Abstract
The favorable responses of crop species to low-dose γ irradiation can help to develop cultivars with increased productivity and improved stress tolerance. In the present study, we tried to reveal the candidate metabolites involved in growth stimulation of barley seedlings after applying low-dose γ-radiation (60Co) to seeds. Stimulating doses (5-20 Gy) provided a significant increase in shoot length and biomass, while relatively high dose of 100 Gy led to significant inhibition of growth. Gas chromatography–mass spectrometry metabolomic analysis uncovered several compounds that may take part in radiation hormesis establishment in irradiated plants. This includes molecules involved in nitrogen redistribution (arginine, glutamine, asparagine, and γ-aminobutyric acid) and stress-responsive metabolites, such as ascorbate, myo-inositol and its derivates, and free amino acids (
Introduction
Due to their sessile nature, plants have to develop effective adaptive responses to changes in the environment that might affect their growing capacity. Crop yields are destabilized by abiotic stress factors 1 and improving plant tolerance is a major goal of crop improvement programs. 2 However, the intensity of abiotic stress factors and changes in growth and development of plants do not usually have a linear relationship. A significant number of studies have revealed that biphasic dose–response often occurs in the range of low concentrations and low intensities of various stress factors. 3,4 Responses of crop species to low-dose stress exposure can also include a broad range of stimulating agriculturally beneficial effects. 5,6
The favorable responses of crop species to low-dose γ-irradiation are relevant for the areas of crop biotechnology and space biology, providing the clues for developing of cultivars tolerant to abiotic stresses and having increased productivity. 7 Such favorable responses, depending on the conditions, include an increase in linear sizes of plant organs and biomass, stimulation of germination, and acceleration of development. 5,6,8 -10 Hormetic effects in plant species seem to depend on the induction of general adaptive response to moderate stress conditions, 11 which includes chaperons and proteasomes activation and general response of antioxidant, repair, and hormonal systems. 12,13
Seedlings play a pivotal role in the life cycle of plants, forming the bridge between the heterotrophic embryo and the established photoautotrophic plant, 14 and their successful development under unfavorable conditions is critical for the effective crop production. In the present study, we used gas chromatography–mass spectrometry (GC-MS) approach to reveal the candidate biochemical pathways involved in growth stimulation of barley seedlings after applying low-dose γ-radiation to seeds. In order to exclude the confounding factors, plants were grown on distilled water in the dark, suggesting the metabolic reallocation of endosperm resources after radiation exposure. The research uncovered a number of compounds that are probably involved in radiation stimulation of growth after irradiation of barley seeds, including major components of nitrogen metabolism and stress-responsive metabolites.
Materials and Methods
Barley Cultivar and Irradiation Conditions
The experiment was carried out using Nur cultivar of spring barley (Hordeum vulgare L). Several experiments on this cultivar showed the induction of growth stimulation in a certain dose range. 10,15 The analyses were performed using seeds harvested in 2017. γ-Irradiation of dry barley seeds was conducted using doses 5, 10, 15, 20, and 100 Gy (dose rate 60 Gy/h) at the Russian Institute of Radiology and Agroecology on a γ-facility GUR-120 (60Co). Seedlings of nonirradiated seeds were used as control.
Growth Conditions and Morphological Assessment
Control and irradiated barley seeds were surface sterilized using 10% H2O2 during 5 minutes. Within 1 hours after irradiation, control and irradiated seeds were sown in paper rolls imbibed in distilled water and incubated at 20°C in the dark (MIR-254; Sanyo, Osaka, Japan). For each condition, 6 paper rolls (each containing 100 seeds) were used. The lengths of seedlings’ roots and shoots were measured after 7 days (168 hours) of germination. After 1 day of air-drying, the weights of roots and shoots were estimated for 100 plants (per paper roll) using analytical scales (PA213C; OHAUS, Parsippani, NJ).
Root and shoot lengths were assessed individually for each plant in a paper roll, and data were normally distributed. Root and shoot weights were assessed per all plants in a paper roll (six paper rolls per condition), thus nonparametric approach was used for calculations. Root and shoot lengths are presented as mean values ± standard error, while weight values are shown as medians and quartiles. For comparisons between lengths of control and irradiated samples, we used t test, for comparisons between weights of control and irradiated samples, we used Mann–Whitney U test Microsoft Office Excel 2013 and Statistica 8.0 software were used for calculations.
Gas Chromatography–Mass Spectrometry Metabolomic Analysis
For metabolomic analysis, four replicates were used per experimental condition. Sampling was performed after 7 days of germination, while a seedling had one true leaf and several roots. A replicate was composed of the root (or shoot) tissue of the biggest seedling from a paper roll. For each replicate, 50 to 60 mg of shoot or root tissue were weighted and immediately frozen in liquid nitrogen. After that, samples were freeze-dried and delivered for GC-MS analysis.
Metabolite extraction, derivatization, GC-MS analysis, and data processing were performed according to Fiehn. 16 The freeze-dried samples were ground using metal beads and resuspended in 1 mL of frozen (−20°C) water:acetonitrile:isopropanol (2:3:3) solution, containing ribitol at 4 µg·mL−1 and extracted for 10 minutes at 4°C by shaking at 1400 rpm in an Eppendorf thermomixer (Eppendorf, Hamburg, Germany). Insoluble material was removed by centrifugation at 20 000g for 5 minutes. A 100 µL of supernatant were collected and dried for 4 hours in a Savant Speed-Vac (Thermo Fisher Scientific, Waltham, MA) and stored at −80°C.
Three blank tubes underwent the same steps as the samples. Quality controls were prepared by pooling the same amount of each sample. They were injected at the beginning, the middle, and the end of the injection series to ensure the stability of the derivatization. After drying for 1 hour after −80°C storage, 10 µL of 20 mg·mL−1 methoxyamine in pyridine were added to the samples. The reaction was performed for 90 minutes at 28°C under continuous shaking in the Eppendorf thermomixer. Then 50 µL of N-methyl-N-trimethylsilyl-trifluoroacetamide (Sigma M7891 10 × 1 mL; Sigma-Aldrich, St. Louis, MO) were added, and the reaction continued for 30 minutes at 38°C. After cooling down, 45 µL of the derivatized sample were transferred to an Agilent vial for injection. Metabolites were analyzed by GC-MS 4 hours after derivatization. One microliter of the derivatized sample was injected in split and splitless modes in Agilent 7890A gas chromatograph coupled to Agilent 5975C mass spectrometer, column Rxi-5SilMS from Restek (30 m with 10 m Integra-Guard column; Bellefonte, PA). The oven temperature ramp was 70°C for 7 minutes, then 10°C/min to 330°C for 4 minutes (run-length 36.5 minutes). Helium constant flow was 0.7 mL/min in splitless mode and 1 mL/min in split mode. Temperatures were as follows: injector = 250°C; transfer line = 300°C; source = 250°C, and quadripole = 150°C. Samples were randomized. An alkane mix (C10, C12, C15, C19, C22, C28, C32, and C36) was injected in the middle of the queue for external retention index calibration. Five scans per second were acquired.
Metabolomic Data Processing
Raw Agilent data files were converted to NetCDF format and analyzed with AMDIS 32. 17 A home AMDIS retention index/mass spectra library, built from the NIST, Golm, and Fiehn databases and standard compounds, was used for metabolite identification. Peak areas were then determined more precisely using the Target-Lynx software (Waters, Guyancourt, France) after conversion of the NetCDF file to MassLynx format. Metabolites were normalized to the ribitol internal standard and to dry weight. A subset of metabolites was quantified absolutely using a response coefficient in both injection modes to ensure the absence of saturation. Primary statistical analysis was done with Multiple Experiment Viewer version 4.8.1. 18 Hierarchical clustering and principal component analysis were done in order to identify possible outliers and to establish metabolic clusters. Kruskal–Wallis test was used to identify significant metabolites. For calculations, the median values among replicates were used. Spearman’s correlation was calculated in Statistica 10.0. For identification of relevant metabolic pathways, the Plant Metabolic Network database was used. 19 Metabolic scheme was prepared in CorelDraw ×8 and Morpheus software.
Results and Discussion
Phenotype of Irradiated Plants
All doses of γ-radiation led to a significant decrease in the mean root length (Figure 1A) in comparison to the nonirradiated plants. The rate of this decrease in the dose range 5 to 15 Gy remained constant, without significant deterioration along with dose increment. Irradiation of 5, 15, and 20 Gy significantly improved shoot lengths (Figure 1B). The application of the relatively high dose (100 Gy) to seeds led to significant inhibition of growth reflected as dramatically decreased linear length of organs (Figure 1A and B). Root (Figure 1C) and shoot weights (Figure 1D) changed in a biphasic manner, showing a significant increase of a shoot weight after γ-irradiation of seeds at 15 Gy.

Phenotypic measurements of control (0 Gy) and irradiated (5-100 Gy) plants. A, Root length (t test); (B) shoot length (t test); (C) root weight per 100 plants (U test); (D) shoot weight per 100 plants (U test); (E) root:shoot ratio, length; and (F) root:shoot ratio, weight.
An integrative physiological parameter, root:shoot (R/S) ratio, changed in a similar way for weight and length of irradiated plants (Figure 1E and F), being maximal at 100 Gy, close to nonirradiated control at 5 and 10 Gy, and significantly lower at 15 and 20 Gy, indicating an increase in shoot length and biomass. During the development of plant, there is a tendency to reduce R/S ratio. 20 This result is in a good agreement with our previous work, 15 where we hypothesized the forward developmental shift of plant irradiated in stimulating doses.
Previously it has been shown that nonlinear dose–response relationship is also valid for R/S ratio. 21 However, our results demonstrate that changes in length do not necessarily correlate with changes in weight (Figure 1E and F), which can be explained by cell expansion without induction of cell proliferation in irradiated barley plants. This indicates a necessity to consider a specificity of the end points used to assess hormetic effect.
General Metabolomic Changes in Irradiated Plants
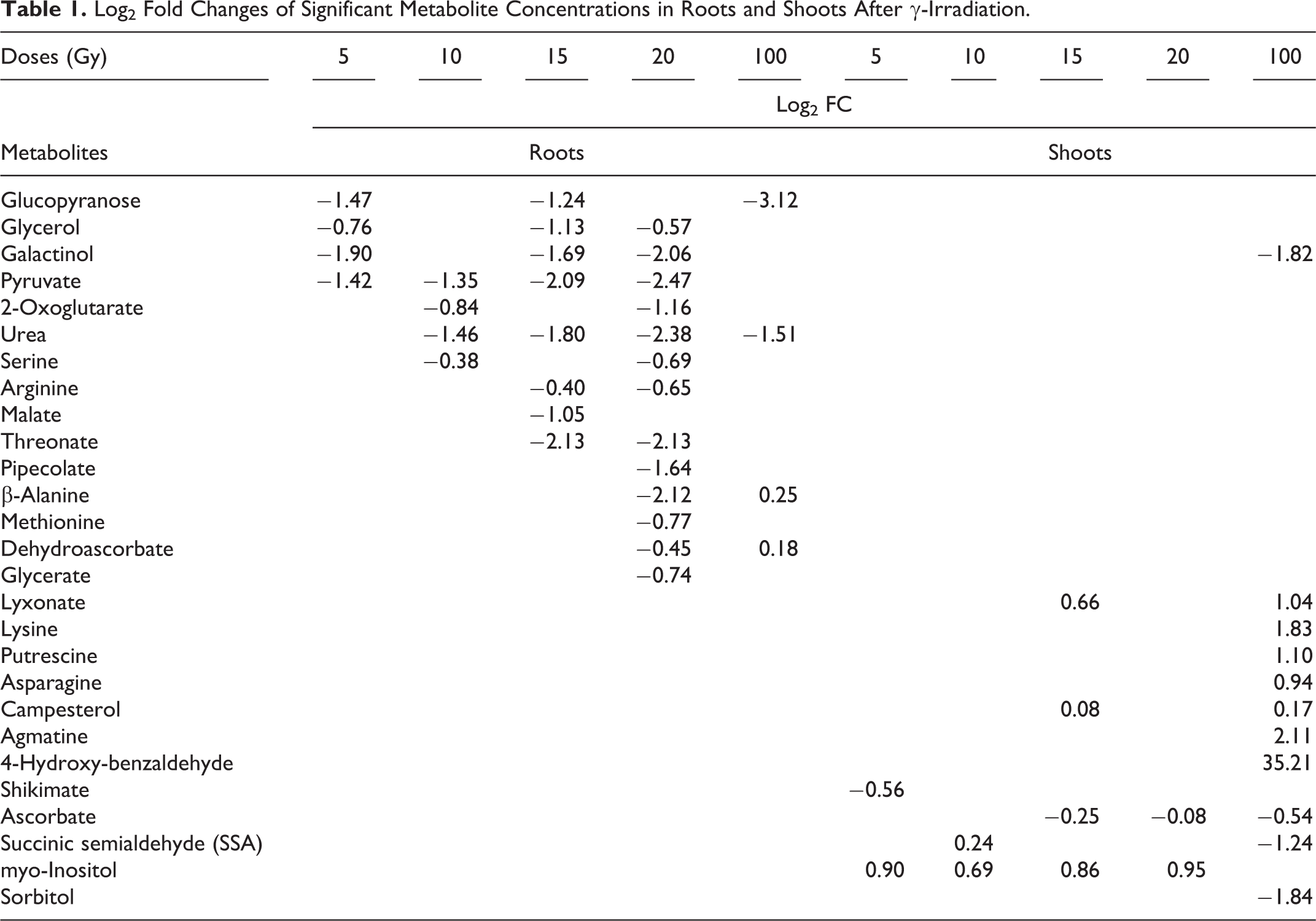
Log2 fold changes of metabolite concentrations are given in Table 1. Only metabolites that were considered significant after Kruskal–Wallis test were included in the subsequent presentation. Generally, significantly different metabolites in roots showed a decrease in concentration, while in shoots, an increase was observed. Except galactinol, no metabolite was significantly different from control in both shoots and roots samples. Among stimulating doses, 15 and 20 Gy were characterized by the highest number of significant changes in metabolite concentrations (Table 1). The simplified scheme of metabolic changes is given as a heat map (Figure 2). Table 2 represents significant correlations between radiation-induced growth changes and metabolite concentrations.
Log2 Fold Changes of Significant Metabolite Concentrations in Roots and Shoots After γ-Irradiation.

Schematic heat map reflecting interconnections between significant metabolites in roots and shoots.
Significant (P < .05) Correlations Between Radiation-Induced Growth Changes and Metabolite Concentrations.

Abbreviations: r, metabolite concentration in roots; s, metabolite concentration in shoots; SSA, succinic semialdehyde.
Nitrogen Metabolism in Seedlings Is Affected by Low-Dose Irradiation of Seeds
Disturbances in nitrogen metabolism under stress conditions are well-known consequences of stress exposure. 22 Under stress conditions, the number of specific nitrogen-containing compounds with diverse functions can accumulate in cells, including amino acids, amides, diamines, and polyamines. 23
In our data, only one significantly different from control metabolite showed correlation with the dose applied to seeds (Table 2). Concentrations of urea in roots decreased with the dose increment, suggesting the decrease of arginine catabolism intensity. 24 Among the 21 proteinogenic amino acids, arginine has the highest nitrogen to carbon ratio, which makes it especially suitable as a storage form of organic nitrogen. 25 The concentration of arginine was sufficiently increased in shoots, especially after irradiation at doses 15 and 20 Gy (Figure 2). Therefore, the data suggest radiation-induced redistribution of available nitrogen from roots to shoots.
2-Oxoglutarate concentrations were also decreased in roots (Figure 2), showing inverse correlation with the R/S ratio by length and direct correlation with the R/S ratio by weight, pointing to the longer and lighter roots when the concentrations are low (Table 2). 2-Oxoglutarate locates at the interface between C and N metabolisms and is essential for glutamine synthesis. A reversible enzymatic reaction involves the assimilation of ammonium into glutamine and the deamination of glutamine into 2-oxoglutarate and ammonium, 26 which also suggests nitrogen redistribution after irradiation due to the decrease of 2-oxoglutarate concentrations in the range of stimulating doses (10-20 Gy, Figure 2). The deamination of glutamine might probably be suppressed, while the metabolic route would go through arginine and γ-aminobutyric acid (GABA).
γ-Aminobutyric acid is also involved in the general nitrogen metabolism and in the storage and transportation of nitrogen. 27 During its catabolism, GABA is converted to succinic semialdehyde (SSA). In our data, SSA concentration was decreased in roots after irradiation in doses 10 to 15 Gy after 7 days of germination and increased in all other conditions (Figure 2), demonstrating opposite pattern with shoot concentrations and having negative correlation with root length (Table 2). It was hypothesized that the degradation of GABA could limit the accumulation of reactive oxygen species (ROS) under oxidative stress conditions. 27 Catabolism of GABA could be used to rapidly generate succinate and energy (as NADPH) via the TCA cycle after removal of the stress factor. 28 This suggests the role of GABA catabolism under radiation exposure as a possible source of energy and/or ROS production limiter. Interesting to note, the protective effect of the application of GABA or its isomer, β-aminobutyric acid, leads to enhance of abiotic stress tolerance in plants. 29
Shoot weight showed reverse correlation with the amount of asparagine in shoots (Table 2) that plays a primary role in nitrogen recycling, storage, and transport in developing and germinating seeds and in vegetative and senescence organs. 30 Smaller concentrations of asparagine in shoots can lead to bigger shoot biomass through more effective usage of nitrogen.
To sum up, several pieces of evidence point to effective nitrogen redistribution between its storage and transport molecules (arginine, glutamine, asparagine, and GABA), which in certain conditions can lead to phenotypic effects of radiation hormesis.
Stress-Responsive Metabolites After Low-Dose Irradiation
Excessive concentrations of ROS slightly exceeding the basal level may be beneficial in reaching the maximum capacity of the biological plasticity, while a high level of ROS beyond the limits of the biological plasticity is harmful to plants. 4 Our previous work showed the excess of hydrogen peroxide concentrations in seedlings of barley undergoing pre-sowing irradiation. 15 This excess of control level of ROS can be one of the sources of growth stimulation; however, the possible reaction of stress-responsive metabolites remained unclear.
Metabolomic analysis allowed revealing an excess of myo-inositol in shoots of irradiated plants (Table 1). An inverse correlation between R/S ratio (length) and the amount of myo-inositol in shoots (Table 2) suggests the occurrence of longer shoots when this compound accumulates. The important role of this compound in the synthesis of cell wall components and root elongation was previously described. 31 Myo-inositol-derived galactinol and associated raffinose-family oligosaccharides are emerging as antioxidants and putative signaling compounds too. 32 It is known that galactinol negatively regulates seed germination, 33,34 thus significantly lower concentrations of galactinol in roots (Table 1 and Figure 2) can reflect its role in faster development of irradiated seeds and eventually in growth stimulation. Similarly, root weight showed an inverse correlation with the amount of galactinol in roots (Table 2).
Concentrations of another stress-responsive metabolite, β-alanine, were sufficiently decreased in both shoots and roots of plant irradiated in stimulating doses (Figure 2). β-Alanine concentrations in roots inversely correlated with shoot length, suggesting the increase of shoot linear sizes, and directly correlated with both R/S ratios (Table 2). The mechanism of possible shoot growth stimulation during β-alanine catabolism, if any, remains unclear. There are several functions of β-alanine unique to plants. It is accumulated as a general stress response molecule involved in protecting plants from extreme temperature, hypoxia, drought, heavy metal shock, and some biotic stresses. 35 Indirectly, it serves as a precursor to the compounds pantothenate and CoA, which are involved in a variety of functions. 31 Thus, low concentrations of β-alanine can point to its usage as an available source of CoA, which is required for Krebs cycle functioning.
Root length was affected by decreased concentrations of lyxonate and ascorbate (AsA) in shoots (Table 2), suggesting the role of ascorbate catabolism in morphological changes after irradiation. Interestingly, high concentrations of ascorbate in roots of irradiated plants were accompanied by low concentrations of ascorbate in shoots (Figure 2). Overall, ascorbate in roots of irradiated plants was reduced faster through dehydroascorbate (DHA), while in shoots the degradation of ascorbate became irreversible through
Mitotic Index in Root Meristem of Irradiated Barley Seedlings in Different Days of Germination. 38

Glycerate concentrations decrease in roots of irradiated plants (Figure 2) can reflect the photosynthetic inhibition: less photorespiration and less glycerate. 42 Inhibition of photosynthetic processes in irradiated plants was also shown on the transcriptional level, 15 which may be connected with ROS overproduction. The decrease of malate in roots showed a correlation with increased root and shoot weights, and a similar relationship was found for pyruvate (Table 2). Pyruvate and malate are assimilatory products of active photosynthesis, 43 and their concentrations in leaves do not change significantly after low-dose irradiation and growing in the dark conditions. The decreased concentrations of these key metabolites can point to the successful and fast reallocation of available endosperm resources, leading to faster metabolism in irradiated plants and eventual growth stimulation.
Low level of glycerol-3-phosphate and its precursors may be the reason for low concentrations of pipecolic acid in roots (Figure 2), which is an osmoprotectant and known player in systemic acquired resistance induction. 44 Concentrations of pipecolate were decreased in plants irradiated in stimulating doses, while irradiation in inhibitory dose 100 Gy led to a dramatic increase of pipecolate concentration in shoots (Figure 2).
Metabolic Responses After Irradiation in Growth-Inhibiting Dose
Drastic changes were observed in seedlings after irradiation in inhibitory dose 100 Gy (Table 1). An increase or a decrease in concentrations of some metabolites under that dose was unique in comparison with other conditions used in the experiment. In roots, a significant decrease of urea and glucopyranose suggested deceleration of nitrogen metabolism and high rates of energy usage, probably for intensification of repairing processes.
In shoots, significant concentrations of 4-hydroxybenzaldehyde (4-HBA) were recorded, which is a product of shikimate metabolism. The accumulation of 4-HBA can lead to inhibition of GABA shunt enzymes, 45 which are involved in transamination of GABA and its conversion to SSA. Indeed, concentrations of SSA are significantly decreased in shoots of 100 Gy irradiated plants (Table 1). Low concentrations of sorbitol (Figure 2) in both shoots (source tissue) and roots (the sink tissue) 46 can point to significant lack of final products of photosynthesis in irradiated plants.
Conclusion
Irradiation of barley seeds in low doses can lead to growth stimulation of seedlings at certain dose points. Moderate damage allows mobilization of endosperm resources even under limited growth conditions. Our data suggest redistribution of available nitrogen among organs of irradiated plants and among metabolic processes. Arginine pathway seems to play an important role in this redistribution, leading to accumulation of nitrogen mainly in leaves. Ascorbate system, myo-inositol and its derivates, amino acids (
Footnotes
Authors’ Note
Volkova P. Yu. designed the experiment, analyzed metabolomics data, and wrote the article; Clement G. performed the GC-MS analysis, relevant data extraction, and primary data analysis; Makarenko E. S., Kazakova E. A., Bitarishvili S. V., and Lychenkova M. A. performed morphological assessments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by Russian Foundation for Basic Research, contract No. 19-04-00152.
