Abstract
Objectives
The current study investigated angiogenesis-arresting attributes of Mesua ferrea oleo-gum resin extract and its underlying molecular mechanisms.
Methods
Series of in vitro, ex vivo and in vivo models were used to assess anti-angiogenic properties.
Results
MTT cell viability experiments showed that oleo-gum resin extract induced moderate cytotoxicity towards EA.hy926 cells (IC50 = 42 µg/mL). Extract-treated cells showed significant reduction in invasion, migration, and tube formation potential. At the protein level, down-regulation in expression of angiopoietin-1 and -2, Tie-2, MMP-1 and -9, VEGF-A, and VEGFR2 pro-angiogenic proteins was observed in extract-treated EA.hy926 cells. Signalling array data indicated a marked down-regulation of transcription factors, i.e., HIF-1α and WNT (−3.68 ± 5.74 and −6.24 ± 6.50 fold-change). Furthermore, extract treatment diminished vessel-sprouting in in vitro 3D spheroids, ex vivo rat aorta ring, and in vivo chick embryo chorioallantoic membrane models. Treatment with extract significantly reduced intracellular ROS and caspases-8 and -9 levels. GC-MS and HPLC analyses of extract indicated the presence of (+)-α-longipinene, isoledene, cedrene and α-elemene. ADMET prediction of detected compounds revealed good intestinal absorption (> 90%) and skin permeability (log Kp < −2.5), making the extract a suitable candidate for the treatment of angiogenesis-associated intestinal and skin disorders.
Conclusion
Overall, these findings suggest that Mesua ferrea extract exhibits anti-angiogenic properties by down-regulating the VEGF/angiopoietin axis, warranting further investigations in treating angiogenesis-associated diseases.

Introduction
Angiogenesis, the formation of new blood vessels, plays a central role in physiology, e.g., wound healing, placenta formation, and embryogenesis. 1 On the other hand, dysregulated angiogenesis is involved in the development and progression of many diseases, including but not limited to autoimmune disorders, cerebral ischemia, cardiovascular disorders, diabetic retinopathy, impaired wound healing, scar formation, arthritis, and tumours.2,3 Clinical studies data showed that diabetic ulcer patients treated with platelet-derived growth factor (PDGF-BB; Becaplermin) were at greater risk of developing cancer 4 supporting the notion that activation of angiogenesis at the site of tissue injury favours tumour cell survival and growth. During angiogenesis various mediators cross-talk with each other to control multiple interlinked cellular and molecular events. Vascular endothelial growth factor (VEGF) and its receptor (VEGFR2), angiopoietin-1 and -2 and their receptors (Tie-2), matrix metalloproteinases (MMP), and IL-1β are over-expressed during angiogenesis and have been extensively studied to target uncontrolled angiogenesis.5,6
Though angiogenesis inhibitors have shown promising results in the clinical settings, but their use is also associated with serious side effects such as bleeding, wound healing abnormalities, thrombosis, hypertension, hypothyroidism, fatigue, proteinuria, oedema, skin toxicity, leukopenia, lymphopenia, and immunomodulation respectively.7-9
In the Ayurvedic medical system, oleo-gum resins obtained from different parts of the plants are used for the treatment of arthritis, bone fractures, diabetes, gout, inflammation, obesity, and loss of memory.10,11 A literature review on the clinicaltrials.gov website revealed that oleo-gum resins derived from various plant species are currently under clinical investigations. For example, myrrh (gum resin obtained from Commiphora species) was investigated for its clinical efficacy in schistosomiasis (Trial ID: NCT01529710). However, this trial demonstrated only a low efficacy of gum resin in managing schistosomiasis compared to praziquantel. 12 Another clinical trial evaluated the efficacy of Boswellia serrata gum resin and its active bioactive molecule, boswellic acid, in the modulation of the immune system in COVID-19 patients (Trial ID: NCT04487964). The trial concluded that it may be considered for use in mild to moderate infections of SARS-CoV-2 or COVID-19 variants. 13 The efficacy of Ferula assa-foetida oleo-gum resin was evaluated in various dental disorders in a double-blinded clinical trial, and its use as an efficient herbal mouthwash for improving gingival health was recommended. 14 Additionally, herbal products are available in Belgium (Flexofytol® PLUS, Tilman Laboratory) that contain oleo-gum resin of Boswellia serrata, and are recommended for the treatment of inflammatory joint disorders. These studies indicated the medicinal potential of oleo-gum resin extracts.
The plant family Guttiferae (also known as Clusiaceae) is well-known for its medicinal importance. For example, latex obtained from Malaysian Calophyllum lanigerum and C. teysmannii of this family was reported to have significant anti-HIV activity. 15 Mesua ferrea L. is another medicinally important member of this family. It is traditionally used to treat several human ailments. For example, its flowers, when mixed with butter and sugar, are used in the treatment of bleeding piles and burns. Leaves and flowers have antidote effects against snake bite and scorpion stings. 16 M. ferrea L. is also one of the active ingredients of herbal formulations used as an immune booster, expectorant, cough suppressant, anti-emetic, anti-inflammatory, analgesic, cardiac tonic, and anticancer agent. Scientifically, it is proven to have anti-inflammatory, analgesic, anti-microbial, anti-cancer, anti-ulcer, and antioxidant properties. 17 Although extensilvey studied scientifically, to the best of our knowledge, there is no study available which reported the anti-angiogenic property of M. ferrea. Therefore, the current study was designed to explore the angiogenesis-inhibiting properties of the oleo-gum resin extract of M. ferrea L. and its underlying molecular mechanisms.
Materials and Methods
Preparation of Oleo-Gum Resin Extract
Cuts were made on the stem of a well-identified Mesua ferrea tree (voucher number 11535) located in the Universiti Sains Malaysia (USM), Malaysia. Oleo-gum resin was collected in glass air-tight containers and stored at 4°C as reported previously. 18 Based on the biological activity of a previous study, 18 the n-hexane oleo-gum resin extract was evaluated for its anti-angiogenic potential. A detailed procedure is mentioned in the supplementary file.
Chemical Characterisation of Oleo-Gum Resin Extract
The oleo-gum resin extract was chemically characterised using GC-MS and HPLC techniques as reported previously 18 .
Prediction of Pharmacokinetic and Toxicity Profile of Reported Phytoconstituents
Online ADMET tools, Deep-PK (https://biosig.lab.uq.edu.au/deeppk/), pkCSM (https://biosig.lab.uq.edu.au/pkcsm/prediction), CardioToxCSM (https://biosig.lab.uq.edu.au/cardiotoxcsm/) were used to predict the various pharmacokinetic and toxicological properties of compounds reported previously in the GC-MS screening of oleo-gum resin extract of Mesua ferrea i.e., α-longipinene, isoledene, cedrene, and α-elemene. SMILES and other relevant information for these compounds were taken from PubChem.19-21
Cell Viability Analysis
In vitro MTT cell viability assay was employed to study the effect of oleo-gum resin extract treatment on the viability of human endothelial cells (EA.hy926; CRL-2922-ATCC). In brief, cells (2.5 × 10^3) were treated with 6 different concentrations of oleo-gum resin and betulinic acid (reference) for 48 h. Cells treated with 0.5% DMSO served as the control. At the end of the experiment, pictures of the cells were taken using an inverted microscope, and IC50 values were calculated following the reported protocol. 22 Based on the IC50 values, 3 different concentrations of the oleo-gum resin extract, viz., 21, 42, and 71 µg/mL were screened in further cell culture assays.
Measurement of Intracellular Reactive Oxygen Species (ROS)
The reported method was used to study the effect of oleo-gum resin treatment on the generation of intracellular ROS. 23 In brief, cells were treated with 21, 42, and 71 µg/mL of oleo-gum resin extract and 8 µg/mL of betulinic acid for 24 h and a cell-permeable fluorescent dye, i.e, 2′,7′-dichlorofluorescein diacetate (DCFH-DA) was used to assess the generation of intracellular reactive oxygen species (ROS) in EA.hy926 cells. The results are expressed as mean ± SD of fluorescent intensity (n = 3).
Caspase-8 and -9 Levels Estimation
The effect of oleo-gum resin extract on the activity of caspase-8 and -9 was studied using Caspase-Glo kits, following the manufacturer’s recommended methods. Luminescence was measured using a microplate reader, and the results are presented as the mean ± SD of the relative fold change in caspase activity in the treatment groups (n = 3). A detailed procedure is mentioned in the supplementary file.
In vitro Anti-Angiogenic Evaluations
Cell Invasion Assay
The Boyden chamber method, with slight modifications, was performed to evaluate the effect of the oleo-gum resin extract on the invasion capability of EA.hy926 cells. Photos of 05 randomly selected fields per well were taken at 10 × magnifications. The number of cells invading the matrix was counted, and the percent (%) inhibition of cell invasion was calculated. The results are presented as mean ± SD of percent inhibition of cell invasion in the treatment groups (n = 3). 18 A detailed procedure is mentioned in the supplementary file.
Cell Migration Assay
A scratch method was used to assess the ability of oleo-gum resin extract to arrest the migration of EA.hy926 cells following reported protocols. Photographs of the scratched areas were taken at 0 h (before treatment) and 24 h (at the end of treatment) from 6 different points of each well using an inverted microscope (EVOS fl, Digital microscopy group, USA) and cell-free area at 0 h. and 24 h. was measured using QWin Leica image analysis software (Leica Microsystems Inc., Buffalo Grove, Illinois). The percent inhibition of cell migration was calculated using a reported formula, and results are presented as mean ± SD of percent inhibition of cell migration (n = 3). 24 Moreover, representative images of each group (24 h) were also analysed using an online version of Wimasis image analysis software and cell-free area and other parameters were calculated (https://www.wimasis.com). A detailed procedure is mentioned in the supplementary file.
Tube Formation Assay
A reported method with minor alterations was used to study the effect of oleo-gum resin treatment on the differentiation of EA.hy926 cells into a tubular organisation on a matrigel matrix. 25 Pictures of formed tubes in each treatment group were taken at 4× magnification using an inverted microscope. The free version of Wimasis image analysis software was used to study the effect of treatment on various tube formation parameters (https://www.wimasis.com). A detailed procedure is mentioned in the supplementary file.
Estimation of Anti-Sprouting Efficacy in 3D Spheroid Model
The reported protocol was adopted to study the effect of oleo-gum resin extract treatment on the sprouting of tube-like vessels from EA.hy926 spheroids. 26 Spheroids were treated with 42 µg/mL of oleo-gum resin extract and 8 µg/mL of BA for 5 days. Media with 0.5% DMSO was used as a control. Photographs of spheroids were taken using an inverted microscope (EVOS fl, Digital microscopy group, USA) on the alternate days, and were analysed for the origination of sprouting in each group (n = 3).
Ex vivo Anti-Angiogenic Evaluations
Rat Thoracic Aortic Ring Model
Thoracic aortas were excised from healthy male Sprague–Dawley rats aged 10-12 weeks and placed in cell culture Petri dishes containing M199 medium without serum and 1% antibiotics (penicillin-streptomycin). Thoracic aortas were carefully cut into 1 mm thick rings after removing the extra fibro-adipose matter. Details for the preparation of the treatment medium and treatment protocols are provided in the supplementary file. 25 Length of vessels as well as other parameters were measured using QWin Leica image analysis software (Leica Microsystems Inc., Buffalo Grove, Illinois) and online version of Wimasis software (https://www.wimasis.com). The inhibition of blood vessel outgrowth (%) on the basis of vessel length (px) in experimental groups was calculated using the reported formulas. The animal handling procedures were approved by the animal ethics committee of Universiti Sains Malaysia [Approval number: USM/Animal Ethics Approval/2014/(94) (599) and (94) (600)].
In vivo Anti-Angiogenic Evaluations
Chicken Chorioallantoic Membrane (CAM) Assay
In brief, fertilised eggs of white leghorn chickens, 3-4 days of age, were purchased from the local market. Eggshells were cleaned with 70% ethanol and incubated at 37°C with 60% humidity for 24 h. Later, under sterile conditions, approximately 4-5 mL of egg albumin was aspirated from the pointed end of the egg using a 21-gauge needle. This allowed CAMs to detach from the shell. Subsequently, a small window was created in egg shells using sterile scissors and observed for stable CAM development. Only eggs with intact and well-developed chorionallantoic membranes (CAMs) were used in the current study. Sterile Whatman’s grade I filter paper discs loaded with 50 and 100 µg of oleo-gum resin extract or 0.5% DMSO were placed in the centre of CAMs. Eggshells were again sealed using a sterile paraffin tape and re-incubated in a cell culture incubator for 24 h. At the end of treatment, the windows were reopened, and pictures of CAMs were taken. 27 The images were analysed using free version of Wimasis software (https://www.wimasis.com).
Determination of the Vascular Endothelial Growth Factor (VEGF) Concentration
The concentration of the angiogenic cytokine (VEGF-A) in a cell culture supernatant was estimated using a human VEGF-A platinum ELISA kit (eBioscience, USA). The linear regression (R2) equation of the standard VEGF-A dose-response was used to calculate the amount of VEGF released by cells treated with oleo-gum resin extract. The results are presented as picograms per millilitre of VEGF in each group (n = 3). A detailed procedure is mentioned in the supplementary file.
Human Angiogenesis Proteome Profiler
The effect of oleo-gum resin extract treatment on the expression of major proteins involved in angiogenesis was investigated using a G-series human angiogenesis antibody array (Raybiotech, USA). The assay was conducted according to the manufacturer’s protocol on EA.hy926 cells using a 8-well antibody-coated glass slide array. A comparison was made between protein expression of target proteins in the 0.5% DMSO-treated and oleo-gum resin-treated cells (42 μg/mL) respectively. The results are presented as mean ± SD of fold change values (n = 2).
Expression of Major Cancer Signalling Pathways
The modulatory effects of oleo-gum resin extract on the expression of 10-major signalling pathways were studied using Cignal finder reporter array (Qiagen, USA). The relative levels of transcription factors, viz., TCF/LEF, RBP-Jk, p53, NF-κB, HIF-1a, SMAD2/3/4, E2F/DP1, Myc/Max, Elk-1/SRF, and AP-1 representing WNT, Notch, p53, NF-κB, HIF-1α, TGF-β, cell cycle, Myc/Max, MAPK/ERK, and MAPK/JNK pathway in EA.hy926 cells were estimated in a 96-well plate format according to the manufacturer’s protocol. The results are presented as mean ± SD of relative fold change in activity (n = 3).
In vivo Toxicity Study
The OECD 425 (up and down procedure) guideline was adopted to study the acute toxicity profile of oleo-gum resin extract. 28 In brief, 05 healthy 12 to 14 weeks old female Sprague–Dawley (SD) rats were obtained from the ARASC facility at Universiti Sains Malaysia (USM) and were kept in an animal transit room located at the School of Pharmaceutical Sciences, USM. For oral administration, the oleo-gum resin extract was triturated with Tween 80 using a porcelain mortar and pestle, and water was added in a drop-wise manner. This trituration resulted in the formation of a milky suspension of oleo-gum resin. Due to its less solubility in water, 2000 mg/kg extract was given in 4 divided doses at 6 h. intervals. The animals were observed for 48 h. for any signs of toxicity and/or mortality, and then again on day 7 and 14. Different parameters, including body weight, food and water consumption, behavioural changes (vomiting, diarrhoea, salivation, level of alertness), and changes in fur colour and lacrimation, were studied.
Statistical Analysis
Findings are represented as mean ± SD (n = 3). GraphPad Prism (San Diego, CA, USA) software was used to conduct a t test and one-way ANOVA (post hoc-turkeys’) to test differences among groups. A P-value less than 0.05 was considered statistically significant.
Results and Discussion
In the current study, the anti-angiogenic potential of an oleo-gum resin extract of Mesua ferrea (L.) was screened in a series of in vitro, ex vivo, and in vivo models. The EA.hy926 cell line derived from human umbilical vein endothelial cells is commonly employed to study the effect of test substances on angiogenesis.
29
The selection of concentrations to be used in in vitro assays was based on the IC50 value of the oleo-gum resin extract against EA.hy926 cells in the MTT cell viability assay. The treatment with oleo-gum resin extract for 48 h. induced 50% inhibition (IC50) at a concentration of 42 ± 0.10 µg/mL (Figure 1). Various studies have categorised the plant extracts into 4 different groups depending upon IC50 values, viz., plant extracts having IC50 value ≤ 20 µg/mL are considered cytotoxic, moderately toxic if the IC50 value is = 20-100 μg/mL, weakly toxic if the IC50 value is between 100-1000 μg/mL, and non-toxic if the IC50 value is above 1000 μg/mL, respectively.
30
According to this criterion, oleo-gum resin extract induced moderate toxicity in EA.hy926 cells. Based on this IC50 value, 3 different concentrations of oleo-gum resin extract were selected for detailed experiments viz., 21 (half IC50), 42 (IC50), and 71 (approximately IC75) µg/mL. Noteworthy, this IC50 was calculated based on the exposure of cells to oleo-gum resin extract for 48 h. while in the subsequent models, cells were exposed to oleo-gum resin extract for 24 h. Thus, this further reduces the chances of significant cytotoxicity. This notion is also supported by the data of the in vivo acute toxicity study, where no signs of toxicity were observed in female albino rats treated with a single dose of 2 g/kg of oleo-gum resin extract (Table 1). In addition, toxicity profiling of reported phytoconstituents of oleo-gum resin extract using ADMET tools showed broad spectrum non-toxic effects supporting the data of in vivo oral acute toxicity (Supplemental Table S1 and S2). Effect of Oleo-Gum Resin Extract Treatment on Viability of EA.hy926 Cells. Representative Photos Were Taken at 4× Magnifications (Scale Bar 1000 μm) Acute Toxicological Observations in Female SD Rats Treated With 2000 mg/kg of Oleo-Gum Resin Extract No visible signs of toxicity were observed in animals treated with oleo-gum resin extract. The (+) sign indicates the presence of toxicity signs, and the (−) sign indicates the absence of any abnormality.

Furthermore, to rule out the possible involvement of cytotoxicity in the inhibition of EA.hy926 cells transformation, the effect of oleo-gum resin extract treatment on the induction of executioner caspases i.e., caspases 8 and 9 in EA.hy926 cells was studied. The data obtained indicated a significant (P < 0.05) decrease in the levels of both executioner caspases, i.e., caspase 8 and 9, indicating a non-cytotoxic effect of the oleo-gum resin extract at the tested concentrations. Relative fold change values of caspases 8 in cells treated with 21, 42, and 71 µg/ml of oleo-gum resin extract and 8 µg/ml of BA were 0.82 ± 0.05, 0.72 ± 0.05, 0.66 ± 0.07 (P < 0.05) and 0.69 ± 0.41 respectively. While, relative fold change values of caspases 9 in cells treated with 21, 42, and 71 µg/ml of oleo-gum resin extract and 8 µg/ml of BA were 1.03 ± 0.06, 1.03 ± 0.05, 0.99 ± 0.03 and 0.11 ± 0.02 (P < 0.05) respectively (Figure S1). Noteworthy to mention that both oleo-gum resin extract and BA treatments result in the down-regulation of both the executioner caspases.
Having established the non-toxic nature of the extract, the study was further extended to explore its anti-angiogenic properties using a series of well-established in vitro, ex vivo, and in vivo models. A 3D hanging drop assay was performed to predict the effect of oleo-gum resin treatment on EA.hy925 cells sprouting in in vivo environment. It is reported that capillary-like sprouts formed in this assay mimic the physiological and programmed arrangement of endothelial cells in vessels.
31
Data obtained indicated the potent capacity of extract to arrest vessel sprouting (Figure 2). Noteworthy, the extract did not kill cells in the spheroids but arrested their capacity to grow. In Vitro Effects of Oleo-Gum Resin Treatment on EA.hy926 Cells Sprouting in 3D Hanging Drop Assay. Cells Were Treated With 42 μg/mL of Oleo-Gum Resin Extract and 8 μg/mL of Betulinic Acid. Marked Inhibition of Vessels Sprouting was Observed in Oleo-Gum Resin Extract and Betulinic Acid Treated Spheroids. Red Coloured Arrows Show Disruption of Vessel Network (Scale Bar 400 μm)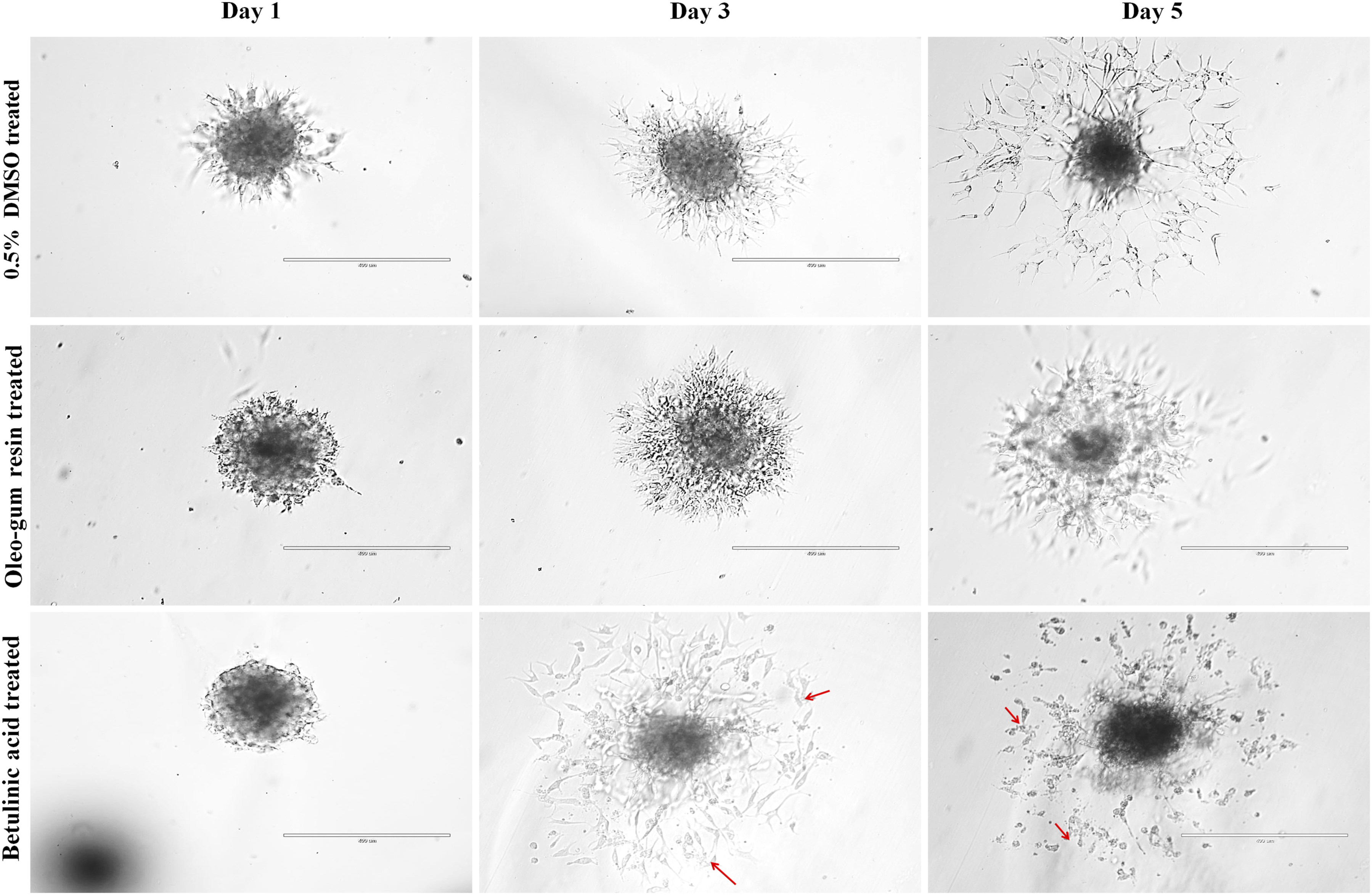
The rat aortic ring assay is one of the most commonly used ex vivo models for screening the anti-angiogenic properties of test drugs. New vessels sprouting from thoracic aortic rings resemble the characteristics of blood vessels formed during angiogenesis.
32
In the current study, different concentrations of oleo-gum resin extract and 20 µg/mL of betulinic acid were tested. Length of vessels in aortic rings treated with 0.5% DMSO, 6.25, 12.5, 25, 50 and 100 µg/mL of oleo-gum resin extract and 20 µg/mL of betulinic acid was 708 ± 80, 585 ± 74.1, 546 ± 72.5, 544 ± 167, 410 ± 165, 84.5 ± 50.6, and 56.4 ± 17.9, respectively. Significant reduction in vessel length was observed in aortic rings treated with 50 (P < 0.01) and 100 µg/mL (P < 0.001) of oleo-gum resin extract and 20 µg/mL of betulinic acid (P < 0.001). The percentage reduction in micro-vessels sprouting from aortic rings treated with 6.25, 12.5, 25, 50 and 100 µg/mL of oleo-gum resin extract and 20 µg/mL of betulinic acid was 17.31 ± 10.47%, 22.87 ± 10.24%, 34.75 ± 17.45%, 42.02 ± 13.35%, 88.05 ± 7.14%, and 92.03 ± 2.52%, respectively. The IC50 value of oleo-gum resin, calculated from the linear regression equation of the dose-response curve (y = 0.9037x + 0.6917, R2 = 0.9921), was determined to be 52.79 ± 2.50 µg/mL. Vessel network analysis also showed a marked reduction in vessel sprouting, sprouting area, and sprouting thickness. Microscopic inspection of the vascular network in the oleo-gum resin extract-treated rings revealed a sparse and irregular network with multiple discontinuities in the chief vessels. 0.5% DMSO treatment did not alter vasculature, i.e., vessels were dense and covered a significant portion of the plate (Figure 3, Figure S3 and Table S3). The findings of the current study are in line with another research report where treatment of aortic rings with Kaempferia galangal similarly inhibited blood vessel outgrowth.
33
Ex Vivo Anti-angiogenic Effects of Oleo-Gum Resin Extract in Rat Thoracic Aortic Ring Model. Where A = 0.5% DMSO Treated, B = 12.5 μg/mL, C = 25 μg/mL, D = 50 μg/mL, E = 100 μg/mL of Oleo-Gum Resin Extract Treated and F = Betulinic Acid 20 μg/mL. Significant Reduction in Different Studied Parameters was Observed in Aortic Rings Treated With 50 and 100 μg/mL of Oleo-Gum Resin Extract. Representative Photos Were Taken at 4 × Magnifications (Scale Bar 1000 μm)
Migration and invasion are the characteristic features of the endothelial cells during angiogenesis. In the current study, treatment with oleo-gum resin extract induced dose-dependent inhibition of EA.hy926 cell invasion across a growth factor-enriched Matrigel matrix, mimicking the tissue basement membrane. Percent inhibition of invasion in 21, 42 and 71 µg/mL of oleo-gum resin extract and 8 µg/mL of BA treated cells was 47.67 ± 18.62% (P < 0.001), 48.24 ± 16.89% (P < 0.001), 74.48 ± 16.89% (P < 0.001), and 92.25 ± 4.02% (P < 0.001), respectively. The IC50 of oleo-gum resin extract in the cell invasion assay was calculated to be 13 µg/mL (Figure 4). Photomicrographic Representation of Anti-invasive Effect of Oleo-Gum Resin Extract Towards EA.hy926 Cells. Where A = 0.5% DMSO, B = 21 μg/mL, C = 42 μg/mL, D = 71 μg/mL, E = Betulinic Acid 8 μg/mL. Significant Reduction in Invasion of EA.hy926 Cells was Observed in Cells Treated With 21, 42 and 71 μg/mL of Oleo-Gum Resin Extract and 8 μg/mL of BA. Red Coloured Arrows Indicate Invaded Cells Across Matrigel Matrix. ** = P < 0.01 and *** = P < 0.001. Photos Were Taken at 10 × Magnification (Scale Bar 400 μm)
Summary of Anti-tubulogenic Effects of Oleo-Gum Resin Extract
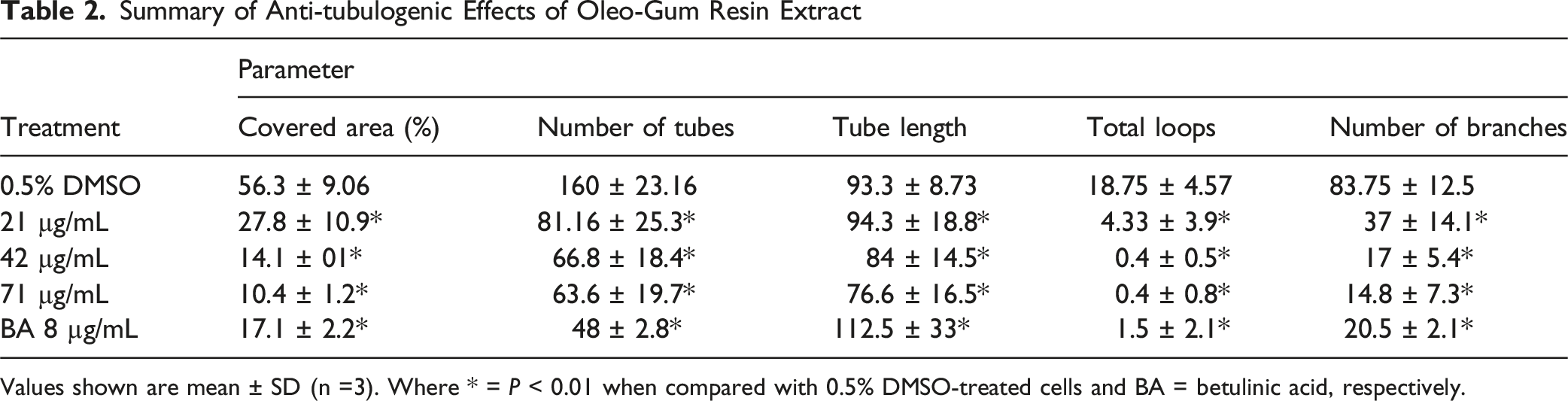
Values shown are mean ± SD (n =3). Where * = P < 0.01 when compared with 0.5% DMSO-treated cells and BA = betulinic acid, respectively.

Photomicrographic Representation of Effect of Oleo-Gum Resin Extract on EA.hy926 Tube Formation Potential. The Blue Area Represents Total Area Covered by the Tubes and Red Lines Represent Tubes and Tube Length. White Dots and Arrows Represent Branching Points and Black Arrows Highlight Loops Formed Respectively. Photos Were Taken at 4 × Magnifications (Scale Bar 1000 μm). * = P < 0.01
Effect of Oleo-Gum Resin Extract Treatment on the Expression of Proteins of the Angiogenic Cascade

Values shown are mean ± SD of the signal obtained from 2 spots of each target protein (n = 2). Raybiotech® analysis tool was used for data aligning, sorting, and normalization against the background. A (−) sign indicated down-regulation in the expression. The order of down-regulation was Tie-2 > VEGFR2 > IL-1α > uPAR > MMP-1 > IL-1β > MMP-9 > Ang-1 > Ang-2. Significant down-regulation in the expression of proteins was observed in oleo-gum resin extract-treated cells as compared with the 0.5% DMSO-treated cells. Where * = P < 0.05, and ** = P < 0.01 respectively.

Effect of Oleo-Gum Resin Extract Treatment on the Expression of Various Proteins Involved in the Angiogenesis Pathway. Targets Were Identified Using Protein Map provided With the Analysis Tool
Angiogenesis is a tightly regulated phenomenon controlled by a balance between pro- and anti-angiogenic signals. 35 Pathological angiogenesis has been identified as one of the core aetiologies responsible for the development and progression of chronic life-threatening diseases, including atherosclerosis, age-related macular degeneration, cancer, diabetic retinopathy, rheumatoid arthritis, psoriasis, and many more.36,37 The pro-angiogenic signals secreted by endothelial cells, fibroblasts, smooth muscle cells, platelets, inflammatory cells, and cancer cells include cytokines such as angiopoietins (Angs), epidermal growth factor (EGF), erythropoietin (EPO), fibroblast growth factors (FGFs), granulocyte-macrophage colony-stimulating factor/granulocyte colony-stimulating factor (GM-CSF)/ G-CSF), hepatocyte growth factor/scatter factor (HGF/SF), interleukin 8 (IL-8), placental growth factor (PGF), platelet-derived growth factor (PDGF), tumour necrosis factor-alpha (TNF-α), transforming growth factor-beta (TGF-β), and vascular endothelial growth factor (VEGF). These cytokines are known to promote various stages of angiogenesis, including activation, migration, invasion, tube formation, and maturation of neo-vasculature. 37 In the current study, HCT116 colon cancer cells were used to investigate the effect of oleo-gum resin extract on the release of VEGF-A. Data obtained indicated that cells treated with 8 (2360 ± 7.9 pg/mL, P < 0.05), 16 (2167 ± 8.3 pg/mL, P < 0.01) and 28 (1955 ± 5.1 pg/mL, P < 0.01) µg/mL of oleo-gum resin extract and 8 µg/mL of betulinic acid (1646 ± 2.4 pg/mL, P < 0.01) released significantly less VEGF as compared with the cells treated with 0.5% DMSO (2807 ± 21.29 pg/mL). Percent inhibition of VEGF secretion in oleo-gum resin extract (8, 16, and 28 µg/mL) and betulinic acid (8 µg/mL) treated cells was observed to be 16.54 ± 9.98%, 25.91 ± 9.68%, 30.02 ± 9.03%, and 40.65 ± 6.21%, respectively (Table S4).
Interaction of VEGF with VEGFR2 receptors results in the activation of endothelial cells, leading to their proliferation, migration, and sprouting.
38
In the current study, in addition to inhibiting VEGF-A release, oleo-gum resin extract treatment (42 µg/mL) significantly down-regulated the expression of VEGF receptors, specifically VEGFR2, on EA.hy926 cells (Table 3). The activated endothelial cells also secrete various degrading enzymes including matrix metalloproteinases (MMPs). MMPs digest the basement membranes making it possible for endothelial cells to invade and migrate toward the chemotactic signal.
36
Moreover, higher levels of MMPs and VEGF in the cancer patients indicate a more aggressive nature of tumours.
39
In the current study, oleo-gum resin extract-treated EA.hy926 cells had low levels of MMP1 and -9 indicating an anti-invasive potential of the extract (Figure 6). This notion is also supported by the findings of an in vitro assay, where the oleo-gum resin extract-exposed cells invaded the Matrigel matrix significantly less compared with untreated cells (Figure 4). Finally, angiopoietin 1 (Ang 1) stabilizes the newly formed blood vessels followed by pericytes incorporation in the basement membrane.
36
However, Ang 2 which is mainly expressed on the surfaces of the endothelial cells plays a critical role during pathological angiogenesis as it destabilizes endothelial-pericyte junctions and promotes endothelial cell survival, migration, and proliferation, especially in tumours.
40
Hence, our findings indicate that the oleo-gum resin extract attenuates the expression of both pro-angiogenic proteins (Ang 1 and 2), as represented in Figure 6, thus causing a dose-dependent reduction in the migratory potential of EA.hy926 cells (Figure 7). Anti-migratory Effect of Oleo-Gum Resin Extract Towards EA.hy926 Cells. In Raw Images Straight Lines Represent Cell Free Area before and after Exposure to Different Concentrations of Oleo-Gum Resin Extract while Circle Points out the Closure of Wound in 0.5% DMSO Treated Cells (i.e., after 12 h). Green Area in the Processed (Analysed) Images Represent Cell Covered Area and Red Lines Represent Distance Between Two Edges of Wound
Moreover, a higher Ang-2 to Ang-1 ratio predicts worse clinical outcomes.41,42 VEGFA and other pro-angiogenic cytokines are also known to regulate the expression of Ang-2.
43
In addition, up-regulated Ang-2 then acts on its receptors, i.e., the Tie-2 receptor located on the surfaces of endothelial cells. Ang-2/Tie-2 interaction activates endothelial cells and promotes angiogenesis via an autocrine loop.
44
The oleo-gum resin extract decreased the levels of VEGFA in the current study which could be one of the possible reasons for the down-regulated expression of Ang 2 and its Tie-2 receptors (Figure 6). This array data is also supported by the findings of an in vivo angiogenesis assay where a marked reduction in blood vessel density was observed in CAMs treated with 50 and 100 µg of oleo-gum resin extract (Figure 8, Table S5). In Vivo Anti-angiogenic Characterisation of Oleo-Gum Resin Extract of Mesua ferrea L. in Chick Choroallantoic Membrane Assay. Representative Images Were Analysed Using Free Version of Wimasis Image Analysis Software. The Blue Area Represents the Total Area Covered by the Tubes and Red Lines Represent the Total Length and Number of Tubes Formed. White Arrows Point Out the Total Number of Branching Points in Each Group Respectively. Marked Reduction in Vessel Network Length, Branching Points and Total Nets was Observed in CAMs Treated With Oleo-Gum Resin Extract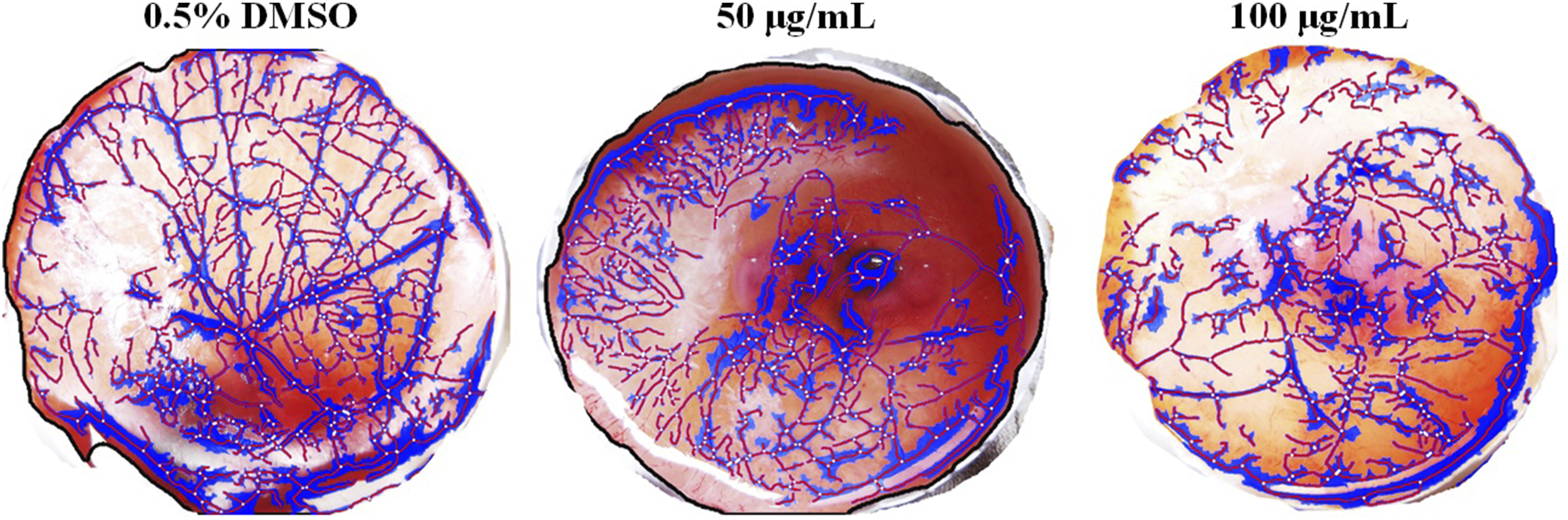
The Effect of Oleo-Gum Resin Extract on Intracellular ROS Levels

Values shown are Mean ± SD (n = 3). Where ns = P > 0.05 and * = P < 0.001.
Having identified protein targets modulated by exposure to oleo-gum resin extract in EA.hy926 cells, our study was further extended to explore the effect of the oleo-gum resin extract on associated signalling pathways. The oleo-gum resin extract significantly attenuated the expression of 2 major signalling pathways, viz., HIF-1α (−3.68 ± 5.74-fold change) and WNT (−6.24 ± 6.50-fold change). HIF-1α, an upstream regulator of the WNT signalling pathway, has been recognised as the master switch of angiogenesis. Under hypoxic conditions, HIF-1α is activated with the subsequent release of VEGF from a variety of cells. 47 Increased levels of reactive oxygen species (ROS) are known to stimulate angiogenesis through both VEGF-dependent and independent pathways. Moreover, ROS are also identified as a positive regulator of HIF-1α. 48 In the current study, treatment with 21, 42, and 71 µg/mL of oleo-gum resin extract and 8 µg/mL of BA resulted in a significant decrease in intracellular ROS levels compared with cells treated with 0.5% DMSO (Table 4, Figure S2). The findings of this study are in contradiction with our previous study where treatment of colon cancer cells (HCT 116) with oleo-gum resin extract resulted in increased ROS levels with subsequent activation of apoptotic machinery. 23 Further studies using a variety of endothelial and cancer cell lines are warranted to establish the exact molecular reason for these opposing effects of oleo-gum resin extract. Moreover, keeping in view the limited number of replicates used to screen the effect of gum resin extract treatment on the expression of proteins and transcription factors regulating the angiogenic cascade, additional studies are recommended using at least triplicates (n = 3) to confirm the hypothesis drawn based on data of the present study. Further, gene knockdown studies to confirm the exact molecular targets are also warranted.
GC-MS and HPLC analyses of oleo-gum resin extract revealed the presence of various terpenes including isoledene, α-longipinene, cedrene, and, α-elemene. which are suggested to be responsible for the observed anti-angiogenic properties of the oleo-gum resin extract (Figure 9, Table 5). The findings of the current study are supported by results of another study reporting anti-cancer properties of a terpene viz., acetyl-11-keto-beta-boswellic acid (AKBA) obtained from the oleo-gum resin of Boswellia serrata through modulation of expression of multiple cancer-related biomarkers including those involved in angiogenesis.
49
Similarly, anti-angiogenic effects of a methanol extract of oleo-gum resins obtained from Pistacia vera, Pistacia khinjuk, and Rhus coriaria have been reported which support the findings and rationale of oleo-gum resin selection for the present study.50,51 Another issue with single molecule therapies is the mutation of the drug targets during disease progression which makes the treatment less or even ineffective. Natural products simultaneously target multiple aberrantly expressed molecular pathways which might facilitate the better and more efficient management of cancer.
24
Intriguingly, we have observed linear dose-response effects of the oleo-gum resin extract (Figure 1), which might be targeting multiple molecular pathways. However, non-linearity in dose-response is a critical problem in the treatment of cancer and therefore, a comprehensive evaluation of the oleo-gum resin extract using appropriate dose-response models and algorithms encompassing linear to non-linear must be carried out in further pre-clinical and clinical settings.52-54 GC-MS (1A and B) and HPLC (2A and B) Analyses of Oleo-Gum Resin Extract of Mesua ferrea L. Where Peak A = Longpinene, B = Isoledene, C = Cedrene, D = α-elemene and E = Cyclohexene, 6-Ethenyl-6-methyl-1-(1-methylethyl)-3-(1-methylethylidene)-, (S)-, F = (−) Alloaromadendrene Respectively. Note: Panel 1A, and 2A & B Used With the Permission of Biomedicine and Pharmacotherapy and Permissionfrance The GC-MS Chemical Profile of Oleo-Gum Resin Extract Compounds with similarity greater than 90% with NIST library are reported in the present study. Five compounds with a similarity of more than 90% were identified in the n-hexane extract. The order of percentage was Isoledene > alpha-elemene > Cedrene > (+)-α-Longipinene.

The mean of peak areas obtained from 3 replicate injections of oleo-gum resin extract and linear regression equation (y = 1.861x + 11.85, R2 = 0.999) obtained from standard isoledene curve (concentration vs peak area of 7 concentrations) was used to calculate the isoledene contents of oleo-gum resin extract following a validated HPLC method. Data obtained from the peak area showed that a total of 170.57 ± 1.37 mg of isoledene was present in each gram of oleo-gum resin extract.
Lastly, the pharmacokinetic properties of oleo-gum resin extract compounds were estimated using AMDET tools. Interestingly, all 4 screened compounds were predicted to have good intestinal absorption, i.e., > 90% making them suitable candidates to be further studied in pathological angiogenesis-associated intestinal diseases (e.g., Ulcerative colitis, Crohn’s disease) (Table S6). However, pharmacokinetic optimisation studies and detailed pre-clinical data are still required to establish this hypothesis.
Conclusion
Overall, this is the first study which explored the detailed anti-angiogenic effects of the oleo-gum resin extract of Mesua ferrea L. in a battery of well-established and interconnected models. Based on the data generated, it is concluded that treatment with this oleo-gum resin extract induced anti-angiogenic effects through down-regulation of the WNT/ HIF-1α regulated VEGF-angiopoietin axis in EA.hy926 cells. Further studies are recommended to investigate the anti-angiogenic effects of oleo-gum resin extract in different angiogenesis-associated disorders (diabetic retinopathies, rheumatoid arthritis, haemangioma, ulcerative colitis and many more) and to develop the safety database of oleo-gum resin before formulation development and clinical studies.
Supplemental Material
Supplemental Material - Mesua ferrea L. extract Attenuates Angiogenesis Through HIF-1α and WNT Mediated Blockage of VEGF/Angiopoietin Axis
Supplemental Material for Mesua ferrea L. extract Attenuates Angiogenesis Through HIF-1α and WNT Mediated Blockage of VEGF/Angiopoietin Axis by Fengwei Zhang, Mohamed Farouk Elsadek, Shafqat Rasul Chaudhry, Nojood Altwaijry, Shamsuddin Sultan Khan, Seyedeh Fatemeh Jafari, Aman Shah Abdul Majid, Chern Ein Oon, Amin Malik Shah Abdul Majid, Thomas Efferth, Muhammad Asif in Dose-Response.
Footnotes
Acknowledgments
The authors would like to extend their sincere appreciation for funding this work through the Ongoing Research Funding Program (ORF-2025-349), King Saud University, Riyadh, Saudi Arabia. We are also very thankful to Biomedicine and Pharmacotherapy and Permission France for allowing us to re-use data for Figure 9 from our published research article (doi:![]() ) for the current study
) for the current study
Ethical Considerations
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Universiti Sains Malaysia (Ethics approval number: USM / Animal Ethics Approval / 2014 / (94) (599) and (600))
Author Contributions
Conceptualisation, methodology, software, and formal analysis: FZ, MA, SCR, MFE, AMSAM, CEO, TE and ASAM. Investigation: MA, and SSK. Resources: AMSAM, MFE and MA. Writing—original draft preparation: MA, FZ, SRC, and CEO. Mechanism drawing: MA, SSK, SFJ and NA. Writing—review and editing: SRC, NA, and TE. Supervision: AMSAM, ASAM, CEO. All the authors have read and agreed to the final version of the manuscript. Moreover, the authors declare that all data were generated in-house and that no paper mill was used.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was carried out with funds provided by Ongoing Research Funding Program (ORF-2025-349), King Saud University, Riyadh, Saudi Arabia, the Institute of Postgraduate Studies, Universiti Sains Malaysia (USM Fellowship: PFD0009/12(R)), and University Grant (RUT1001/PFARMASI/851001), Universiti Sains Malaysia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study can be obtained from Mr. Muhammad Asif on a reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
