Abstract
The wound dressings fabricated by polymers and oregano essential oil (OEO) can be very effective as a hydrogel. The current study has been focused on fabricating the hydrogel membranes of oregano oil encapsulated as an antibacterial agent into sodium alginate (SA) solution by solvent casting method and then evaluated the antibacterial, antioxidant activity, and physicochemical performance of SA/OEO-based polymeric membranes. The polymeric interactions, surface morphology, water absorption capability, thermal stability, and encapsulation efficiency were investigated by FT-IR, SEM, swelling ratio, DSC, and encapsulation efficiency. The percentage encapsulation efficiency of essential oil was 40.5%. FTIR validated the presence of molecular interaction between individual components. SEM images showed a rough and porous appearance for hydrogel membranes. Moreover, DSC showed that the fabricated membranes were thermally stable. The inclusion of more content OEO decreased swelling ratios. The antioxidant test was carried out by DPPH assay and antibacterial test through disc diffusion method against microbes. The results revealed that membranes containing the highest content of OEO had more excellent antioxidant and antibacterial efficacy. Therefore, the polymeric membranes of sodium alginate loaded with oregano essential oil can be employed as an effective wound-healing candidate.
Keywords
Introduction
The global population is quickly increasing, and obesity and diabetic cases are also rising, leading to a stunning rise in chronic wounds. The injuries to the epithelial layer can be cured by re-epithelization. However, the body cannot adequately repair partial or complete disruption to the dermis cells, and the recovery process is mainly slowed. A good dressing material with antibacterial characteristics is necessary in such a situation.1-6 Dressings are crucial in preventing infection and boosting growth factors for faster healing.7,8 Traditional wound dressings are primarily dry and challenging to apply, and they cannot heal patients. 9
Moreover, they are limited due to their therapeutic efficacy, high price, and biological safety issues.10-13 Increased financial and social strain is placed on healthcare organizations, caregivers, patients, and their families when a condition is not cured, and wound-healing treatments are prolonged. Active wound coverings that stop microorganisms from infiltrating the wound, regulate inflammatory reactions, and encourage tissue regeneration have been developed to allay these worries. Hydrogels have fulfilled the primary requirements for caring for injuries because of their high water content and their biocompatibility. 14 The insoluble hydrophilic structure exhibits a remarkable ability to absorb wound exudes and facilitates the diffusion of oxygen to accelerate wound healing. There are several advantages to applying hydrogels as wound dressing, such as protection against secondary infection, providing a moist microenvironment, managing wound bed’s hydration, exhibiting low adherence, and soothing the skin, which may result in a significant reduction in pain. 15
Recently, there has been much research into biomaterial-based constructs made of synthetic and natural polymers for future clinical use. Natural polymers exhibit the best characteristics: resistant potencies, easy availability, cell proliferation ability, and regenerative tissue features. They also possess antibacterial, immune-suppressive, cell proliferation, and angiogenesis properties to fabricate a suitable environment that helps in wound healing. Polysaccharides, proteoglycans, and proteins are the most common natural biopolymers used in wound healing.16-18
Sodium alginate is a poly-anionic polymeric compound obtained from brown seaweeds, mainly comprised of β-D-mannuronate (M) and α-L-guluronate (G) residues that are interconnected through 1, 4 glycosidic linkages (Aderibigbe & Buyana, 2018). Sodium alginate is abundantly available, eco-friendly, and relatively inexpensive to produce; therefore, it is extensively used in numerous healthcare accomplishments, including drug delivery, medical dressing, dentistry impression materials, food, and paper industries.19,20 The salts of alginic acid are utilized to heal wounds and burns because of their hemostatic properties, biocompatibility, and flexibility. They can also enhance angiogenesis and cell migration, lowering the concentration of pro-inflammatory cytokines in the injured area during recovery.17,21-23 Alginate could effectively show gelation with multivalent cations forming hydrogel that offer a moist micro-environment, absorb wound exudates due to its hydrophilic nature, improve the hemostatic efficacy, and help to repair damaged tissues and less painfully remove than the conventional dressing materials. Moreover, SA would have no antibacterial intervention to prevent wound infection. Polyphenol phytoconstituents derived from medicinal herbs can be antibacterial agents. 24
Essential oils derived from plant extraction are natural microbial agents; immiscible with water, volatile, and easily degraded through sunlight, oxygen, elevated heat, and high pH, restricting their applicability. Their integration into the polymeric matrix improves its water solubility, stability, and vapor permeability, decreases lipid oxidation and controls bioactive chemical delivery at selected areas.25-29 Oregano oil is extracted from the oregano plant that belongs to the Lamiaceae class of aromatic plants, also called the mint family. 30 The vital components found in oregano oil are carvacrol and thymol. 31 γ-terpinene, p-cymene, and β-fenchyl alcohol are also metabolites of OEO. They have various antibacterial, antifungal, anesthetic, antioxidant, and anti-inflammatory properties. They have been used in various items, including cosmetics, food flavor, and pharmaceutical industries.
In this research, we reported oregano essential oil incorporation in various concentrations into SA to fabricate variable hydrogel membranes of SGCaT-80-OEO. Producing alginate hydrogels with an oily component is emulsification, crosslinking with CaCO3 and solvent casting. The effects of OEO concentration on encapsulation efficiency, water absorption, surface morphology and characterization, and antioxidant and antibacterial potential of OEO/SA hydrogel membranes have been studied.
Material and Methods
Sodium alginate (SA) was purchased from Sigma Aldrich, Germany, calcium carbonate was received from (ICI, USA), and glycerol was received from Antares Chemical Pvt. Limited, India. DPPH was received from (Sigma-Aldrich, Germany), ethanol and methanol (Sino-pharm Chemical Reagent Co., Ltd, China), oregano essential oil, Tween-80 (ICI, USA), nutrient broth (Oxoid, UK), norfloxacin (10 µg), Gram-positive and Gram-negative bacteria, and distilled water. All chemicals have an analytical grade and are used without further process.
Preparation of SA/Glycerol/CaCO3/TW-80/OEO Hydrogel Membranes
Composition of Fabricated Hydrogel Membranes.

Characterizations of Synthesized Hydrogel Membranes
Encapsulation Efficiency
To calculate encapsulation efficiency, 15 mg of oregano essential oil-loaded hydrogel membranes was dissolved in 10 mL of ethanol, and the resulting solution was stored overnight. After that, the solution mixture of hydrogel membranes was filtered, and the absorbance was measured with a UV-Vis spectrophotometer at 276 nm.
32
The equation (1) determined the percentage encapsulation efficiency
FTIR Analysis
FT-IR spectroscopy is a characterization technique used to obtain absorption, emission, and photoconductivity spectrum for solids, liquids, and gases by recording transmittance and absorption of radiations (Sindhu, Binod, and Pandey, 2015). This technique is used to characterize functional groups. Alpha-ІІ FTIR-ATR by BRUKER INTERNATIONAL USA was used to determine the FTIR spectrograph. The spectra were obtained between 650 and 4000 cm−1.
SEM-EDX Analysis
The sodium alginate membranes were mounted on aluminum stubs using double-sided adhesive carbon tabs in order to permit correct imaging and guarantee sample stability, which was coated with gold (35 s DC mA). The stub should be conductive or coated with a conductive material to minimize charging during the analysis. The characterization assay used to determine and evaluate surface modification and porosity of materials is scanning electron microscopy by examining the material with an intense energy beam of electrons. SEM-EDX is a typical technique to capture hydrogels’ particular “network” structure and guarantees that the hydrogel keeps its structure.33,34 For SEM-EDX analysis, NOVA-450 Nano SEM by USA was used.
Thermal Analysis
DSC is a method used to calculate the enthalpy changes that occur in polymers, polymer blends, and composites as a function of temperature or time and that are caused by changes in the materials' physiochemical properties. DSC calculates heat flow in a specimen to analyze melting point, glass transition temperature, and crystallization when the sample is heated or cooled. 35 For DSC analysis, Shimadzu DSC-60 by Japan was used.
Antioxidant Activity
DPPH radical scavenging method is a significant way to determine the antioxidant ability of hydrogels. .05 g hydrogel membrane of each sample was dissolved in 10 mL of ethanol, and the samples were shaken overnight at room temperature in a shaking water bath. The mixture was filtered, and after that, 1 mL from every solution was taken and put into a 3 mL solution of DPPH and tightly closed the test tubes with aluminum foil. The mixture was allowed to stand for 1 h in low light conditions. The absorbance was detected by UV-Vis spectroscopy at 517 nm.
36
The antioxidant activity can be calculated as shown in equation (2)
Antibacterial Activity Evaluation
Bacterial Growth Medium, Cultures, and Inoculum Preparation
Pure cultures were maintained on nutrient agar medium in the slants and Petri plates. For the inoculum preparation, 13 g/L of nutrient broth was suspended in distilled water, heated, mixed well, distributed homogenously, and then autoclaved. A pure (10 µL) culture of bacterial strain was mixed with a medium and placed for 24 h at 37°C in a shaker at 120 rpm. All the inoculum was prepared by the same pattern and stored at 4°C. The inoculum with 1 × 108 CFU/mL was used for further analysis.
Antibacterial Assay by Disc Diffusion Method
28 g/L of nutrient agar was suspended in distilled water, mixed well, and distributed homogeneously. The medium was sterilized by autoclaving at 121°C for 15 min. Before the medium was transferred to Petri plates, inoculum (100 μL/100 mL) was added and poured into sterilized Petri plates. After this, different synthesized membranes and antibiotic discs (norfloxacin) having catalogue number (CT0434 B) of size 5 mm were laid flat on a growth medium containing. The Petri plates were then incubated at 37°C for 24 h for the growth of bacteria. With antibacterial activity, the extracts inhibited bacterial growth and formed clear zones. The inhibition zones were measured in millimeters using a zone reader. 38
Swelling Studies
At an ambient temperature, a fixed-mass membrane was soaked in a beaker containing the solvent, that is, distilled water. After a specific time, interval, the excessive solvent was discarded, and the weight of the puffed membrane was computed. The membranes were immersed in the solvent again until they reached equilibrium. The swelling at equilibrium can be determined by the equation (3)
39
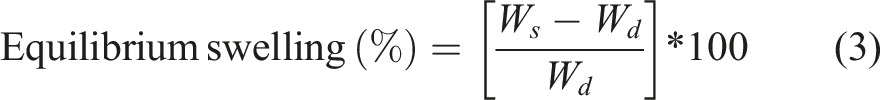
Results and Discussion
In this research, oregano oil-encapsulated sodium alginate hydrogel membranes were successfully fabricated by using sodium alginate as a biodegradable and biocompatible raw material, and OEO was used as an antibacterial and antioxidant agent to improve the role of SA. Glycerol was added as a plasticizer to induce plasticity and TW-80 as a surfactant to improve the dispersion of OEO. The prepared membranes have a yellowish color. All the fabricated membranes show good mechanical stability and homogeneity.
Encapsulation Efficiency
The EE (%) is the pharmaceutical or oil encapsulation into the microspheres, nanoparticles, and hydrogels. The samples may have been kept overnight to give the encapsulation procedure enough time to complete. Some encapsulation techniques need the oil to diffuse into the hydrogel matrix and create the enclosed structure over a specific amount of time. The samples should be kept overnight to give the encapsulation process enough time to finish before any additional testing or analysis is done. Thyme, clove, and Satureja hortensis essential oils had loading encapsulation efficiency of 85, 24.7, and 52%–66%.32,37,40 In our study, the highest encapsulation efficiency was 40.5% for SGCaT-80-1.0, and the lowest encapsulation efficiency was 20.4% at .5% v/v concentration of oregano oil, as shown in Figure 1. Effect of OEO concentration on percent encapsulation efficiency.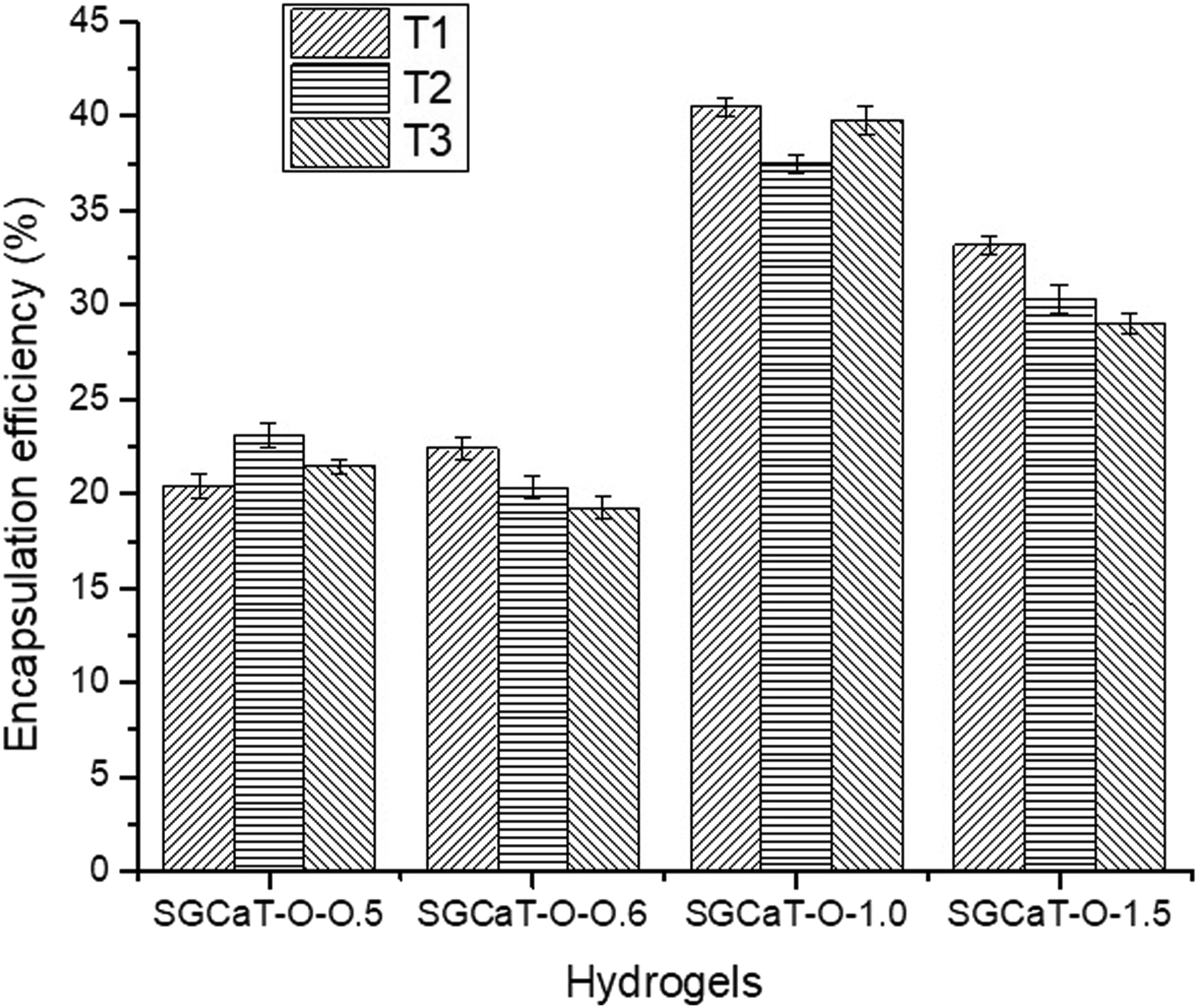
Encapsulation Efficiency of the Prepared Hydrogels.

The crosslinking process may also influence % EE. In our study, SA to CaCl2 ratio was 9% (w/v) to 1% (w/v), and crosslinking time was 2 h. Excessive concentration of sodium alginate might raise the viscosity of the solution lowering the encapsulation efficiency. 42 According to the previous study, the highest %EE was achieved for thyme oil when sodium alginate to CaCl2 concentration was 2% to .5% and crosslinking time was 20 min. 43
Functional Group Analysis
The Functional Group Analysis of the Synthesized Membranes.
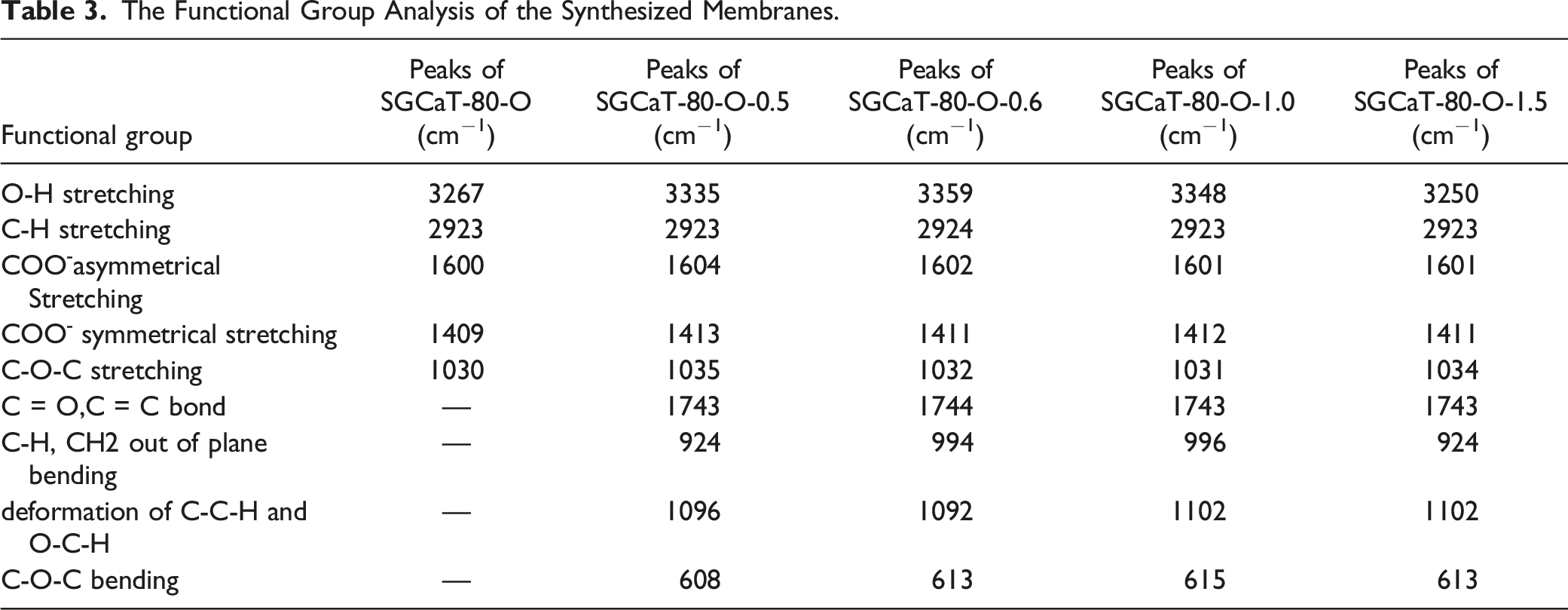
The FTIR spectra of all membranes (Figure 2) showed the same significant peaks; however, the amplitude of peaks varied depending on the concentration of oil encapsulated in sodium alginate hydrogel. All hydrogel membranes displayed a distinctive broad absorption peak at 3500–3200 cm−1 and an intense peak at about 2900–2930 cm−1 because of O-H and C-H str vibrations. As the broad single absorption band of O-H at 3267 cm−1 in SGCaT-80-O, 3335 cm−1 in SGCaT-80-O-0.5, 3359 cm−1 in SGCaT-80-O-O.6, 3348 cm−1 in SGCaT-80-O-1.0, and SGCaT-80-O-1.5 appears at 3250 cm−1, this shift in O–H stretching vibrations proposed specific interactions between sodium alginate and OEO. FTIR spectrum of (A) SGCaT-80-O, (B) SGCaT-80-O-0.5, and (C) SGCaT-80-O-1.0.
The asymmetrical and symmetrical str. vibrations of COO− appears at about 1600 and 1409 cm−1 in SGCaT-80-O, 1604 and 1413 (cm−1) in SGCaT-80-O-0.5, 1601 and 1411 (cm−1) in SGCaT-80-O-0.6, 1601 and 1412 (cm−1) in SGCaT-80-O-1.0, and 1601 and 1411 cm−1 in SGCaT-80-O-1.5 MBN. This small shift in the wavenumber of carboxyl groups indicated ionic interaction between Ca2+ and sodium alginate chains. 48 The absorption peaks of a six-membered ring of Na-alginate appear at about 1200–960 cm−1. 49 Additionally, the pyranose ring (C-O and C-C) vibration absorption bands at 1092, 1096, and 1101 (cm−1) are caused by the C-C-H and O-C-H deformations. The characteristic glycosidic bond absorption at about 1030 cm−1 is observed, or the carbonyl group (C = O) str bond appears at about 1743 cm−1.
According to these findings, the encapsulation of OEO in a specific concentration caused a shift in absorption peaks of the hydroxyl group, COO asymmetrical and symmetrical and C–O–C str due to ionic binding of SA with Ca2+, and intra-molecular and intermolecular hydrogen bonding, which gives the hydrogel membrane its overall texture. Although several studies showed that adding other substances to the alginate hydrogel membrane results in no chemical binding between components, 50 no chemical interaction was observed between sodium alginate and Frankincense oil. 51
Surface Morphology and Elemental Analysis
The structural characterization of membranes was investigated using the scanning electron microscopy technique. The microstructural analysis revealed its influence on cell adhesion, migration, rapid tissue growth, and proper functioning during wound remedy.
52
The pure alginate had a smooth and homogenous surface, but the addition of OEO caused changes in the surface morphology of the films.
50
The SEM images reveal oil particles on the surface because oils are hydrophobic and immiscible in water, confirming OEO's incorporation into the SA membrane. The surface roughness increases as the oil concentration increases, making the hydrogel membranes good candidates for cell attachment, like fibroblasts and keratinocytes that help in wound curing. Figure 3 shows SEM micrographs of fabricated hydrogel membranes with different content of OEO. SEM images of SGCaT-80-O-0.5 (A, B, C) and SGCaT-80-O-1.0 (D, E, F).
The main components of OEO are carvacrol and thymol; they have a phenolic group, three CH3, and 1 O-H group. Agglomeration begins as the oil content in polymeric membranes rises due to three CH3 moieties that become insoluble in organic solvents. The rough architecture was also noticed for PVA/starch-based membranes by incorporating OEO.
47
The surface has minute porosity and permeability ranging from millimeters to macro cavities. This porous architecture is due to the evaporation of EO from the surface of the membrane. The increased porosity influences certain features like mechanical potencies and H2O retention capabilities. Koosehgol revealed similar findings and fabricated chitosan/polyethene glycol fumarate/thymol films.
53
The SEM-EDX micrographs explain the surface modification, porosity, and adherence of constituents.
54
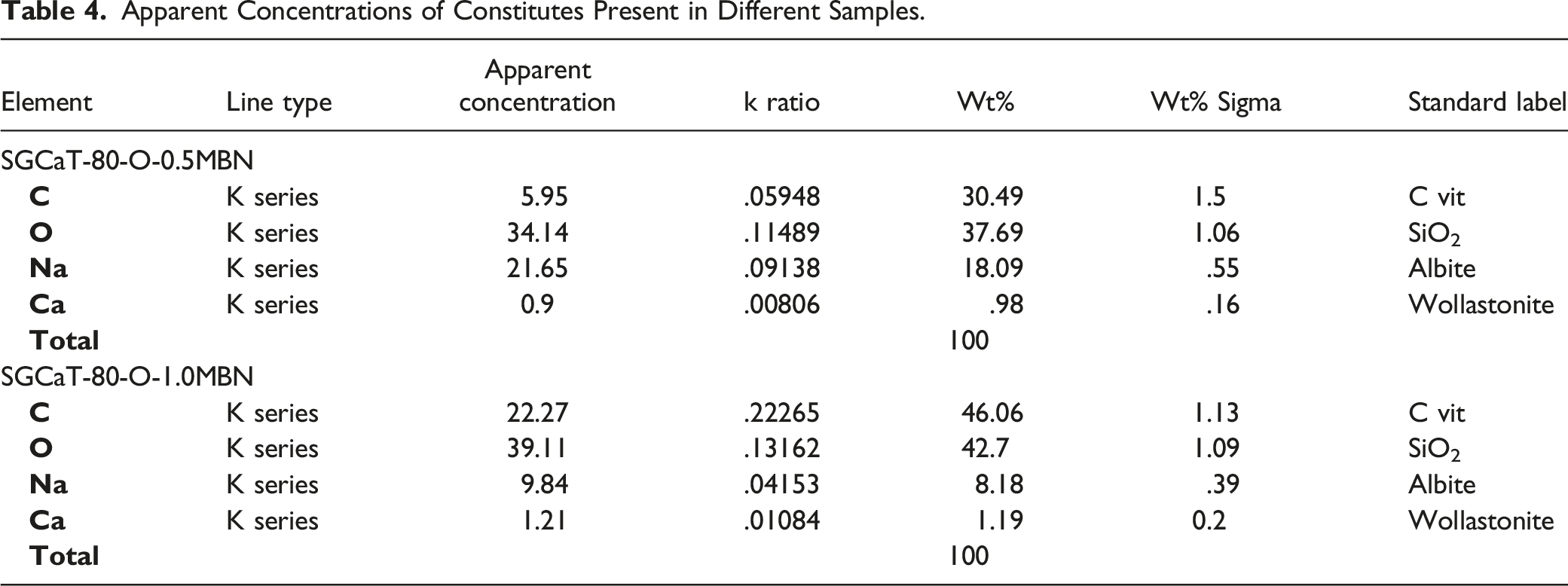
The EDX graph of fabricated polymeric membranes displayed peaks for carbon and oxygen which are the major constituents of natural polymers and OEO (Figure 4). The peaks for sodium and calcium are due to sodium alginate and calcium carbonate (Table 4). Elemental analysis of (A) SGCaT-80-O-0.5 and (B) SGCaT-80-O-1.0 MBN. Apparent Concentrations of Constitutes Present in Different Samples.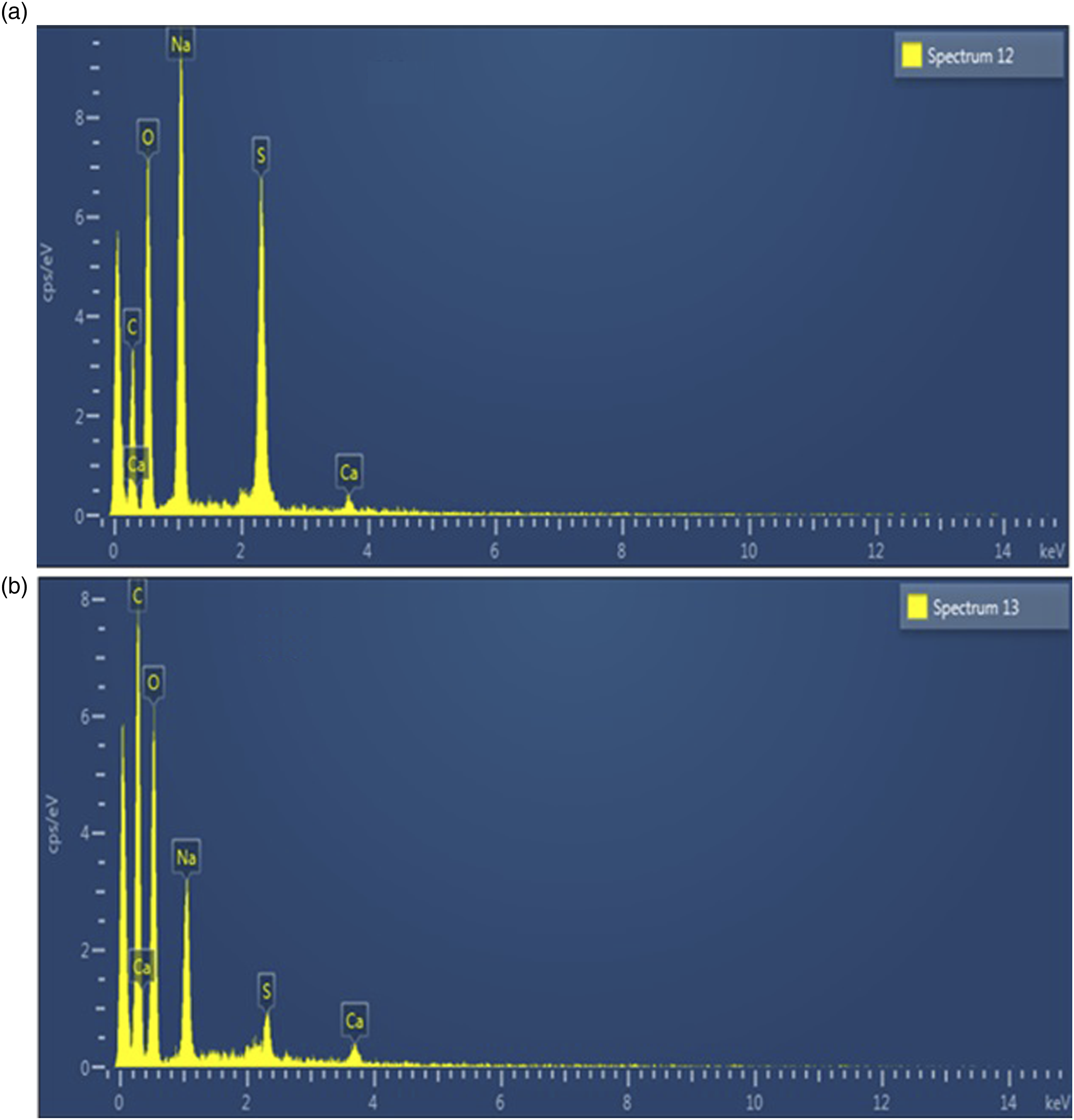
Thermal Analysis
The thermal properties of fabricated hydrogel membranes were investigated by differential scanning calorimetry. SA is characterized by an exothermic peak at 246°C due to the degradation of the polymer.
55
Polysaccharides are composed of carboxylate or R-COOH moieties. Hence, thermally, separation of carboxylate moieties and CO2 formation from the associated carbohydrate chain could be a possible way for thermal changes. The minor decomposition of the polymer chains peaked at 250°C and primary decomposition generally at about 300–400°C.
32
The DSC curves for SGCaT-80-O-0.5 and SGCaT-80-O-1.0 are shown in Figure 5. Samples SGCaT-80-O-0.5 and SGCaT-80-O-1.0 showed an endothermic peak at 60°C, which was ascribed to loss of H2O molecules. It is most likely owing to Na-alginate's hydrophilicity. In DSC thermo-grams, there is no exothermic peak for polymer degradation due to the ionic interaction of calcium ions with the alginate polymer chain. The endothermic peaks at 160°C and 164°C indicate the melting temperature of hydrogel membranes. Increasing the OEO concentration in the membrane increased the melting point from 160°C to 164°C. Similar results were shown by Phaiju, who encapsulated cinnamon essential oil into POLY (Ɛ−CAPROLACTONE) that gave melting peaks at 60.70°C and 60.54°C.
7
The result showed that the EO was dispersed into a polymeric matrix, and the oregano essential oil-encapsulated hydrogel membrane was thermally stable enough to be employed as a wound dressing. Thermal analysis of (A) SGCaT-80-O-0.5 and (B) SGCaT-80-O-1.0 MBN.
Antibacterial Activity
The antibacterial activities of Na-Alg hydrogel membranes encapsulated with different concentrations of OEO were investigated according to the disc diffusion approach against microorganisms (Figure 6). The antiseptic components present in OEO are carvacrol and thymol as they move out from membranes into bacterial growth media and show resistance against bacteria.
47
The antibacterial efficacy of EO’s would be related to the hydrophobic phenolic components. The hydrophobic EO's takes part in the phospholipid of the cell barrier and the membrane would deteriorate; due to this structural distortion, its permeability would increase.
56
The results showed that sodium alginate membrane without oregano oil was ineffective against the tested bacteria. The hydrogel membranes fabricated by incorporating OEO are more potent towards Gram-positive and Gram-negative microbes. The highest antibacterial activity was achieved at 1.0% (v/v) OEO concentration, with mean inhibition zone 11.11 ± 1.23, 19.33 ± 4.40, 18.56 ± 2.47, 18.67 ± 2.8, and 15.66 ± 3.78 against P. vulgaris, Pasteurella multocida, E. coli, Staphylococcus aureus, and B. subtilis (Table 5). Previous research suggested that the films containing thymol essential oil have the most potent antibacterial activity and can be employed as a safe and efficient nano-sized wound dressing.
57
Inhibition zones of SGCaT-80-O-0.5, SGCaT-80-O-0.6, and SGCaT-80-O-1.0 MBN towards B. subtilis, E. coli, S. aureus, P. vulgaris and P. multocida. Antibacterial Activity of SGCaT-80-O, SGCaT-80-O-0.5, SGCaT-80-O-0.6, and SGCaT-80-O-1.0 Against Tested Bacteria. Bacterial name: P. vulgaris = Proteus vulgaris, P. multocida = Pasteurella multocida, E. coli = Escherichia coli, S.A = Staphylococcus aureus, B. subtilis = Bacillus subtilis. The values are mean + SD of triplicate samples; the standard drug used as control at a conc. of 1 mg/mL.

Antioxidant Activity
The DPPH radical scavenging measures antioxidant potential, mostly related to a compound’s ability to supply hydrogen.
58
Free radical scavenging of DPPH is used to assess antioxidant activity because of its speed, efficiency, simplicity, and reliability. The phenolic compounds in EO’s show antioxidant action against biological and non-biological oxidants. Carvacrol and thymol are the major phenolic components in OEO that are responsible for the DPPH radical neutralization. The phenolic compounds can transfer hydrogen atoms of hydroxyl groups to free radicals, in this way stopping other compounds from being oxidized.
59
Therefore, antioxidant agents may be useful in improving the non-healing phase of chronic wounds.
60
The degree of DPPH radical neutralization affects by the content of oil and incubation time. The highest antioxidant activity was recorded at a maximum oil concentration and after 60 min of incubation. Figure 7 shows the antioxidant activity at various concentrations of OEO. The results confirmed that the sodium alginate membrane without OEO showed certain antioxidant activity, and DPPH radical scavenging activity increased with increasing OEO concentration. The highest % of DPPH scavenging could reach 83% for sample SGCatT-80-O-1.5 MBN (Table 6). Previous studies have shown that encapsulating EO’s into alginate-based films can improve antioxidant properties. The DPPH activity increased from 42.40%–80.60% and 55% due to the presence of Zataria multiflora and thymus EO’s.
36
DPPH free radical scavenging of fabricated membranes. DPPH Free Radical Scavenging of Fabricated Membranes.

Swelling Behavior
Swelling is an essential and fundamental feature of a hydrogel for assessing the sustained release of incorporated substances, lesion adhesion, and capacity to absorb excess wound fluids to enhance tissue growth and epithelial cell migration.
60
% age swelling for different membranes prepared with the addition of OEO into sodium alginate at different periods for H2O contact time has been depicted in Figure 8. The swelling ratio of SGCaT-80-O, SGCaT-80-O-0.5, SGCaT-80-O-0.6, SGCaT-80-O-1.0, and SGCaT-80-O-1.5 MBN has been decreased to 328%, 299%, 280%, 249%, and 215% for the initial ten minutes and 399%, 388%, 375%, 360%, and 315%, respectively, during 30 min (Table 7). As the OEO concentration increases from .5 to 1.5, the swelling behavior decreases—the swelling behavior of films is affected by the type of components and their hydrophilicity and hydrophobicity index. Hydrophilic substances could increase, and hydrophobic molecules could decrease the swelling.
57
Swelling behavior of fabricated membranes as a function of contact time. Swelling Behavior of Hydrogels With Standard Deviation.

SA hydrogel membrane had the highest swelling ratio, but the encapsulation of hydrophobic EO remarkably reduced the swelling ratio of the hydrogel membrane. The inclusion of non-polar components decreases the swelling ratio by inhibiting water uptake. EO's precipitate at higher concentrations well over hydrogels, causing voids inside the membrane. The swelling ratio can also be lower due to the rise in gel fraction % that indicates a rise in crosslink stiffness by incorporating EO’s. Different factors such as ionic strength, heat, the type of constituents, lesion microenvironment, and the extent of crosslinking may be influenced the swelling behavior of hydrogels. 47 Based on these findings, the membrane with less OEO concentration became more solvent and reflected better swelling.
Conclusion
The current study has displayed the antibacterial and antioxidant potentiality of oregano oil incorporated into sodium alginate to develop blended membranes. The change in OEO concentration in blended solution showed a noticeable effect on morphological and physicochemical features. The encapsulation efficiency (%) of essential oil shows improvement when the initial concentration of oil is raised to an optimal level. However, when concentrations exceed this optimum range, there is a potential decline in EE. OEO-incorporated membranes exhibited good antioxidant and antibacterial activity against tested bacteria, an essential principle for potent wound dressing. These findings suggested that the OEO-encapsulated membranes could be employed as an antioxidant and antibacterial agent to cure wounds and as a viable choice for producing novel biomaterials for wound dressing. Hydrogels possess promising characteristics such as antioxidant, antibacterial, and swelling properties, making them suitable for various applications. They have unique properties that find utility in biomedical engineering, drug delivery, tissue engineering, and personal care products.
Footnotes
Acknowledgements
The authors extend their appreciation to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R165), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R165), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
