Abstract
Purpose
This work was the first study to show the impact of γ- radiation on adropin levels in the serum and liver tissue of male albino rats.
Methods
Liver tissue and blood samples of rats were collected at 1, 3, 7 and 14 days after whole-body exposure to 7.5 Gy of γ-radiation.
Results
Irradiated groups revealed a marked decrease in adropin associated with a significant increase in STAT3 in the serum and gene expression. Furthermore, lipid profile (cholesterol, T.G, HDL, LDL, VLDL), liver function (AST, ALT, albumin and total protein), complete blood count (RBCs, WBCs, PLT, Hb, Hct%, MCH, MCV, WBCs differential), glucose and insulin were exhibited more noticeable alterations at all time periods of the experiment. In addition, data exhibited an obvious elevation in some inflammatory markers (IL-6) and TOS accompanied by a decline in the TAC.
Conclusion and future scope of work
γ- radiation has adverse effects on adropin that related inversely with STAT3, leading to further damage to liver cells as well as disturbances in lipid and glucose metabolism. Therefore, adropin could be used in people exposed to radiation such radiotherapy to control the serious effects of radiation. Further study is needed to confirm these results.

Introduction
Ionizing radiation (IR) refers to radiation that passes through a material and has enough energy to remove the orbital electrons of the atoms and separate these atoms into positive ion molecules and free electrons. So, IR has the ability to change the chemical composition of the material it interacts with, leading to protein changes and disruption of DNA, RNA and cell membranes.1,2 The final stage of radiation damage is characterized by visible functional and then morphological changes, arrest of cell division, chromosome breakage, cell death. The molecular proceedings followed radiation are complex and extends to a variety of biologic processes, including senescence,3,4 oxidative stress, 5 inflammation, and fibrosis. 6 Oxidative stress (OS) is a consequence of an imbalance between ROS formation and antioxidant protection processes. It affects the intracellular molecules. 7 The elevation of OS causes heart ailment, 8 liver injury, 9 cancer, 10 kidney, 11 and neurological diseases. 12
The liver regulates and makes many hormones and molecules that aid in physiological functions in the body. Radiation causes acute or chronic injury in the liver. Early effects of IR include DNA damage, oxidative stress and ROS formation, resulting in hepatocellular apoptosis and acute inflammatory responses in irradiated areas. 9 Kupffer cells (KCs), sinusoidal endothelial cells (SECs) and hepatic stellate cells, are radiosensitive cells which release various substances that promote liver fibrosis, contributing to distorted liver structure and function during radiation. 13 Liver damage is affected by the nature of the radiation, the total radiation dose, the dose level, and the exposed part of the body. 3 Hepatokines are hormone-similar proteins produced by hepatocytes that are liberated in the circulation and can impact the metabolic and inflammatory mechanisms. 14
Adropin considers as an important hepatokine protein that has a potential function in the adjustment of metabolic ailments such as diabetes mellitus and non-alcoholic fatty liver disease. 15 Moreover, adropin is a released peptide forming from 76 amino acids and is encoded by (Enho) gene that considers as energy stability. 16 It is found in the liver, brain, heart, kidney, and central nervous system, 17 where it can control angiogenesis, raise blood flow, increase capillary density and maintain endothelial cells. 18 Adropin levels vary in biological and physiological conditions such as sclerosis, 19 COVID-19, 20 gestational diabetes, 21 obstructive sleep apnea, 22 rheumatoid arthritis, 23 coronary artery ectasia, 24 and diabetic nephropathy. 25 Previous researchers have described that adropin levels may be linked to diabetes, obesity, hypertensive syndrome, cardiovascular problems, metabolic disorders, and multiple cancers.26,27
The STAT3 gene (signal transducer and activator of transcription 3) is part of the STAT gene family that carries instructions for the production of proteins. STAT proteins bind to specific regions of DNA to control regions adjacent to genes that allow proteins to control the transformation of these genes. Previous study reported that STAT3 is involved in the genetic regulation of adropin, increasing the levels of circulating adropin and promoting Enho expression in the livers of diabetic rats. 28
Under physiological circumstances, it mediates cell proliferation, survival, apoptosis, immune function, angiogenesis, and other essential mechanisms through the stimulation of cytokines and growth factors. 29
Methodology of the Study
This research article discusses the impact of gamma rays on adropin levels in the serum and liver tissues of rats. The study team selected 25 adult male Wistar albino rats (120-150 g) for the experiment and used gamma rays at a single dose of 7.5 Gy based on prior studies. Rats were irradiated and then were sacrificed on days 1, 3,7 and 14 post-irradiations to prepare blood and liver tissue samples for hematological and biochemical analysis. Adropin levels and the other parameters such as STAT3, ALT, AST, albumin, total protein, total lipids, glucose, insulin, and IL-6 were measured. Additionally, complete blood count (CBC) was examined in the blood.
Our assessments revealed strong evidence that radiation can cause a significant decrease in the adropin levels throughout the experiment. These results led the study team to conclude that people exposed to radiation such as radiotherapy often suffer from a substantial decline in the performance, and this may be due to low levels of adropin. Therefore, adropin treatment could control the serious side effects of radiation exposure.
Material and Methods
Gamma- Irradiation
Rats were irradiated by Gamma cell 40 bioradiation device using Cesium137 (Cs137) at National Centre for Radiation Research and Technology (NCRRT) by Atomic Energy of Canada Limited, Ontario, Canada source. The rats were exposed to a single dose of whole-body gamma radiation (7.5 Gy) 30 with a dose rate of 0.587 rad/sec. The irradiation process and dose rate calculation were done according to the Dosimetry and Protection Department at NCRRT at the time of the experiment.
Experimental Animals
Adult male Wistar albino rats (120-150 g) were obtained from the animal house belonging to the National Centre for Radiation Research and Technology (NCRRT), Cairo, Egypt. Rats were maintained under standard conditions of light, ventilation, temperature (25°), and humidity (60%) and allowed fed standard pellet diet, containing all nutritive elements.
Ethics Approval
This study protocol has been reviewed and approved by Zagazig University - Institutional animal care and Use committee (ZU-IACUC)” according to the U.K. Animals (Scientific Procedures) (OFFIC 1986) and the National Research Council’s Guide for the Care and Use of Laboratory Animals (Care and Animals 1986) and in compliance with the ARRIVE guidelines, “The approval number: ZU-IACUC/3/F/142/2024”
Experimental Design
The experiment was carried out for 2 weeks. Animals were separated into two main groups. Group I (n = 5): control group, in which rats were administrated with saline; Group ii (n = 20): The rat’s whole body was exposed to 7.5 Gy applied in a single shot dose at a dose rate of 0.587 rad/sec. Irradiated group was divided into four subgroups (5 rats each). Rats of all groups were sacrificed on days 1, 3, 7 and 14 post-radiation.
Sample Preparation and Biochemical Analysis
Rats of all groups were anesthetized with urethane (1.2 g/kg, intraperitoneally) according to the method,
31
urethane was obtained from Sigma-Aldrich Chemical Co. (St Louis, Missouri, USA), product Number U 2500. Blood samples were withdrawn from each rat on days 1,3,7 and 14 by heart puncture using the sterilized syringe, the blood was divided into two parts one part was placed on ethylene diaminetetra acetic acid (EDTA) from Sigma Aldrich Chemical Co. (St Louis, Missouri, USA), for hematological analysis. The second part of blood was centrifuged at 3000 RPM (run /min) for 15 minutes to obtain sera that were stored at −20C
Hematological and Biochemical Investigation
Complete blood cells were assessed according to method 33 using Hematology analyzer BC-20s (Mindray-Chinese) device. Adropin amounts in the serum were evaluated via Rat adropin ELISA Kit (My BioSource, Inc. USA), Catalog No: MBS760446. STAT3 is measured using Rat STAT3 ELISA Kit (My BioSource, Inc. USA), Catalog No: MBS760293. Level of serum glucose was measured due to procedure 34 using commercial kits supplied by Bio diagnostic. The level of serum insulin was determined using Rat Insulin ELISA Kit (Crystal Chem. Europe), Catalog No. 90010. Serum triglyceride (T.G) level was determined according to method. 35 Cholesterol (TC) level was valued according to method. 36 HDL level was measured due to the procedure. 37 LDL level was determined as: LDL = TC-HDL-TG/5. 38 VLDL) level was calculated as: VLDL= T.G/5. 35 Commercial kits (Biodiagnostic Co., Egypt) were used in total lipid profile estimation. ALT and AST levels were measured by. 39 Total protein and albumin were measured via colorimetric assay kit.40,41 respectively and the kits purchased from Biodiagnostic, Egypt. Serum IL-6 level was measured using ELISA Immunoassay Product No. SEA079 Ra and the kit is a sandwich enzyme immunoassay for in vitro quantitative measurement of IL6 in the rat serum. TOC and TOS were estimated, using commercial kits supplied by Biodiagnostic Co, Egypt. Calculation of OSI: The OSI (arbitrary unit) = TOS (μmol H2O2 Eq/L)/TAC(μmol uric acid Eq/L). 42
Detection of Adropin and STAT3 Gene Expression via Real Time-Polymerase Chain Reaction (RT-PCR)
Total ribonucleic acid was extracted from hydrolyzed tissues with Direct-zol RNA Miniprep Plus (Cat# R2072, ZYMO RESEARCH CORP. USA) and then the amount was evaluated by Beckman dual spectrophotometer (USA). Superscript IV One-Step RT-PCR kit (Cat# 12594100, Thermo Fisher Scientific, Waltham, MA USA) was applied for reverse transcription of the extracted RNA and then by PCR in the same step. The result was expressed in Cycle threshold (Ct) next to the RT-PCR run. The PCR records sheet includes Ct values of assessed gene [stat3 and adropin (Enho)] vs the corresponding the house keeping gene (GAPDH). A standard sample should be used to measure the specified genes expression. The RQ (The relative quantification) of every specific gene is measured and standardized to housekeeping gene via the calculation of delta-delta Ct (ΔΔCt). We examined the RQ of every gene by picking 2-∆∆Ct
Statistical Analysis
Data will be presented as mean ± S.D. Statistical significance will be determined by one way analysis of variance (ANOVA) followed by LSD test, P values less than .05 will considered to be significant. The correlations between parameters will be analyzed using Pearson’s correlation. In statistical analysis, SPSS version 22 program for Windows (SPSS Inc. Chicago, IL, USA) will be used.
Results
Effect of Radiation (7.5 Gy) on the Level of Adropin and STAT3 in the Serum and Liver Tissue of the Studied Groups at Different Times Post- Irradiation.

Values are expressed as the means ± standard deviation (n = 5). Values between brackets show the percentage of change from control. Differences between means were considered significant at P ≤ 0.05.
aSignificance vs control group.
Effect of Radiation (7.5 Gy) on the Inflammatory and Redox State Markers in all Studied Groups.
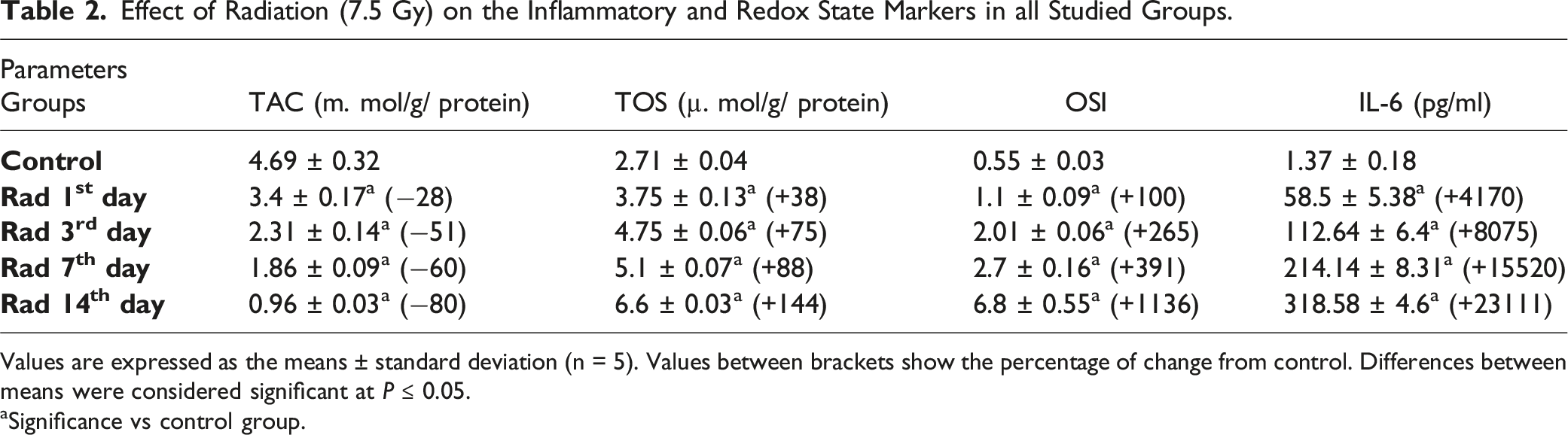
Values are expressed as the means ± standard deviation (n = 5). Values between brackets show the percentage of change from control. Differences between means were considered significant at P ≤ 0.05.
aSignificance vs control group.
Effect of Radiation (7.5 Gy) on the Level of Some Biomarkers for Liver Damage in all the Studied Groups.

Values are expressed as the means ± standard deviation (n = 5). Values between brackets show the percentage of change from control. Differences between means were considered significant at P ≤ 0.05.
aSignificance vs control group.
Effect of Gamma Radiation (7.5 Gy) on the Level of Lipid Profile, Glucose and Insulin in all Studied Groups.

Values are expressed as the means ± standard deviation (n = 5). Values between brackets show the percentage of change from control. Differences between means were considered significant at P ≤ 0.05.
aSignificance vs control group.
Effect of Radiation (7.5 Gy) on CBC in all Studied Groups.

Values are expressed as the means ± standard deviation (n = 5). Values between brackets show the percentage of change from control. Differences between means were considered significant at P ≤ 0.05.
aSignificance vs control group.
Pearson’s Correlation Coefficient (r) Between Adropin Level and Some Studied Parameters at Different Times Post-radiation.


Scatter Dot Plot Showing Correlation Between Adropin and STAT3 (A), TOS (B), TAC (C), IL-6 (D), AST (E), ALT (F), Cholesterol (G), T.G (H), HDL (I), LDL (J), Glucose (K) Among Studied Groups.
Primers Sequence of Adropin and STAT3 Genes.

Effect of Radiation (RAD) on Adropin (A) and STAT 3 (B) Gene Expression in the Liver. Results are Expressed as Mean ± SD (n = 5). (a) Significance vs Control Group.
Discussion
Exposure to IR activates oxidative stress in different organs. Radiation can cause acute or chronic damage to the liver. The early effects of radiation include DNA damage, oxidative stress and the production of ROS leading to liver cell apoptosis and acute inflammation. 43 In this experiment, whole body exposure of rats to γ-rays has initiated oxidative stress and stimulated alterations in the redox system of the liver. Data showed a marked liver dysfunction in the rats after radiation exposure manifested by significant elevations in the level of TOS, STAT3, IL-6, TC, TC, T.G, LDL, AST, ALT and glucose associated with depletion of adropin, TAC, insulin, HDL, albumin, total protein levels, as well as changes in the blood components. In addition, the present study found a decrease in adropin gene expression associated with an increase in STAT3 gene expression in the liver.
Adropin is a hepatokine that has a major effect on the liver and is known to have a number of biological and biochemical actions that occur in cells to maintain metabolic regulation. It also has regulatory effects on glucose, and lipids as well as protective properties against insulin resistance and vascular dysfunction. 44
The results obtained showed that total body irradiation at 7.5 Gy resulted in a significant gradual decrease in serum adropin level and its gene expression (Enho) in the liver tissues from day 1 to day 14 after radiation, compared to the control groups. Here we propose that low levels of adropin post-irradiation might be attributed to liver injury, which is responsible for adropin secretion, and this depletion might lead to increased secretion of free radicals in the liver tissue, causing further liver damage. This explanation is supported by a clear decrease in the level of adropin gene expression (Enho) associated with an increase in total oxidant in the liver tissue. Our final result is in line with previous findings that adropin intervention could significantly stop overproduction of ROS in the myocardium induced by radiation.45-47 similar results have also been shown that adropin-deficient mice had enhanced hepatic steatosis, while receiving synthetic adropin attenuated the development of NASH (non alcoholic steatohepatitis) in mice. 48 In addition, from previous studies, adropin activates the nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway, a key regulatory factor of cellular antioxidant responses, leading to activation of antioxidant enzymes, and reduced production of reactive oxygen species from liver mitochondria. 48 Therefore, in our present study, we suggest that the decrease in adropin level and its gene expression (Enho) post-radiation might be led to over production of total oxidant in the liver.
Furthermore, we suggest that the decrease in adropin level might be attributed to the elevation in STAT3, which is activated by IL6 that was elevated in our study after irradiation. Besides, oxidative stress is a vital factor in stimulating STAT3 receptors to activate STAT3 which may act as a pro-inflammatory mediator, leading to increased free radicals and liver damage. Our study found a negative correlation between adropin and STAT3, and IL-6 in irradiated rats, a finding is supported by similar researchers who reported that low adropin levels were negatively correlated with markers of inflammation in patients with liver disease. 49
Exposure to ionizing radiation provoked a significant augmentation in the activity of both ALT and AST from 1st to 14th day and this might be attributed to the facilitation of the passage of these enzymes due to increased membrane permeability. 50 Also, the data exhibited a remarkable reduction in total protein content in irradiated rats at all times of the experiment and this might be due to an imbalance permeability of liver, kidney and other tissue cells as well as modification in the biochemical steps. 51 Our study also revealed an obvious elevation in the level of glucose associated with a reduction in insulin level and this was explained by the previous researchers who ascribed the increase to the hepatic and pancreatic cells damage post- radiation, leading to glycogenolysis.52,53 We suggest that low levels of adropin might affect glucose levels because it helps in the uptake of glucose into cells, and this suggestion is consistent with other authors who have noted that adropin has a significant effect in modulating glucose metabolism, regulating insulin sensitivity and glucose utilization in the various tissues. Besides, adropin has been shown to inhibit hepatic gluconeogenesis, and interacts with other metabolic regulators to a greater extent. 16
Adropin directly affects lipid metabolism via 5'-adenosine monophosphate-activated protein kinase (AMPK), a regulatory factor in maintaining energy balance in cells. Its activation increases fat consumption by stimulating lipolysis and inhibiting lipogenesis. 54 The current work exhibited a decline in the gene expression of adropin (Enho) and this is similar with the previous findings that found excess cholesterol inhibits hepatic expression of Enho mRNA leading to decreased adropin production which in turn causes greater increases in total lipid. 55 Furthermore, the hypercholesterolemia observed in our work might be due to the increased rate of cholesterol formation in the tissues of irradiated rats and the degradation of cell membranes. 56 The increase can also be explained by the breakdown of stored fat from adipose tissue being released into the blood and by changes in the metabolism in the hepatocytes and blood lipoprotein. 57 High levels of triglycerides might be due to inhibition of lipase (an enzyme that hydrolyses triglycerides and releases fatty acids) and release of triglycerides from adipose tissue. 58 Adropin has a crucial effect in modifying fat production pathways by inactivating the activity of synthase, a vital enzyme responsible for fat resynthesis and thus, it reduces the conversion of excess carbohydrates into fatty acids and their accumulation in the form of triglycerides, thus reducing fat storage.16,44 Therefore, we suggest that decreased levels and expression of adropin might contribute to lipid disorders, and the data are consistent with the previous.17,18
In addition, our findings indicated abnormalities in all blood components, which could be explained by direct destruction of the entire circulatory system. Radiation could also lead to bone marrow injury, which in turn leads to a decrease in hematopoietic stem cells and failure to regenerate or replace lost cells.59,60
This study has some limitations, as the animal size was not calculated and justified.
Conclusion
According to the final data in this experiment, radiation-induced oxidative stress resulted in liver toxicity, decreased adropin levels and gene expression (Enho), and abnormal complete blood count. Furthermore, the experimental study showed a negative correlation between adropin and STAT3, TOS, IL-6, AST, ALT, cholesterol, T.G, LDL and glucose, while a positive correlation was observed between adropin and TAC and HDL. Therefore, we suggest the use of adropin to control the risks resulting from radiation exposure. This study has been approved by Zagazig University-Institutional animal care, number: ZU-IACUC/3/F/142/2024.
Footnotes
Acknowledgments
The authors are very grateful to the Egyptian Atomic Energy Authority for supporting the authors until the work has been carried out.
Author contributions
Nadia Abdel-Magied, Amira MK Abouelella, Hayam Mostafa, Hebatallah E. Mohamed and Nehal I. Hendy contributed to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
