Abstract
Introduction
Rheumatic ailments as well as chronic degenerative ailments that affect tissues and joints are associated with constant, recurring, or indeed persistent pain, inflammatory condition, and physical disability. 1 They harm the locomotor mechanism that contributes to impairment of the patient which impacts healthcare, social services, and reduces the individual’s living standard2,3 that also influence about 30 to 40 % population in Europe. 4 Such abnormalities include muscle pain, weakness, limited mobility, inflammation, or even diminished standard of living are associated with this condition. The incidence of rheumatic illnesses including muscle pain varies approximately 14 and36 % in advanced countries. 5 Treatment of inflammatory rheumatic conditions that consist of synthetic disease modifying antirheumatic drugs (sDMARDs) and glucocorticoids is generally effective as well as aimed at improving the illness. These commercially available treatment options are just causing economic exploitation and are also associated with severe adverse effects.6,7 For example, methotrexate also produces ulcers of the mouth. 8 For that reason, the herbal medicine or natural medicine, having plant origin, may play effectual role in the treatment of arthritis and other inflammatory conditions.6,7
Furthermore, because of the efficacy and cost-effectiveness of herbal medicines, people from developing countries such as India, Bangladesh, and Pakistan have relied on conventional medicinal systems to treat several ailments as alternate for healthcare. Local practice of medicinal herbs for treating numerous sicknesses is really popular in various areas of Pakistan. 9 The developing world today is indeed prone towards alternative and complementary medicines originating primarily from natural sources. Over one quarter of certified synthetic drugs are initially expected to have been extracted from herbs. 10 Herbs for medical purposes are being used throughout history as a treatment option for pain relief. 11
There are many medicinal plants which are being used traditionally in clinical practice to treat arthritic conditions, for example, Azadirachta indica (leaves), Pipper nigrum (seeds), Zingiber officinale (rhizomes), Withania somnifera (arial parts) etc. 12 Colchicum autumnale (Colchicum) is a perennial herb commonly known as Colchicum or Suranjan tulkh which belongs to the family Colchicaceae and have alkaloids mainly colchicine, colchicoresin, demo-colchicine those which have anti-inflammatory, analgesic, phlegmagogue effects and therefore commonly used to treat arthralgia, gout, headache etc. 13 Strychnos nux-vomica (Nux-vomica) belongs to Loganiaceae family having various alkaloids (brucine and brucine N-oxide) traditionally used for inflammatory diseases including arthritis. 14 Aloe barbadensis (Aloe-vera) commonly called Aloe-vera belongs to the family Asphodelaceae have many valuable active constituents like vitamins, enzymes, anthraquinone glycosides, phenols, sugars, amino acids, hormones, and salicylic acid. Pharmacological properties of aloe-vera are widely acknowledged worldwide which includes wound healing, anti-aging, anti-inflammatory, antioxidant, and immuno-modulatory effects. 15
Therefore, the present work was focused to evaluate the in-vitro anti-arthritic and anti-inflammatory activities of Colchicum (Colchicum autumnale), Nux-vomica (Strychnous nux-vomica), and Aloe-vera (Aloe barbadensis). Primarily, the aqueous-ethanolic extracts of these plants were phytochemically screened followed by Fourier Transform Infrared (FTIR) analysis.
Material and Methods
Procurement of Plant
Corms of Colchicum and seeds of Nux-vomica were collected from the local market of Bahawalpur and leaves of Aloe-vera were collected from the lawns of Khwaja Fareed Campus, the Islamia University of Bahawalpur, identified from the Department of Pharmacognosy, the University of Karachi. Voucher numbers for identified plant specimens were A-132, A-134, and A-131. The study was approved by the ethical committee of University College of Conventional Medicine, Faculty of Medicine and Allied Health Sciences, the Islamia University of Bahawalpur (1460/UCCM Dated 23-04-2019).
Extraction Process
The collected plants materials were washed with double distilled water and dried under shade. The seeds of Nux-vomica were immersed in excess of water for 5 days, to make them detox, in milk for 2 days followed by their boiling in milk, for its detoxification. Colchicum and Nux-vomica were grinded and weighted in specific proportions to sock in ethanol for making 70:30 aqueous ethanolic extract. The filtrate was placed for 72 hours with occasional stirring. Freshly cut leaves of Aloe-vera were taken and the epidermis was removed with knife. Pulp was separated and small pieces were added into ethanol and distilled water in specific proportions and agitated on sonicator for one hour followed by filtration, first with mucilin cloth, second with Whattman’s filter paper grade no. 01. For preparation of extract, evaporation of ethanol was done with the help of rotary evaporator with revolution 70 rpm and temperature 45°C. Aloe-vera filtrate was lyophilized and the extract was placed at room temperature for three to 4 days for desiccation. Dried extracts were placed in jars and preserved for later analysis.
Preliminary Phytochemical Analysis of Plant Extracts
Preliminary qualitative phytochemical screening was carried out to identify the active constituents of plant extracts such as alkaloids, flavonoids, cardiac glycosides, terpenoids, carbohydrates, protein, fats, steroids, phenols, saponins, tannins, and anthraquinones with the following methods Dragondroff’s test, lead acetate test, Legal’s test, copper acetate test, Molish and Benedicts test, ninhydrin test, Salkowski test, ferric chloride test, froth test, and gelation test, respectively.16,17
Fourier Transform Infrared
Dried extracts of Colchicum, Nux-vomica, and Aloe-vera were inserted into the spectroscope (Shimadzu, Japan) having a scanning range 400-4000 cm−1 along with a resolution of 4 cm−1.
In Vitro Anti-Arthritic Activity by Protein Denaturation Method
In vitro evaluation of anti-arthritic activity by protein denaturation method was assessed as previously described methods.18,19 The reaction mixture was prepared by adding egg albumin 0.2 mL, phosphate buffer saline (PBS, pH 6.4) 2.8 mL, and different concentrations of plant extracts or Diclofenac sodium 2 mL (200 µg, 400 µg, and 600 µg/mL) to make total volume up to 5 mL. Distilled water 2 mL, buffer 2.8 mL, and 0.2 mL albumin were taken as control. The mixture was incubated at 37°C for 15 minutes and further heated at 70°C for 5 minutes. Absorption was taken at 660 nm while using spectrophotometer followed by blank. The inhibitory percentage was analyzed by the under given formula.
Percentage inhibition = [Abs control – Abs sample ÷ Abs control] x 100 where Abs stands for absorption.
In-Vitro Anti-Inflammatory Activity by Human Red Blood Cell Membrane Stabilization Method
In-vitro anti-inflammatory activity by HRBC membrane stabilization method as reported previously with little modifications were carried out. 20 Blood was taken from healthy human volunteers and Alsever solution was added in equal volume. Blood was centrifuged at 3000 rpm for 30 minutes. 10% V/V RBC suspension was prepared with iso-saline solution. Plasma was decanted and packed cells were washed with iso-saline. Reaction mixture is prepared by adding phosphate buffer, RBC suspension, hypotonic saline solution, and different concentrations of plant extracts (125, 250, and 500 µg/mL). Total volume = 3 mL. Same is prepared for different concentrations of Diclofenac sodium. Control is 2 mL distilled water with phosphate buffer and RBC suspension. The reaction mixtures are incubated at 37°C for 30 min. Centrifugation was done at 3000 rpm for 20 min. Absorption was taken at 570 nm from UV visible spectrophotometer and spectra were taken. Percentage protection was calculated by following formula 19
Percentage protection = 100 – [(absorption sample ÷ absorption control) x 100]
Statistical Analysis
The data was expressed as Mean with standard error of Mean (SEM), analyzed by SPSS version 20. One-way analysis of variance (ANOVA) was performed by using post-hoc Tukey test.
Results
Phytochemical Screening
Summary of Phytochemical Analysis of Medicinal Plants.
Fourier Transform Infrared Analysis
Colchicum
Figure 1 shows the FTIR spectra of aqueous-ethanolic extract of Colchicum. The peaks at 3959.89 to 3730.20 show the presence of organic acids (–COOH group). The peak at 3340.92 and 3155.48 refers to the presence of amine group (NH2). The peak from 2923.46 to 2169.72 denotes the presence of aromatic compounds with C–H bonds or aliphatic compounds with –CH bonds. The peaks from 1943.13 to 1587.99 show the presence of ester bonds which can be C = N, C = C or O. The peaks at 990.3 to 628.94 refer to the presence of halogens with carbon group C–X. Fourier Transform Infrared spectra of aqueous-ethanolic extract of Colchicum autumnale.
Nux-vomica
Figure 2 shows the spectra of aqueous-ethanolic extract of Nux-vomica. The peak at 3896.13 and 3615.04 shows the presence of alcoholic groups (-OH group) with phenolic compounds. The peak at 3401.04 and 3356.82 shows the presence of NH2 (amine group). The peak from 3123.38 to 2772.89 denotes the presence of aromatic compounds with –CH bonds or aliphatic compounds with –CH bonds. The peak from 1933.20 to 1516.42 is referred as ester peaks (C=N, C=C, O). The peak from 1098.06 to 628.17 shows the presence of halogen with carbon group (C–X). Spectra of aqueous-ethanolic extract of Strychnous Nux-vomica.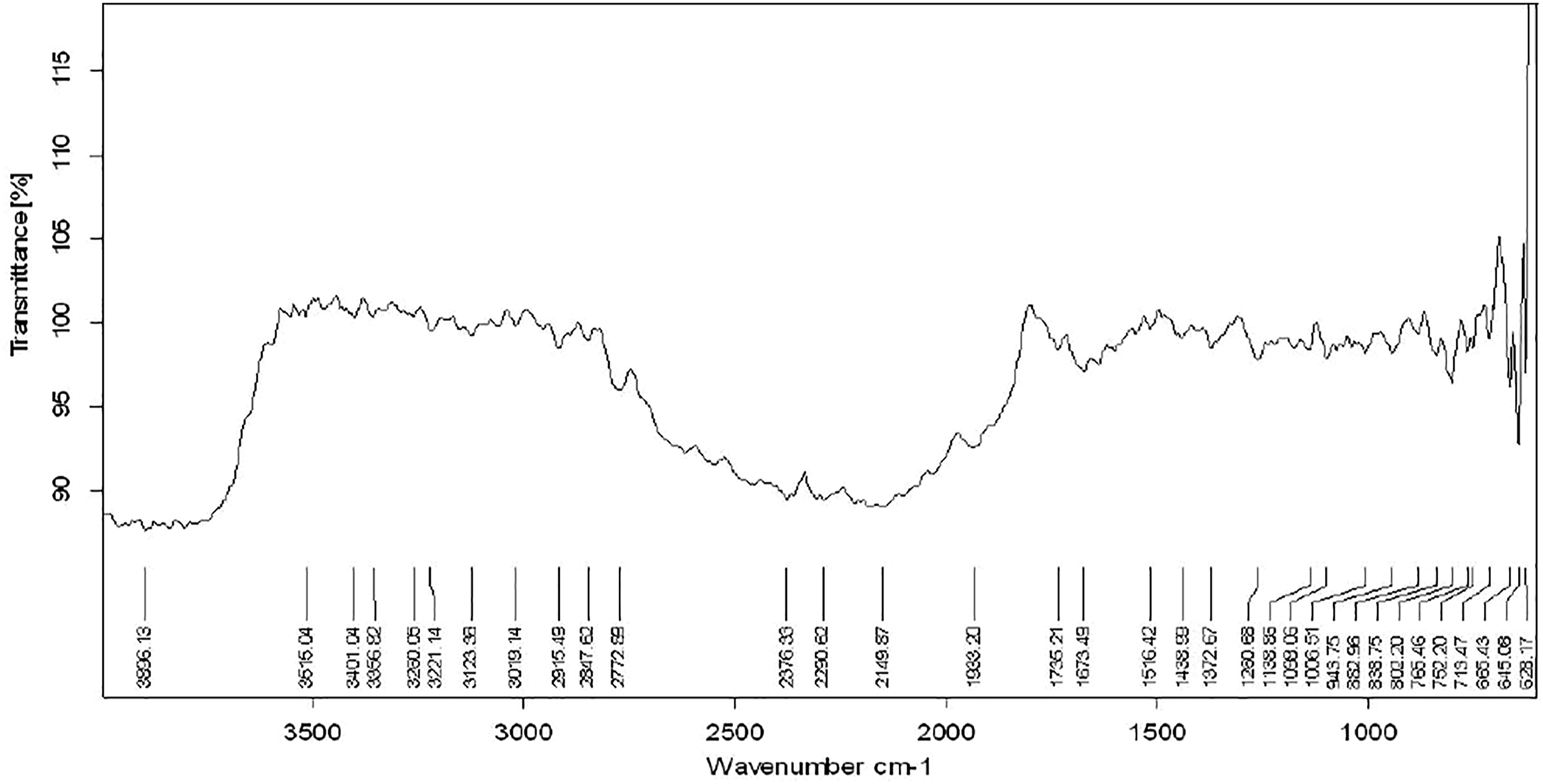
Aloe-vera
Figure 3 shows the spectra of aqueous-ethanolic extract of Aloe-vera. The peak at 3927.50 shows the presence of organic acids (-COOH) group. The peaks at 3739.73, 3643.39 and 3626.53 show the presence of –OH group (alcoholic compounds with phenolic group). The peak at 2921.65 and 2853.56 refers to the presence of aromatic compounds –CH bonds or aliphatic compounds with –CH bonds. The peaks at 1706.67, 1636.32, and 1587.88 are referred as ester peaks (C = N, C = C, O). The peaks at 989.86 to 620.31 denotes the presence of halogen compounds with carbon group (C–X). Spectra of aqueous-ethanolic extract of Aloe barbadensis.
In Vitro Antiarthritic Activity
Percentage Absorption and Inhibition by Anti-Arthritic Activity.

In vitro anti-arthritic and anti-inflammatory activities of Colchicum autumnale, Strychnous Nux-vomica, and Aloe barbadensis (“A” Percentage of inhibition by egg albumin protein denaturation method, “B” Percentage of stabilization by human red blood cell membrane stabilization method).
In Vitro Anti-Inflammatory Activity
The anti-inflammatory activity was determined by HRBC membrane stabilization method and percentage of protection was calculated as summarized in Table 2. Dose dependent increase in the percentage of protection was observed and all the extracts showed maximum inhibition at the concentration of 500 µg/mL. Strongest effect was observed by Colchicum and Aloe-vera followed by Nux-vomica as compared to the standard diclofenac sodium as shown in Table 2 and Figure 4.
Discussion
The medicinal plants for therapeutic purposes are used since ancient times and are the principal source of many modern medicines for the treatment of different diseases. 21 Aqueous-ethanolic extracts of three plants, that is, Colchicum, Nux-vomica, and Aloe-vera were screened phytochemically and the presence of different chemical constituents were confirmed. It was found that Colchicum possesses alkaloids, flavonoids, glycosides, fats, proteins, saponins, tanins, terpenoids, phenols, steroids, and anthraquinones. Nux-vomica contains alkaloids, phenols, saponins, terpenoids, and anthraquinones, whereas Aloe-vera comprises of alkaloids, terpenoids, tanins, anthraquinones, proteins, and fats (traces). Alkaloids are reported to have anti-inflammatory effect 21 and colchicine in Colchicum; strychnine and brucine in Nux-vomica are alkaloids in nature.22,23 Similarly, barbaloin is glycoside and Aloe emodin in Aloe-vera is anthraquinone in nature 24 that are reported to have anti-arthritic and anti-inflammatory potential. 25
We also performed FTIR analysis to find out functional groups present in these plant extracts and found the presence of organic acids, amine groups, aromatic compounds with C-H bonds or aliphatic compounds, esters and halogen compounds with carbon group (C-X), and phenolic compounds. As the chemical compounds found in the extracts are reported to have antiarthritic and anti-inflammatory activity, we sought to investigate these crude extracts to determine the aforementioned pharmacological effects in vitro.
Previous literature reported that one of the factors which is involved in the causation of rheumatoid arthritis is denaturation of proteins. 26 These findings prompt us to evaluate the anti-arthritic potential by protein denaturation method for 125, 250, and 500 µg/mL of concentration of aforementioned medicinal plants derived crude extracts. For Colchicum, we observed the 88.4, 96.0, and 98.5% of inhibition and Nux-vomica expressed 83.83, 90.55, and 92.30 % inhibition of protein denaturation while Aloe-vera revealed 10.2, 75.72, and 66.16 % inhibition, respectively. All the extracts showed comparable effects as of standard drug, that is, Diclofenac sodium having 30.7, 50.74, and 74.98 % of inhibition. Previously, H. Rehman et al. 18 worked on Oryza sativa (variety: Joha Rice) and found comparable 75.0, 80.31, and 84.15 %, respectively, at same concentration that are in line with our findings. Mechanism of denaturation can be due to changes in hydrogen, electrostatic, disulfide, and hydrophobic bonding in structure of peptides. It is evident from various studies that many anti-inflammatory drugs exhibit dose-dependent ability of inhibition for thermally induced protein denaturation. 27 Protein denaturation method in our study is expected to involve such mechanism.
The cell vitality depends upon the integrity of their membranes. Exposure of RBCs to injurious substances, such as hypotonic medium, causes the lysis of membrane which leads to hemolysis and oxidation of hemoglobin. The hemolytic effect of hypotonic solution is related to excessive fluid accumulation within the cell, resulting in the rupture of membrane. Such kind of injury to membrane causes secondary damage through lipid peroxidation due to free radicals. 28 It is thus anticipated that compounds possessing membrane-stabilizing properties should offer significant cell membrane protection against injurious agents. 29 Thus, taking in view the critical role of cell membrane protection, we aimed to investigate the membrane stabilizing effect of Colchicum, Nux-vomica, and Aloe-vera crude extracts by HRBC membrane stabilization method at the concentration of 125, 250, and 500 µg/mL. Results showed that percentage protection of Colchicum is 30.31, 31.37, and 40.20% of protection. Similarly, Nux-vomica expressed remarkable 30.58, 34.36, and 35.67% of protection while Aloe-vera revealed 29.33, 35.58, and 40.17% of protection and was comparable with the standard drug, that is, Diclofenac sodium that had 15.89, 17.05, and 29.36% of protection. Compounds with membrane-stabilizing properties are considered reliable for their ability to restrict the release of phospholipases that are responsible for triggering the formation of inflammatory mediators. 30 In this study, absorbance of hemoglobin is taken by spectrophotometer in HRBC membrane stabilization method. When RBCs are exposed to hypotonic medium, hemoglobin inside the cell membrane is lysed. 31 Due to the addition of membrane stabilizing agents, the cell membrane stability is increased, and thus, less absorbance is noted by spectrometer. The medicinal plant extracts used in our study, namely, Colchicum, Nux-vomica, and Aloe-vera has shown comparable membrane stabilizing property with the standard that may be due to the inhibition of the release of phospholipases. 32
Conclusion
We concluded that aqueous-ethanolic extract of Colchicum, Nux-vomica, and Aloe-vera were found to be more potent in inhibition of egg albumin denaturation and protection of HRBC membrane stabilization and is comparable with Diclofenac sodium suggesting that these plants can be used as alternative options in the management of inflammatory and arthritic conditions. Further studies are warranted to find out the phytochemicals responsible for the potential and their molecular targets underlying disease recovery process.
Footnotes
Acknowledgments
Authors are thankful to University College of Conventional Medicine for their financial as well as moral support to accomplish this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
