Abstract
Background
Cyperus rotundus L., commonly known as nutsedge or purple nutgrass, is a resilient herb in the Cyperaceae family with notable pharmacological potential due to its diverse secondary metabolites. This study aimed to identify these metabolites and evaluate the antioxidant and enzyme inhibitory activities of the crude methanol extract of C. rotundus.
Methods
The crude methanol extract of C. rotundus was prepared, and its antioxidant activity was assessed using the DPPH scavenging assay. Additionally, in vitro inhibitory effects against the enzymes: α-amylase and α-glucosidase were evaluated. The secondary metabolites present in the extract were identified using ESI-MS/MS analysis.
Results
ESI-MS/MS analysis revealed the presence of various secondary metabolites, including rutin, quercetin, β-sitosterol, limonene, and beta-pinene. The crude methanol extract exhibited strong antioxidant activity and significant inhibition of α-amylase and α-glucosidase enzymes, which play crucial roles in carbohydrate digestion.
Conclusion
The findings indicate that the crude methanol extract of C. rotundus possesses notable antioxidant properties and the potential to inhibit key enzymes involved in carbohydrate metabolism. This study enhances our understanding of the bioactive compounds in C. rotundus and suggests its therapeutic applications for managing conditions related to carbohydrate metabolism.
Introduction
Cyperus rotundus L. is commonly known as purple nutgrass or nutsedge, which is a widespread and persistent weed belonging to the Cyperaceae family. It has thin, flaky crawling rhizomes that are swollen at the base and grow individually. The rhizomes can range from 1-3 cm in length and are accompanied by tubers. The tubers of C. rotundus L. are typically black on the outside or reddish white inside and have a distinctive smell. The nuts produced by the plant are oval shaped and three shaped when fully mature and exhibit a combination of black and yellow colors. C. rotundus L. was discovered in subtropical, temperate, and tropical climates worldwide, although it originated in India. 1 The rhizomes of C. rotundus L. have been traditionally used in Asian countries as folk remedies for various bowel and autoimmune conditions.2-4 It is considered a conventional herbal remedy with a wide range of uses, including sedation, analgesia, antimalarial, antispasmodic properties, and the treatment of stomach disorders.5,6 Moreover, C. rotundus L. tuber is one of the earliest known medicinal herbs used to address menstrual irregularities and dysmenorrhea. Decoctions made from this herb have been employed to treat fever, pain, and dysentery; act as an emmenagogue (stimulating menstrual flow); and alleviate other types of digestive discomfort. 7
C. rotundus contains a wide variety of chemical constituents, including flavonoids, alkaloids, sitosterol, starch, tannins, glycosides, monoterpenes, furochromones, sesquiterpenes, fatty oil, myristic acid, glycerol, stearic acid, and linolenic acid.8-12 The essential oil and rhizome extracts of C. rotundus contain several active components.13-18 The essential oil of C. rotundus is composed primarily of sesquiterpene hydrocarbons, monoterpenes, ketones, aliphatic alcohols, and epoxides, which contribute to the distinct flavor and aroma of the herb. Additionally, the herb contains the ketone cyperadione and the monoterpenes cineole, limonene, and camphene. C. rotundus also possesses various triterpenes, such as sitosterol and oleanolic acid, along with flavonoids, minerals, and sugars. 19
In this work, we first extracted C. rotundus and then analyzed the crude extract via ESI‒MS/MS, revealing the presence of various secondary metabolites. The phenolic acids in C. rotundus include 3-hydroxy-4-methoxybenzoic acid, ferulic acid, p-coumaric acid, caffeic acid, chlorogenic acid, and ellagic acid. Additionally, flavonoid compounds such as luteolin, quercetin, rutin, and catechin have been identified in plants. These findings suggest the potential medicinal properties of C. rotundus extracts, which contain a diverse range of secondary metabolites. These compounds contribute to the biological activity of C. rotundus and its potential therapeutic applications. In this study, we aimed to investigate the presence of secondary metabolites via ESI‒MS/MS analysis, evaluated the antioxidant activity of C. rotundus L. and explored its potential inhibitory effects on enzymes involved in the digestion of carbohydrates. By conducting in vitro enzyme inhibition assays, we assessed the ability of the plant extract to inhibit these enzymes. The findings of this study provide valuable insights into the secondary metabolite profile of C. rotundus L. and its potential therapeutic applications.
Materials and Methods
Chemicals and Reagents
Methanol, chloroform, acetone, n-hexane, n-butanol, potassium dihydrogen phosphate, Dragendorff’s reagent, potassium mercuric iodide (Mayer’s reagent), sodium nitroprusside, ferric chloride, gelatin solution, Conc. H₂SO₄, HCl, sodium hydroxide, acetic acid, acetic anhydride, nitric acid, potassium iodide (Wagner’s reagent), ninhydrin reagent, silica gel, 2,2-diphenyl-1-picrylhydrazyl (DPPH), butylated hydroxytoluene (BHT), porcine pancreatic amylase, starch, acarbose, α-glucosidase, 5,5′-dithiobis (2-nitrobenzoic acid) (DTNB), dimethyl sulfoxide (DMSO), phosphate buffer, and Tris-HCl of analytical grade were used in this study.
Plant Collection
C. rotundus L. whole plants were collected from the plant nursery at Faisalabad and identified by Dr Iqbal Hussain, Department of Botany, Government College University Faisalabad. The whole plant was dried in the shaded area at approximately 25 °C.
Extraction
This study was conducted from November 2021 to July 2022. The dried plants were crushed and sieved to obtain a fine powder, a total of 2 kg of this powder was collected for extraction, and the solvent used for extraction was pure methanol (15 L). First, 0.5 kg of plant powder is deposited within a jar; methanol (5 L) is immersed on top until the plant material is entirely coated. After that, the jar was sealed and retained for seven days. The contents were mixed regularly, but if they were deposited within a flask, they were mixed occasionally to achieve maximum extraction. The filtrate was extracted from the mixture via the filtration or decantation method with Whatman filter paper at the end of the extraction method. Afterward, the extract was collected from the solvent by evaporation through a rotary evaporator at 40 °C. The process was repeated three times to ensure that enough C. rotundus extract was collected. This process was repeated three times to ensure that enough C. rotundus extract was collected. To determine the percent yield of the extraction, the researchers used the following formula:
Fractionation
Fractionation was performed through a separating funnel method employing four distinct solvents: n-hexane, chloroform, ethyl acetate and n-butanol. The extracted mixture was introduced into the separating funnel and agitated to initiate the process. After settling, the layers were separated on the basis of solvent polarity. The least polar solvent, n-hexane, was added to an equal volume of water and stirred, and the resulting fraction was transferred to a small beaker. This step was carried out iteratively until no significant extract remained after the addition of n-hexane or stirring. The same sequential procedure was applied for the n-butanol, chloroform, and ethyl acetate fractions. Each solvent, added in equal volume to the mixture, was stirred and separated until no substantial extract remained in the separating funnel. Each fraction comprised compounds more soluble in the corresponding solvent, enabling detailed analysis and investigation of specific components. This process enhances comprehension of the chemical composition and potential bioactivity of the extract.
Column Chromatography
In this procedure, a column was used to separate and fractionate the extract on the basis of the polarity of the compounds. Silica gel was employed as the stationary phase, whereas a liquid, in this case, the mobile phase, was used to carry the extract through the column. The procedure began by packing 30 g of silica gel (specifically, 70/35 grade) into an 80 cm long and 5 cm diameter translucent glass column. Care was taken to avoid the introduction of air bubbles during the packing process. Once the column was prepared, the extract to be fractionated was introduced from the upper end of the column. As the mobile phase, the lowest polar solvent, n-hexane, was initially introduced into the column. The mixture was allowed to flow through and remain in the confined column for 1 hour to facilitate separation. At regular intervals, different sections of n-hexane were collected from the bottom of the column. Other solvents, namely, n-butanol, chloroform, acetone, and additional portions of n-hexane, were subsequently introduced into the column as separate mobile phases. Each solvent was allowed to flow through the column, and distinct sections were collected at different time intervals. These collected sections were then characterized and used for the identification and analysis of secondary metabolites present in the extract.
Sample Preparation for MS/MS Analysis
To characterize the secondary metabolites, an extract obtained from the n-butanol fraction of C. rotundus L. was used. Specifically, 1.5 mL of methanol and 500 µL of chloroform were used to dissolve the calculated amount of the extract. Once the extract was dissolved in the solvent mixture, 500 µL of this mixture was transferred into separate vials for further analysis. The ESI‒MS‒MS analysis was carried out via a C18 column (Agilent Zorbax Eclipse, 2.1 × 50 mm, 1.8 µm) with a methanol‒water gradient (5% to 95% methanol over 15 minutes) at a flow rate of 0.3 mL/min.
General Conditions for Sample Analysis by Mass Spectrometry

In vitro Characterization
Free Radical Scavenging Activity
We aimed to evaluate the free radical scavenging potential of C. rotundus extracts via the use of a DPPH radical scavenging biomarker. For this purpose, the free radical scavenging potential of the C. rotundus extract was evaluated via the use of a DPPH radical-scavenging biomarker. The ability of the plant extract to donate hydrogen atoms, an indicator of its antioxidant capacity, was determined by measuring the decolorization of a methanol solution of DPPH. First, a stock solution of 0.1 mM DPPH was prepared in methanol. Additionally, a stock solution of C. rotundus extract was prepared in methanol. The extract was prepared at various concentrations ranging from 12.5 to 150 µg/mL. In the experiment, 2.4 mL of the DPPH stock solution was combined with 1.6 mL of the C. rotundus extract at specified concentrations. The resulting mixture was thoroughly mixed by vortexing and then kept in a dark environment at room temperature for 30 minutes. After the incubation period, the absorbance of the resulting mixture was measured at a wavelength of 517 nm via a microplate reader, specifically the BioTek Elx800 TM model. To evaluate the percentage of DPPH radical scavenging activity, the following formula was used:
The percentage of inhibition was calculated via absorbance values. A0 represents the absorbance of the control (without extract), whereas A1 represents the absorbance of the extract or standard (in this case, often BHT, a reference compound). The experiment was performed in triplicate at each concentration to ensure accuracy and reproducibility. Finally, the percentages of inhibition values obtained at different concentrations were plotted to generate a graph. The IC₅₀ value, which represents the concentration of the extract required to scavenge 50% of the DPPH radicals, can be determined from the graph.
Determination of the in vitro Inhibitory Activity of α-amylase
The following steps were used: 0.5 M Tris-HCl buffer solution with a pH of 6.9 was used. To make the substrate solution, 0.01 M CaCl₂ (0.2 mL) was added to 2 mg of starch. The substrate solution was transferred into test tubes. The test tubes were boiled for 5 minutes, while the other tubes were preincubated at 37 °C for 5 minutes. Two grams of C. rotundus extract was dissolved in 50 mL of DMSO. The extract solution was prepared at different concentrations ranging from 1 to 1000 µg/mL. A total of 0.2 mL of C. rotundus extract solution at various concentrations was added to the test tubes containing the substrate solution. Porcine pancreatic amylase (1 mL in Tris-HCl buffer at a concentration of 2 U/mL) was then added to initiate the reaction. The reaction mixture was incubated at 37 °C for 10 minutes. After 10 min, the reaction was stopped by adding 0.5 mL of 50% acetic acid to each test tube. The mixture was then centrifuged at 4 °C and 3000 rpm for 5 minutes to remove any precipitate. The optical density of the supernatant was determined at a wavelength of 595 nm via a microplate reader. The equation is as follows:
A1 represents the absorbance of the sample (C. rotundus extract at various concentrations).
This was accomplished by establishing the α-amylase inhibitory effect for multiple dilutions and determining the concentration at which 50% inhibition occurred. The entire process was repeated in triplicate for each concentration of the C. rotundus extract to ensure the accuracy and reproducibility of the results. This method allowed for the evaluation of the α-amylase inhibitory potential of the extract and comparison with the control compound, acarbose.
Determination of the in vitro Inhibitory Activity of α-glucosidase
In this method, the objective was to determine the α-glucosidase inhibitory effect of the C. rotundus extract. The following steps were used: the C. rotundus extract was dissolved in 50 mL of methanol. A total of 50 µL of the C. rotundus extract solution was taken at various concentrations ranging from 1 to 1000 µg/mL. The extract solution was mixed with a solution containing 10 µL of α-glucosidase (maltase 1 U/mL) and 125 µL of 0.1 M PBS (pH 6.8). The mixture was incubated for 20 minutes at 37 °C. To start the reaction, 20 µL of 1 M pNPG (4-nitrophenyl-β-D-glucopyranoside) substrate was added to the mixture. The reaction mixture was further incubated for 30 minutes. To stop the reaction, 50 µL of 0.1 N Na2CO3 was added to the mixture. The optical density of the resulting solution was measured at 405 nm via an ELISA reader. The equation is as follows: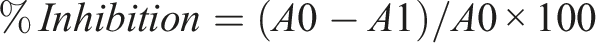
A0 represents the absorbance of the control (100% enzyme effect).
A1 represents the absorbance of the sample (C. rotundus extract at various concentrations).
The IC₅₀ values, which indicate the concentration required to inhibit 50% of the α-glucosidase, were determined for both the acarbose (a control sample and an α-glucosidase inhibitor) extract and the C. rotundus extract. This was accomplished by establishing the α-glucosidase inhibitory effect for multiple dilutions and determining the concentration at which 50% inhibition occurred. The entire process was repeated three times for each concentration of the C. rotundus extract to ensure the accuracy and reproducibility of the results. Acarbose was used as a control sample in this method.
Statistical Analysis
For the data analysis, the results are presented as the means ± SDs. The mean readings were calculated on the basis of data obtained from a minimum of three replications. Statistical significance was assessed via P values. A threshold of 0.05 or less (P < 0.05) was considered statistically significant. This means that if the calculated P value was equal to or less than 0.05, the observed differences or effects were considered statistically significant, indicating that they were unlikely to occur by chance alone.
Results
ESI‒MS/MS Analysis
ESI‒MS/MS analysis of the n-butanol fraction of C. rotundus revealed the presence of various secondary metabolites. Among these compounds, essential oils, terpenoids, flavonoids, sitosterol, sesquiterpenes, cyperol, cyperene, nootkatone, and valencene were identified as significant phytochemicals in C. rotundus L. These compounds are known for their antioxidant and free radical scavenging properties.
Analysis of the Positive Mode of Full-MS
The n-butanol fraction of the C. rotundus extract was analyzed via full MS, and the results revealed the presence of numerous secondary metabolites (Figure 1). The full MS spectrum displayed multiple peaks, each corresponding to different compounds identified in the extract. The presence of these diverse secondary metabolites in the n-butanol fraction of the C. rotundus extract suggests the potential bioactive and pharmacological properties associated with this plant. Each compound may contribute to the overall therapeutic potential of the extract and could be further investigated for its specific effects and applications. ESI‒MS/MS spectrum of positive mode full-MS (50--2000) revealing the presence of secondary metabolites ((-)-pelletierine, chrysoeriol, (2Z,5Z,8Z,11Z,14Z,17Z)-icosa-2,5,8,11,14,17-hexaenoic acid, ptaeroxylol, rogeasin, koparin, (3R,5 R)-1-(4-hydroxyphenyl)-7-phenyl-3,5-heptanediol, alnusin, 28-Norurs-12-en-3beta-ol, pteroside Q, luteolin-4'-O-glucoside, chrysanthemin, ureferin, 5,7,2',6'-tetrahydroxyflavone 2'-O-glucoside 8-C-beta-D-glucopyranosylkaempferol, pectolinarigenin 7-glucuronide, luteolin 3',4'-dimethyl ether 7-glucuronide, hemibrevetoxin B, etc.).
ESI‒MS/MS Analysis
The secondary metabolites detected at 236.17 m/z were cyperadione, chrysanthediol B, 5-hydroxylucinone, 2β-(5-oxypentyl)-2β-methyl-5β-isopropenylcyclohexanone, 2α-(5-oxopentyl)-2β-methyl-5β-isopropenylcyclohexanone, cyperolone, van derol 4α, and cyperusol A₂ (Figure 2). ESI‒MS/MS spectrum of full MS2 at 236.00, revealing the presence of secondary metabolites such as cyperadione, chrysanthediol B, n-butyl-β-D-fructopyranoside, 5-hydroxylucinone, cyperolone, 2β-(5-oxypentyl)-2β-methyl-5β-isopropenylcyclohexanone2α-(5-oxopentyl)-2β-methyl-5β-isopropenylcyclohexanone, Vellerdiol 4α, 5α-Oxido eudesm-11-en-3α-ol and Cyperusol A₂.
In addition, ESI‒MS/MS analysis of 391.20 ions (m/z ratio) revealed the presence of piceid, with a peak observed at 391.33 (Figure 3). Piceid has a chemical formula of C₂₀H₂₂O₈. The main fragment peak of piceid appeared at 373.25, which was formed by the elimination of an OH group. This fragmentation pattern provides insight into the structure of piceids and aids in their identification. ESI‒MS/MS spectrum of full MS2 at 391.20 m/z, revealing the presence of the secondary metabolite piceid.
Moreover, ESI‒MS/MS analysis of 413.50 ions (m/z ratio) revealed the presence of β-sitosterol and behenic acid monoglyceride, with a peak observed at 413.33 (Figure 4). β-Sitosterol has a chemical formula of C₂₉H₅₀O, whereas behenic acid monoglyceride has a formula of C₂₅H₅₀O₄. The main fragment peak of these compounds appeared at 301.17, which was formed by the elimination of the circled part of the β-sitosterol and behenic acid monoglyceride structures. This fragmentation pattern aids in the characterization and identification of β-sitosterol and behenic acid monoglycerides. Overall, ESI‒MS/MS analysis provides valuable information about the fragmentation patterns and structural features of the detected compounds, including Britanlin E, piceid, β-sitosterol, and behenic acid monoglyceride. This analysis contributes to the understanding of their chemical composition and aids in their identification within the C. rotundus extract. ESI‒MS/MS spectrum of the full MS2 spectrum of 413.33 v secondary metabolites, including β-sitosterol and behenic acid monoglyceride.
Analysis of the ESI‒MS/MS Spectrum in Negative Mode
ESI‒MS/MS analysis in negative mode revealed the presence of various secondary metabolites in the sample (Figure 5). Multiple peaks were observed in the full-MS spectrum, indicating the presence of different compounds. Among these peaks, several secondary metabolites were identified, including limonene, beta-pinene, alpha-pinene, sabinene, salicylic acid, terpinolene, p-hydroxybenzoic acid, camphene, (R)-(+)-limonene, p-coumaric acid, retinal, methyl stearate, and myristic acid. These compounds are known for their diverse biological activities and are commonly found in plants. Limonene, β-pinene, and α-pinene are monoterpenes known for their aromatic properties. Sabinene is another monoterpene with antimicrobial and antioxidant properties. Salicylic acid is a phenolic compound recognized for its anti-inflammatory and analgesic effects. Terpinolene is a terpene that contributes to the aroma of many plants and possesses potential antioxidant properties. P-hydroxybenzoic acid is a phenolic acid with antioxidant and antimicrobial properties. Camphene is a monoterpene found in many essential oils, and it has been studied for its potential antimicrobial and antioxidant activities. (R)-(+)-Limonene is an enantiomer of limonene that shares similar properties. P-coumaric acid is a phenolic acid known for its antioxidant and anti-inflammatory effects. The retina is a derivative of vitamin A that plays a crucial role in vision. Methyl stearate and myristic acid are fatty acid derivatives that are commonly found in various plant oils. The presence of these secondary metabolites indicates the complex chemical composition of the sample and highlights the potential bioactive compounds present in the C. rotundus extract. The identification of these metabolites provides valuable information for further research on their potential pharmacological activities and health benefits. ESI‒MS/MS spectrum in full MS negative mode (50-2000) revealing the presence of secondary metabolites, including limonene, β-pinene, α-pinene, sabinene, salicylic acid, terpinolene, p-hydroxybenzoic acid, camphene, (R)-(+)-limonene, p-coumaric acid, retinal, methyl stearate and myristic acid.
ESI‒MS/MS Analysis by Fragmentation Technique
Myristic acid has a chemical formula of C₁₄H₂₈O₂, and its molecular weight is 228.37 g/mol. ESI‒MS/MS analysis in negative mode revealed a characteristic peak at 227.30 m/z, indicating the presence of myristic acid. The main fragment peak observed at 183.08 m/z was formed by the elimination of the COOH group from the myristic acid molecule (Figure 6). Full-scan MS2 spectrum of 227.30, revealing the presence of myristic acid.
Moreover, the MS/MS analysis at 283.30 m/z demonstrated the presence of retinal at the peak of 283.08. The main fragment peak observed at 162.92 m/z was formed by the elimination of the circled part from the retinal compound (Figure 7). Finally, the ESI‒MS/MS analysis at 297.00 m/z revealed the presence of methyl stearate at the peak of 297.17. The main fragment peak observed at 183.00 m/z was formed by the elimination of the circled part from the methyl stearate compound (Figure 8). These ESI‒MS/MS analyses provide valuable information about the fragmentation patterns and structural characteristics of the identified secondary metabolites. By understanding the fragmentation behavior of these compounds, it becomes possible to identify and confirm their presence in the sample. This knowledge contributes to the overall characterization and understanding of the chemical composition of the sample, allowing further investigations into the potential biological activities and applications of these compounds in various fields. ESI‒MS/MS spectrum of the full-scan MS2 of 283.30 revealing the presence of the secondary metabolite retinal. ESI‒MS/MS spectrum of full-scan MS2 297.17 revealing the presence of the secondary metabolite methyl stearate.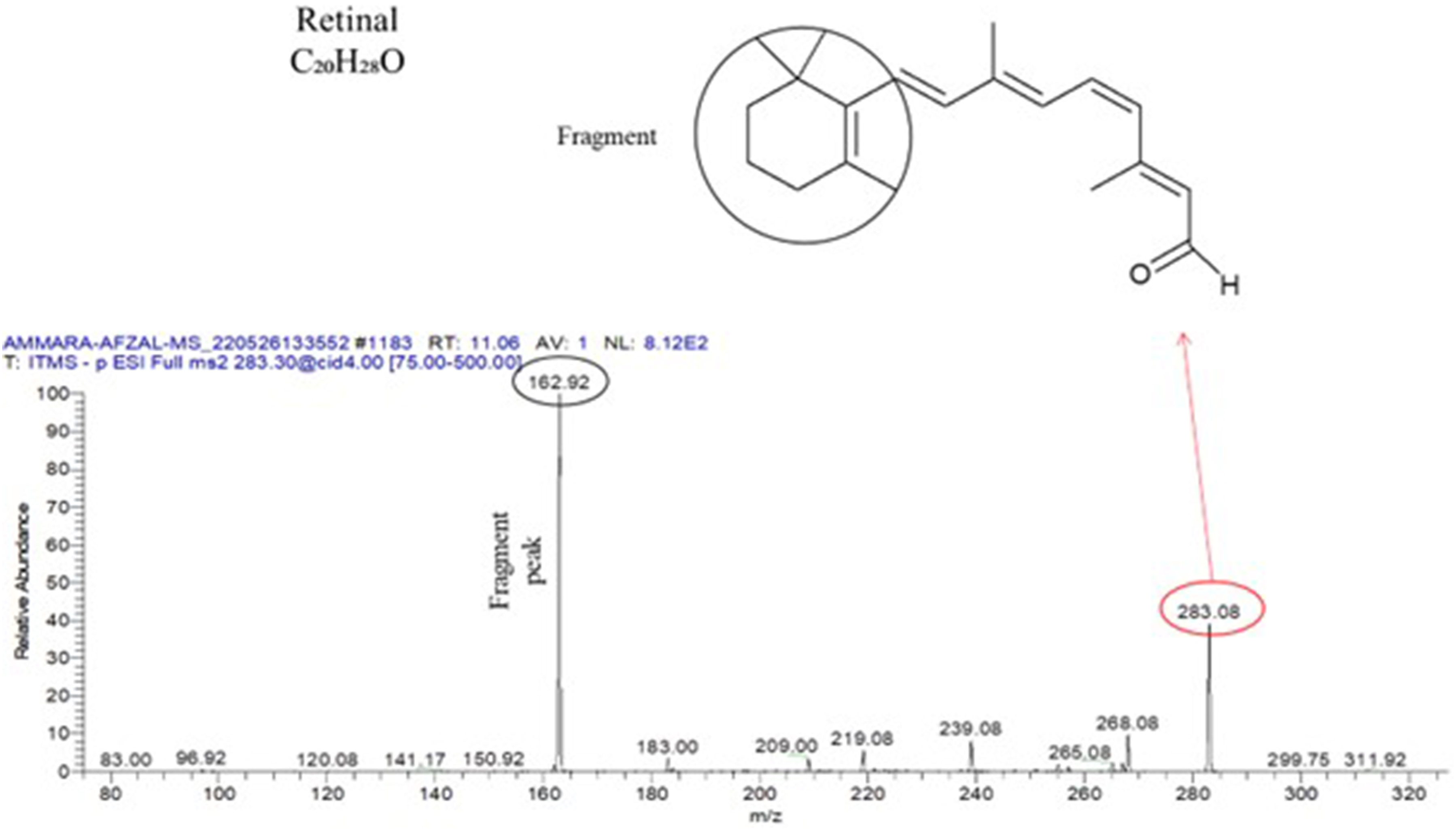
DPPH Activity
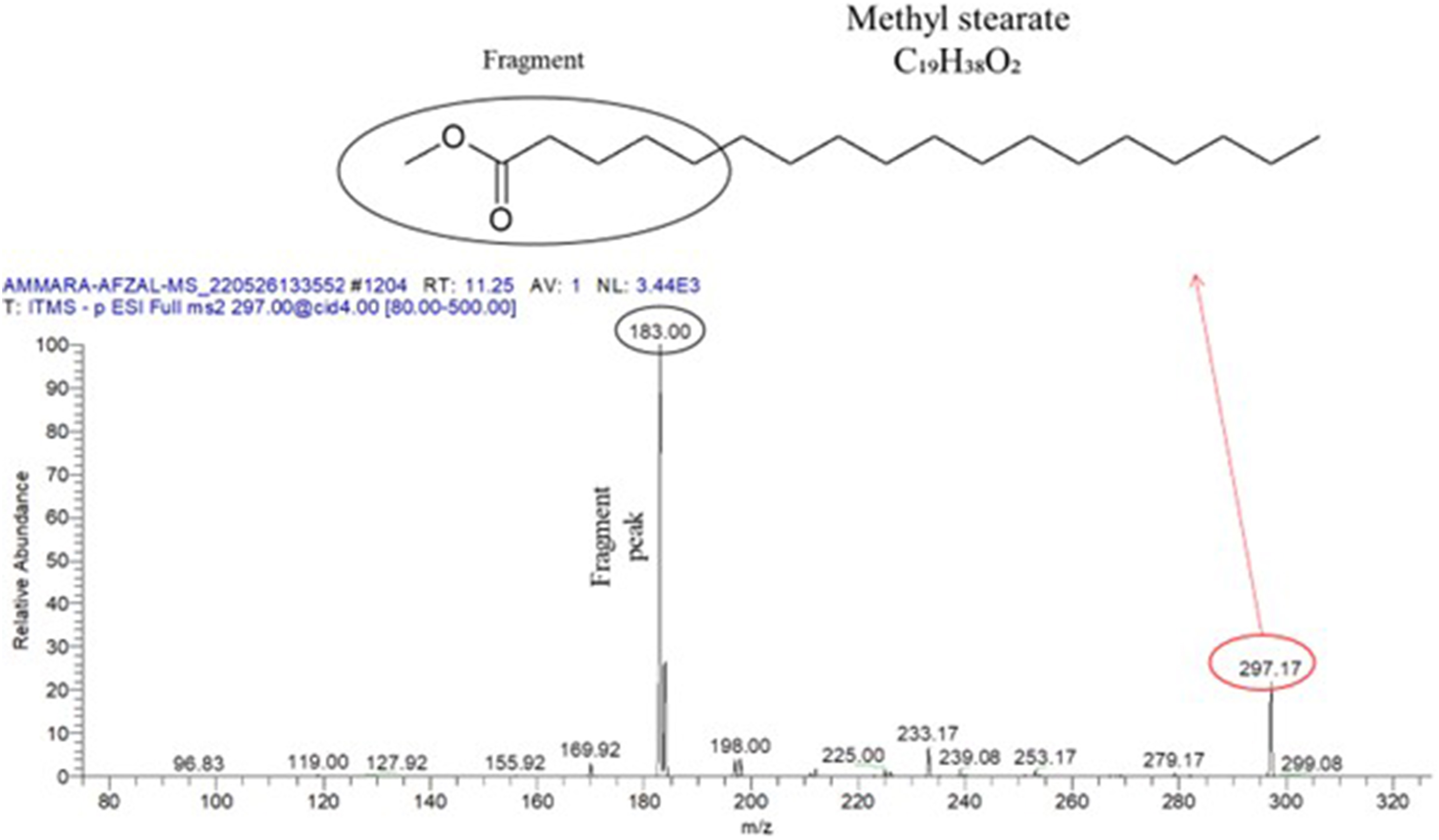
For the C. rotundus extract, a DPPH assay was conducted to evaluate its free radical scavenging potential. The results indicated that compounds with lower IC₅₀ values possess greater antioxidant properties. At a concentration of 160 µg/mL, the free radical scavenging activity of the C. rotundus extract was 86.67 ± 0.058%, whereas that of BHT was 71.42 ± 0.06%. The IC₅₀ value, which represents the concentration of the compound required to scavenge 50% of the DPPH radicals, was determined to be 25.92 µg/mL for the C. rotundus extract and 16.88 µg/mL for BHT. These values indicate that, compared with the C. rotundus extract, BHT was more potent at scavenging DPPH radicals (Figure 9(A)). DPPH free radical scavenging assay (A) of the CR extract at different concentrations, such as 2.5, 5, 10, 20, 40, 80, and 160 µg/mL, and determination of the α-amylase (B) and (C) α-glucosidase activity of the C. rotundus extract and acarbose (standard) at various concentrations, such as 1, 3, 7, 15, 31, 62, 125, 250, 500, and 1000 µg/mL.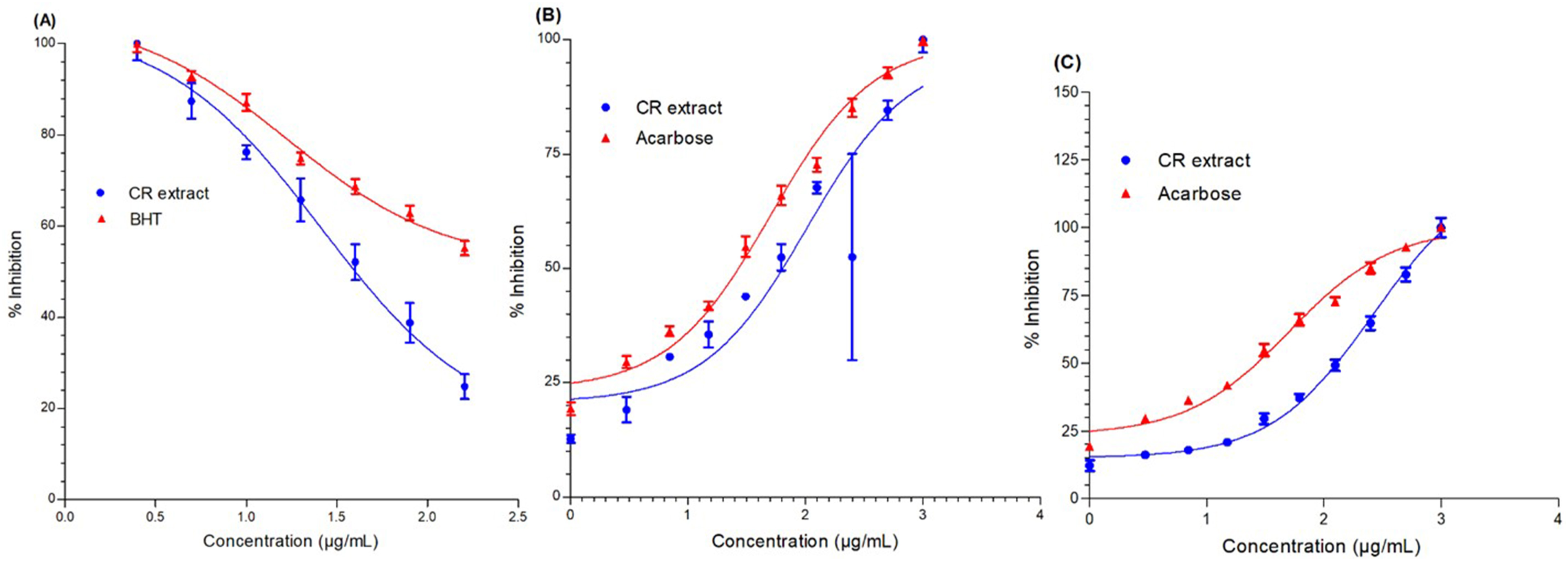
In vitro Assay for α-amylase Inhibition
Figure 9(B) shows a comparison of the inhibitory effects of the C. rotundus extract and acarbose (a known α-amylase inhibitor) on enzyme activity. The inhibition observed appeared to be dose dependent, indicating that the potency of the extract directly influenced the extent of enzyme inhibition. The determined IC₅₀ value, which represents the concentration required to inhibit α-amylase activity by 50%, indicated that compared with the C. rotundus extract, acarbose resulted in greater α-amylase inhibition. The IC₅₀ value for acarbose was measured to be 50.47 µg/mL, whereas the C. rotundus extract had a higher IC₅₀ value of 102.3 µg/mL. This suggests that the plant extract may contain impurities or other components that contribute to a lower potency in inhibiting α-amylase activity. The higher IC₅₀ value for the C. rotundus extract implies that it was less effective in inhibiting α-amylase than was acarbose.
In vitro Assay for α-glucosidase Inhibition
C. rotundus L., also known as C. rotundus, is widely used in Ayurvedic medicine for the treatment of diabetes and other related disorders. The α-glucosidase inhibitory activity of the extract obtained from C. rotundus was evaluated, which is relevant for managing postprandial blood sugar levels. The IC₅₀ value, which represents the concentration required to inhibit 50% of the α-glucosidase activity, was compared to that of acarbose, a known α-glucosidase inhibitor. The results indicate that the C. rotundus extract exhibited mild α-glucosidase inhibitory behavior, as evidenced by its higher IC₅₀ value than that of acarbose (Figure 9(C)). The IC₅₀ value of the extract was significantly greater than that of acarbose, suggesting that acarbose was more potent in inhibiting α-glucosidase activity. In other words, the extract of C. rotundus was less effective at inhibiting the enzyme responsible for breaking down complex carbohydrates into simple sugars than the reference drug, acarbose. These findings imply that while C. rotundus extract may possess some level of α-glucosidase inhibitory activity, it is relatively mild compared with the standard medication acarbose. However, the potential therapeutic benefits of this extract in the context of diabetes and other disorders may extend beyond its α-glucosidase inhibitory activity alone, as it is commonly used in Ayurvedic medicine because of its overall medicinal properties.
Discussion
C. rotundus is recognized for its phytochemical constituents. Studies have confirmed the presence of phenolic compounds, flavonoids, terpenoids, and steroidal compounds in C. rotundus extracts,19,20 which are linked to the following pharmacological activities: antioxidant, anti-inflammatory, antimicrobial, and anticancer effects. MS/MS analysis revealed the presence of diverse secondary metabolites in the C. rotundus extract, revealing its rich phytochemical profile. The detected compounds included essential oils, terpenoids, flavonoids, sitosterol, sesquiterpenes, cyperol, cyperene, nootkatone, and valencene, which is consistent with prior studies. The abundance of phenolics and flavonoids, which are known for their antioxidant and radical scavenging properties, offers potential health benefits. Phenolic acids (e.g., 3-hydroxy-4-methoxy-benzoic acid, ferulic acid, loganic acid, p-coumaric acid, caffeic acid, chlorogenic acid, and ellagic acid) indicate the diverse phenolic contents of the extract. Flavonoids (e.g., luteolin, quercetin, rutin, and catechin) further enhance flavonoid composition. These findings align with previous C. rotundus studies highlighting phenolics and flavonoids. 21 These compounds are associated with biological activities such as antioxidant, anti-inflammatory, antimicrobial, and anticancer effects.
MS/MS qualitative analysis revealed additional compounds in the C. rotundus extract. These diverse compounds emphasize the complex nature of secondary metabolites in C. rotundus extracts. This wide range suggests potential bioactivity and health benefits. These compounds exhibit various pharmacological activities, such as antioxidant, anti-inflammatory, antimicrobial, and antitumor effects, 22 which aligns with the traditional uses of C. rotundus in folk medicine. Full MS/MS analysis of the n-butanol fraction revealed numerous secondary metabolites, indicating the complex phytochemical composition of C. rotundus. Each peak in the full MS/MS spectrum corresponds to a different identified compound, providing valuable insights into the diversity and potential bioactivity of secondary metabolites in the n-butanol fraction. These diverse secondary metabolites in the n-butanol fraction of the C. rotundus extract indicate potential bioactive and pharmacological properties. Each compound has a unique structure and may possess distinct biological activities. Studies have linked these compounds to antioxidant, anti-inflammatory, antimicrobial, anticancer, and neuroprotective effects. 21 Negative mode full MS/MS analysis of the C. rotundus sample revealed various secondary metabolites, revealing its complex chemical composition. These metabolites provide valuable insights into potential bioactive compounds and their pharmacological activities.23,24
The DPPH assay was used to evaluate the free radical scavenging potential of the C. rotundus extract in this study. The results demonstrated the significant scavenging activity of the extract. These findings indicate that, compared with the C. rotundus extract, BHT was more potent at scavenging DPPH radicals. The higher IC₅₀ value for the extract suggests that it may have lower efficacy in inhibiting DPPH radicals than the standard antioxidant BHT does. Notably, the DPPH assay provides a general indication of antioxidant activity and may not fully reflect the overall antioxidant capacity or potential health benefits of the extract. Further research is necessary to investigate the specific mechanisms and bioactive compounds responsible for the observed antioxidant activity of C. rotundus extracts.
In the α-amylase inhibition in vitro assay, the ability of the C. rotundus extract to inhibit the activity of the α-amylase enzyme involved in starch digestion was tested. The results demonstrated dose-dependent inhibition by the extract, with higher concentrations resulting in greater effects. However, the IC₅₀ value for the C. rotundus extract (102.3 µg/mL) was greater than that of acarbose (50.47 µg/mL), a known α-amylase inhibitor, indicating that acarbose exhibited stronger inhibition. Similarly, in the α-glucosidase inhibition in vitro assay, the ability of the C. rotundus extract to inhibit the α-glucosidase enzyme involved in carbohydrate breakdown was assessed. The IC₅₀ value of the extract was significantly greater than that of acarbose, indicating that acarbose was more potent. These findings suggest that the C. rotundus extract has mild α-amylase and α-glucosidase inhibitory activities, but its potency is lower than that of acarbose. Importantly, the therapeutic potential of C. rotundus in diabetes and related disorders may extend beyond α-amylase and α-glucosidase inhibition alone. This extract is widely used in Ayurvedic medicine for its overall medicinal properties, warranting further research to explore its comprehensive effects and mechanisms of action in these diseases.
This study has certain limitations. First, the methanol extract of C. rotundus contains a complex mixture of compounds, making it difficult to identify the specific bioactive components responsible for its antioxidant and enzyme inhibitory effects. Second, while in vitro assays for α-amylase and α-glucosidase inhibition were conducted, their relevance to in vivo conditions remains uncertain due to the lack of pharmacokinetic data. Third, the extract’s lower potency compared to acarbose raises concerns about its clinical effectiveness without further refinement. Additionally, toxicity and long-term safety assessments were not performed, limiting conclusions on its therapeutic potential. Further research is required to isolate active compounds, evaluate in vivo efficacy, and ensure safety.
Conclusion
The C. rotundus extract contained significant levels of secondary metabolites, including flavonoids, phenolic acids, and terpenoids, which are known for their antioxidant and free radical scavenging properties. Furthermore, the inhibitory effects of the extract on key enzymes involved in digestion, such as α-amylase and α-glucosidase, were evaluated. The results demonstrated that the extract exhibited moderate α-amylase inhibitory activity, although acarbose displayed greater potency in this regard. Similarly, the extract showed mild α-glucosidase inhibitory behavior compared with the reference drug acarbose. While the extract may not exhibit the same level of inhibitory activity as acarbose does, it is important to consider that the C. rotundus extract is traditionally used in Ayurvedic medicine because of its holistic medicinal properties. Therefore, the extract may offer additional therapeutic benefits beyond its specific enzyme inhibitory effects. Overall, these findings contribute to the understanding of the potential pharmacological properties of C. rotundus extracts and support their traditional use in Ayurvedic medicine. Further research is warranted to explore the mechanism of action of the extract, identify its active components, and investigate its potential for clinical application in the management of various disorders, including diabetes.
Footnotes
Acknowledgments
The authors acknowledge and extend their appreciation to the Researchers Supporting Project Number (RSPD2024R1005), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

