Abstract
This study focuses on the investigation of the significance of polymers in drug delivery approaches. The carboxymethyl cellulose (CMC), polyvinyl alcohol (PVA) and dextrin-based hydrogel membrane were prepared and employed for the sustained release of third-generation oral antibiotic (cefixime). Different proportions of CMC, PVA and dextrin were blended and hydrogel membranes were fabricated via solvent casting method. The prepared membrane was characterized by FTIR, SEM, UV-visible, TGA and swelling analysis. Cefixime drug was incorporated in the CMC/PVA/dextrin matrix and drug release was investigated. The sustained release of the tested drug (cefixime) was investigated and the drug was released in 120 min in the phosphate-buffered saline (PBS) solution. The antibacterial activity of the prepared membrane was promising against Proteus vulgaris, salmonella typhi, Escherichia coli and Bacillus subtilis strains. The swelling capabilities, thermal stability and non-toxic nature of the prepared CMC/PVA/dextrin membrane could have potential applications for cefixime drug in delivery in a controlled way for the treatment of infectious diseases.
Introduction
In recent years, the development of advanced drug delivery systems has become imperative to optimize the therapeutic efficacy of pharmaceutical agents while minimizing potential side effects.1,2 Controlled release systems, particularly polymeric membranes, have emerged as promising platforms for achieving sustained and targeted drug delivery.3–6 Among the wide array of antimicrobial agents, cefixime, a third-generation oral cephalosporin, stands out for its potent antibacterial activity against a broad spectrum of gram-positive and gram-negative microorganisms. However, its short half-life and frequent dosing requirements necessitate the exploration of innovative drug delivery strategies to enhance its therapeutic effectiveness.7–9 Cefixime exhibits good oral bioavailability (around 40-50%), and controlled release systems can further enhance its absorption and protect the drug from enzymatic degradation. 10 It has a moderate half-life (around 3-4 hours), making it suitable for controlled release systems aiming for extended drug release over 6-12 hours.11,12 Its solubility in water facilitates its incorporation into various dosage forms and release mechanisms. Various controlled release technologies can be employed, including polymers,13,14 matrices, microspheres, liposomes etc. depending on the desired release profile and the target site. Cefixime’s pharmacokinetic, stability, therapeutic, and design-friendly properties make it a promising candidate for controlled drug delivery systems.15,16 Careful consideration of its properties and potential challenges can guide the development of effective and patient-centric interventions.17,18
To overcome these challenges, the present research introduces a novel polymeric membrane composed of carboxymethyl cellulose (CMC), polyvinyl alcohol (PVA), and dextrin for the controlled release of cefixime. This tri-component system consists of three unique molecules that work together to improve the drug-delivery capabilities of the membrane. Carboxymethyl cellulose19–21 is used for supporting the membrane’s structural integrity and enabling long-term drug release. Polyvinyl alcohol 22 is well-known for its film-forming and biocompatibility qualities. 23 Because of its high mechanical strength and hydrophilic character, polyvinyl alcohol works well with CMC to limit drug diffusion and improve membrane stability.24,25 The combination of CMC, PVA, and dextrin for film preparation offers a synergistic approach to address various challenges in controlled drug delivery. CMC contributes to film-forming ability and biocompatibility, while PVA enhances mechanical stability and mucoadhesive properties. Dextrin acts as a plasticizer, improving flexibility and drug release kinetics. Together, these polymers provide films with enhanced adhesive qualities, regulated drug release, and desirable mechanical characteristics. This polymer combination plays a crucial role in maintaining sustained release profiles of drugs like cefixime, ensuring prolonged therapeutic levels in the body.26,27 Enhanced adhesion improves drug retention at the application site, facilitating longer contact and absorption, while controlled release kinetics maximize antibacterial activity against pathogens and minimize side effects. The selection of polymers also affects the stability and solubility of the drug in the film matrix, further influencing its antibacterial efficacy and release profile. Overall, the blend of CMC, PVA, and dextrin offers a balanced solution to overcome individual polymer limitations and achieve controlled drug release with improved therapeutic outcomes.28–30
To address delayed diabetic wound healing, a novel multifunctional hydrogel was developed, incorporating carboxymethyl chitosan, 2,3,4-trihydroxybenzaldehyde, copper chloride, and graphene oxide-N, N'-di-sec-butyl-N, N'-dinitroso-1,4-phenylenediamine (GO-BNN6). This hydrogel demonstrated stability, self-healing, conductivity, antioxidant, and antibacterial properties. 31 An enhanced biodegradability is promoted by the water-soluble carbohydrate derivative dextrin, which also modifies drug release kinetics.25,32 This recent study introduces self-adaptive wound dressings by incorporating MoS2-loaded nanozyme into pH, NIR, and temperature-responsive cryogels. The cryogels exhibit acid-triggered bacterial capture capacity and enhance photodynamic antibacterial efficiency. They enable controllable on-demand delivery of reactive oxygen species (ROS) and nitric oxide (NO), effectively eliminating MRSA biofilm through NO-assisted photodynamic and photo-thermal therapy. These multi-stimuli-responsive cryogels reduce wound infection, alleviate oxidative stress, and accelerate healing processes, offering a promising solution for infected wound treatment. 17 This research aims to create a flexible and effective drug delivery system by combining polymers into a single membrane. Glutaraldehyde is added to form a three-dimensional network within the polymer matrix, enhancing mechanical strength and durability. Controlled release of cefixime from this matrix is expected to prolong therapeutic benefits, reduce dosing frequency, and minimize side effects due to consistent drug concentrations. The membrane is also anticipated to exhibit increased antibacterial activity, ensuring optimal medication levels at infection sites for an extended duration.
Based on the aforementioned facts, this research represents a significant step forward in the design and fabrication of polymeric membranes for drug delivery applications. The synergy between CMC, PVA, and dextrin, coupled with the controlled release of cefixime, holds the potential to revolutionize the treatment of bacterial infections by providing a more efficient and patient-friendly therapeutic approach. Glutaraldehyde acts as a crosslinking agent that forms covalent bonds between the polymer chains of CMC, PVA, and dextrin. The fabrication process, characterization and the CMC/PVA/dextrin-based polymeric membrane for controlled release of cefixime were investigated.
Materials and Methods
Chemicals and Reagents
Polyvinyl alcohol (PVA) (Mw ∼ 30,000-70,000; viscosity. 4-6 cP, 4% in H2O (20°C), carboxymethyl cellulose (Mw ∼ 700,000) were purchased from Sigma Aldrich. All other materials were procured from the specified named supplier. Dextrin from corn starch (Mw ∼ 504.438) from Fisher Scientific, Glutaraldehyde, 50% solution; (Mw ∼ 100.12) from Junsei Chemical Co., Ltd. Sodium hydroxide and sulfuric acid were purchased from Sigma-Aldrich. Deionized water was used for the preparation of the aqueous solutions.
Preparation of Membranes without Drug Loading
Detail of Membranes Sample Preparation Without Drug Loading.

Preparation of Membranes with Drug Loading
The drug is loaded in CPDG#2 with the same composition of CMC, PVA, and dextrin, as mentioned above. Dextrin solution was prepared by the addition of 3 g of dextrin in 30 mL distilled water with constant stirring and heated at 70°C for an hour then cefixime drug (10 mg) was added onto this solution of dextrin. When the drug was dissolved in the dextrin solution the prepared solution of dextrin was added to CMC, PVA solution, which was also prepared as the procedure was adopted for CPDG#1 fabrication. Cross-linker (GA) .1 mL was also added to this solution and continuous stirring was done at 70°C for 5 h (Figure 1). Then, poured the mixture in a Petri plate and dried for 24 hours at 40°C. The membranes containing 25 mg and 50 mg drugs were also prepared by the same procedure, which was named CPDG#3 and CPDG#4.
34
Schematic for the fabrication of CMC/PVA/dextrin-based polymeric membrane for Controlled release of cefixime.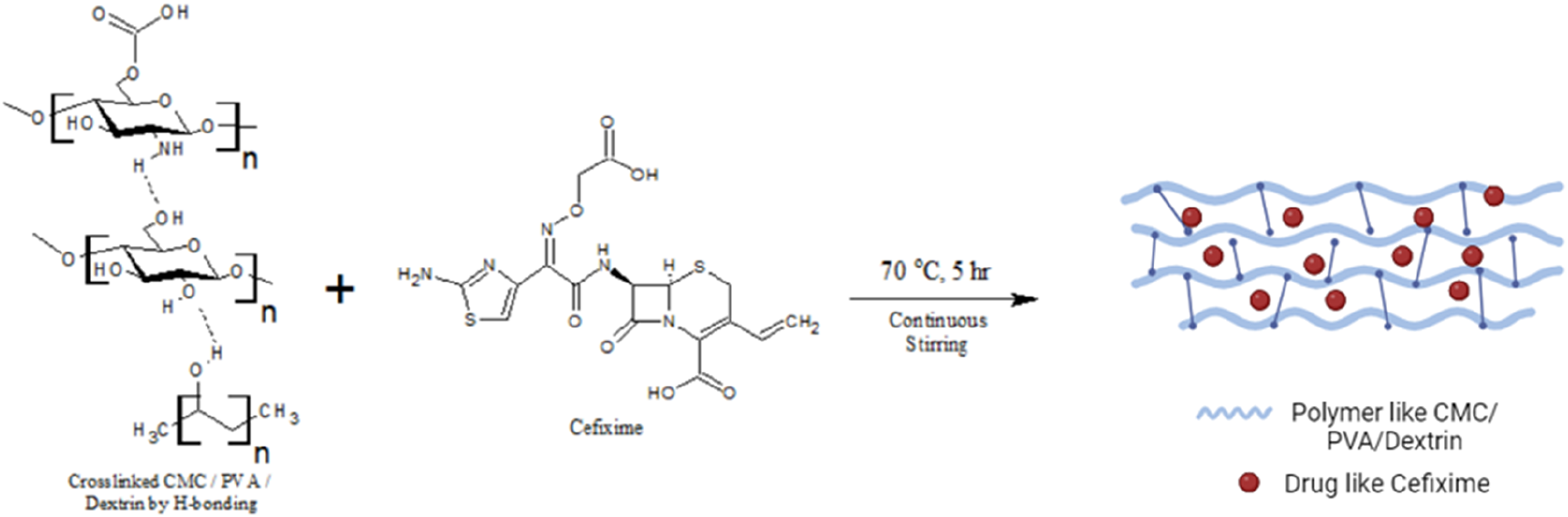
Characterization Techniques
Hydrogel characterization was performed by FTIR, UV-visible, TGA and SEM techniques. The prepared hydrogels were also studied for their antibacterial activity and cytotoxicity analysis. FTIR spectroscopy was used for detecting the functional groups in a blended membrane as well as probable molecular interactions among chemical compounds in the membrane. FTIR analysis of membrane was carried out using a Bruker Alpha-II spectrometer through a resolution of 16 cm−1 in a wavenumber range from 500-4000 cm−1. A UV-Vis spectrophotometer was used to check the absorbance of supernatant solutions and aliquots (Shimadzu, model: UV-1800). Using the Beer-Lambert law (A = ƐCL), the technique is commonly employed in quantitative analysis to calculate the concentration of an absorbing species in solution. 35 TGA is a thermal analysis method that measures the mass of a sample through time as the temperature varies. Using TGA, we analyzed the membrane which was carried out by Thermogravimetric Analyzer TGA Q50 V20.10 Build 36 Model under a nitrogen environment, a TGA Heat Exchanger system with a heating rate is about 10 °C/min was used. TGA is a valuable method for assessing the heat stability of materials polymers. 36 The SEM is one of the techniques to analyze the surface morphology of the material. SEM analysis was employed to evaluate the morphology. 37 A Zeiss SEM-type Ultra-Plus scanning electron microscope was utilized to analyze the morphology of the hydrogel membrane.
Drug Release Analysis
The rate of drug release would be proportional to the hydrogels’ equilibrium swelling ratio. Immersing 50 mg drug-loaded hydrogel membrane in 100 mL phosphate buffer solution of pH ∼ 7.4 was kept at 37°C, which was shaken for regular intervals and used for in vitro drug release investigation as a function of time. A 5 mL of solution was withdrawn and absorbance was measured at 293 nm at regular time intervals. The volume was maintained by replacing the buffer solution (3 mL) each time.
Antibacterial Activity
Using a nutrient agar medium, a pure culture of bacterial strains was established. For this investigation, 1.2 × 108 CFU/mL inoculum of all identified microorganisms was utilized. In 1 mL sterilized distilled water, 10 mg membranes (CPDG#1, CPDG#2) were dissolved. 20 mL of sterilized nutrient agar was added as well as 100 μL of fresh bacterial culture with each sterilized disposable petri plate, mixed well, or set aside for solidification over 20 min at room temperature. The wells were induced in each Petri plate using a sterilized process, and then 100 μL containing membrane sample and control (ciprofloxacin 1 mg/mL) were poured into each well. After 10 min, petri plates were transferred into incubate at 37°C for 24 h for bacteria growth and sample showing their antibacterial potential, clear zone was recorded using a zone reader in reported in millimeter (mm).
Biodegradation Study
In vitro biodegradation of hydrogel samples (50 mg) was evaluated by immersing them in phosphate-buffered saline (PBS) solution. The amount of weight lost was observed for each formulation for different time intervals. At every time interval, the hydrogel samples were taken out of the PBS solution, and weight loss was noted. The weight loss that was seen over time was indicative of the hydrogels’ biodegradation process. This approach enabled the evaluation of the in vitro biodegradation behavior of the hydrogels, which is useful for its biomedical applications.
Cytotoxicity Analysis by Hemolytic Assay
The cytotoxicity studies of the hydrogel loaded with cefixime was analyzed by hemolytic assay, a method with some modification.
38
Three mL of freshly obtained bovine blood was added in heparinized tubes to avoid coagulation and gently mixed, poured into a sterile 15 mL falcon tube and centrifuged for 5 min at 850 × g. The supernatant was poured off and RBCs were washed three times with 5 mL of chilled (4°C) sterile isotonic phosphate buffer saline (PBS) solution, adjusted to pH ∼ 7.4. The washed RBCs were suspended in the 20 mL chilled PBS. Erythrocytes were counted using a hematology analyzer and RBC count (4.068 ×106/μL) was used for this study. After dissolving 10 mg of the membrane (CFX 50, CPDG#2) into 1 mL water, 50 μL of the stock solution of (CPDG#1, CPDG#2) was mixed with 180 μL of red blood cell (4.068 ×106/μL). The reaction samples were incubated at 37°C for 30 min with continuous agitation. After the incubation was finished, the tubes were quickly cooled on ice for 5 min and then centrifuged for 5 min at 1310 × g. The supernatant, 100 μL was collected and diluted with 900 μL of phosphate buffer saline (PBS), the whole process was done under a control condition to minimize the cell lysis from external factors. After that, a 200 μL sample was poured into 96 well plates along with a negative control of phosphate buffer saline and a positive control of .1% Triton x-100. Using a micro-plate reader absorbance was recorded at 576 nm (BioTek, Winooski, VT, USA). The percentage of cell lysis was recorded using the relation shown in equation (1).
Results and Discussion
Properties of the Membranes
The material’s functional groups are identified using Fourier transform infrared spectroscopy. FTIR analysis of CMC/PVA/dextrin hydrogel membranes was recorded at room temperature in 500-4000 cm−1 and results are shown in Figure 2. The IR spectra of pure PVA have three main regions. The first region is broad and the strong absorption band appears at 3266.62 cm−1 which is linked to the hydroxyl group stretching O-H. The next region displays the slightly strong bonding at the wavelength of 2859-2882 cm−1. In the pure PVA structure, the peak that appeared in this region is of C-H stretching. The third region, which is related to C–O stretching, exhibits weak bonding and is revealed at 1050-1500 cm−1 range.
39
FTIR spectra of synthesized hydrogel membranes (CMC, PVA and dextrin).
The IR spectra of CMC indicate the stretching peaks including a broad peak for O-H stretching at 3230 cm−1, C-H stretching at 2891 cm−1, 1588 cm−1 peak due to C = O stretching and C-O stretching at 1016 cm−1 was observed. 40 The IR spectra of dextrin were found in the 3860-3230 cm−1 range, which refers to the O–H bond and the band at 2928 cm−1 indicates the vibration of the C–H bond. The band detected at 1540-1430 cm−1 is due to H-O-H bending vibration. The bands for C-O and C-O-C groups appear at 1160 and 1028 cm−1. 39
The IR spectra of CPDG#1 revealed the peak, which indicates the presence of interaction between PVA, CMC and dextrin. The broad and strong absorption band of O-H stretching appeared at 3293.46 cm−1 and for C-H stretching from the alkyl group, the peak was observed at 2918.75 cm−1. The carbonyl (C = O) peak intensity was decreased as the PVA content was increased. This demonstrates that when the concentration of PVA increased, cross-linking decreased. 26 At the wavelength range of 1242.05 cm−1 C-O stretching of acid (carboxylic acid) and C-O stretching also appeared at 1020.70 cm−1. Characteristics peak due to CH2 bending at 1417.68 cm−1, C-O-C stretching vibrational peak at 1078.80 cm−1 was also detected.
Absorption Values of the Blended Membrane in FTIR Spectra.

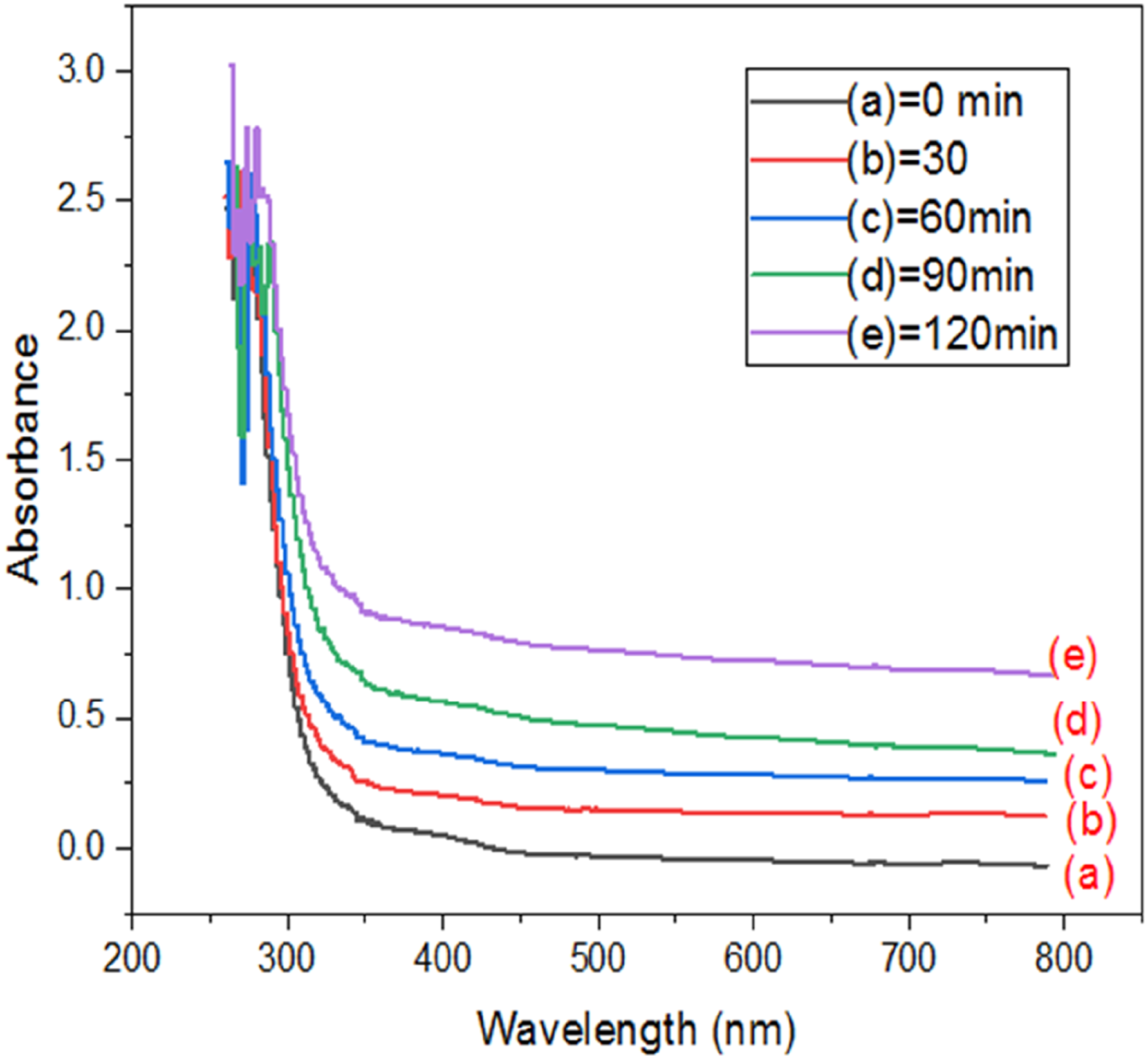
UV-vis absorption spectra hydrogel membranes (PVA, CMC, dextrin).
Thermogravimetric Analysis
Thermal gravimetric analysis (TGA) spectra generally exhibit three discernible phases of weight loss in carboxymethyl cellulose (CMC) and polyvinyl alcohol (PVA). The initial phase, which corresponds to the evaporation of moisture absorbed by the samples, takes place at relatively moderate temperatures, reaching 200°C for CMC and 150°C for PVA. During this phase, volatile components may also be lost in the case of CMC. The primary decomposition process occurs during the second stage, which occurs at elevated temperatures (150-450°C for CMC and 300-450°C for PVA). The polymer chains degrade during this phase, liberating gaseous byproducts including carbon dioxide and water vapour. Several variables affect the specific temperature range and decomposition rate, including the degree of hydrolysis for PVA and the heating rate and other unique properties of the polymer. Ultimately, exceedingly high temperatures result in the formation of a plateau, which symbolizes the remaining mass of non-volatile constituents that persist after decomposition. The residual weight of the polymers offers valuable information regarding their thermal stability and purity (Fig. S1).
The TGA curves are shown in Figure 4 which differentiates the loaded and the unloaded membrane. The weight loss phases of the hydrogels can be observed by thermograms. Firstly, both membranes showed evaporation of the moisture content followed by weight loss at 200°C due to readily degradable components like glycerol.
41
The TGA curve of the CPDG#2 unloaded membrane indicated two main weight losses. The first weight loss indicates the weight loss appeared between 250 and 400°C because of the breaking in polymer chain linkages like hydrogen bonding in both polymers (PVA and CMC). Thermal degradation or depolymerization of both PVA as well as CMC, and also dehydration for CMC saccharide rings. The second indication of weight loss was observed at temperatures between 400 and 550°C, which may be linked to the formation of PVA and CMC as byproducts of the thermal breakdown process.
42
TGA analysis (A) unloaded membrane PVA, CMC and dextrin (B) cefixime drug loaded membrane of PVA, CMC and dextrin.
In the case of drug-loaded membrane, the TGA curve of polymer blends revealed a small weight loss at 60-150°C, which is due to water molecule evaporation, the second (150-200°C) phase was attributed to polymer degradation and at third phase (200-250°C), the degradation was due to polymeric chain. Thermal stability was slightly low due to a change in crystallinity of the membrane and thermal stability was observed at 400°C. 43 Furthermore, thermograms of both loaded and unloaded hydrogels indicated that the presence of the drug-loading membrane enhances thermal stability. This enhancement suggests a stronger interconnection between the polymers, possibly due to crosslinking, which contributes to the improved thermal stability observed.
Surface Morphology
The hydrogel surface is one of the essential factors influencing drug release behavior.
42
SEM analysis was used to analyze the surface morphology of the prepared membranes.
37
The SEM images of drug-loaded and unloaded hydrogel membranes were obtained and compared (Figures 5 and 6). The loaded and unloaded hydrogels revealed a significant change in surface morphology. The unloaded hydrogel membrane has a smooth surface which indicates excellent polymer blending and the loaded hydrogel membrane showed cracks and a spherical shape inside the matrix. The results also revealed that the formation of spherical structures indicated the cefixime drug in the loaded membrane, which is useful for its application in drug delivery
39
and water molecules may absorb easily because of the hydrogel network’s interconnected cracks, affecting the rate of drug release. These cracks are also predicted to be water permeation zones and external stimuli interaction sites, such as temperature, pH ionic strength etc. Surface morphology hydrogel membrane without drug loading at different magnifications, (A) 100Kx, (B) 50Kx and (C) 25Kx. Surface morphology of drug-loaded hydrogel membrane at different magnifications, (A) 100Kx, (B) 50Kx and (C) 25Kx.

Biodegradation Study
The results of a biodegradation experiment on the prepared hydrogels submerged in PBS solution are shown in Figure 7. On days 1, 3, and 6, the CPDG#2 sample showed weight reductions of 35.5%, 26.3%, and 16.2%, respectively. The CPDG#3 samples showed similar biodegradation patterns, although CPDG#3 biodegradation was slightly lower than the CPDG#2 samples. These results suggest that there may be differences in the crosslinking density or structural makeup of the hydrogel formulations, which could influence how easily they break down chemically or enzymatically in PBS. The distinct hydrogel formulations’ differing biodegradation rates highlight how formulation parameters affect degradation behavior. These revelations provide important details regarding the hydrogels’ biodegradation kinetics. Hydrogel formulations customized for particular biomedical applications may be made easier with more research into the fundamental mechanisms causing these variations in biodegradation rates. The biodegradation of hydrogels based on CMC/PVA/dextrin can be influenced by various factors. The hydrogel’s composition, specifically the ratios of PVA, CMC and dextrin, can affect how susceptible it is to biodegradation. Differentiating degradation rates may also be caused by the polymers’ chemical makeup and susceptibility to the medium. The degree of crosslinking affects the hydrogel network’s stability and resistance to breaking down. Slower rates of biodegradation are generally the result of higher crosslinking densities, which also improve network stability.
17
In addition, the hydrogel’s physical properties, like pore size and distribution, can influence the biodegradation mode. Larger pore sizes and increased interconnectivity could enhance the degradation by making it easier for degrading agents to enter polymer chains.
27
Biodegradation behavior of hydrogel membranes in Phosphate-buffered saline.
Drug Release Analysis
The drug-loaded membranes, CPDG#2 and CPDG#3 were submerged in a solution of 100 mL phosphate-buffered saline (PBS) at pH ∼ 7.4 and kept at 37°C to examine the drug’s release behavior (Figure 8). The drug release was monitored for 120 min and in the case of CPDG#2, the drug release (10.8%) was started within the first 10 min and increased linearly over the next 60 min, reaching 98.15% at the end of 120 min. Comparably, for CPDG#3, the drug release was observed to be 14.2% during the first 20 min and to increase over the next 110 min, reaching 90.6%. According to the analysis of the data, the rate of drug release was first rapid and then gradually decreased over time. It is possible to deduce from the results that the CMC/PVA/dextrin membrane has the potential to deliver drugs depending on pH. Stimuli-responsive hydrogels can react to outside stimuli, like variations in temperature, exposure to light, or changes in pH.
29
These stimuli are useful for controlling and enabling on-demand release mechanisms by modulating drug release rates.
26
The prepared membranes that are stimuli-responsive can react to outside stimuli, like variations in temperature, exposure to light, or changes in pH. These stimuli are useful for controlling and enabling on-demand release mechanisms by modulating drug release rates.
28
The release mechanism of cefixime from the CMC/PVA/dextrin membrane involves several factors. Upon exposure to aqueous environments, the membrane matrix swells, allowing water molecules to penetrate and interact with the polymer network, thereby creating pathways for drug diffusion. Concentration gradients drive the diffusion of cefixime molecules through the swollen membrane. Various factors, including the size and charge of the drug molecules, as well as the porosity and mesh size of the membrane matrix, influence the rate of diffusion.27,30 Additionally, the integrity of the membrane may degrade over time due to hydrolysis or enzymatic breakdown, potentially leading to the release of encapsulated drug molecules.26,44 The drug release percentage from the drug-loaded hydrogels as a function of time.
Antibacterial Activity
Using the well diffusion method, the antibacterial activity of (CPDG#1, CPDG#2) was evaluated against a panel of microorganisms (Proteus vulgaris, salmonella typhi, Escherichia coli and Bacillus subtilis) are shown in Figure 9. Antibacterial activity of the sample reduced bacterial load in culture medium and resulting clean zones recorded using zone reader and results of growth inhibition potential of cefixime loaded in hydrogel was measure in mm.
45
Using the disc diffusion method, the antibacterial activity of membranes against gram-positive and gram-negative bacteria was determined and given in Table 3. For centuries ago, various infectious diseases caused by different pathogenic bacteria have been a main anxiety to the progress and existence of humans. These diseases are a global burden and the leading cause of death around the world. Hydrogel-based drug delivery systems have emerged as the most effective strategy for eradicating various bacterial infectious diseases.
31
The best antibacterial activity of the PVA/CMC/dextrin in hydrogel films could be explained by the efficient release of cefixime in the medium. It was concluded that gram-negative bacteria E. coli showed the best antibacterial activity with synthesized hydrogel films. Antibacterial activity (zone of inhibition in mm) of PVA, CMC, dextrin and cefixime membrane against (A) Proteus vulgaris (B)Salmonella typhi (C) Escherichia coli and (D) Bacillus subtilis. Antibacterial Activity of the Membrane Against Gram-Positive and Gram-Negative Bacterial Strains. Bacterial name: P. vulgaris: Proteus vulgaris, S. typhi: Salmonella typhi: E. coli: Escherichia coli: and B. subtilis: Bacillus subtilis. The values are mean ± SD of triplicate samples (n = 3); the standard drug Ciprofloxin (1 mg/mL).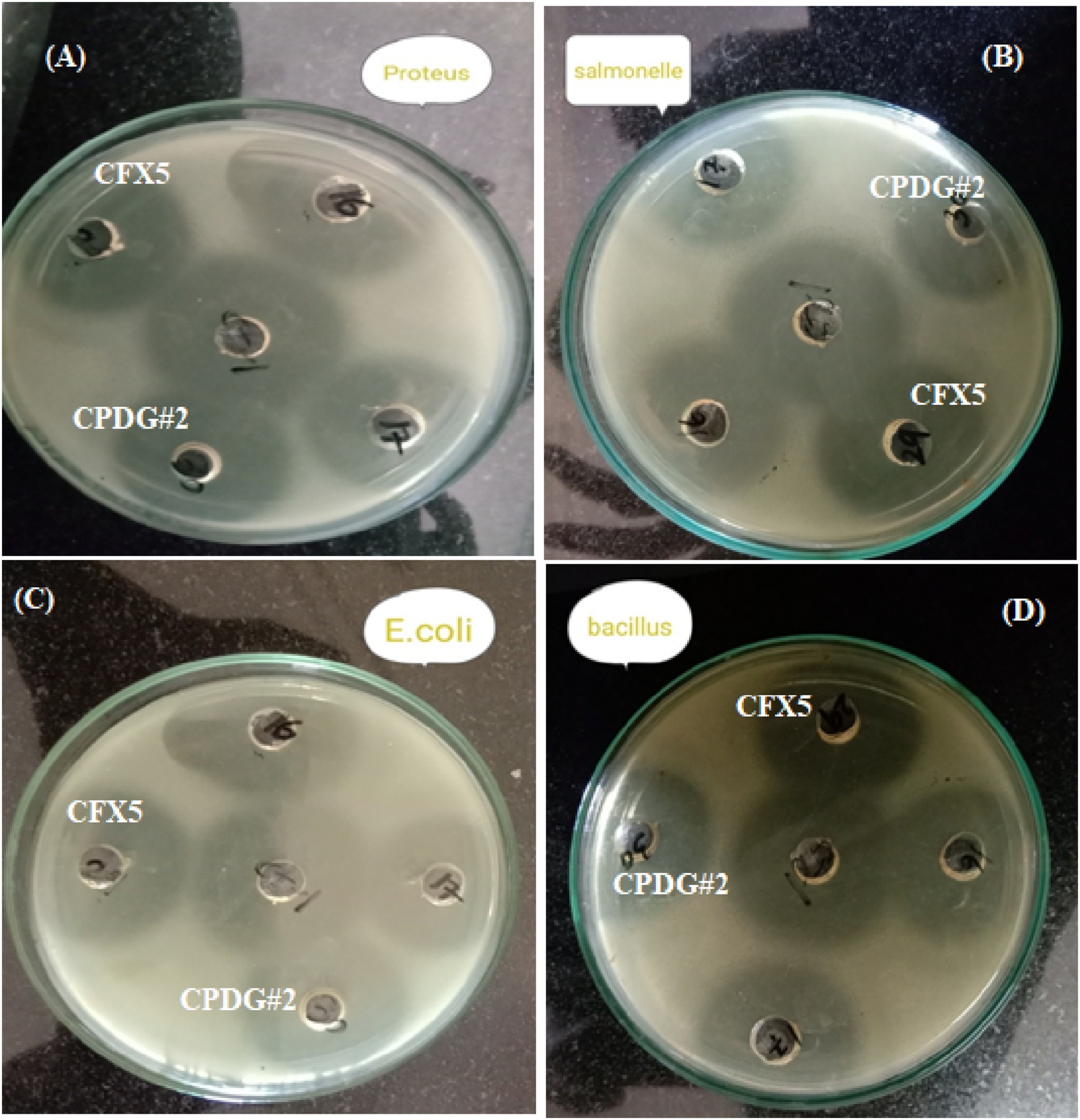

Cytotoxicity Studies
In-vitro cytotoxicity (%) Against Red Blood Cells (RBCs).

A sterile chilled buffer, Phosphate buffer saline (pH ∼ 7.4) was used as the negative control (0% background RBCs cell lysis) and Triton x-100 (.1%) was used as a positive control (100% RBCs cell lysis). The values are mean ± SD of triplicate samples (n = 3).
The Na-CMC/PVA microsphere has been prepared using water in an oil emulsion crosslinking process. 49 The use of chitosan to encapsulate Na-CMC/PVA microspheres was successful in slowing FBP release in the gastro-stomach area. The coating was further corroborated by SEM pictures, which revealed the smooth surfaces of the drug-loading coated microspheres. It was revealed that the coating approach is particularly useful for extending the release of pharmaceuticals with a short plasma half-life, increasing drug loading, and avoiding the commonly seen burst release of medications. Also, 50 evaluated the physicochemical properties of CMC and PVA films by cross-linking them with amino acids. The prepared sample showed promising antibacterial activity against B. cereus and E. coli. The L-histidine cross-linked CMC/PVA film is an appropriate antibacterial agent. The membrane fabricated the guar gum, polyvinyl alcohol and chitosan. 34 The change in concentrations of guar gum (GG) and chitosan has a promising impact on the swelling performance or permeability of these films. Membrane (B-2) of specific composition showed excellent swelling, permeability and absorption. The B-2 membrane showed promising activity against microorganisms. Cytotoxic analysis testing also revealed that the prepared membranes have potential for biomedical applications.
The fabrication of a CMC/PVA/dextrin-based polymeric membrane for the controlled release of cefixime with enhanced antibacterial activity represents a promising advancement in drug delivery systems. However, despite its potential, the study encounters several limitations that warrant consideration. Although the CMC/PVA/dextrin-based polymeric membrane for controlled drug delivery is thoroughly examined for its antibacterial activity and release kinetics, its scalability and repeatability during manufacture are not examined. Furthermore, the assessment of antimicrobial activity is predominantly conducted in controlled laboratory settings, which may constrain the generalizability of results in real-world contexts. By addressing these issues, the viability of the membrane for real-world uses could be better understood. Overall, the study shows promising results; however, in order to accurately assess the potential of the CMC/PVA/dextrin-based polymeric membrane for controlled drug delivery applications, these limitations must be addressed.
Conclusion
The CMC/PVA/dextrin was used to fabricate the hydrogel membrane by solvent casting method. These prepared membranes were characterized and their cefixime controlled release investigation revealed that the drug was released in 120 min in the phosphate-buffered saline (PBS) solution. The surface morphology and thermal stability of the prepared hydrogel membrane were changed significantly between drug-loaded and unloaded membranes. The hydrogel biodegradation was studied at pH ∼ 7.4, which was also affected due to drug loading in the membrane. The loaded membrane also showed promising antibacterial activity against a panel of microorganisms. In response to enhanced properties and controlled cefixime drug release. The findings conclude that the prepared hydrogels could be used for the controlled release of drugs for the treatment of infectious diseases.
Supplemental Material
Supplemental Material - Fabrication of CMC/PVA/Dextrin-Based Polymeric Membrane for Controlled Release of Cefixime With Enhanced Antibacterial Activity
Supplemental Material for Fabrication of CMC/PVA/Dextrin-Based Polymeric Membrane for Controlled Release of Cefixime With Enhanced Antibacterial Activity by Arif Nazir, Mazhar Abbas, Dure Najaf Iqbal, Fadia Ameen, Samiah H. Al-Mijalli, Naveed Ahmad, and Munawar Iqbal in Dose-Response.
Footnotes
Acknowledgments
The authors express their gratitude to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R158), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Authors Contribution
Arif Nazir, Fadia Ameen, Dure Najaf Iqbal: Investigation: Roles/Writing – original draft; Conceptualization; Supervision; Methodology, Samiah H. Al-Mijalli, Mazhar Abbas: Project administration; Software; Resources Munawar Iqbal, Naveed Ahmad: Visualization; Data Curation; Writing – review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their gratitude to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R158), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
