Abstract
Since ancient times, medicinal plants have been used as traditional medicine to treat a variety of ailments. Aloe vera (AV) gel’s therapeutic potential is one of the most effective approach in the fabrication of functional materials. The current study aimed to prepare the AV and chitosan (CS) membranes using various cross-linkers that were characterized using X-ray diffraction (XRD), scanning electron microscopy (SEM), Fourier transform-infrared (FT-IR) spectroscopy, thermal gravimetric analysis (TGA), and ultraviolet-visible (UV-Visible) techniques, as well as swelling ratio and antimicrobial studies. SEM analysis revealed that the membrane is porous, with interconnected pores. The inclusion of AV contents in the membrane improved thermal stability and crystallinity. The swelling ratio of the ACPG-3 membrane with a 2:1 CS to AV ratio was 366%. The membranes showed promising antimicrobial activity against Escherichia coli, Staphylococcus aureus, Bacillus subtilis, and Pasteurella multocida strains. The findings revealed that polymeric CS/AV membranes have effective potential for use in the biomedical field.
Introduction
Wound healing is a complex and ongoing process in which injured tissues are repaired and an efficient system is required to improve the healing process. There are numerous ointments and bandages available for treatment. Commonly used synthetic polymeric bandages have poor healing efficacy for chronic wounds.1-3 The significant limitations of skin injury treatment formulations such as gels and ointments include their inability to sustain the release of medicine for an extended period of time at wet injury sites due to their short-lived existence.4,5 Furthermore, these dressings do not provide adequate moisture, pH, or gas exchange to injured skin. These conditions promote the growth of bacteria and the spread of infection which slows the healing process. 6 As a result, many efforts have been made to create high-quality wound dressing materials that are natural and biodegradable in order to speed up the healing process.7-10 Recently, medicinal plants have been used as a medicine substitute.11,12 The phytochemicals derived from plants have medicinal properties that are used to treat a variety of infections. Over the past few years, aloe vera has been the focus of extensive scientific research due to its therapeutic benefits.13,14 It is known as “the healing and wonder plant.” Due to its anti-inflammatory, antibacterial, and wound-healing characteristics, aloe vera has long been used to treat digestive issues as well as skin injuries (burns, wounds, insect bites, and eczemas). The goal of research on this medicinal plant was to understand its mode of action and to pinpoint the chemicals responsible for these effects. 7 The active components of aloe vera gel are vitamins (A, B12, C, E, choline, and folic acid), enzymes (basic phosphatase, aliases, amylase, carboxypeptidase, bard kinase, catalase, lipase, cellulase, and peroxidase), minerals (copper, chromium, calcium, selenium, manganese, magnesium, zinc, sodium, and potassium),8-10 carbohydrate (fructose, glucose, and glucomannan),11,12 anthraquinones, folic acids (cholesterol,β-sitosterol, lupeol, and camp sterol), hormones (gibberellins and auxins), amino acids, salicylic acid, lignins, and saponins. The active ingredients that have received the greatest research are acemannan, aloe-emodin, aloin, aloes, and emodin. 13
The majority of in vitro research on skin protection examined that how Aloe vera and its active ingredients aid in wound healing. The most popular cell lines are fibroblasts, primary normal human epidermal keratinocytes (HEKa), and immortalized human keratinocytes (HaCaT). These research studies have shown that aloe vera and its active ingredients (aloes, aloin, and emodin) exert their protective effects primarily through anti-inflammatory and antioxidant processes to safeguard the body.14-16
Additionally, chitosan, a bio-polymeric material, had been broadly utilized as a base substance for wound therapy due to its distinctive set of biological features, which include biocompatibility, biodegradability, and low toxicity. Chitosan also has mucoadhesive, hemostatic, and antibacterial characteristics and may speed up the healing of wounds.17-19
In order to create a new class of compounds known as biosynthetic polymeric blends or bio-artificial materials with better mechanical strength, thermal stability, and biocompatibility as compared to individual components that are likely to be administered for many medicinal purposes, the natural polymeric blends are combined with synthetic polymers because they exhibit poor mechanical efficacies. Bio-artificial mixing’s goal is to create semi-synthetic blends with specific structural characteristics and tensile strengths depending on specified biopolymer and synthetic polymer attributes. 20 The polyvinyl alcohol (PVA) is a man-made, cost-effective, nontoxic, and water-miscible polymer. The film of PVA has superior quality and possesses good tensile strength and elastic nature with diverse implementations in various disciplines, such as artificial muscle, contact lenses, ignited wound bandages, and vocal band reformation.21,22 The phenomenon of wound curing is very complex that can show several irregularities. So, the choice of proper wound bandage is very crucial to guide and enhance the curing action. 23
Therefore, the objective of the present work is to develop a synergic relationship between chitosan and aloe vera gel to design a polymeric blended bio-membrane through solution casting process that is likely to be utilized as an effective skin lesion bandage for providing a novel approach for wound therapy. The potentiality of various cross-linkers inclusion on physicochemical performance of AV/CS-based polymeric membranes was also assessed. The fabricated membranes were characterized by UV-visible, FT-IR, XRD, SEM, and TGA techniques. The antimicrobial activity and swelling ratio of AV/CS-based films were also investigated.
Material and Methods
Chemicals and Reagents
Chitosan has been obtained from GmbH Germany, CH3COOH, Tween 80, and polyvinyl alcohol from ICI American, Inc. Glycerol from Antares Chemical Pvt. Limited, India, and glutaraldehyde from Sigma-Aldrich, Germany. All the chemicals were of analytical grade and were used without further processing.
Aloe Vera Gel Extraction
For the extraction of Aloe vera gel, 2 years old aloe vera plants are selected from the local garden of Lahore. The aloe vera leaves have been separated from the plant and cleaned with distilled H2O to eliminate dirt particles superficially. The dermis is removed with the help of a sharp knife. The specimens were washed vigorously with water to clean the surface. The AV gel is put in a blender for homogenization and subsequently filtered. The homogenized mixture is heated at 60°C for 30 min for stabilization.24,25
Chitosan and Aloe vera Membranes Preparation
CS/AV Membranes Composition with Sample Code.


Schematic presentation for the preparation of AV-gel-based membrane.
Characterization of the Membranes
SEM investigation of fabricated films was employed with FEI-NOVA-450 Nano SEM (FE-SEM), USA. XRD analysis was studied by AXS-D8 advanced X-ray diffractometer (Bruker, USA), with a range from 0 to 80, 2 Theta. UV-visible analysis was performed by Cecil-Aquarius 7400 Ce, UK. FT-IR (Alpha II, Bruker, USA) is used to characterize functional groups at scanning range of 4000–625 cm−1. TGA-250 (Bruker, USA) at heating rate 10°/min under nitrogen atmosphere was used to investigate thermal stabilization. Prepared hydrogels were subjected to the subsequent method for swelling examination. The membrane of fixed mass was soaked in a beaker containing the distilled water at an ambient temperature. After a specific time, excessive solvent was discarded and weight of puffed membrane was computed. The membranes were immersed in the solvent again till it achieved equilibrium state. The swelling and equilibrium swelling was estimated using equations 1 and 2
26


Antimicrobial Activity
Bacterial Strains
Following Gram-positive and Gram-negative bacterial strains, 1 E. coli, 2 P multocida, 3 S. aureus, 4 and B. subtilis were used to study the antimicrobial activities of chitosan/AV-based polymeric membranes.
Bacterial growth medium, cultures, and inoculum preparation
Pure cultures were maintained on a nutrient agar medium in the slants. For the inoculum preparation, 1.3 g/100 mL of nutrient broth (Oxoid, UK) was suspended in distilled water, heated, mixed well, and distributed homogenously and autoclaved. Pure (100 μL) culture of a bacterial strain was mixed with the medium and placed for 24 h at 37°C in a shaker 120 r/min. All the inoculum was prepared by the same pattern and stored at 4°C. The inoculum with 1 × 108 CFU/mL was used for further analysis.
Antibacterial Assay
Antimicrobial activity of chitosan/AV-based polymeric membranes was determined using disc diffusion method (CLSI, 2011). 2.8 g/100 mL of nutrient agar (Oxoid, UK) was suspended in distilled water, mixed well, and distributed homogenously. The medium was sterilized by autoclaving at 121°C for 15 min. Before the medium was transferred to petri plates, inoculum (100 μL/100 mL) was added to the medium and poured in sterilized petri plates. After this, different synthesized membranes and antibiotic discs (norfloxacin; 10 μg, catalogue number CT0434 B) of size 5 mm were laid flat on the growth medium containing medium. The petri plates were then incubated at 37°C for 24 h for the growth of bacteria. The extracts having antibacterial activity, inhibited the bacterial growth and clear zones were formed. The zones of inhibition were measured in millimeters using a zone reader. 27
Results and Discussion
Aloe vera/chitosan-based membranes have been synthesized successfully by using different cross-linkers. Aloe vera enhances collagen production due to the interlinking of glucomannan with gibberellin (growth receptor) present in the fibroblast. 28 More amount of collagen Ш is produced and crosslinking degree of collagen is also enhanced. 29 All these factors facilitate skin lesion contraction and amplify the cracking power of resultant scar tissues to help wound healing. 30 Chitosan has been extensively employed in various medicinal applications owing to its distinctive features such as bio adsorbable, nonpoisonous, antimicrobial, and bio-acceptable. It is of great interest to use chitosan as a wound curing agent because it encourages hemostasis and tissue reformation. It has been used for the treatment of burns, ulcers, and wounds. 31
Functional Group Analysis of the Membrane
It is a way of chemical characterization for the functional class identification of polymeric, inorganic, and organic substances.
32
The investigation of functional moieties in fabricated hydrogels based on AV and chitosan was examined by FTIR spectroscopy. The generating characteristic peaks and responses are presented in Figure 2. The spectra of all blended membranes specify the particular peaks of AV and chitosan which are directly related to the individual content ratios in the membrane. The N-H and C = O peaks which appear between 1645 and 1670 cm−1 are occupied by a single peak at 1656 cm−1 in ACGG-3, 1652 cm−1 in ACGG-2, 1657 cm−1 in ACPG-3, 1640 cm−1 in ACPG-2, 1652 cm−1 in CAT-3, and 1650 cm−1 in CAT-2 which proposed certain interactions between AV and CS. Various researchers have manifested that chitosan makes a bond with selected anionic molecules like nucleic acid, proteins, and polysaccharides.
33
AV gel consists of complex dissolute of proteins and polysaccharides.
34
Distinct interactions like H-bonding can exist between AV gel and CS to describe the noticed bond shift.
24
(a) FTIR analysis of ACGG 2-3, ACPG 2-3, and CAT 2-3 samples.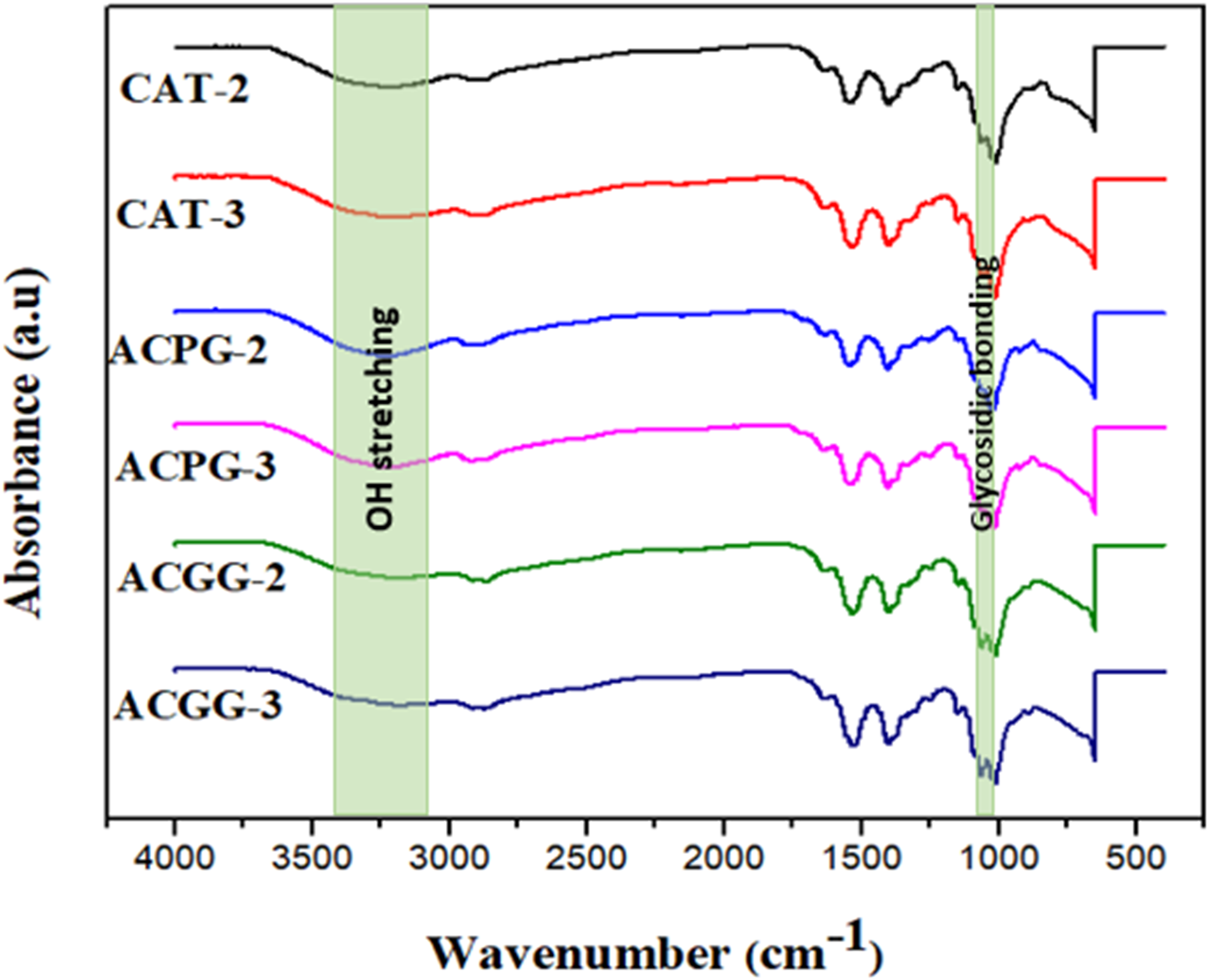
The IR spectra of ACGG-2 and ACGG-3 point out the existence of new bonds between AV, CS, and glutaraldehyde as the spectra exhibit characteristic peaks at a particular area. A wide peak at about 3400 cm−1 is noticed, which is the stretching vibration mark for the O-H group. The bending vibration peak at 1330 cm−1 for O-H of chitosan is reduced to the smallest which specifies the crosslinking with glutaraldehyde. 35 The IR spectra of ACPG-2 and ACPG-3 demonstrated a slight peak shift which stipulated fabrication of the membrane. It displays C-H str at 2860–2930 cm−1, and a wider peak at 3200–3400 cm−1 is responsible for O-H str. An exact band at 1640 cm−1 indicates N-H ben, C-H ring absorption at 950 cm−1, C-O-H ben at 1440–1450 cm−1, ether C-O absorption at 1150 cm−1, and a characteristic glycosidic bond absorption at 1040–1070 cm−1 is observed.
The IR spectra of CAT-2 and CAT-3 manifest the synthesis of blend based on CS, AV, and TW. The characteristic broad peaks observed as O-H str at 3200–3400 cm−1 corroborate that such groups are responsible for H-bond between individual components of blend. The interfacial intermolecular H-bond is further authenticated by the existence of very broad bands of O-H ranging between 3100 and 3600 cm−1. 36 A sharp peak originated at 1650 cm−1 indicating N-H ben. A characteristic peak at 1430–1435 cm−1 is responsible for C-O-H ben. An exact absorption peak at 1250 cm−1 indicates a glucan unit. A small peak at 1155 cm−1 of ether C-O is observed, 2 absorption peaks of glycosidic bonding occur at 1020 cm−1and 1070 cm−1, and CH ring absorption exists at 955 cm−1 and 960 cm−1 in CAT-3 and 2, respectively.
UV-Vis Analysis
Light transmittance is an important characteristic for membranes during wound curing applications.
37
The spectra of ACPG-3 and ACPG-2 point out the typical absorbance peaks ranged between 315–517 nm and 310–511 nm (Figure 3). The absorbance peak at 420 nm was indicated for CS membranes cross-linked with tween 80.
38
The absorbance peak of CS with more AV contents is shifted towards a higher wavelength. Certain active constituents in AV are responsible for absorbance in the UV region.
39
The absorbance peaks for ACGG-3 and ACGG-2 are observed at 336–565 nm and 330–580 nm. The sinapate esters and flavonoid compounds present in AV gel assist in UV absorption. Furthermore, glucosamine and N-acetylglucosamine units in CS act as chromophore groups that help in UV absorption.
40
The spectra of ACCG-3, ACCG-2, ACPG-3, ACPG-2, CAT-3, and CAT-2 point out 90, 82, 89, 88, 37, and 34 (%) transmittance, respectively. Consequently, AV addition in membranes may provide good protection against UV-light for wound therapy.
41
UV-Visible analysis of ACGG 2-3, ACPG 2-3, and CAT 2-3 samples.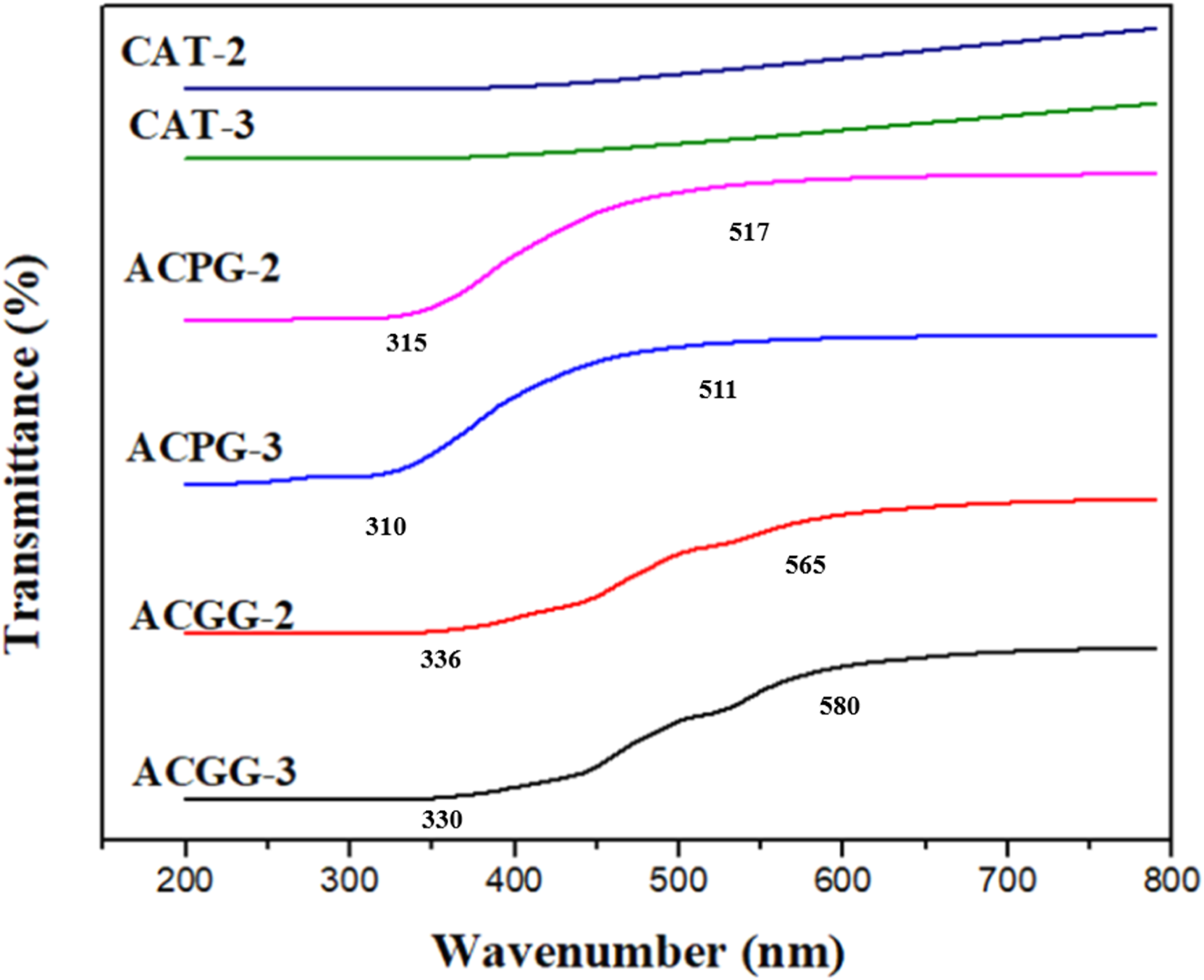
Thermal Analysis
Thermal stabilization and polymeric properties have been assessed by thermogravimetry.
42
TGA profile of fabricated membranes displayed mass reduction in four phases (Figure 4). The first phase (71–124°C) and second phase (250–308°C) correspond to glycerine volatalization and H2O evaporation of physically and H-bonded water with chitosan.43,44 Polysaccharides possess high H2O affinities and perhaps, they show disordered structural arrangements in solid states, so such macromolecules are possibly hydrated with ease. Polysaccharide’s water interaction is dependent upon the basic and molecular structure, and bounded water also exerts a strong impact on the nature of polymorphic macromolecule. The bound H2O evaporation causes molecular and chemical changes during the de-acetylation process.
45
The third phase (312–635 C) corresponds to decomposition of branched and linear glucose chains, N-acetyl glucosamine, acetyl glucosamine units, and AV multicomponent (enzymes, minerals, anthraquinones, vitamins, fatty acid, sugars, and hormones) decomposition.13,46 The fourth phase (668–725 C) designates to pyranose ring destruction and residual carbon decomposition.
43
(a) TGA of ACPG-2, ACPG-3, ACGG-2 samples and (b) TGA of ACGG-3, CAT-2, and CAT-3 samples.
The temperature between 523 and 723 C was attributed to the total mass reduction of ammonia, water, carbon dioxide, carbon monoxide, methane, and acetic acid. The -NH2 group on glucosamine unit could be released by 2 ways, that is, release of NH3 and formation of hetero-aromatic ring that confirm the bio-polymeric degradation through C-O-C bond breakage. 43 The CS, AV membranes demonstrated more thermal stability than CS membranes. These conclusions proposed that thermal stability of CS membranes is possibly enhanced by AV addition due to formation of glycosidic bonding as indicated in the FT-IR spectrum.
Surface Morphology and Elemental Composition
The microstructural analysis revealed its influence on cell adhesion, migration, rapid tissue growth, and its proper functioning during wound remedy.
36
The SEM images of CS cross-linked membrane demonstrated smooth morphology which revealed good blending between individual constituents of the membrane (Figure 5). The SEM image of AV gel showed an interlinked channel network structure. By the addition of AV gel in CS membrane, agglomerates formation takes place. It displayed porous and rough morphology for CS, AV-gel blended membrane. The porous architecture was also noticed for CS, AV, and PVA films.
36
The enhanced porosity and roughness have been observed for AV, CS membranes cross-linked by genipen.
47
Similar findings had been indicated by Silva, proposing that the existence of AV-gel could encourage macromolecular rearrangement within the blended network, which is expressed as changes in membrane roughness Representative SEM micrographs of AV-gel-based membranes showing their morphology.
Structural Analysis
The XRD pattern (Figure 6) described the crystallinity or amorphous form of blended coating membranes because such physical nature affects polymeric properties like H2O absorption and biodegradation.
48
CAT membranes show 2θ values at 33.31° for CS,
38
36° for CS,
49
39° for AV,
46
and 62.61° for CS. XRD profile of CAT films displayed a slight displacement in 2θ values which indicated development of new bonds between C = O of AV and ˗NH2 of CS, consequently confirming the bonding between CS, AV, and TW.
24
The diffractogram suggested amorphous nature for CAT-2 and CAT-3. The diffractive index of ACPG membranes indicated 2θ values at 36° for CS, 44.52°, 65° for AV, 38.36° for PVA.
50
The diffractogram of ACPG-2 and ACPG-3 shows a trivial shifting of peaks that specified the formation of new bonds between ˗OH of PVA, C = O of AV, and -NH2 of CS to exhibit successful bonding of individual constituents of blended membrane.
36
XRD analysis of ACPG-2 and ACPG-3 indicated that the increase of amorphous region and crystalline region was partially developed with well extensive chain formation. The diffractive index of ACGG-2 pointed out 2θ values at 33.31° for CS and
38
36° for CS.49,51 The XRD of ACGG-2 pointed out the synthesis of blend based on AV, CS, GG, and glutaraldehyde by showing the existence of ester linkage between ˗NH2 of CS and ˗OH of GG with glutaraldehyde.
35
In ACPG-2, decrease in peak lengths depicted more disarray of chain alignment after crosslinking with glutaraldehyde and exhibited amorphous structure. ACGG-3 membrane displayed 2θ values at 28.66° for CS, 47° for AV, 56.27° for AV,
46
33.14° for CS,
38
and 70° for GG.
52
Therefore, the diffraction peaks are increased by PVA crosslinking, which changes intramolecular hydrogen bonding to intermolecular hydrogen bonding. The diffractogram of ACPG-2 and ACPG-3 shows a trivial shifting of peaks that specified the formation of new bonds between -OH of PVA, C = O of AV, and -NH2 of CS to exhibit successful bonding of individual constituent of blended membrane.
36
The overlapped patterns are found because cross-linked PVA is more crystalline than non-cross-linked and is showing larger peak area.
53
The graph indicated blending between AV, CS, GG, and glutaraldehyde by representing a minimal shift in 2θ values. The presence of more AV contents in ACGG-3 created more compactness to cause more favorable H-bonding and consequently increasing crystallinity. (a) XRD patterns of ACGG2, CAT-3, and CAT-2 membranes (b) ACGG-3, ACPG-3, and ACPG-2 samples.
Swelling Behavior of the Membranes
Swelling is the basic feature of a hydrogel. Swelling features of natural materials rely upon medium nature and diffusion of solvent in the material from an extra-cellular medium. The ACPG-2, ACPG-3, ACGG-2, ACGG-3, CAT-2, and CAT-3 membranes attained the saturation level of 329, 366, 156, 238, 211, and 344 (%), respectively, during 30 min (Figure 7). Likewise, the swelling (%) increased progressively with the increase of AV contents from 10 to 40%. The hydrophilic nature of AV polysaccharides in CS membranes might be responsible for such behavior. At swelling equilibrium, the membranes containing more than 30% AV gel resulted in a loss of structural integrity and shape due to the extremely hydrophilic behavior of AV-gel. This may cause the weakening of intermolecular interactions of CS, AV, and cross-linkers.
54
Based on such findings, the optimum AV gel content in the blended membrane was fixed as 30% w/v. The ACPG-3 sample with less CS concentration and more AV amount demonstrated the best swelling potential amongst others. The membranes with less CS contents and higher AV-gel amount became more porous and hold more solvent. So, this membrane reflects better swelling. Swelling pattern of AV-gel-based membranes in aqueous medium.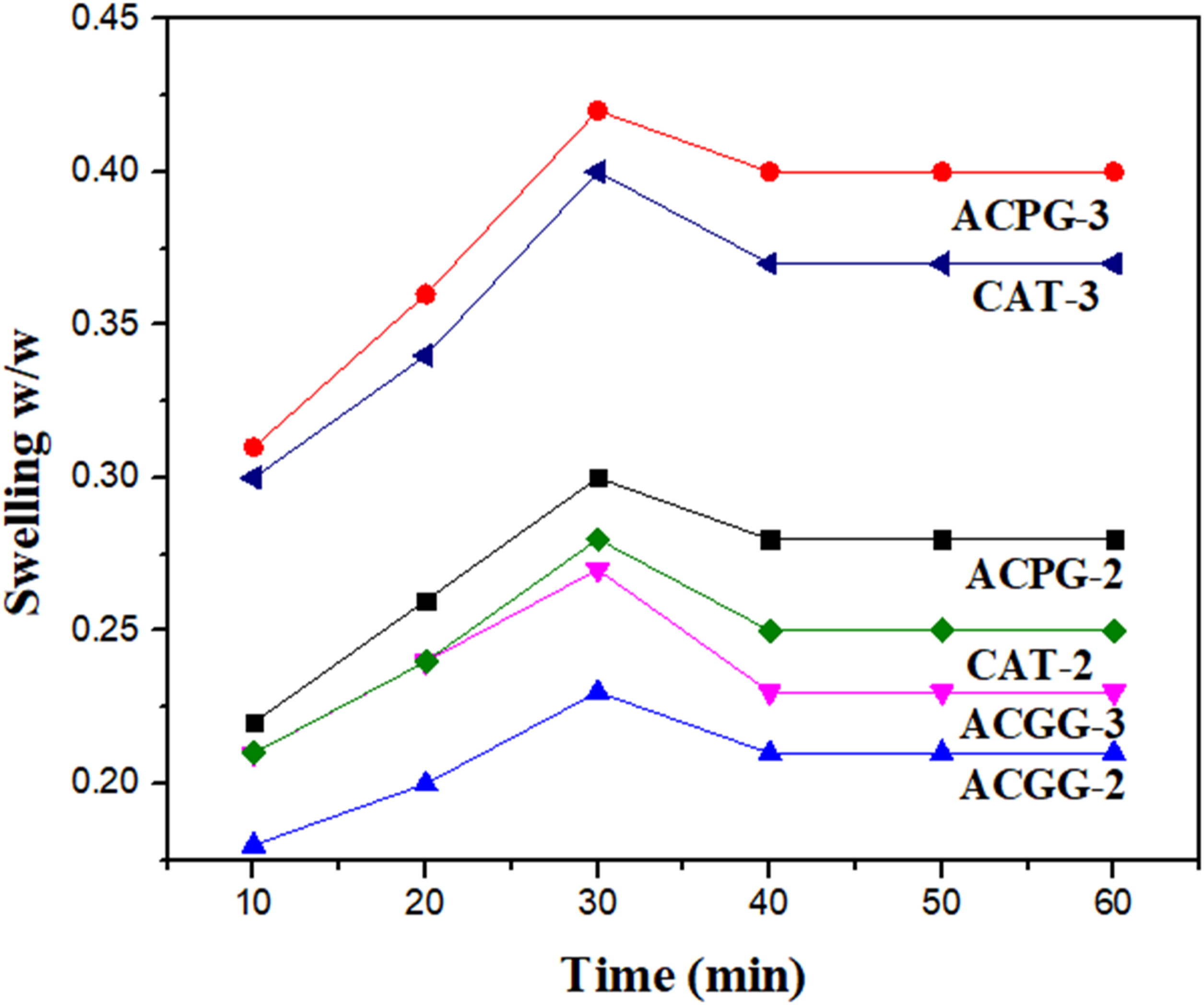
The swelling ratios of blended membranes were also observed in different pH of phosphate buffers. The swelling behavior of blended membranes was changed in different pH media due to protonation and deprotonation of functional groups. All the formulated membranes have free amino groups, polysaccharides units, and hydroxyl groups. The amino groups show attraction for protons. The concentration of protons is increased in acidic media, so the blended membranes display more swelling. 55 The availability of protons is less in neutral media, so the swelling behavior was decreased. Minimal H2O absorption was noticed for membranes in basic media due to decrease in protonation degree for amino groups. 56 Consequently, the best swelling was observed at skin pH. Increased H2O holding capability of fabricated membranes is a favorable aspect for cell attachment and growth. Similarly, the H2O holding character could facilitate nutrient transport from membrane to cell. During wound-healing application, such membranes could prevent loss of fluids from the body at wound site. 54
Antimicrobial Activity
Antibacterial Activity of AV-Gel-Based Membranes Against Gram-Positive and Gram-Negative Bacteria.

Bacterial name: E coli: Escherichia coli: S aureus: Staphylococcus aureus: B subtilis: Bacillus subtilis, and P. multocida: P multocida. The values are mean ± SD of triplicate samples (n = 3); the standard drug norfloxacin-10 μg CT0434 B) is used as control.
Conclusions
In the present study, different cross-linkers were used to prepare the aloe vera (AV) and chitosan (CS) membranes. These membranes were characterized using XRD, SEM, FT-IR spectroscopy, TGA, UV-Visible, swelling ratio, and antimicrobial studies. The membrane properties were significantly affected by the addition of aloe vera. The ACPG-3 was found better among all prepared membranes with respect to absorption, swelling, and stability. The AV gel contents addition has really improved the surface structural properties. The antimicrobial activity was tested against Es coli, S. aureus, B subtilis, and P multocida strains and membranes showed promising antimicrobial activity. Based on these data, the prepared membranes have potential applications in the biomedical field.
Footnotes
Acknowledgments
The authors extend their appreciation to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R165), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R165), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
