Abstract
The current study was planned to investigate the pharmacological basis of Solanum virginianum extract (SV.CR) pertaining to anxiolytic, antidepressant and memory-enhancing effects in rats. The SV.CR was analyzed in-vitro for phytoconstituents, antioxidant potential and anticholinesterase activity. The rats treated in a dose-dependent manner (25, 50 and 100 mg/kg of SV.CR) were subjected to behavioral tests for anxiety, depression and memory judgment followed by biochemical studies. A notable dose-dependent anxiolytic potential of SV.CR was observed in elevated plus maze and open field tests (P < 0.05). The decreased immobility time of the treated rats in the forced swim test (P < 0.01) unveiled the plant’s potential to reduce depression. Moreover, SV.CR treatment also reversed scopolamine-impaired cognition (P < 0.05) in various deployed memory and learning tasks. Biochemical studies of brain homogenates of SV.CR treated animals demonstrated decreased anticholinesterase activity and lipid peroxidation levels whereas increased levels of superoxide dismutase and glutathione peroxidase (P < 0.05 vs scopolamine group) were noted. The scientific validation of the study supported the use of Solanum virginianum in reducing anxiety, depression and amnesia in experimental models. Phytoconstituents in SV.CR such as oleanolic acid and caffeic acid might have played a significant neuroprotective role via modulation of oxidative stress and neurochemical aspects.
Keywords
Introduction
Neurological disorders have been imposing a huge burden on people’s health globally. Unfortunately, this delinquency is claimed to grow exponentially in developing countries during the next decade. 1 The etiology of brain diseases may include congenital origin 2 or acquired at some stage of life due to some chemical imbalance, infection, trauma, cerebral and vascular disarrays. 3 Among numerous brain diseases, Alzheimer’s disease (AD), anxiety and depression are reported to predominantly affecting individuals and their families. 4
Alzheimer’s disease (AD) is a prevailing illness presenting memory loss in elder individuals that has affected 24 million populace worldwide. 5 The disease encompasses an initial decline in cognition that gradually influences all intellectual functions resulting in an individual’s dependency on others for basic daily activities. 6 It involves progressive neurodegeneration resulting in dementia that is continuous and irrecoverable with behavioral fluctuations, disorientation and cortical dysfunction. 7 The pathophysiology includes amyloid beta and neurofibrillary tangle formation, mitochondrial dysfunction, a mutation in tau protein and elevated oxidative stress leading to brain shrinkage and cortical and hippocampus atrophy. 8 The deteriorated cholinergic neurotransmission is also crucial in the development of AD as evidence reveals that cholinergic neurons present in the amygdala, nucleus-basalis and neocortex are responsible for cognitive processes. 9 One of the pharmacological approaches to treat AD is to inhibit acetylcholinesterase (AChE) in order to recompense the damage of cholinergic neurons. 10 -12 Unfortunately, this therapeutic approach has certain limitations due to the shorter half-lives of most of the available anticholinesterase drugs and their usage is associated with several adverse effects. 13,14
Depression and anxiety are recurrent stress-related psychiatric illnesses that may cause life-long incapacity and early demise. 15 Approximately 20% of the global population becomes the victim of these psychological challenges at some point in their lifespan. 16 These 2 disorders were dealt and managed separately for decades using amine reuptake inhibitors for depression and benzodiazepines for anxiety. But in recent medical practices, the drugs to alleviate depression are slowly replacing benzodiazepines for the management of various anxiety disorders. 15,17,18 Benzodiazepines have undoubtedly been beneficial but their features of being sedative and muscle relaxant as well as the associated physical dependence make them less-preferred drugs. 19 Depression is resulted due to altered brain levels of monoamines, especially serotonin and norepinephrine, and their reuptake inhibitors are the treatment of choice. 20 Irrespective of the availability of different drugs for depression and anxiety, it is still a challenge to minimize the symptoms of these ailments to the fullest due to encountered adverse effects, less efficacy and poor tolerance. 21
Plant-based preparations have been tremendously adopted as an alternative option for the management of a range of health issues including neurological ones. 22 The researcher’s efforts on medicinal plants to discover new therapeutic molecules are continuously yielding evidence of their pharmacological usefulness in different animal models. 23 -25 The herbal remedies i.e. Ginkgo biloba, Grewia asiatica and Mentha piperita are reported to be more friendly due to cost-effectiveness, lesser side-effects and easy accessibility. 26 -28 Therefore, they have been opted for healthcare by 80% of the world’s population and people’s consistency with natural remedies has significantly increased over the past 3 decades. 29
Solanum virginianum, a wild eggplant bearing spines, commonly known as “Kantakari” belongs to the family “Solanaceae.” 30 It comprises numerous remedial constituents such as glycosides, anthraquinones, flavonoids, sterols, saponins and alkaloids. 31 This traditionally used medicinal herb has been reported to own cardio-protective, 32 hepatoprotective, 33 antidiabetic, 34 anti-asthmatic, 35 anti-inflammatory 36 and anti-cancer properties. 37
The current study was intended to explore pharmacologically important constituents in the methanolic extract of S. virginianum (SV.CR) through a set of in-vitro experimentation. Moreover, the ameliorating potential of a chronically administered SV.CR on anxiety, depression and scopolamine-induced amnesia was demonstrated by incorporating a range of behavioral studies. These behavioral outcomes were further authenticated by undertaking a series of biochemical studies on isolated brain tissues of animal model. We aimed to reveal the phytochemical and antioxidant characteristics of this plant to relate them with its capacity to ameliorate the anxiety, depression and cognitive deficit.
Materials and methods
Preparation of Plant Extract
All aerial parts of Solanum virginianum except fruit were gathered in May from Multan, Pakistan. The plant was verified by an expert taxonomist and a voucher R.R. Stewart 644 was issued. The collected parts of the plant were carefully cleaned to eradicate contaminants. After shade-drying for 2 weeks, coarsely ground powder was macerated in the methanol (80% v/v) in an amber-colored container. The macerated material was frequently shaken at room temperature for 1 week. This entire mixture was filtered and the whole procedure of maceration and filtration was done twice to allow the maximum extraction of constituents. The filtrate was then dried in a rotary evaporator and 14% extract was yielded. For in-vitro studies, the extract was dissolved in methanol and for in-vivo studies; the extract was dissolved in 0.9% saline.
Animals
Sprague Dawley male rats (150 to 250 g) were purchased from the National Institute of Health Islamabad and harbored in polycarbonate cages in the animal house of the Faculty of Pharmacy, B.Z University Multan, Pakistan under controlled temperature (23-25 °C) and humidity (50%) with a 12 h L/D cycle. All animals were supplied with standard rodent chow and water ad libitum. All behavioral experiments were carried out from 8:00 am to 6:00 pm. Before each behavioral experiment, the animals were acclimatized to the experimental room atmosphere and handling. All procedures of animal studies were in agreement with the Institute of Laboratory Animal Resources (ILAR), National Research Council (NRC, 1996) and approved by the Ethical Committee of B.Z.U, Multan, Pakistan (EC/04-PHL-2017 dated 20-03-2017).
Drugs and Chemical
Chemical with a high percentage of purity was used in the present study. Piracetam, scopolamine and most of the chemicals were purchased from Sigma-Aldrich, USA. The procurement of diazepam and fluoxetine was done from Roche Pharma (Switzerland) and Hilton Pharma (Pakistan) respectively. All pure available drugs were freshly dispensed in normal saline prior to the experiment.
In-Vitro Experiments
GC-MS analysis and compound identification
The methanolic extract of Solanum virginianum was subjected to phytochemical analysis through GC-MS (Turbomass, PerkinElmer, Inc., Waltham, MA, USA). The initial temperature was set to 40 °C for 2 min and gradually increased to 200°C at a rate of 5°C per minute and sustained for 2 min. Subsequently, the temperature was raised from 200 to 300°C in a similar fashion and kept for an additional 2 min. The eluted phytocomponents of test extract were determined by comparing their mass spectra with the reference mass spectra in the National Institute of Standard and Technology and WILEY Spectral libraries. The mass spectra of SV.CR compounds were also screened with the spectra similar compounds in the Adams Library (Adams, 2007) and the Wiley GC/MS Library. 38
HPTLC analysis of caffeic acid and oleanolic acid
The quantification of caffeic acid and oleanolic acid in SV.CR was done by using an NP-HPTLC plate (10 × 10 cm) (Merck, Germany). The stock solution of caffeic acid and oleanolic acid (1 mg/mL) were made using methanol and later diluted to 20 to 140 μg/mL solutions. All prepared dilutions of caffeic acid and oleanolic acid along with SV.CR (10 μL) were applied to the HPTLC plate through a microliter syringe connected with TLC Sampler-4 (CAMAG, Switzerland). A 6 mm wide band of each sample was applied at a speed of 160 nL/sec to provide a linearity range of 200-1400 ng/band. After application, the TLC plate was later developed in a pre-saturated twin-trough glass chamber (Automatic Development Chamber-2, CAMAG, Switzerland) at 25 ± 2°C temperature and 60% humidity. The developed HPTLC plate was first quantified at 254 nm wavelength to evaluate the presence of caffeic acid. The plate was further derivatized with vanillin-sulphuric acid to give clear and compact spots of oleanolic acid and quantitatively analyzed at 530 nm wavelength in absorbance mode.
Estimation of Total Phenolic and Flavonoid Contents of SV.CR
The Standard Folin-Ciocalteau method was used to estimate the total phenolic content of the SV.CR as described previously. 39 Briefly, 0.1 ml of Folin-Ciocalteau reagent (0.5 N) was added to the 0.5 ml of extract and mixed vigorously. Later, 2.5 ml of Sodium carbonate (Na2CO3) was added and the whole mixture was incubated for 30 min. The optical density of this solution was spectrophotometrically measured at 760 nm and total phenolic contents were expressed as mg gallic acid equivalents per gram of the extract.
For total flavonoid content, the standard aluminum chloride spectrophotometric method was used. 40 Briefly, 1 ml of SV.CR (1 mg/ml) was added to 10% AlCl3 and the contents were mixed thoroughly. After incubating for 30 min, the absorbance was measured at 415 nm. Flavonoid concentration in each sample was calculated through a standard calibration plot generated by known concentrations of quercetin and were expressed as mg quercetin equivalent unit per gram of the extract.
Antioxidant Activity
DPPH radical-scavenging activity
The antioxidative activity was estimated by DPPH (2, 2-diphenyl-1-picrylhydrazyl) method. 41 Various concentrations of SV.CR (10, 50, 100, 500 and 1000 μg/mL) were prepared. After that, 0.5 mL of each concentration was mixed with 0.125 mL DPPH and 0.375 mL methanol and incubated for 30 min. The optical density of each solution was measured at 517 nm using ascorbic acid as a positive control. Radical scavenging activity was calculated by the following formula:
ABTS radical cation scavenging activity
The antioxidative activity of SV.CR was further estimated by utilizing ABTS method 42 with minor modification. Aqueous solutions of ABTS (7 mmol/L) and potassium persulfate (2.45 mmol/L) were prepared. After incubating for 12 h in darkness, the 2 solutions were mixed and incubated for 30 min. Later, the mixture was kept in the refrigerator for 24 h and then diluted in ethanol. Variable concentrations of the extract (10–1000 μg/mL) were incubated with ABTS solution 50 µg/ml (1:1) in triplicates and the absorbance was measured at wavelength 734 nm employing ethanol (95%) as a blank while ascorbic acid as a standard. The antioxidant capacity of the extract was determined using the following formula. 43
Acetylcholinesterase (AChE) Inhibitory Activity
The SV.CR was evaluated for AChE inhibitory potential according to the method described. 44 250 µl of each plant extract (25-400 µg/ml) was mixed with 20 µl of DTNB (10 mM) and 10 µl 6.67 U/ml AChE in 1710 µl of 50 mM Tris-HCL buffer (pH 8.0). The absorbance of the resulting mixture was noted at 412 nm taking galantamine as a positive control. Later, 10 µl of ATC was added to the cuvette and the change in absorbance was noted for 5 min. The % inhibition was calculated by formula 45 :
Behavioral Studies
Animal grouping with drug treatments
The animals under study were randomly divided into different groups (n = 10). 1st group was considered as a negative control to which normal saline (1 ml/kg) was administered. Group 2nd-4th were designated as test groups receiving a dose of 25, 50 and 100 mg/kg of SV.CR (100 mg SV.CR dissolved in 1 ml normal saline) respectively. Animals of 1st-4th groups were treated with their specific treatments via oral gavage once a day for 20 consecutive days. The detailed animal grouping and designated treatments are depicted in Figure 1.

The detailed animal grouping (n = 10) and group-wise treatments.
For the behavioral tests for anxiety, 5th group (n = 10) was incorporated as a positive control to which diazepam (2 mg/kg, i.p.) was administered 60 min before the test. For the forced swimming test, the 5th group (n = 10) taken as a positive control was treated with fluoxetine (20 mg/kg, i.p.) for 1 week (17th–23 rd days). While for the behavioral tests for memory and learning, the 5th group (n = 10) reserved as a positive control was treated with piracetam (200 mg/kg, i.p.) for 1 week (20th–26th days). During behavioral tests for memory, animals of all 5 groups (piracetam, SV.CR 25, 50 and 100 mg/kg) were treated with scopolamine (1 mg/kg, i.p.) about 0.5 h before experimentation to induce amnesia and the outcomes were compared with another group (group 6) of animals (n = 10) receiving scopolamine (1 mg/kg i.p.) 0.5 h before experiment.
Behavioral Studies for Anxiety
Elevated plus maze (EPM) test
This is a behavioral study utilized broadly to evaluate the impact of test compounds on anxiety. 19,21 The test equipment consisted of open and closed arms constituting a plus-shaped maze (110 × 10 cm) which was lifted to a height of 50 cm. On the 21st day of study, rats were first treated with their specific treatments. After 1 h of drug treatments, they were tested individually for 5 min by placing in the middle of the maze while facing an open arm. Time spent in open and close arms were noted to reveal the anxiolytic potential of plant as an increased preference of exposed areas is considered as a sign of reduced anxiety in rodents. 15
Open field test (OFT)
This test is famous for the evaluation of the impact of test compounds on exploratory behavior and anxiety. 21,25 On the 22nd day of study, the test was conducted in an acrylic square-shape (80 × 80 cm) open field surrounded by the 40 cm high wall to avoid animal’s escape. After 60 min of specific treatments, rats were individually exposed to the arena of open field and their capability to explore the maze was recorded for 5 min. The number of entries and time spent in the center zone was noted for animals as the increased preferences for the central zone is an indicator of reduced anxiety. Besides these, the number of rearings and line crossings were also noted to depict the impact of SV.CR on animal locomotion.
Behavioral Studies for Depression
Forced swimming test (FST)
This test is widely utilized by scientists to evaluate the effect of test treatment on depression. 46 The test was conducted in a glass tank (34.5-23.5 cm) filled with water (25 °C) up to the level that the animal can float without touching its paws to the bottom. A forced swimming test was conducted on the 23 rd day of study. On test day, the animals of all groups were treated with their respective treatments an hour before experimentation taking fluoxetine as a positive control. Each rat was tested individually for 5 min by releasing in test apparatus and their movement was noted as decreased animal’s mobility is the sign of depression-like behavior in forced swimming test.
Behavioral Studies for Memory and Learning
After forced swimming test, the animals were given 2 days (25th-26th) as washout period to minimize any possible confounding impact of aversive stimuli (water) on the outcomes of next behavioral test.
Y-maze test
This test was conducted on the 27th day in an apparatus comprising 3 arms (A, B and C) arranged at 1200 constituting a Y-shaped maze (50 × 10 × 15 cm). Animals were first exposed to group-wise treatment and after 30 min, scopolamine was administered to induce amnesia. 47 Each rat was placed individually in any of 3 arms and allowed to explore the maze for 5 min. The sequence of arm visits (ABC, BAC, CAB etc.) was noted to calculate the spontaneous alternation (SAP) behavior of the animal. The spontaneous alternation behavior increases if the animal recalls the previously visited arm and prefers the new one and works as an indicator for his preserved memory. 48 % SAP is calculated through the formula given:
Novel object recognition (NOR) test
This test utilizes the rodent’s innate behavior of exploring the novel surroundings to assess his memory. 10,49 On the 28th day of study, the test was conducted in an acrylic field (80×80 cm) and the rodent’s inclination toward a novel object was taken as a characteristic of efficient memory. An hour before experimentation, each animal was first treated with specific treatment and after 30 min, amnesia was induced through scopolamine administration. The animal was first familiarized with the equipment atmosphere by placing it into the arena for 5 min. After that, 2 identical shaped objects were introduced in apparatus and animal was allowed to explore them for 10 min. Later, one of those objects was replaced by a novel object and animal’s interaction with old and new item was observed for 5 min. Discrimination index (DI) was calculated by under given formula to assess the memory and increased discrimination index depicts improved memory.
Passive avoidance test (PAT) (Step-through)
The test was conducted on 29th-30th days of study by using an apparatus that consisted of inter-connected light and dark compartments (20 × 20 × 20 cm). 50,51 The illuminated compartment was equipped with a light source (100 W) while the dark compartment was comprising a stainless steel made grid floor to deliver an electric shock of 0.5 mA (Coulbourn animal shocker) for 2 sec. The both compartments were connected through a manually operated door. Each rat was first positioned in an illuminated compartment from where he naturally tends to exit due to innate preference of dark places. On entering the dark compartment, he was treated with foot shock as an unfavorable stimulus to remember. After 30 min of specific treatment, scopolamine was given to rats to make them amnesic. Each rat was tested for 5 min by placing in an illuminated compartment after 1 h (acquisition trial) and 24 h (retention trial) of specific treatment to evaluate the impact of the test substance on learning and retention memory respectively.
Morris water maze (MWM) test
This behavioral test is extensively used to evaluate learning and memory including spatial and working memory. 52,53 The test apparatus comprised of a circular glass-fiber tank of 150 cm diameter and 50 cm height consisting a square platform (10 × 10 cm). The tank was divided into 4 poles (N, W, S, E) and 4 quadrants (NW, SW, SE, NE) in which proximal cues were displayed at the inner surface of the tank. 23,54 The distal cues were displayed exteriorly to the water tank and the arrangements was kept same throughout the experiment. The tank was filled with water (22-25 °C) opacified with some dye up to 32 cm and the platform (10×10 cm) was submerged 2 cm below the water surface. The platform was placed in the same quadrant throughout the experiment of 6 consecutive days (31st-36th days of study). During the first 2 days, rats were trained for 60 seconds twice daily with a platform in the tank. For the next 3 days, the experimental trials were conducted comprising 2 trials (with an interval of 30 min) each day in which each animal was allowed to locate the platform within 2 min. If he fails to find it in a specified time, he was slightly pushed toward the platform and permitted to stay there for 30 seconds. On the 6th day, the animals were tested without platform and their activity was recorded by Logitec HD camera to monitor their remembrance of the platform quadrant. The animals were administered with specific treatment an hour before first trial on each day while scopolamine was administered after 30 min of previous treatments.
Biochemical Analysis
Preparation of brain homogenate
After completion of the Morris water maze test, the randomly selected animals (n = 4) were decapitated after anesthetizing them with phenobarbital (50 mg/kg) and immediately brains were extracted on ice. The separated brains (cerebellum removed) were homogenized with phosphate buffer (pH 7.4; 0.3 g of the brain in 3 ml buffer) and centrifuged at 4 °C, 12000 rpm and for 15 min. The supernatant was separated and stored at −80 °C for further biochemical analysis.
Analysis of MDA, SOD, AChE and GPx
The resulting brain homogenate was evaluated according to a previously adopted method. 55 for the analysis of levels of malondialdehyde (MDA), superoxide dismutase (SOD), acetylcholinesterase and glutathione peroxidase (GPx).
Malondialdehyde Assay
100-500 µl of the homogenate was mixed with 2 mL mixture of 0.38% thiobarbituric acid and 15% trichloroacetic acid. The combined mixture was initially boiled for about 20 min followed by chilling it at a reduced temperature of 4 °C. The mixture was centrifuged at 3500 rpm for 15 minutes and absorbance was measured at 532 nm.
Superoxide Dismutase Assay
0.5 ml of brain homogenate was mixed with mixture comprising 24 µM NBT (0.4 mL), 50 mM sodium bicarbonate (1 mL), and 0.1 mM of EDTA (0.2 mL). Subsequently, 1 mM of hydroxylamine hydrochloride (0.4 mL) was added to initiate the reaction, and absorbance was monitored at 560 nm for 5 min.
Acetylcholinesterase Activity
0.4 mL of brain homogenate was combined with 100 µL DTNB and 2.6 mL of 0.1 M phosphate buffer having pH 8. The mixture was evaluated for absorbance at 412 nm followed by the addition of 5.2 µL ATC. The change in absorbance was noted for 10 min.
Glutathione Peroxidase Assay
The brain homogenate (0.3 mL) was mixed with 10 mM sodium azide (0.1 mL), 0.1 mL of H2O2 (0.1 mL), 0.1 M phosphate buffer of pH 7.4 (0.3 mL), and 2 mM reduced glutathione (0.2 mL). After incubation for 0.25 h at 37 °C, 5% TCA (0.5 mL) was added to stop the reaction. The mixture was centrifuged at 1500 rpm for 5 min followed by the removal of the upper layer. The 0.1 mL of the separated layer was reacted with 0.7 mL of DTNB and 0.2 mL of Phosphate buffer and absorbance was measured at 420 nm.
Statistical Analysis
All data were interpreted in terms of mean ± SEM. The data’s normality was first checked by using Shapiro-Wilk test using Graph Pad Prism (version 6). One-way ANOVA followed by a post-Dunnett’s test was employed for analysis of all test parameters. The 2-way ANOVA followed by a Tukey’s multiple comparison test was applied for the evaluation of escape latencies, step-through latencies and time spent in light (parameters for MWM and PAT). P ≤ 0.05 was considered to be statistically significant.
Results
In-Vitro Studies
GC-MS analysis for phytoconstituents
GC-MS analysis identified 17 different phytocompounds in S. virginianum extract which are 98.99% of the total extract. The retention time, molecular formula, molecular weight and concentration (Area %) of these phytocompounds are presented in Table 1 and Figure 2A. Phytol (25.5%), Hexadecyl acrylate (25.1%) and P-cresl octanoate (24%) were the primary constituents of this extract Figure 2B.
Phytoconstituents Identified in Methanolic Extract of S. virginianum Through GC-MS.

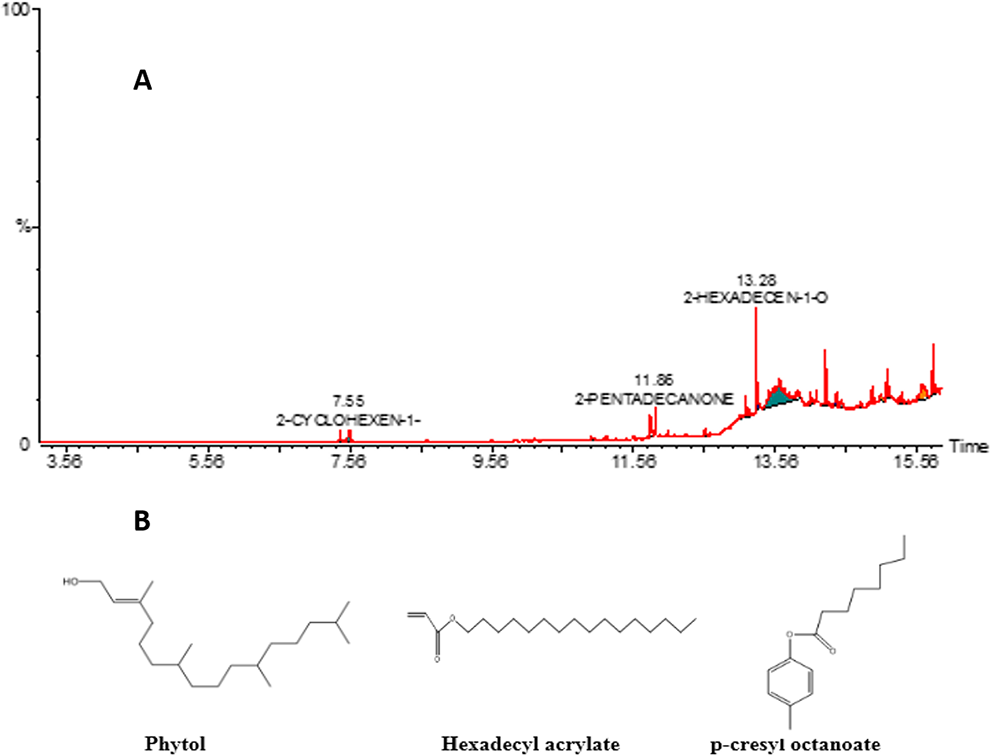
GC-MS analysis of methanolic extract of Solanum virginianum. (A) GC-MS chromatogram and (B) major constituents identified in SV.CR.
Analysis of caffeic acid and oleanolic acid in SV.CR by HPTLC method
The mobile phase to be used in HPTLC for appropriate quantification of caffeic acid and oleanolic acid in Solanum virginianum extract was chosen after trying several combinations of solvents. Among all those, the mobile phase comprising toluene, ethyl acetate and formic acid (7: 3: 0.1; v/v/v) was found most suitable. It furnished strong peaks of caffeic acid and oleanolic acid at Rf = 0.27 and 0.67, respectively (Figure 3A and B) and separated the standards as well as different phytoconstituents of S. virginianum very clearly (Figure 3C). The regression equation (Y)/correlation coefficient (r 2 ) for caffeic acid and oleanolic acid were found as 5.899X+1161/0.9978 and 5.380X+256.59/0.9993, respectively as shown in (Figure 3D). 3D of all tracks at 254 nm is shown in (Figure 3E). The outcomes revealed the presence of caffeic acid and oleanolic acid as 2.1 mg/g and 5.2 mg/g of dried weight of extract respectively.

Chromatogram of standards (A) caffeic acid (Rf = 0.27 ± 0.002) and (B) oleanolic acid (Rf = 0.67 ± 0.001) [mobile phase: toluene: ethyl acetate: formic acid (7:3:0.1, V/V/V)] (C) and chromatogram of estimated caffeic acid and oleanolic acid in S. virginianum hydroalcoholic extract (D). Standard calibration curve for caffeic acid and oleanolic acid (E) 3D spectrum of all tracks at 254 nm.
Total Phenol and Flavonoid Content
The evaluation of the total flavonoid content of SV.CR revealed the presence of 22.6 ± 4.3 mg QU/g total flavonoid content and 80.9 ± 6.4 mg gallic acid/g of total phenolic content in the methanolic extract of S. virginianum (Figure 4).

Total flavonoid content (TFC) and total phenolic content (TPC) of methanolic extract of S. virginianum.
Antioxidant Activity by DPPH and ABTS Method
S. virginianum showed dose-dependent scavenging activities with IC50 455 µg/ml in the DPPH scavenging activity test and IC50 631 µg/ml in the ABTS test as presented in Table 2.
% Scavenging Activities of S. virginianum Extract.

AChE Inhibitory Activity
The SV.CR demonstrated AChE inhibitory potential with IC50 of 386.25 ± 24.21 µg/ml and % inhibition was increased with concentration. The maximum enzyme inhibition (61.13 ± 1.15) was observed at the highest dose employed (400 µg/ml) when compared to galantamine as standard as shown in Table 3.
% Acetycholinesterase Inhibitory Activity of SV.CR With IC50 Values.

Behavioral Studies for Anxiety
Elevated plus maze test
The animals treated with different doses of SV.CR (25, 50 and 100 mg/kg) were exposed to the arena of the elevated plus maze. The outcomes revealed the significant inter-group differences for all observed parameters. The overall assessment exposed that SV.CR treated rats frequently visited open arms [F(4,35) = 16.75, P < 0.0001] and spent more time there [F(4,35) = 16.79, P < 0.0001] as shown in Figure 5A and C. Furthermore, the SV.CR treated animal was lesser anxious as evident from reduced entries [F(4,35) = 8.18, P < 0.01] and shorter duration of stay [F(4,35) = 8.35, P < 0.001] in the closed arms of the maze (Figure 5B and D, respectively). Furthermore, The post-hoc test revealed that animals treated with 50 and 100 mg/kg of SV.CR had reduced anxiety (P < 0.05) as compared to the control group but the animals treated with 25 mg/kg did not show any significant anxiolytic outcomes.
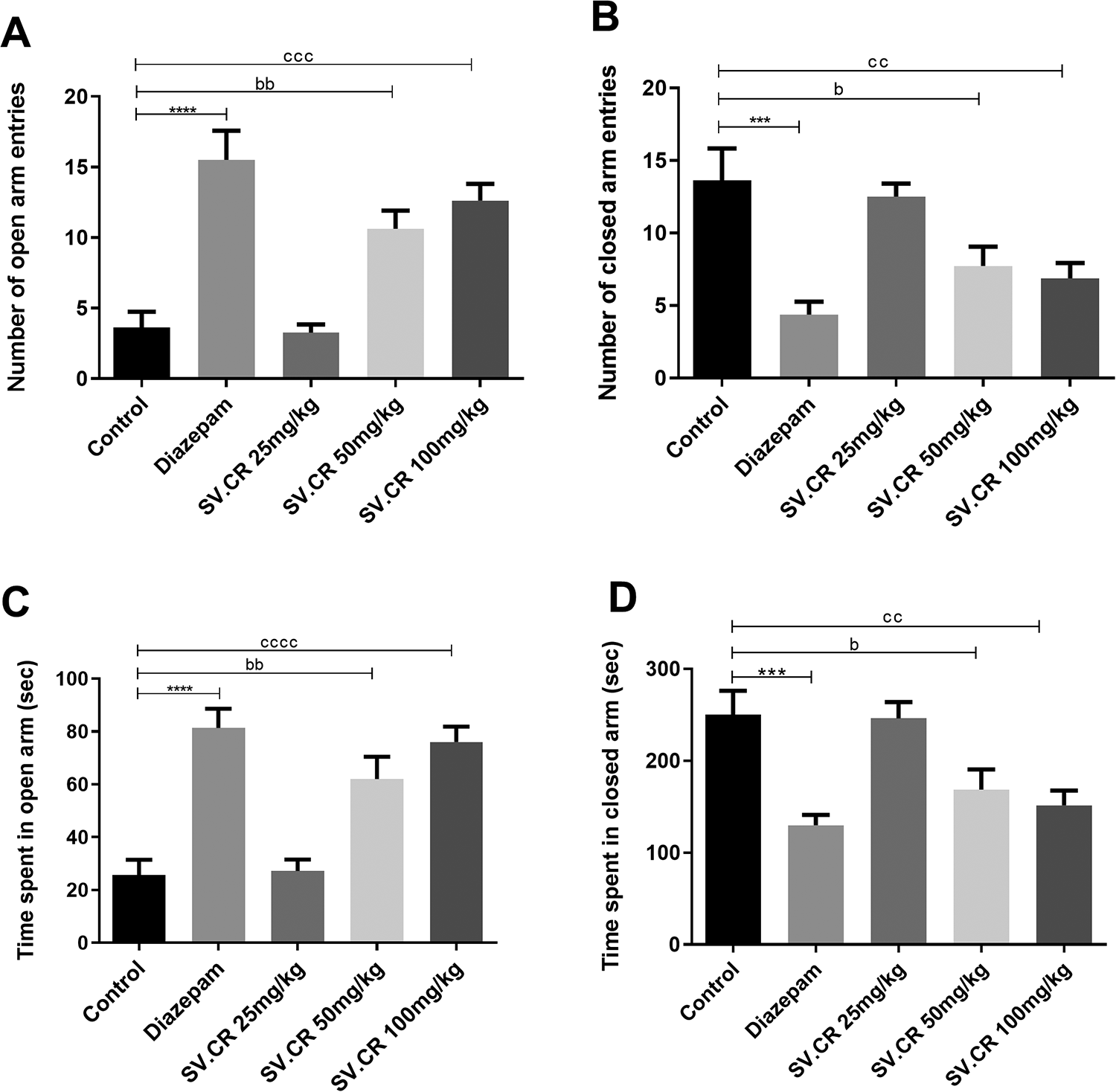
Effect of SV.CR on the assessment of anxiety in rats. In this experiment, the rats treated with 25, 50 and 100 mg/kg of SV.CR were observed for behavioral anxiety by elevated plus maze test for 5 min. The (A) number of open arm entries, (B) number of closed arm entries, (C) time spent in open arms and (D) time spent in closed arms were determined and compared with control, using diazepam as standard. Data is evaluated by one-way ANOVA followed by Dunnett’s test and whole data is expressed as mean ± SEM (n = 8). ***P < 0.001,****P < 0.0001 comparison between control and diazepam groups, bP < 0.05, bbP < 0.01 comparison between control and 50 mg/kg SV.CR treated animals, ccP < 0.01, cccP < 0.001 comparison between control and 100 mg/kg SV.CR treated animals.
Open field test
On the 22nd day, the animals were tested in an open field after 60 min of administration of the dose of SV.CR (25, 50 and 100 mg/kg) and a single dose of diazepam to rats (positive control group). There was significant differences among all groups for number of entries in open areas [F(4,35) = 15.20, P < 0.0001] (Figure 6A) and duration of stay there [F(4,35) = 27.57, P < 0.0001] (Figure 6B). The post-hoc evaluation demonstrated that the administration of SV.CR induced a dose-dependent reduction in animal’s anxiety toward an open area. The animals treated with SV.CR showed an increased preference for the central zone at 50 (P < 0.05) and 100 mg/kg (P < 0.01) as compared to the control group.

Effect of SV.CR on the assessment of anxiety in rats. The rats treated with 25, 50 and 100 mg/kg of SV.CR were observed for behavioral anxiety by open field test for 5 min. The (A) number of center zone entries, (B) time in center zone, (C) number of rearings and (D) number of line crossings were monitored and compared to control, using diazepam as standard. Data is evaluated by one-way ANOVA followed by Dunnett’s test and whole data is expressed as mean ± SEM (n = 8). **P < 0.01,****P < 0.0001 comparison between control and diazepam groups, bP < 0.05, comparison between control and 50 mg/kg SV.CR treated animals, cP < 0.05, ccP < 0.01, cccP < 0.001 comparison between control and 100 mg/kg SV.CR treated animals.
The number of rearings and line crossings were also varied among groups significantly with [F(4,35) = 16.26, P < 0.0001] and [F(4,35) = 13.18, P < 0.0001] respectively. The administration of SV.CR (50 and 100 mg/kg) led to an increased number of rearings (P < 0.05) and line crossings (P < 0.05) as compared to the control group as depicted in Figure 6C and 6D respectively. Though, these parameters were dominantly decreased (P < 0.01) in animals treated with diazepam.
Behavioral Studies for Depression
Forced swimming test
The animals treated with SV.CR were forced to swim to expose any anti-depressant activity of test plant using fluoxetine as a positive control. The One-way ANOVA described significant inter-group variation for immobility time [F(4,25) = 7.99, P < 0.001] (Figure 7). The comparison of SV.CR treated groups with control showed prominent anti-depressant activity at 50 (P < 0.01) and 100 mg/kg (P < 0.001) with no beneficial outcomes at 25 mg/kg. The rats were less immobile after plant’s treatment and their behavior was comparable to the fluoxetine treated animals.
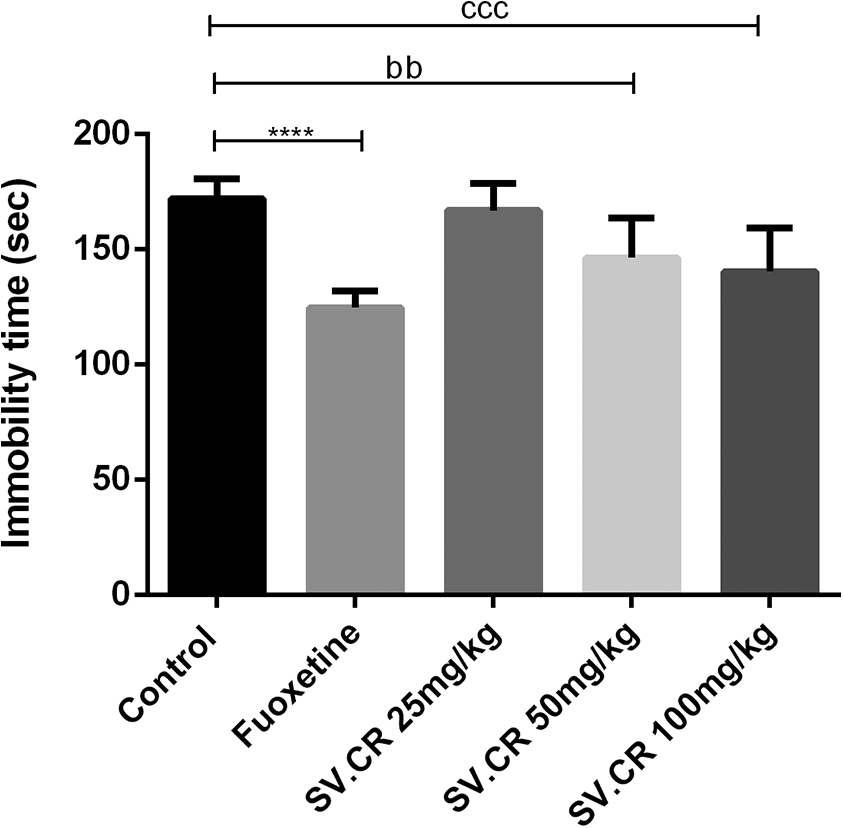
Effect of SV.CR on depression in rats. The rats treated with 25, 50 and 100 mg/kg of SV.CR were observed for depression by forced swimming test for 5 min. The immobility time was noted and compared to control, using fluoxetine as standard. Data is evaluated by one-way ANOVA followed by Dunnett’s test and whole data is expressed as mean ± SEM (n = 8). ****P < 0.0001 comparison between control and fluoxetine group, bbP < 0.01 comparison between control and 50 mg/kg SV.CR treated animals, cccP < 0.001 comparison between control and 100 mg/kg SV.CR treated animals.
Behavioral Studies for Learning and Memory
Y-maze test
The impact of SV.CR on spontaneous alternation behavior was estimated in amnesic rats by exposing them to Y-maze on the 27th day. The One-way ANOVA revealed a significant overall variation of %SAP between all treatment groups [F(5,42) = 9.14, P < 0.0001]. The post-hoc test disclosed that the administration of scopolamine led to a prominent decline in memory. The scopolamine-treated rats had reduced remembrance of the previously visited arm as compared to the control group as evident from the declined %SAP (P < 0.001). However, animals treated with 50 and 100 mg/kg of SV.CR exhibited increased inclination toward new arms of maze. Hence, the amnesia induced by scopolamine was significantly corrected back in animals treated with 50 (P < 0.05) and 100 mg/kg (P < 0.001) of SV.CR the way as piracetam did (Figure 8). Furthermore, the animal’s behavior was still amnesic at 25 mg/kg unfolding the dose-dependent memory-improving influence of Solanum virginianum.

Effect of SV.CR on memory and learning in rats. The rats treated with 25, 50 and 100 mg/kg of SV.CR were evaluated for memory through y-maze test for 5 min. The % spontaneous alternation behavior was calculated and compared to the scopolamine group, using piracetam as standard. Data is evaluated by one-way ANOVA followed by Dunnett’s test and whole data is expressed as mean ± SEM (n = 8). ***P < 0.001 comparison between control and scopolamine treated groups, ####P < 0.0001 comparison between scopolamine and piracetam treated groups, bP < 0.01 comparison between scopolamine and 50 mg/kg SV.CR treated animals, cccP < 0.001 comparison between scopolamine and 100 mg/kg SV.CR treated animals.
Novel object recognition test
The animals were tested for their capacity to discriminate the novel object from older ones on 28th day of study to estimate the memory-improving potential of plant. The outcomes demonstrated that the animals from all treatment groups had significantly different discrimination index [F(5,42) = 8.28, P < 0.0001]. The treatment with scopolamine brought memory deficit in rats as the animal became less capable to identify the novel object as compared to the control group (P < 0.01). However, the treatment with SV.CR was dose-dependently efficient in retaining the animal’s memory as they demonstrated enough recognition of familiarized objects. The amnesic influence of scopolamine was noticeably reversed by SV.CR at the dose of 50 and 100 mg/kg with P < 0.05 and P < 0.01 respectively and outcomes were comparable to those yielded by piracetam (Figure 9A). These SV.CR treated animals were also noted for enhanced time spent in exploring the novel object as compared to the amnesic rats. This parameter was also improved in animals treated with higher doses of SV.CR (P < 0.05) and animal remained distracted at 25 mg/kg (Figure 9B).

Effect of SV.CR on memory and learning in rats. The rats treated with 25, 50 and 100 mg/kg of SV.CR were evaluated for memory through novel object recognition test for 5 min. The animal’s interaction with new and old objects was monitored for 5 min to calculate the (A) discrimination index and (B) time spent with novel object behavior as compared to scopolamine group, using piracetam as standard. Data is evaluated by one-way ANOVA followed by Dunnett’s test and whole data is expressed as mean ± SEM (n = 8). *P < 0.05, **P < 0.01 comparison between control and scopolamine treated groups, ###P < 0.001 comparison between scopolamine and piracetam treated groups, bP < 0.05 comparison between scopolamine and 50 mg/kg SV.CR treated animals, cP < 0.05, ccP < 0.01 comparison between scopolamine and 100 mg/kg SV.CR treated animals.
Passive avoidance test
The results of the passive avoidance test demonstrated the memory-enhancing capacity of SV.CR. The 2-way ANOVA revealed a significant overall difference in step-through latencies among all groups [F(5,42) = 132.6, P < 0.0001]. The scopolamine treated animals presented declined latencies to enter the dark compartment (P < 0.0001) as compared to the control group in both acquisition and retention trials which reveals the memory deterioration induced by scopolamine. However, the administration of piracetam and SV.CR (50 and 100 mg/kg) worked to enhance animal learning (acquisition trial) and memory retention (retention trial) after 1 and 24 h of shock stimuli. These animals had better rememberance of aversive stimuli delivered in the dark compartment. The animals treated with 25mg/kg showed poor remembrance of foot shock and behaved the was as a scopolamine-treated animal did. Anyhow, the step-through latencies were prominently increased in rats treated with extract at 50 (P < 0.05) and 100 mg/kg (P < 0.01) (Figure 10A). Similarly, the time spent in the light compartment was also increased by scopolamine-amnesic animals on SV.CR (50 and 100 mg/kg) treatment with P < 0.01 and P < 0.001 (Figure 10B).

Effect of SV.CR on memory and learning in rats. The rats treated with 25, 50 and 100 mg/kg of SV.CR were evaluated for memory through a passive avoidance test for 5 min after 1 (acquisition trial) and 24 hrs (retention trial). The (A) step-through latency and (B) time spent in light compartment were monitored and compared to scopolamine group, using piracetam as standard. Data is evaluated by 2-way ANOVA followed by Tukey’s test and whole data is expressed as mean ± SEM (n = 8). ****P < 0.0001 comparison between control and scopolamine treated groups, ####P < 0.0001 comparison between scopolamine and piracetam treated groups, bP < 0.05, bbP < 0.01, bbbP < 0.001, bbbbP < 0.0001 comparison between scopolamine and 50 mg/kg SV.CR treated animals, ccP < 0.01, cccP < 0.001, ccccP < 0.0001 comparison between scopolamine and 100 mg/kg SV.CR treated animals.
Morris water maze test
Morris water maze test was employed to evaluate the impact of plant extract on spatial learning and memory in rats. During 3 consecutive training days, the animals of the control group successfully located the hidden platform. But, the administration of scopolamine led to amnesia resulting in poor navigation and thigmotaxic behavior in rats as evident from their longer escape latencies [F(1,14) = 782.1, P < 0.0001] as compared to the control group . However, the piracetam treated rats demonstrated a significant reversal of this memory deterioration [F(1,14) = 1205, P < 0.0001] as the animal was efficient enough to locate the hidden platform. Similarly, the animals treated with 50 and 100 mg/kg were also quick in locating the platform as evident from the dose-dependent decline in escape latencies depicted in tracings as well (Figure 11). The 2-way ANOVA revealed the significant difference of escape latencies among scopolamine vs SV.CR 50 [F(1,14) = 82.04, P < 0.0001] and 100 mg/kg and [F(1,14) = 157.8, P < 0.0001] treated groups in a dose-dependent manner (Figure 11A). On probe day, the animals were monitored for their remembrance of the platform quadrant by monitoring their entries in that quadrant and time spent there. The scopolamine-treated rats had poor memory as they presented minimum partiality for the platform-stationed quadrant by showing a decreased number of entries (P < 0.01) and duration of stay (P < 0.001) there as compared to control animals. However, theses outcomes were significantly reversed in SV.CR (50 and 100 mg/kg) treated rats as they depicted an increased number of entries (P < 0.05) and prolonged duration of swimming (P < 0.05) in the quadrant where the platform was located previously as shown in Figure 11B and C, respectively. However, these results were found nonsignificant at 25 mg/kg of SV.CR.

Effect of SV.CR on memory and learning in rats. The cognition of rats treated with 25, 50 and 100 mg/kg of SV.CR was evaluated for 3 consecutive days by monitoring the capability to locate the hidden platform in 2 min. The (A) animal’s escape latencies with tracings, (B) time spent in platform zone and (C) time spent in platform zone were noted and compared to the scopolamine group, using piracetam as standard. Data for escape latencies is evaluated by 2-way ANOVA followed by Tukey’s test. While one-way ANOVA followed by Dunnett’s test was used to evaluate other mentioned parameters and whole data is expressed as mean ± SEM (n = 8). **P < 0.01, ***P < 0.001, ****P < 0.0001 comparison between control and scopolamine treated groups, ###P < 0.001 ####P < 0.0001 comparison between scopolamine and piracetam treated groups, bP < 0.05, bbbP < 0.001, bbbbP < 0.0001 comparison between scopolamine and 50 mg/kg SV.CR treated animals, cccP < 0.001, ccccP < 0.0001 comparison between scopolamine and 100 mg/kg SV.CR treated animals.
Biochemical Analysis
Acetylcholinesterase assay
The ANOVA revealed a significant difference [F(5,18) = 58.71, P < 0.0001] of enzyme inhibition capacity among all evaluated groups as shown in Figure 12A. The post-hoc test described the enzyme levels were significantly higher (P < 0.0001) in rats exposed to scopolamine. However, these elevated AChE levels were normalized by piracetam (P < 0.0001). Similarly, the plant extract treated rats demonstrated a significant reduction in levels of acetylcholinesterase at a dose of 50 (P < 0.001) and 100 mg/kg (P < 0.0001).

Biochemical analysis showing levels of (A) acetylcholinesterase, (B) superoxide dismutase, (C) glutathione peroxidase and (D) malondialdehyde in brain homogenates. Data is evaluated by one-way ANOVA followed with post-hoc Dunnett’s test and whole data is expressed as mean ± SEM (n = 4). **P < 0.01, ***P < 0.001, ****P < 0.0001 comparison between control and scopolamine treated groups, ###P < 0.001, ####P < 0.0001 comparison between scopolamine and piracetam treated groups, bP < 0.05, bbP < 0.01, bbbP < 0.001 comparison between scopolamine and 50 mg/kg SV.CR treated animals, cP < 0.05, ccP < 0.01, cccP < 0.001, ccccP < 0.0001 comparison between scopolamine and 100 mg/kg SV.CR treated animals.
Superoxide dismutase assay
The assay was performed to evaluate the levels of superoxide dismutase. It is an important enzyme that combats the reactive oxygen species and provides protection against various diseases. The results described the significant intergroup difference [F(5,18) = 28.65, P < 0.0001] with a prominent enzyme reduction in scopolamine treated rats (P < 0.001). This deterioration was significantly reversed by piracetam (P < 0.0001) and SV.CR (P < 0.05) (Figure 12B).
Glutathione peroxidase assay
Glutathione peroxidase is an endogenously present defense enzyme that catalyzes the reduction of harmful peroxides to friendly products. The one-way ANOVA depicted a notable variation in enzyme levels after different treatments [F(5,18) = 13.4, P < 0.0001]. The levels of GPx were significantly reduced due to scopolamine treatment showing the increased oxidative stress due to scopolamine (P < 0.001). The SV.CR at dose 50 and 100 mg/kg was efficient enough to elevate the enzyme levels with P < 0.01 (Figure 12C).
Malondialdehyde assay
Malondialdehyde is a marker of lipid peroxidation due to oxidative stress. The ANOVA revealed a significant difference in MDA levels among all groups [F(5,18) = 11.63, P < 0.0001]. The scopolamine led to increased oxidative stress that is indicated in results through elevated MDA levels (P < 0.01). This lipid peroxidation was significantly reduced by piracetam (P < 0.001) and SV.CR at dose 50 (P < 0.05) and 100 mg/kg (P < 0.05) as depicted in Figure 12D.
Discussion
The inhabitants of emerging countries like Pakistan are increasingly trusting on natural resources for a range of ailments. 29 People are showing more interest in natural remedies due to their immediate accessibility, safety profile and economic aspects. 56 The various phytoconstituents owned by plants are known to exert remedial properties for different health complaints including neurological disorders.
In the present study, the methanol extract of the whole plant of Solanum virginianum was initially explored for phytochemical constituents through different in-vitro studies. Terpenoids (Phytol, carvone, Tetracosane, etc.) were among the major constituents detected in the methanolic extract of Solanum virginianum through GC-MS analysis. Terpenoids are previously reported to possess multi-faceted neurological properties because of their antioxidant properties. 57 Phytols, the major detected terpenoids, are testified to exert prominent antioxidant action by activating superoxide dismutase and restoring the glutathione metabolizing enzyme system. Furthermore, recently it has been reported that phytol, can mitigate the scopolamine-induced cognitive deficit by repressing the activity of cholinesterases in the brains of Wistar rats. 58 Similarly, the outcomes of the HPTLC analysis of SV.CR confirmed the presence of oleanolic acid and caffeic acid in the plant. Oleanolic acid (OA), a pentacyclic oleanane triterpenoid, 59 is endowed with the extraordinary antioxidant potential through direct radical-scavenging as well as indirect biological effects. 60,61 Oleanolic acid has been reported to activate Nrf2 protein that is an important cellular defense mechanism against oxidative stress. 61 Under increased oxidative stress, Nrf2 translocates to the nucleus and induces the transcription of various cellular defense genes including Glutathione peroxidase. 62
Similarly, the other detected phytomolecule, caffeic acid (CA) is a polyphenol that is reported to interrupt the development of free radicals (primary antioxidant) as well as metal-chelating agent thus inhibiting the decomposition of peroxides free radicals (secondary antioxidant). It comprises a catechol group with an unsaturated carboxylic group that interacts with oxidizing species. 63
Additionally, the total phenolic and flavonoid content estimation testing revealed the presence of flavonoids and phenols in SV.CR. It is well-established that these constituents are strongly linked to the antioxidant characteristics as they are known to play role in radical scavenging. 64 These antioxidant characteristics were authenticated through DPPH and ABTS assays where the SV.CR demonstrated concentration-dependent radical scavenging potential. These characteristics might be attributed to the enrichment of the plant with oleanolic acid, caffeic acid, high phenolic content and flavonoids.
For possible neuromodulatory potential, the Sprague Dawley rats were chronically administered with SV.CR for 20 days after which they were undergone to a series of behavioral experiments for anxiety, depression and memory. The animals were tested for anxiety through EPM and OFT in which they showed a dose-dependent anxiolytic effect of SV.CR. The animals treated with 50 and 100 mg/kg were daring enough to visit open arms and central area in the elevated plus maze and open field respectively and conclusions were analogous to diazepam. However, the results of the number of rearings and the number of line crossings were in contradiction with the outcomes of diazepam. The number of rearing (index of vertical motion) and the number of line crossings (index of horizontal motion) were decreased in animals treated with diazepam revealing its sedative properties. But, these parameters were significantly improved by SV.CR revealing its anxiolytic activity with CNS stimulating effects. 15 The anxiolytics are known to enhance GABAergic neuro-inhibition through the opening of chloride channels. The flavonoids are the phytoconstituents known to induce neuronal inhibitory impact through modulation of GABA levels in the brain. 65
The animals were examined for depression through a forced swimming test using fluoxetine, a selective serotonin reuptake inhibitor, as a positive control. The animals treated with SV.CR (50 and 100 mg/kg) showed decreased immobility, a generalized accepted parameter of FST for the assessment of antidepressant effect. The ample flavonoid content in SV.CR may be linked to this anti-depressant potential of the plant. Flavonoids are compounds reported to improve brain-derived neurotrophic factor (BDNF) levels in the brain 66 which is reported to decline in depressed patients. 67 BDNF is also reported to play a critical role in synaptic and structural plasticity that if compromised are allied with depression. 68 Additionally, flavonoids also play a key role in the management of depression by inhibiting the monoamine oxidases thus elevating monoamines in the brain. 69
To explore the impact of SV.CR on animal’s learning and memory behavior, the scopolamine-amnesic rats were tested in y-maze, novel object recognition test, passive-avoidance test and morris water maze test. Scopolamine is known to mimic dementia seen in AD by crossing the blood-brain barrier (BBB) quickly. It leads to the weakening of cholinergic neurotransmission that is the characteristics of age-related memory impairment. 70 The outcomes of all experiments were found promising in reversing the scopolamine-induced memory impairment. The SV.CR showed a dose-dependent increase in spontaneous alternation and discrimination index on y-maze and NOR tests respectively. The scopolamine-increased step-through and escape latencies were also significantly reversed by SV.CR (50 and 100 mg/kg). The plant-treated animals depicted improved remembrance of the platform quadrant on probe day in MWM as their number of entries and stay was enhanced as compared to scopolamine-treated rats. The in-vitro anti-acetylcholinesterase assay in the present study also demonstrated the enzyme inhibitory activity of SV.CR predicting that may act to increase in brain acetylcholine thus improving cognition and memory. 9 Polyphenols are reported to possess anticholinesterase activity that makes them beneficial in the alleviation of disorders resulting from Ach deficiency. Due to this property of polyphenols, these have been reported to be useful in the treatment of Alzheimer’s disease. 71 This anticholinesterase capacity of the plant was further confirmed on assaying the enzyme inhibitor activity of SV.CR on isolated brain homogenates at the end of behavioral experiments.
Polyphenols are prevalent phytoconstituents that are believed to possess extraordinary health benefits. 72 The polyphenols and their metabolites can modulate various enzymes critical for signal transduction i.e. protein kinase and lipid kinase pathway. 73 They exert a neuroprotective effect by modulating various signaling pathways crucial for neuronal survival 74 as well as by hindering the neuropathological events. 75 They also play role in regulating the cerebrovascular blood flow that ultimately affects synaptic plasticity and improves cognition. 76 The phenols and flavonoids can also exert neuroprotection by activating caspase-3 that imparts anti-apoptotic effects. 77 Moreover, they encourage neuronal proliferation by promoting the liberation of various neurotrophic factors that are beneficial for hippocampus neurogenesis. 78
Researchers have revealed the ameliorative impact of oleanolic acid in the scopolamine-amnesic mice model. 79 The researchers claimed that OA reversed memory impairment through TrkB (tyrosine kinase B) activation and modulation BDNF-ERK1/2-CREB pathway. BDNF (brain-derived neurotrophic factor) is known to play a critical role in regulating synaptic structure and function through TrkB. 80 The augmented BDNF/TrkB leads to activation CREB (cAMP response element-binding protein) that is a well-known regulator of cognition-linked gene expression in the hippocampus. 81 So, these vital phytoconstituents owned by S. virginianum might be responsible for observed neuropharmacological potential.
Reactive oxygen species (ROS) are continuously produced as a by-product of regularly happening numerous biological reactions. 82 These ROS, if overproduced, may deplete endogenous antioxidant reservoirs and oxidize essential biomolecules resulting in different diseases. Hence, the biochemical evaluation of the dissected brains of scopolamine-treated rats revealed the reduction of SOD and Gpx and elevation of MDA. The brain is enriched with polyunsaturated fatty acids and is highly prone to oxidative stress which plays role in the pathogenesis of various neurological disorders. 83 The administration of scopolamine in rats gave rise to increased production of reactive oxygen species. 84 These ROS might deteriorate structural and functional neuronal components resulting in apoptosis that leads to impaired learning processes. 85 These damages were significantly reversed in rats treated with SV.CR revealing prominent In-vivo antioxidant activity of detected phytoconstituents that further validated the in-vitro radical scavenging action of SV.CR. Several scientists have stated the increased ROS are associated with the development of anxiety, 86 depression 87 and cognitive decline. 88 Thus, terpenoids specifically phytol and oleanolic acid and polyphenol like caffeic acid present in Solanum virginianum might be combating the oxidative stress through multiple mechanisms behind observed neuroprotective benefits.
Conclusion
The outcomes of present study have revealed the prominent neuropharmacological potential of Solanum virginianum. The chronic administration of its methanolic extract depicted anxiolytic, antidepressant and memory boosting potential in rats. The results were further validated through Ex-vivo biochemical assays. These beneficial outcomes could be related to the antioxidative properties of various phytoconstituents detected in GC-MS and HPTLC analysis. The terpenoids and phenolic compounds present in the plant might be exerting neuroprotective action through multiple mechanisms, alleviating the observed neurological impairments. The outcomes of this study can be further validated through the isolation and characterization of these plant constituents and the evaluation of their actions at molecular levels.
Footnotes
Authors’ Note
Usman Javaid, Sana Javaid, and Waseem Ashraf contributed equally. UJ, SJ, WA, MFR, FA and II intellectualized the study design, performed the experiment and analyzed the data and figures. SJ, MFR, OMN, ASA, AM, WS, and FA contributed in the assessment of data and statistical analysis. UJ, SJ, WA, OMN and THA performed the biochemical assay. All authors contributed equally in the preparation of the manuscript and approved the final manuscript with consent. All animal studies were performed after availing authorization from “Department of Pharmacology Ethical Committee” BZU, Multan (EC/04-PHL-2017 dated 20-03-2017) and were accomplished by following instructions of the “Institute of Laboratory Animal Resources” (ILAR), Commission on Life Sciences, National Research Council (NRC, 1996). The data of the current study are available from the corresponding author on reasonable request.
Acknowledgments
We are thankful to Mr. Muhammad Imran (Animal house Attendant) who devotedly worked for the welfare of laboratory animals. Furthermore, the authors extended their appreciation to Distinguished Scientist Fellowship program at King Saud University, Riyadh, Saudi Arabia for funding this work through Research Supporting Project Number (RSP-2020/131).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Distinguished Scientist Fellowship program at King Saud University, Riyadh, Saudi Arabia through research supporting project Number (RSP-2020/131).





