Abstract
Colorectal cancer is the most common malignant cancer in developing countries. Canarium odontophyllum, also known as “Dabai” or “Borneo Olive“ is among the natural plants that can potentially be used as an anticancer agent. This study aims to determine the antiproliferative activities and cytotoxicity effects of acetone extract from C. odontophyllum stem bark against human colorectal cancer cell lines HCT 116 and HT 29. Acetone extract of C. odontophyllum stem bark exerted a significant cytotoxic effect on HCT 116 and HT 29 cells determined by MTT assay at the concentration of 12.5 μg/mL to 200 μg/mL for 24, 48, and 72 hours treatment. It was found that acetone extract of C. odontophyllum stem bark inhibited proliferation of HCT 116 with an IC50 value of 184.93 ± .0 μg/mL, 61.24 ± .1 μg/mL, 79.98 ± .029 for 24, 48 and 72 hours respectively. The findings also showed that acetone extract of C. odontophyllum stem bark revealed a lower inhibitory effect against HT-29 with an IC50 value of more than 200 μg/mL for 24, 48 and 72 hours. However, acetone extract of C. odontophyllum stem bark at similar concentrations and time points did not show any cytotoxic effect to normal colorectal fibroblast cell CCD18-Co. In conclusion, the acetone extract of C. odontophyllum stem bark exhibited more sensitivity against HCT 116 than HT 29. Its antiproliferative ability towards HCT 116 and HT 29 cells provides insight that this extract may serve as an anticancer agent against colorectal cancer.
Keywords
Introduction
The third most common cancer globally is colorectal cancer. This cancer has been identified as one of the leading causes of death especially in Western countries. 1 Colorectal cancer can be defined as uncontrolled cell growth in the digestive system especially in the large intestine. 2 This cancer usually begins with a growth on the wall of the colorectal or rectal. These growths are known as polyps and will turn into cancer when left untreated. In Malaysia, colorectal cancer patient distribution by race mostly involves the Chinese, followed by Malays and Indians. 1 Several factors can invite this case of colorectal cancer. Among them are age factors, family history of colorectal cancer, smoking and unhealthy diet practices, especially diets with high protein and fat content and diets lacking in fiber. 3 Various treatments are available to treat this colorectal cancer. Among them are surgery, radiotherapy, chemotherapy, hormone therapy, immune therapy, sympathetic therapy, and support. 4 Yet this treatment can lead to various side effects such as anemia, hair loss, and diarrhea. 3
Since a long time ago, natural plants have been used to treat a wide variety of diseases. Natural resources also play an important role in treating cancer. 5 Therefore, natural products can be used as an alternative treatment to treat cancer. According to Ref [6], plant extracts can be used as cancer-preventing agents and can inhibit the process of carcinogenesis. Among the drugs produced using natural products are vincristine, teniposide, and vinorelbine. These drugs are anti-tumor agents that are used to treat cancer. 6 Canarium odontophyllum also known as “Dabai” or “Borneo Olive” is one of the natural plants that have the potential to be used as an anticancer agent. The plant distribution in Asia is largely concentrated in Borneo, Malaysia, Brunei, Kalimantan, Sumatera, Indonesia, and Palawan, Philippines. 7 This plant belongs to the “Burseraceae” family and is a native plant found along the banks of the Kanowit, Sarikei, and Kapit rivers in Sarawak. 8 The fruit produced by this plant is purple and oval in shape. It has one hard elongated seed. 9 The stem bark of this tree is gray or yellowish from the outside while the inside is pink with a soft texture and produces a fragrant aroma.10,11 Typically, the harvesting season runs from November until January. Because of the plant’s rapid respiration rate, its fruits have a short shelf life. As a result, this study focuses on the plant’s stem bark, which is more readily available throughout the year.11,12
Various studies have been performed using the leaves, fruits, seeds, and stem bark of this plant.9,10,13-15 According to a study by Refs.[10,14], this plant stem bark has anticancer properties against colorectal cancer continuous cells HCT 116. Several phytochemicals are also found in this plant stem bark namely flavonoids, terpenoids, tannins, saponins, and phenolic compounds that may also contribute to the anticancer activity. Ref. [14] found that acetone extract from Canarium odontophyllum stem bark was the most potent in terms of IC50 values on HCT 116 cell line compared to methanol and aqueous extract. Hence in this study, acetone extract produced from the plant’s stem bark has been collectively decided as the principal cell treatment extract. However, no previous studies have been done on the effect of acetone extract from Canarium odontophyllum stem bark against HT 29. Therefore, this study was designed to determine antiproliferative activities and cytotoxicity effects of acetone extract from the stem bark of Canarium odontophyllum against human colorectal cancer cells, HCT 116, and HT 29. The findings of this study may show that the extract has potential to be used as an anticancer agent in the fight against colorectal cancer.
Results and Discussion
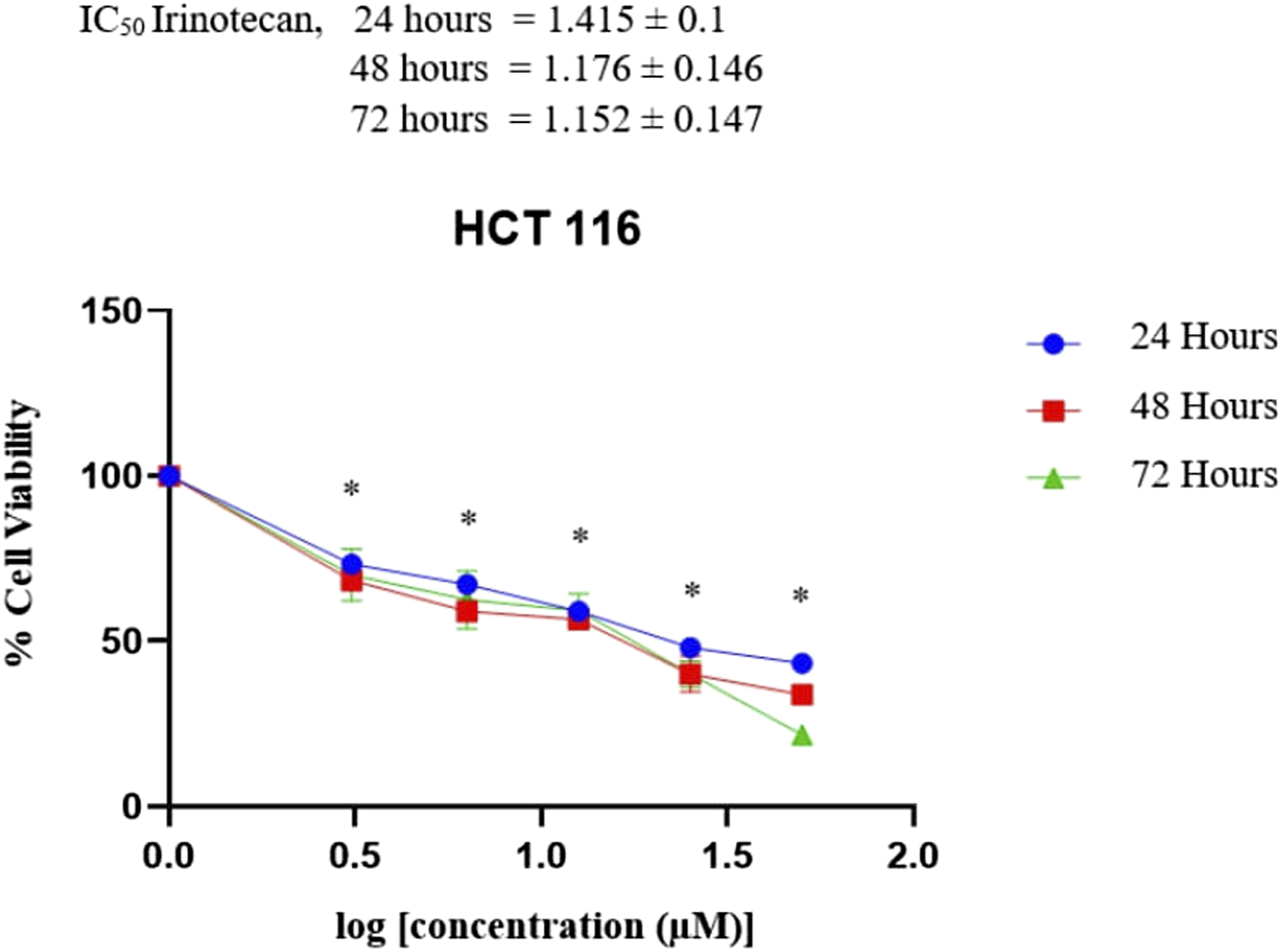
The results showed that acetone extract from C. odontophyllum stem bark exhibited different suppressive effects on colorectal cancer cells. The antiproliferative activities of acetone extract from C. odontophyllum stem bark increase as the concentration of acetone extract level increases from 12.5 μg/ml to 200 μg/ml. The results showed that the highest cytotoxicity effect of acetone extract from C. odontophyllum stem bark that demonstrated the lowest IC50 against HCT 116 cell was shown at 48 hours with IC50 61.24 μg/ml ± .1 compared to the 24 hours and 72 hours treatments which are 184.93 ± .0 μg/mL and 79.98 ± .029 μg/ml, respectively (Figure 1 and Table 1). While, the IC50 of menadione was 10.19 ± .037 μM, 9.40 ± .037 μM and 9.95 ± .002 μM after 24 hours, 48 hours, and 72 hours treatments, respectively (Figure 2 and Table 1). IC50 of irinotecan was shown higher than menadione, 26.00 ± .1 μM, 15.00 ± .146 μM, and 14.19 ± .147 μM after 24 hours, 48 hours, and 72 hours treatments, respectively, against HCT 116 (Figure 3 and Table 1) Percentage viability of HCT 116 cell line after 72 hours of treatment with acetone extract of C. odontophyllum stem bark. Each data was expressed in mean ± SEM and obtained from three separate experiments. (*P <.05 against negative control). IC50 Value of HCT 116 Cell Line After 72 hours of Treatment With Acetone Extract of C. odontophyllum Stem Bark, Menadione And Irinotecan. Percentage viability of HCT 116 cell line after 72 hours of treatment with menadione. Each data was expressed in mean ± SEM and obtained from three separate experiments. (*P < .05 against negative control). Percentage viability of HCT 116 cell line after 72 hours of treatment with irinotecan. Each data was expressed in mean ± SEM and obtained from three separate experiments. (*P < .05 against negative control).


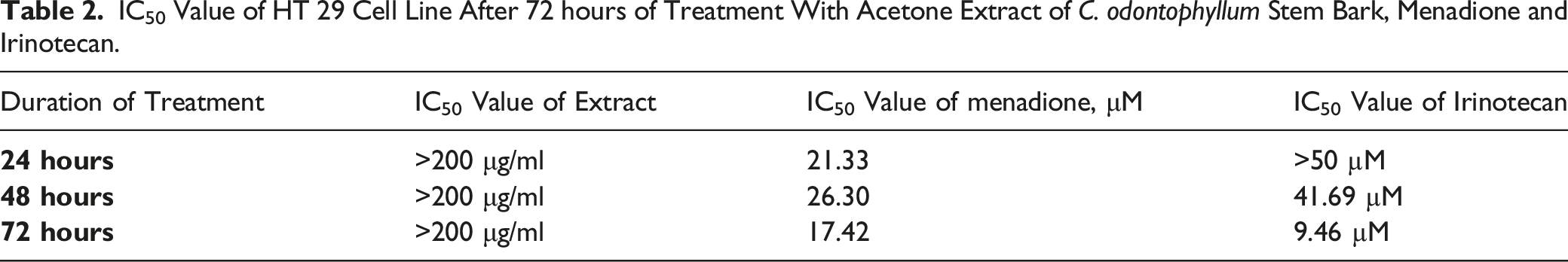
After treatment with acetone extract of C. odontophyllum stem bark, it is shown that this treatment has the ability to inhibit cell proliferation of HT 29 for 72 hours. However, the IC50 values for all three time points were more than 200 μg/ml (Figure 4 and Table 2). While, the IC50 of menadione was 21.33 ± .048 μM, 26.30 ± .019 μM and 17.42 ± .031 μM after 24 hours, 48 hours, and 72 hours treatments, respectively (Figure 5 and Table 2). IC50 value of irinotecan was more than 50 μM for 24 hours, while for 48 hours and 72 hours exhibited IC50 41.69 ± .025 μM and 9.46 ± .108 μM, respectively, against HT 29 (Figure 6 and Table 2). Although this extract revealed showed growth inhibitory potential towards colorectal cancers, the good thing is this acetone extract did not elicit a cytotoxic effect against normal human colorectal cells, CCD18-Co (Figure 7). Based on Figure 8, menadione showed prominent cytotoxic activity against both HCT 116 and CCD-18co cells with IC50 of 10.19 μM and 17.54 μM, respectively. Percentage viability of HT 29 cell line after 72 hours of treatment with acetone extract of C. odontophyllum stem bark. Each data was expressed in mean ± SEM and obtained from three separate experiments. (*P < .05 against negative control). IC50 Value of HT 29 Cell Line After 72 hours of Treatment With Acetone Extract of C. odontophyllum Stem Bark, Menadione and Irinotecan. Percentage viability of HT 29 cell line after 72 hours of treatment with menadione. Each data was expressed in mean ± SEM and obtained from three separate experiments. (* P < .05 against negative control). Percentage viability of HT 29 cell line after 72 hours of treatment with irinotecan. Each data was expressed in mean ± SEM and obtained from three separate experiments. Percentage viability of CCD18-Co after 72 hours of treatment with acetone extract of C. odontophyllum stem bark. Each data was expressed in mean ± SEM and obtained from three separate experiments. (*P < .05 against negative control). Percentage viability of CCD18-Co and HCT 116 after 24 hours of treatment with menadione. Each data was expressed in mean ± SEM and obtained from three separate experiments. (*P < .05 against negative control).




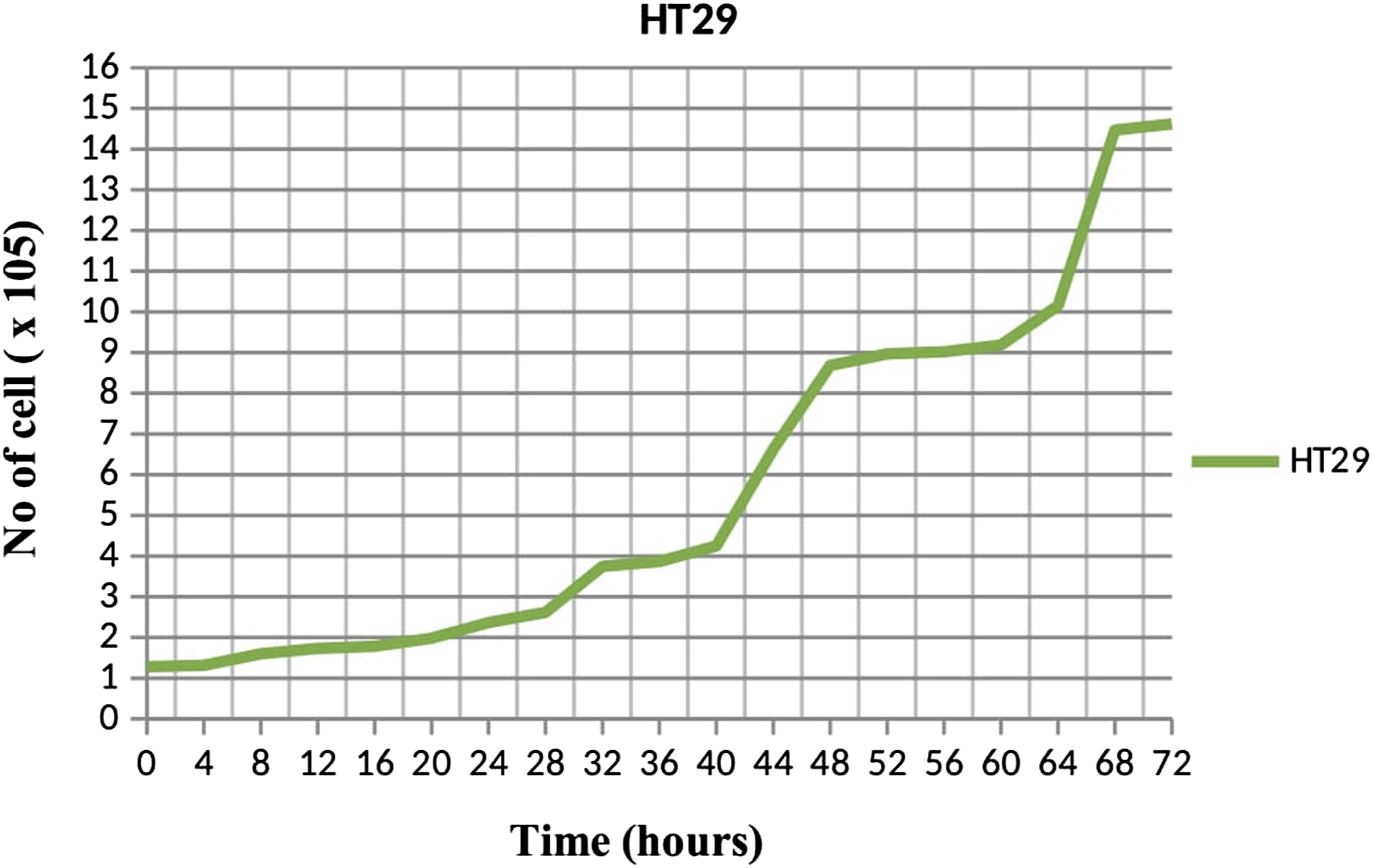
Although this acetone extract from C. odontophyllum stem bark demonstrated an antiproliferative effect with the dose-dependent manner in these two colorectal cancer cells, these results showed that acetone extract was more effective and sensitive toward HCT 116 rather than HT 29 because of low IC50 was exhibited by HCT 116. In other words, HT 29 response was seen more resistant to this C. odontophyllum extract treatment compared to HCT 116. Moreover, this may also be due to the faster doubling time exhibited by HCT 116 which is 20 hours compared to doubling time by HT 29 which is 22 hours that showed on the growth curve graph (Figure 9 and Figure 10). Growth curve graph of HCT 116 after 72 hours. Each data was expressed in mean ± SEM and obtained from three separate experiments. Growth curve graph of HT 29 after 72 hours. Each data was expressed in mean ± SEM and obtained from three separate experiments.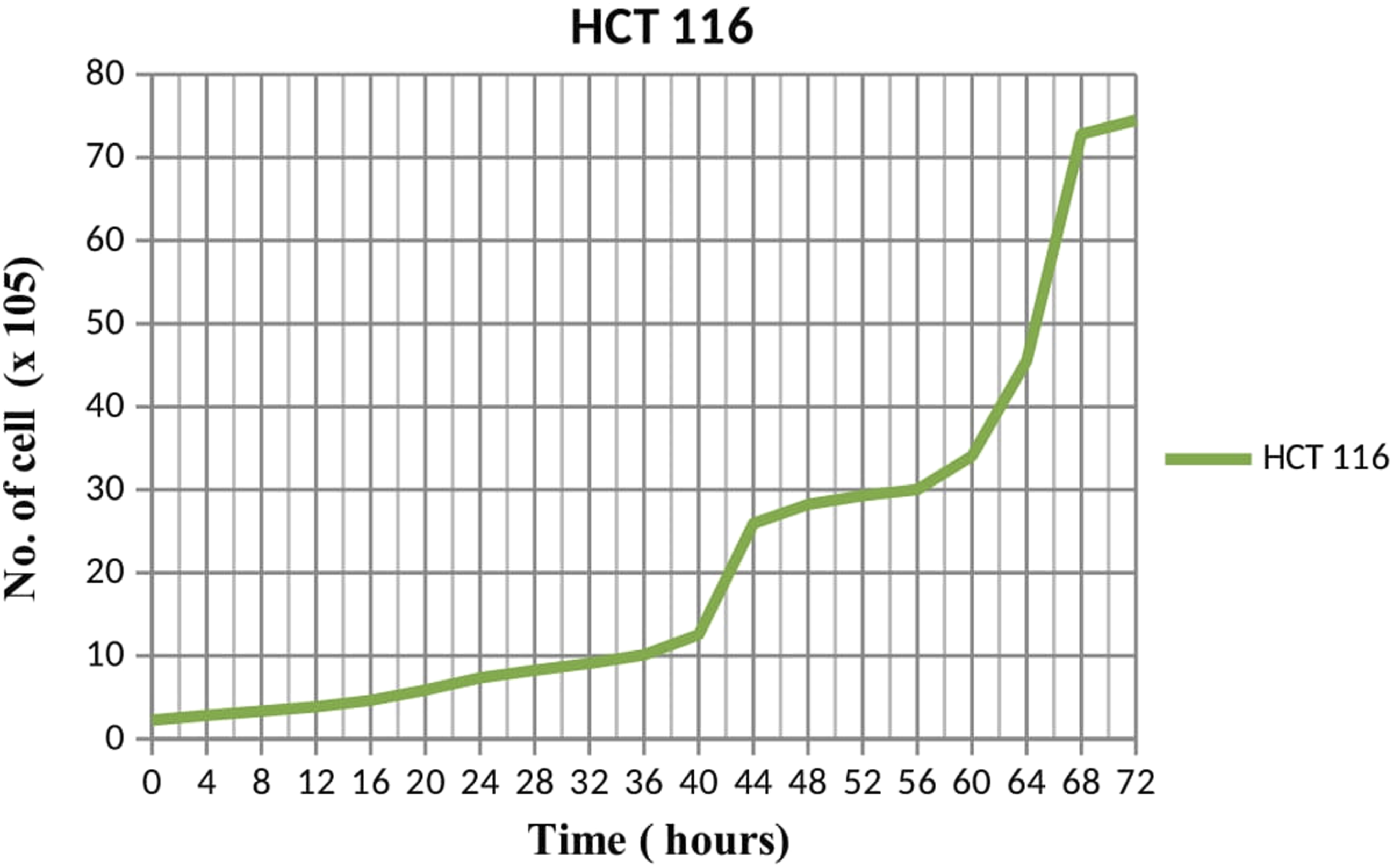
Acetone extract of C. odontophyllum stem bark demonstrated cytotoxic ability in inhibiting the proliferation of HCT 116 and HT 29 cells at all three treatment time points (24 hours, 48 hours and 72 hours) and showed a prominent decrease in viability of HCT 116 at 48 hours. The result had been supported by Ref. [16] demonstrated a drastic reduction of HCT 116 cell viability after 48 hours of treatment with 8-prenylnaringenin. It also reported that the lowest IC50 of 8-prenylnaringenin against HCT 116 was shown at 72 hours but there is no significant difference in IC50 compared to 48 hours. 16 While in this study, higher IC50 of acetone extract against HCT 116 cells were obtained at 72 hours treatment compared to the 48 hours but there is no significant difference (P > .05) indicated between these two values. This might be due to the instability of certain phytochemicals in the extract. A study by Ref. [17] reported that exposing cancer cells at a low dose for a prolonged time does not inhibit the proliferation of cancer cells. This may explain the finding in this study that the prolonged exposure of HCT 116 cells toward the extract no longer inhibits the growth of HCT 116 cells. This acetone extract from C. odontophyllum stem bark demonstrated an antiproliferative effect in a dose-dependent manner against these two colorectal cancer cells. A study from Ref. [18] also reported that viability cells of both colorectal cancer cells (HCT 116 and HT 29) reduce dose-dependently after treatment with hexane and ethyl acetate extracts for 72 hours. Treatment with certain extracts in a dose-dependent manner that inhibited the growth of target colorectal cancer cells indicate that increasing the exposure of concentration will increases the colorectal cancer cell numbers be killed. 18
The IC50 value obtained in this study was slightly lower (61.24 μg/ml ± .1 μg/ml) when compared to the previous study by Ref. [19] (82 ± 9.3 μg/ml) under similar conditions. However, it contradicts another previous study when IC50 value obtained in this study was higher than the study by Ref. [20] (24 ± 3.059 μg/ml) at 48 hours of treatment. The differences in IC50 values can be due to storage conditions and periods that could affect the phytochemical content of the plant extract. A study conducted by Ref. [21] found that the flavonoid compounds and phenolic acids in Salvia tomentosa had decreased after 6 months of storage. In this study, acetone extract of C. odontophyllum stem bark has been stored in Universiti Kebangsaan Malaysia for more than 6 months. Therefore, there could be a slight difference in flavonoid, phenolic acids, and other compounds of C. odontophyllum stem bark sample, resulting in differences in IC50 values. Phytochemical screening conducted by Ref [10] to study the presence of the phytochemicals responsible for the cytotoxic activity of these crude extracts against HCT 116 found that flavonoid, tannin, saponin, terpenoid, and phenolic compounds were present in the acetone extracts of C. odontophyllum stem bark. However, the identification of active compounds in this acetone extract of C. odontophyllum stem bark is needed to be further study.
This study also reported that HCT 116 cells as the most sensitive cell compared to HT 29. In other words, HT 29 response was seen more resistant to this C. odontophyllum extract treatment. This also had been supported by a previous study which demonstrated that HCT 116 were more sensitive to 8-prenylnaringenin by Ref. [16] Annona muricata hexane and ethyl acetate extracts by Ref. [18] and fish singgang extracts by Ref. [22] compared to HT 29. Moreover, this may also be due to the higher and faster growth rate exhibited by HCT 116 compared to HT 29 from the growth curve. A study from Ref. [23] showed that a higher and faster growth rate of HCT 116 cells can lead to short-term effects and high sensitivity against treatment compared to other cell lines which are HT-29 and CCD-18Co. However, some studies reported that HCT 116 cells were more resistant than HT 29 cells after treatment with protopanaxadiol ginsenosides. 24 Besides that, menadione exhibited more sensitivity against both colorectal cells after 72 hours of treatment compared to irinotecan and will be used as a positive control in this study.
Before any further study can be done, it is very important to evaluate SI value for any research on extract or active compound. Any extract or active compound should not affect the normal cells although it can inhibit the growth of cancer cells. So, cytotoxicity against normal cell lines and cancer cell lines must be determined in order to calculate the SI value. 25 In this study, acetone extract did not elicit any cytotoxic effect against normal human colorectal cells although this extract could inhibit the growth of colorectal cancer cells. The evaluation of the selectivity index (SI) value of this extract is more than 3. This indicates that this extract produced a non-toxic effect on normal colorectal cells and was highly selective on colorectal cancer cells. Similar to menadione, positive control in this study was selective against colorectal cancer cells. SI value can be defined as a simple ratio of IC50 calculated between normal and cancer cells26-29 which indicated values more than 1 is selective against cancer cells. Only cancer cells were found to be suppressed by the extract, but not normal cells. This can excite new interest in additional research into possible cancer-cell-targeting compounds.
Conclusion
This acetone extract from C. odontophyllum stem bark demonstrated an antiproliferative effect with the dose-dependent manner in the two colorectal cancer cell lines. However, HCT 116 was more sensitive towards the treatment compared to HT 29. Proliferation of normal colorectal cell CCD18-Co was observed after treatment with the acetone extract despite antiproliferative activities in colorectal cancer cell lines. Evaluation of the selectivity index (SI) value of this extract is 3 (>1) indicating high selectivity towards colorectal cancer cells. Its antiproliferative ability toward HCT 116 and HT 29 cells provides insight that this extract may serve as an anticancer agent against colorectal cancer.
Materials and Methods
Plant Materials
The material used in this study was dried acetone extract from C. odontophyllum stem bark that was prepared in advance by solvent extraction method. C. odontophyllum stem bark was taken from Kuching, Sarawak and the voucher number for C. odontophyllum stem bark was UKMB 40052. A stock solution of 100 mg/mL was prepared by dissolving 100 mg of dried stem bark acetone extract in 100% DMSO. The stock solution was mixed using a vortex for 5 minutes before being centrifuged for 5 minutes at 2500 rpm. The centrifuged stock solution was then filtered using .22 μM nitrocellulose membrane filter. The stock solution was stored at −20°C until further used. Menadione and irinotecan were used in this study to determine which one is the best as a positive control.
Preparation of Cell Culture
HCT 116 and HT 29 human colorectal carcinoma cell line originated from American Type Culture Collection (ATCC® CCL-247 TM & ATCC® HTB-38 TM) was used in this study. The cell line was maintained in complete growth medium McCoy’s 5A media (Sigma Aldrich, USA) enriched using 10% of fetal bovine serum (FBS) and 1% of penicillin-streptomycin. Cells were cultured at 37°C under 5% CO2. These HCT 116 and HT 29 cells are adherent cells. The cells were cultured to reach 70-80% confluence approximately 2–3 days before the next subculture.
Evaluation of Cytotoxic Activity
The cytotoxic activity of the extract was determined using (3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide), MTT assay. This method was to determine the effect of cytotoxic activity of acetone extract from C. odontophyllum stem bark. The C. odontophyllum extract was used to treat HCT 116 and HT 29 cells ranging from 12.5 μg/ml to 200 μg/ml and the seeded cells were incubated for 24 hours prior to treatment. Menadione and irinotecan at a concentration ranging from .781 μM to 12.50 μM were used to determine which one is the best as a positive control, while untreated cells were used as a negative control. All media in each well were discarded and C. odontophyllum use as treatment and menadione was introduced into each well accordingly. Then, the 96-well plate was incubated for 24, 48, and 72 hours at 37°C, 5% CO2. Thereafter, a total of 20 μL of MTT was added to each treated well. This procedure is performed in the dark. The plates were incubated for 4 hours. Then, all media were removed and DMSO was added to each well to dissolve the dark blue formazan crystals at room temperature. The plates were re-incubated in the incubator for 15 minutes. Afterward, the plates were shaken with an automatic microplate shaker for 5 min. The absorption will be read using an ELISA microplate reader at 570 nm. The response dose curve graph was plotted to obtain the IC50 value. The cell viability was calculated as follows
Statistical analysis
All the data obtained were expressed as mean ± SEM of three replicates. Graphs were plotted in a semi-log graph using GraphPad Prism 9.0. Data were analyzed through two way-ANOVA to compare for significance using GraphPad Prism 9.0. The values of P < .05 were considered statistically significant.
Footnotes
Acknowledgments
The colorectal cancer cell lines used in this study were kindly provided by Prof. Dr Saiful Anuar Karsani from the Institute of Biological Sciences, Faculty of Science, Universiti Malaya.
Author Contributions
Assoc. Prof Dr Dayang Fredalina Basri designed and lead the project. Siti Fairuz Ishak performed the experiment and prepared the article. Prof. Dr Fadilah Rajab supervised the experiment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by research code grant Research University Grant GUP-2016-036.
