myeloperoxidase (MPO) in mice serum were measured using commercial kits; (2) The spleen index was calculated; (3) Hematoxylin–eosin (H&E) staining was performed to observe spleen morphology; (4) Splenocyte apoptosis and DNA damage were detected; (5) Immunohistochemistry and Western blotting were employed to detect DNA damage repair (eg, γ-H2AX) and apoptosis-related protein expression in splenic tissues.
Results
LP pretreatment alleviated irradiation-induced impairment of antioxidant capacity in mice, increased SOD, CAT, GPx activities and GSH content, decreased MDA and MPO levels. It reversed irradiation-induced reduction in spleen index. H&E staining showed LP mitigated radiation-caused splenic morphological damage. TUNEL assay demonstrated LP lowered the splenocyte apoptosis rate. Comet assay confirmed LP inhibited radiation-induced DNA damage in splenocytes. Immunohistochemistry and Western blotting results revealed LP down-regulated the expression of γ-H2AX and modulated DNA damage repair-related proteins, regulating apoptosis-related protein levels.
Conclusion
LP can protect mice from radiation-induced splenic damage.
Introduction
Ionizing radiation (IR) has been widely applied to clinical tumor radiation therapy, medical imaging diagnosis. Although radiation brings many benefits to patients, its side effects cannot be ignored. Advanced clinical radiotherapy techniques—such as intensity-modulated radiotherapy (IMRT), image-guided radiotherapy (IGRT), heavy ion therapy, and proton therapy—are widely applied primarily to minimize radiation-induced toxicity and damage to normal tissues. Despite these advances, the radiotoxicity of normal tissues remains a limiting factor in radiation therapy for many diseases. 1 IR can directly or indirectly damage normal tissues and organs by generating various ions, including damage to hematopoietic cells, nerves, lung tissues, etc., and may even cause cancer.2-4 This leads to reactions such as single-strand breaks (SSBs), alkaline oxidation, and depurination, which in turn affect cell function and the stability of genetic material, posing long-term potential health risks to patients. Therefore, during radiotherapy, how to effectively alleviate radiation damage to normal tissues has long been a research focus, driving researchers to continuously explore new protective strategies and treatment regimens.
Radioprotective agents play an important role in mitigating radiation damage, especially natural products. Numerous studies have demonstrated that various bioactive compounds derived from plants, such as polyphenols, saponins, flavonoids and polysaccharides, exhibit notable efficacy in shielding against radiation. Notably, ginseng polysaccharides, ganoderma lucidum polysaccharides, astragalus polysaccharides, and angelica polysaccharides have all been identified as possessing substantial radiation protection properties.5-10 Polysaccharides are ubiquitously present in animals, plants and microorganisms, constituting essential constituents of the fundamental components that comprise living organisms. Lanzhou lily (Lilium davidii var. unicolor) is a unique dual-purpose species in the Lilium genus, valued for both cuisine and medicine in China. It belongs to the Liliaceae family and is a cultivated variant of Lilium davidii, native to the high-altitude, cool regions around Lanzhou, Gansu Province. 11 The bulbs contain bioactive components like polysaccharides, saponins, and colchicine, with Lily Polysaccharide (LP) as the key component (over 20% of dry weight). Structurally, LP is a heteropolysaccharide of D-glucose, D-mannose, and galactose. 12 It offers various pharmacological effects: regulating immunity, antioxidant activity, hypoglycemic, anti-carcinogenic, anti-fatigue, and antibacterial properties.13,14
Ionizing radiation is well-documented to damage the hematopoietic system, thymus, and spleen. It also hinders tissue and cell repair after injury and induces immunosuppression—key issues in radiation-related harm. 15 Building on our preliminary research, which showed that Lily Polysaccharide (LP) from Lanzhou lily may protect human umbilical vein endothelial cells (HUVECs) by inhibiting the PI3K signaling pathway, this study further explored whether LP fragments could exert effective systemic radiation protection. Specifically, we examined their potential to mitigate radiation damage by enhancing immune function and reducing DNA injury. 16 The findings of this study provide a theoretical foundation for the development of Lanzhou lily-derived products and their potential application to address radiation-induced harm.
Materials and Methods
Chemicals
Preparation of LP. First, the polysaccharides were extracted from the bulbs of
Kits for superoxide dismutase (SOD), catalase (CAT), myeloperoxidase (MPO), malondialdehyde (MDA), glutathione (GSH), glutathione peroxidase (GPx), and lactate dehydrogenase (LDH) were purchased from Nanjing Jiancheng Co.Ltd. (Nanjing, China). Analytical grade chemicals were used in all cases.
Experimental Design
Northwest Minzu University approved the use of mice in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health. Laboratory conditions (12-h dark/light cycles, 22°C, 50% humidity) were acclimatized prior to experimentation. Following a 1-week adaptation period under specific pathogen-free (SPF) conditions, sixty healthy male Balb/c mice (8-12 weeks old and weighing 18-22 g) were randomly allocated into five groups: (1) control group, (2) X-ray irradiation (IR) group, (3) different doses of LP (50、100、200 mg/kg.d) + IR. The administration was continued for 7 days. After the last administration, the mice in each group exposed to 4 Gy whole-body irradiation. At a dose rate of approximately 1.38 Gy/min, radiation was administered using the Faxitron Cabinet X-ray System (Model RX-650, 100 kVp, 5 mA, USA). The following experiments were carried out after 24 h irradiation. This study was conducted from June 2022 to August 2023.
Biochemical Analysis
Commercially available kits were used to measure SOD, CAT activities, MDA content, and GSH, MPO, LDH, GPx levels of the mice peripheral blood serum in each group.
Spleen and Thymus Index
After anesthesia, the spleen and thymus of the mice were removed. The masses were measured by analytical balances, and the spleen index of each group of mice was calculated. Spleen weight and thymus weight were expressed as the spleen and thymus index, respectively.
Hematoxylin–Eosin (H&E) Staining
Spleen tissue was fixed in 4% buffered formalin. H&E staining was performed after embedding and fixing the tissues. NIS-Elements D2.30 software (Nikon, Melville, NY, USA) was used to capture the digital images.
Apoptosis was Detected by TUNEL Assay
Deparaffinized spleen sections were rehydrated in graded ethanol solutions, and then a dUTP nick-end labeling assay (TUNEL) was performed in accordance with manufacturer’s instructions (GenScript, Piscataway, NJ, USA). Hematoxylin was used as a counterstain.
DNA Damage Detection by Comet Assay
The spleen cells were prepared by homogenizing spleen homogenate obtained after the mice were sacrificed. Based on Zhao, et al., the cells were embedded in 0.75% low-gelling-temperature agarose and pipetted onto a precoated microscope slide. 17 With CASP software (https://www.casplab.com/), 150 individual comet images from each sample were analyzed for tail moment (TM), DNA content, and percentage DNA in tail. Besides head radius, tail length, and TM, the program can calculate Olive tail moment (OTM) and TM for comets oriented from left to right.
Immunohistochemical Detection
Immunohistochemistry was used to detect the expression of different proteins (γ-H2AX, Ku70, DNA-PKcs, Chk2, P53, p-ATM) after the spleen tissue sections were prepared. DiAmino-Benzidin (DAB Substrate System, Labvision) was used for chromogen staining in conjunction with primary antibodies and a UV detection system for horseradish peroxidase (HRP). Slides were counterstained with Mayers hematoxylin and mounted with Entellan for light microscopy.
Protein Levels by Western Blotting Analysis
After the mice were sacrificed, the spleen tissues were collected for homogenization. Total protein was harvested and quantified by a BCA Protein Assay Kit (Takara Bio, Kusatsu, Japan). Then, 20 mg protein was resolved on SDS-polyacrylamide gels and transferred onto polyvinylidene difluoride (PVDF) membranes. After blocking the membranes with 5% milk, primary antibodies (p-Ku70, DNA-PKcs, p-ATM, Chk2, γ-H2AX, P53, Caspase3, Bax, Bcl-2 and β-actin, all from Abcam) were incubated overnight at 4°C. A secondary antibody labeled with HRP from Abcam was then incubated with the membranes for 1 h at room temperature. Super Signal Substrate kits were used to detect chemiluminescence.
Statistical Analysis
Statistics were analyzed using SPSS version 18.0 software (SPSS Inc., Chicago, Illinois, USA). Data are expressed as mean ± standard deviation (SD). Comparisons among multiple groups were performed using one-way analysis of variance (ANOVA).
Results
LP Treatment Moderates Radiation-Induced Oxidative Stress
LP Pretreatment Enhanced Antioxidant Capacity and Reduced MDA, MPO Content Induced by IR
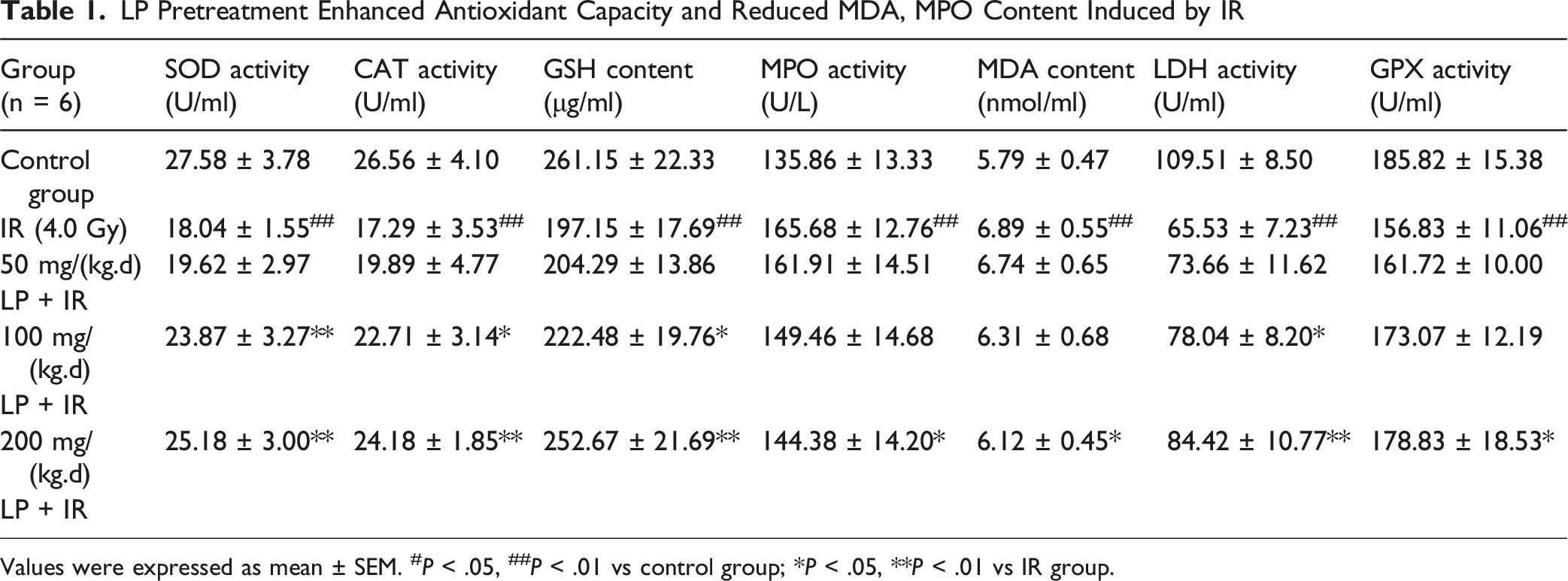
Values were expressed as mean ± SEM. #
LP Decreased Thymus and Spleen Damage Caused X-ray Radiation
LP Pretreatment Decreased Thymus and Spleen Damage Caused by X-ray Radiation

Values were expressed as mean ± SEM. #
LP Treatment Mitigated Radiation-Induced Morphological Changes in the Spleen
Morphological changes of spleen cells were observed by H&E staining. The main morphological features are described as follows. In the normal control group, the boundary between red pulp and white pulp was distinct, lymphocytes were closely arranged, and a large number of red blood cells was seen in the white pulp (Figure 1A). In the IR group, the splenic corpuscle had an abnormal shape and structure, the boundary between the white pulp and the red pulp was indistinct, and the number of germinal centers was reduced or undergoes atrophy (Figure 1B). In the LP groups, with an increase in drug dose, the boundary between red pulp and white pulp gradually became distinct, and the number of red blood cells in the white pulp gradually increased as shown in Figure 1C-E. Morphology of spleen after radiation (100×). A: control group, B: IR (4.0 Gy), C: 50 mg/kg.d LP + IR, D: 100 mg/kg.d LP + IR, E: 200 mg/kg.d LP + IR
LP Reduced Spleen Cell Apoptosis Induced by Radiation
Sections of the mouse spleen were stained with TUNEL and observed by a laser confocal microscope. The results of each group are shown in Figure 2A. The spleen cell nucleus appeared blue and TUNEL-positive cells were green. The overlap between the two represented cell apoptosis. Following 24 h of X-ray irradiation, the number of apoptotic cells in the spleen tissue was significantly higher than that of the normal control group ( Effects of LP on splenocyte apoptosis induced by X-ray irradiation in mice. ##
LP Prevents Radiation-Induced DNA Damage in Mice
Comet assays play an important role in evaluating DNA damage in mammalian cells. Radiation causes DNA strands to break, increasing the growth of short strand fragments that form comet-like shapes and long tails during electrophoresis. A significant increase in DNA damage was observed after irradiation (Figure 3A). We also calculated the TM and OTM with or without LP. As shown in Table 3, the TM and OTM of spleen cell DNA in the irradiated group were significantly higher than those in control group. LP pretreatment significantly reduced the tail length of spleen cells compared with the irradiated group. The results suggested that LP had the potential to prevent DNA damage in irradiated mice. Representative images from the neutral comet assay The Effect of LP on DNA DSBs in Mouse Spleen Cells Values were expressed as mean ± SEM. #

Effects of LP on Radiation-Induced DNA Damage Repair-Related Proteins by Immunohistochemistry
DSBs are important in determining the fate of cells because damaged DNA that is not repaired can lead to cell death. DSBs activate a cascade of DNA damage repair (DDR) signals that control cell cycle arrest, DNA repair, and cell fate. DDR functions by an orchestration of multiple molecules, sensors, transducers, mediators, and effectors, and its transduction pathway is mediated by protein phosphorylation.
18
Therefore, we first analyzed the levels of the DSB marker γ-H2AX by immunohistochemistry to evaluate directly the role of LP pretreatment in the cellular response to irradiation-induced DNA damage. The number of cells with positive γ-H2AX expression in spleen sections of mice in the irradiation group was significantly higher than that in the normal group, whereas pretreatment with LP could decrease the number of positive expression cells as shown in Figure 4. We also detected the expression and phosphorylation of DDR key proteins (Ku70, DNA-PKcs, Chk2, p-ATM, and P53) in this study. As shown in Figure 4, the number of Ku70, DNA-PKcs, Chk2, p-ATM, and P53 positive expression cells in spleen sections of mice in the irradiation group was significantly higher than that in the normal group, whereas pretreatment with LP could decrease the number of cells with positive expression. Effect of LP on DNA damage repair related proteins in the tissues measured by immunohistochemical staining #
Effects of LP on Radiation-Induced DNA Damage Repair-Related Proteins
Further analysis of protein expression levels revealed the possible mechanisms by which LP affects molecule signaling in DNA breaks. The western blotting results in Figure 5 show a significant increase in the protein levels of γ-H2AX, p-Ku70, DNA-PKcs, Chk2, p-ATM, and P53 after irradiation. However, LP treatment significantly reduced the levels of protein expression, which was consistent with the results of immunofluorescence experiment. In addition, we also detected the protein levels of apoptosis-related Caspase3 and Bax, and the results showed that after irradiation, levels of Caspase3 and Bax were significantly increased. In contrast, the expression level of Bcl-2 was significantly decreased. However, LP pretreatment significantly reduced the expression of Caspase3 and Bax and increased the expression of Bcl-2. Effect of LP on DNA damage-related proteins in tissues #
Discussion
Although radiation therapy remains a conventional method for treating malignant tumors, the dose is limited because of the toxicity to normal tissues. IR can cause dysfunction of hematopoietic, nervous system and immune system.19,20 Radiation damage is partly caused by the production of ROS, including hydrogen peroxide (H2O2), superoxide anion (O2
As part of the immune system, bone marrow, the thymus, the spleen, and lymph nodes form an intricate network of cells and organs. It is possible for immune dysfunction to result in autoimmune disorders, inflammatory conditions, and even cancer..23,24 The thymus and spleen are radiation-sensitive organs, which are easily attacked by IR.25-27 Thymus and spleen injury indexes reflect the degree or state of their injuries. Our results showed that the thymus and spleen indexes of irradiated mice decreased, and LP treatment before radiation exposure prevented thymus and spleen damage in mice exposed to radiation.
The immune system is mainly conducted in the white pulp of the spleen, which is formed by lymphoid tissue aggregation. Spleen injury can cause immune system dysfunction and increase vulnerability to infection and other diseases. The morphology of white pulp and red pulp is easy to identify and can be used to evaluate the functional status of the spleen. Because lymphocytes are extremely sensitive to radiation, white and red pulps disappear rapidly after IR exposure. 27 The structure of the spleen has been identified as shown in Figure 1. The white pulp was severely damaged, whereas red pulp cells and surrounding hyperemia decreased after IR. LP pretreatment effectively protects the tissue structure. Inflammatory injury is related to impaired antioxidant capacity of cells. These results suggested that LP may protect spleen from radiation damage through its antioxidant effect.
Numerous pathways can mediate apoptosis, including mitochondria, endoplasmic reticulum, and death receptor pathway, which are reported to be involved in REDOX imbalance. 28 IR produces a substantial amount of ROS. Excessive ROS will place the body in a state of oxidative stress. Moreover, radiation-induced apoptosis is closely related to oxidative stress. 29 It has been found that polysaccharides reduced apoptosis induced by irradiation.30,31 In the present study, there was a significant increase in apoptosis following irradiation, but pretreatment with LP reduced apoptosis in splenic cells dose-dependently.
DNA is the direct target for cell damage caused by radiation. Previous studies have confirmed that comet assay can be used to detect DNA damage caused by radiation.32-34 We used comet assays to evaluate the spleen DNA damage and the protection against radiation offered by LP pretreatment. As shown in Table 3, the values of TM and OTM increased significantly. These results suggested that IR-induced DNA damage in cells was consistent with previous reports. Pretreatment with LP reduced radiation-induced DNA damage, suggesting that LP potentially provided protection against the damage caused by radiation.
DSBs are important in determining the fate of cells because damaged DNA that is not repaired can lead to cell death. The DSB-induced chromatin alterations and subsequent phosphorylation of H2AX near the DSB site are signals for ATM to activate. As a sensitive method for detecting DSBs caused by irradiation, γ-H2AX foci can be used. After irradiation, we observed an increase in the number of -H2AX positive cells in the spleen tissue. Moreover, its levels of protein expression were also significantly increased. DNA-PKcs and Ku70/Ku80 form the core of a multi-protein complex known as DNA-PK, which recruits to DSBs and facilitates synapsis. Additionally, DNA-PK serves as a signal to downstream effectors when DNA is damaged. This is achieved by phosphorylating the conserved Ser139 site at the carboxyl terminus, which flanks the histone H2A.X breakage.35,36 Our results indicated that the protein expression of DNA-PKcs in mouse spleen cells was significantly increased after irradiation as shown in Figures 4 and 5. Combined with the comet assay, our results suggested that X-ray caused DNA DSBs in splenocytes, and that LP prevented irradiation-induced DNA DSBs.
To further demonstrate how LP prevents DSBs, we examined the DNA DDR-related signaling pathway. Ku70 is an evolutionarily conserved DNA repair subunit protein that binds to DNA DSBs and assists in DNA repair. Our results showed that both Ku70 and phosphorylated Ku70 expression were significantly elevated after irradiation. DNA repair, cell death, and cell cycle progression are controlled by DDR via a complex network of proteins. DNA damage triggers ATM, which belongs to the PIKK family. Activation of the ATM can induce the activation of sensor proteins, such as Chk1, Chk2, p53. 37 After irradiation, the expression of ATM, Chk2 and P53 were significantly increased. Pretreatment with LP reduced the expression of the above proteins in a dose-dependent manner. Previous studies have shown that ROS generated by IR acted on mitochondria, leading mitochondrial dysfunction and activating the mitochondrial apoptosis pathway.38-40 Bcl-2 family members and Caspase-3 are key factors in mitochondrial apoptosis pathway. In subsequent experiments, we examined the protein expression associated with mitochondrial apoptotic signaling. We observed significantly increased levels of Bax and caspase3 after irradiation and a significant decrease in the expression of the antioxidant protein Bcl-2. A significant reduction in Bax and Caspase-3 protein expression was observed following LP pretreatment, while an increase in Bcl-2 protein expression was observed.
This study confirmed that LP pretreatment exerts protective effects against radiation-induced damage, such as redox imbalance, immune organ injury, DNA double-strand breaks, and cell apoptosis. However, we have not yet isolated and identified the specific active components of LP that mediate these protective effects. LP may contain a variety of bioactive substances (eg, polysaccharides, peptides, or phenolic compounds), and the relative contributions of individual components remain unclear. Additionally, while we observed that LP regulates the DDR signaling pathway (ATM/Chk2/p53) and the mitochondrial apoptosis pathway (Bcl-2/Bax/Caspase-3), the direct molecular targets of LP (or its active components) in these pathways have not been verified (eg, whether LP directly binds to ATM, Bcl-2, or other key proteins to modulate their activity). This limits a deeper understanding of the intrinsic mechanism underlying the radioprotective effect of LP. This study focused on the impact of pretreatment on the protective effect of LP.
Conclusion
Taking these data together, Lanzhou Lily Polysaccharide exerts a protective effect against radiation-induced splenic damage in mice, and its mechanism is associated with alleviating splenic oxidative stress, preserving splenic structural integrity and immune organ function, inhibiting splenocyte DNA damage, and regulating the splenocyte apoptosis pathway.
Footnotes
Ethical Considerations
Approval of the research protocol by an institutional review board: Northwest Minzu University approved the use of mice in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81560508), Natural Science Foundation of Gansu Province (20YF8FA046, 25YFFA032), the Science and Technology Plan Project of Chengguan district (2022SHFZ0020), Key Laboratory of Environmental Ecology and Population Health in Northwest Minority Areas (MWZD202201).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article.
Animal Studies
XBMZ-YX-20150008.
