Abstract
Industrial waste still present an environmental danger for the nature and survival of all living beings. Among these toxic products, the focus has been on liquid effluents from the baker's yeast industry that cause real environmental problems mainly due to their pollutant load and the release of unpleasant odors. In order to minimize these hazards and to take advantage of these wastes for the sake of our environment, the present work consists on valorizing effluents from the baker's yeast industry on barley (Hordeum vulgare) and pea (Pisum sativum), two important agricultural products of Tunisian north-west. Results showed that this waste is characterized by its richness in organic matter, and the presence of proteins traces with high chemical and biochemical oxygen demand (COD and BOD5) values. Diluted effluent at a dose of 2.5 mg/g significantly improves germination of both plant seeds by germination index (GI) calculation, to reach a maximum of 190 ± 17% and 150 ± 14% for barley and pea, respectively. In fertigation experiment, the use of a lower dose of .62 mg/g of diluted effluent promotes plant length to reach 52 ± 4 cm and 45 ± 1.4 cm, respectively, for H. vulgare and P. sativum. Gas chromatography coupled to mass spectrometry (GC-MS) analysis after derivatization showed significant enhancement of auxin production in pea treated with .62 mg/g of cream compared to control with a concentration of 10.60 ± .81 and 8.16 ± .43 ng/gFW, respectively. In another experiment, the irrigation of pea plants with furfural, as major compound of cream, promotes length and auxin production to reach 9.89 ± .56 ng/gFW for a furfural dose of .31 mg/g. This leads us to valorize baker’s yeast effluent as an environment-friendly natural product in pea and barley agricultural and give insight to its mode of action.
Introduction
Industrial effluents are normally considered as the main industrial pollutants containing organic and inorganic compounds. Among these effluents, liquid waste, which is the least valorized in Tunisia, is directly rejected in nature and endangers its ecological balance. The major raw material used in baker’s yeast manufacturing is molasses, a low cost by-product of sugar manufacturing. The molasses is diluted, clarified, sterilized, and fed into fermentation vessels. The yeast culture is propagated in a series of up-scaling steps to levels of seed fermenters. The yeast is then transferred to production fermenters that are fed with appropriate nutrients and minerals at a controlled rate. After fermentation, the yeast is separated from the effluent using centrifugation. Like most companies in the agri-food sector, the baker's yeast production industries have problems managing their effluents, especially liquids, and continually add large amounts of wastewater containing high levels of nutrients, heavy metals, and hazardous substances to cropland.1,2 These effluents not only increase the level of nutrients but also the excessive tolerance limits and in most cases cause toxicity.3,4 The various metallic and non-metallic elements act as nutrients, but at high concentrations, they have toxic effects on seed germination and seedling growth, ultimately affecting plant growth and yield. On the other hand, various metals/nonmetals may not be toxic to the plant, but the combination of these with other products may cause toxic effects.5,6 Some scientific research studies were limited on measuring toxicological parameters and focused on finding new technologies for treatment of yeast effluents.7,8 But no scientific research dealing with their chemical profiling and their valorization in agriculture has been found in the literature.
Thus, the present investigation deals for the first time with (1) the chemical characterization of baker yeast (Saccharomyces cerevisiae) industrial effluent, (2) assessment of its effect on barley and pea germination and growth, and (3) correlation between the doses of effluent cream or furfural and auxin in treated plants.
Material and Methods
Effluent Collection
The liquid effluent used throughout this work was collected from Rayen Food Industries, a plant specializing in the baker's yeast industry, located in the north-west of Tunisia, Ben Bachir-Bousalem (36°34'14" N; 8°51'27" E). Sample was taken at the end of fermentation process and was stored under adequate conditions to ensure better stability. For biological activities, effluent was concentrated using a rotary evaporator and the cream was recovered.
Liquid Effluent Physicochemical Characterization
The pH was measured using a pH meter (INOLAB) according to the potentiometric method. Electrical conductivity and salinity were measured using an MEAS/Cond 8 conductivity meter. Dry matter determination was carried out by adding 5 g of liquid effluent to 20 g of dry sand. The whole is dried for 2 h in the oven at 105°C. 9 Total nitrogen was determined by the Kjeldahl method. 10
Toxicological Parameter Determination
COD is defined as the amount of oxygen equivalents consumed in oxidizing the organic compounds of samples by strong oxidizing agents. COD analysis used a slight modification of colorimetric method. 11 3.7 mL of COD reagent (BDH Laboratory Supplies, England) mixed with 3.30 g/L of K2Cr2O7 was added to 2 mL of dilute sample, incubated at 150°C for 2 h, and the absorbance was then measured at 600 nm.
BOD5 is a parameter that measures the amount of oxygen consumed by microorganisms for the process of decomposition of the organic matter in the water bodies. It is measured for 5 days. 12 COD and BOD5 are considered two most important quality control parameters of an effluent in wastewater treatment facility.
GC-MS Analysis
Chromatographic separation and identification of the main components of cream was performed on a Trace GC Ultra Gas Chromatograph (Thermo Finnigan, MA) coupled with a Trace DSQ mass spectrometry detector, a split−splitless injector, and an Xcalibur MS platform. 13 The column was a CP-WAX 57CB from Varian (60 m long, .25 mm i.d., and .25 μm film thickness; Varian Inc., USA). The injector and transfer line were at 200°C. The oven temperature was programmed as follows: 50°C (hold 1 min) and then raised to 220°C (3 °C/min) and isothermally hold for 13 min. Helium was the carrier gas at a constant flow rate of 1 mL/min; 1 μL of each sample was injected in the splitless mode (60 s). Mass spectrometry acquisition was carried out using the continuous (EI positive) scanning mode from 45 to 500 amu. Cream components were identified by comparison of their relative retention times and mass fragmentation with those of authentic standards and computer matching against an NIST/EPA/NIH Mass Spectral Library (NIST 08). GC-MS analysis of auxin was performed according to the method of Tada et al. in 2014 with slight modifications using a DB-1 HT fused-silica capillary column (15 m × .25 mm, film thickness of .10 μm; 6890 N, Agilent Technologies, USA). 14 The ion source was held at 220°C, the injector and detector temperatures were set at 250°C, the transfer line was at 290°C, and the column temperature was programmed to rise from 120°C to 240°C at 15°C/min and then from 240°C to 390°C at 8°C/min and finally maintained at 390°C for 6 min.
In vitro Germination Test
Zucconi test is the best way to test the phytotoxicity of compost, effluents, or extracts on plant germination. It consists on calculating the GI which is widely used to test the effect of salinity, soil pathogens, toxic substances, and other physical and chemical properties of effluents and extracts on plant germination.
15
Barley and pea seeds were purchased from a local market in Bou Salem, Tunisia (36°34'12" N; 8°59'30" E) in September 2018. Then, 10 seeds of each species were placed on a screen in a glass petri dishes with dimensions of 110 mm × 20 mm. Seeds were irrigated with .5 mL of the cream recovered from the rotary evaporator, diluted in water from 10 to .31 mg/g, and then capped and kept in a dark incubator at 25°C temperature for 5 days. GI was calculated by counting the grown seeds and determining the average sum of seeds radical elongation in each tested sample by the following formula:
NE: number of germinated seeds irrigated by diluted cream, NT: number of germinated seeds in the control irrigated by water, LE: average length of the radical of germinated seeds for the sample, and LT: average length of the radical of germinated seeds for the control. All the experiments were carried out in triplicates.
Fertigation Effect
The main objective of this experiment is to test the effluent effect on plant growth and to optimize its beneficial concentrations for these species. This essay was conducted in accordance with the natural climatic conditions favorable to the growth of barley and pea. Indeed, all the pots were placed in a greenhouse designed as a growth chamber programmed for a photoperiod of 12 h of light and 12 h of darkness, with a photosynthetic photon flux density of 300 μmol m−2 s−1, temperature of 24 ± 1/18 ± 1°C day/night, and relative humidity of 60/70 ± 3%. 9 The test was carried out in a polystyrene honeycomb plate filled with soil (20% of sand, 2% of clay, and 5% of organic matter, pH = 7 ± .5). Plants aged of 1 week are carefully irrigated with 10 mL of water or diluted cream in water with a dose of 10–.31 mg/g soil for 20 days. In the same period, the measure of plant growth was continually done.
In another experiment, pea plants were irrigated with doses of furfural (Sigma-Aldrich, Milano, Italy), the most abundant compound on baker yeast effluent, from 1.25 to .15 mg/g. Plant growth, furfural dose, and auxin concentrations in leaves were then correlated.
Auxin Characterization in Treated Pea Plants
Leaves of pea plants previously treated with cream or furfural were moved and extracted with water at 4°C as previously reported by Jager et al. 16
Subsequently, the extract was dried on a rotary evaporator. Extracts for auxin characterization were taken up in 30 mL of KHSO4 (.3 N) and distilled water, respectively, and partitioned 3 times with 10 mL of chloroform. The organic phase was then dried under rotary evaporation, transferred to a tapered-bottom vial, and taken to complete dryness in a sample concentrator. Trimethylsilylation was then performed by adding 40 mL N, O-bis (trimethylsilyl) trifluoroacetamide (BSTFA) containing 1% trimethylchlorosilane (TMCS) to the dry sample with 10 mL pyridine to aid dissolution and heated at 80°C for 30 min. Subsequently, the extract was dried under nitrogen and 15 mL BSTFA (1% TMCS) was added. Samples were then placed in an oven at 80°C for further 30 min and then used for gas chromatography analysis.
Samples and standards were dissolved in methanol (.1–1.0 mg/mL) and 1 μl was injected through a split-injector (1/5) in triplicate. MS spectra were detected in EI mode. Furfural and auxin were quantified as percentages of the total peak areas.
Statistical Analysis
All experiments were carried out in triplicates. All values are expressed as mean ± standard deviation (±SD), and the statistical significance of differential finding between experimental groups was determined by one-way ANOVA test. Analysis of variance was done with the software Statistica, using Tukey’s test. Statistical significance is indicated with asterisks (P < .05).
Results
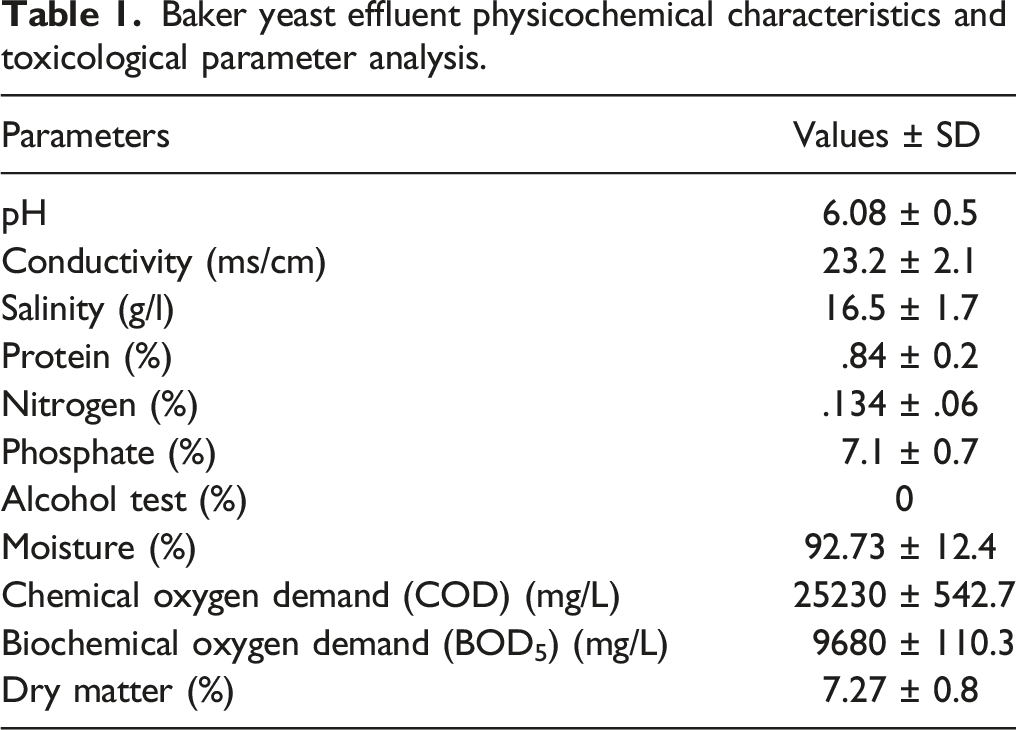
Physicochemical Characteristics, COD, and BOD5 Determination
Baker yeast effluent physicochemical characteristics and toxicological parameter analysis.
Cream Gas Chromatography Analysis
By GC-MS analysis, 14 compounds were detected in cream. Only 5 compounds were identified by comparing to NIST library. Furfural is strongly dominating followed by furfuryl ethyl ether and 1-pentanol (Figure 1). For our best knowledge and literature survey, this is the first report on the characterization of baker yeast liquid effluent. GC-MS analysis of baker yeast cream effluent identified peaks: 
In vitro Germination Test
In another experiment, the determination of the GI of seeds of barley and pea during 5 days of treatment showed that this parameter is significantly higher using diluted cream than water control. It reaches a maximum of 190 ± 17% and 150 ± 14% for barley and pea, respectively, at a dose of 2.5 mg/g, and then gradually decreases (Figure 2). Germination index (GI) of barley and pea seeds using different concentrations of baker yeast effluent cream. All experiments were made in triplicates. Values are expressed as means ± SD. *Indicates significant differences (P < .05). The use of a dose of 2.5 mg/g of cream indicates a significant difference in Barley GI compared to the other used doses and control. The same significant difference using the same dose is noted for pea GI compared to the other used doses and control.
Fertigation Effect
Irrigation with diluted cream showed an increase of stem length average compared to the control. It reaches the maximum of 52 ± 4 cm and 45 ± 1.4 cm for barley and pea, respectively, in plants irrigated by a dose of .62 mg/g (Figure 3). However, below and above this concentration an antagonistic effect resulting in a decrease of plant length was noted, confirming the toxicity of cream at high doses. Barley and pea plant length after 20 days of fertigation by different concentrations of baker yeast effluent cream. All experiments were made in triplicates. Values are expressed as means ± SD. *Indicates significant differences (P < .05). The use of a dose of .62 mg/g of cream indicates a significant difference in Barley GI compared to the other used doses and control. The same significant difference using the same dose is noted for pea GI compared to the other used doses and control.
Auxin Characterization in Treated Pea Plants
Auxin in treated pea plants was characterized by GC-MS analysis after the derivatization step. A significant enhancement in auxin levels was noted in plants treated with .62 mg/g of cream according to control (10.60 ± .81 and 8.16 ± .43 ng/gFW, respectively) (Figure 4). Variation of pea plant length using different concentrations of baker yeast effluent cream and correlation with auxin level. All experiments were made in triplicates. Values are expressed as means ± SD. *Indicates significant differences (P < .05). The use of a dose of .62 mg/g of cream indicates a significant difference in pea plant length compared to the other used doses and control. This significant difference in plant length using the same dose is also associated to a significant difference in auxin production compared to the other used doses and control.
On the other hand, pea plants irrigated with furfural showed an increase in stem length average compared to control. A significant correlation among furfural dose, plant length, and auxin level was noted. It reaches 9.89 ± .56 ng/gFW and 70 ± 6 cm for furfural dose of .31 mg/g (Figure 5). Dose of furfural and response of pea plant length and auxin level. All experiments were made in triplicates. Values are expressed as means ± SD. *Indicates significant differences (P < .05). The use of a dose of .31 mg/g of furfural indicates a significant difference in pea plant length compared to the other used doses and control. This significant difference in plant length using the same dose is also associated to a significant difference in auxin production compared to the other used doses and control.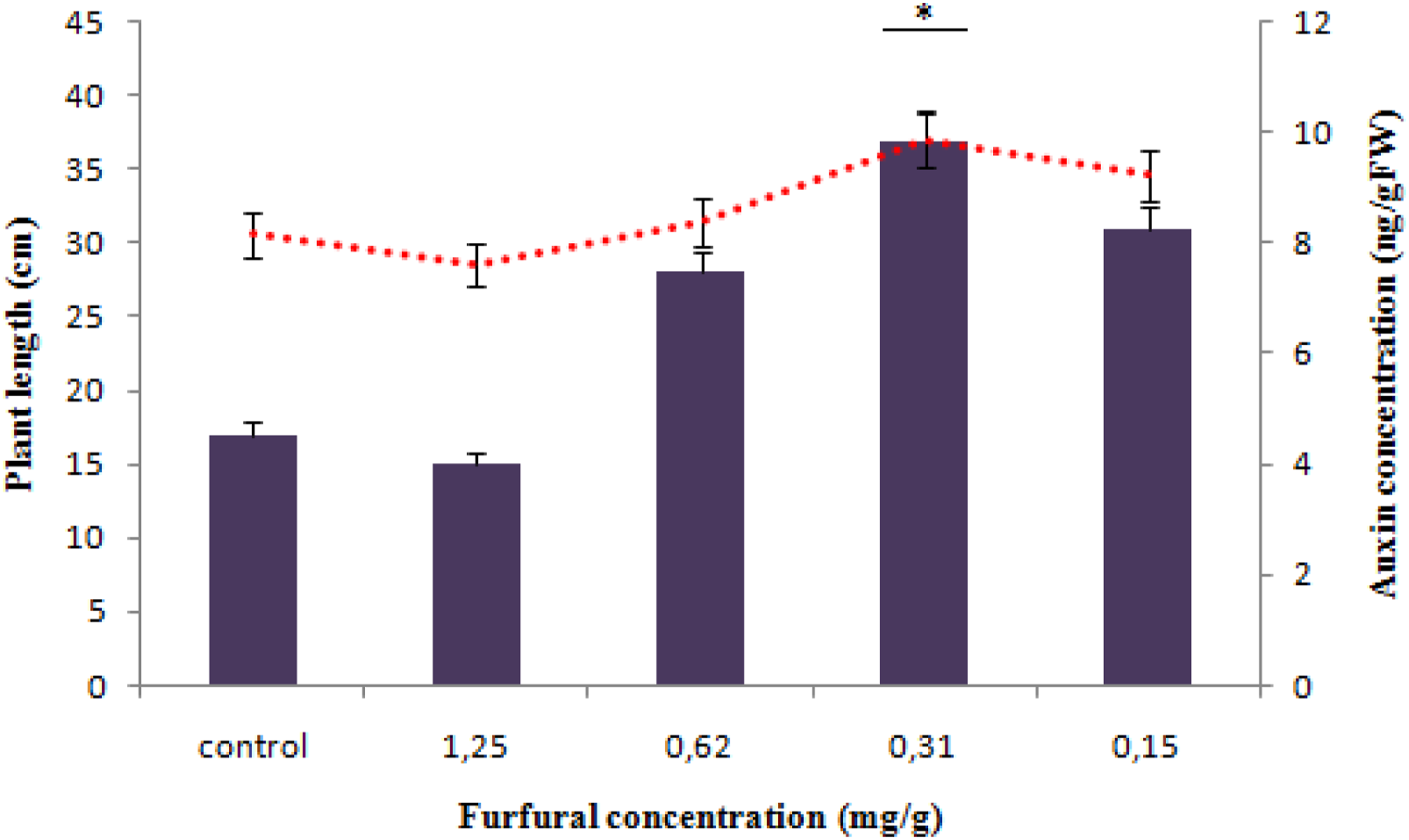
Discussion
The physicochemical characteristics of baker’s yeast effluent depend on the raw material (molasses) and the manufacturing process. The pollutant effect of the latter resulted in a slightly acidic pH of 6.08 and a high dry matter content of 7.27 ± .8%. Similar results were reported by Sirbu and Begea in 2011. 17 This effluent is characterized by the absence of alcohol which could not therefore be at the origin of a possible toxicity. 18 Its mineral composition shows a significant composition of moisture (92.73 ± 12.4%) coupled with a large amount of phosphate (7.1 ± .7%) and conversely low doses of protein (.84 ± .2%) and nitrogen (.134 ± .06%). These results are in an agreement with those reported by the same authors. High COD and BOD5 values of 25230 ± 542.7 and 9680 ± 110.3 mg/L were, respectively, noted confirming high pollution degree of this effluent. These results confirm those found by Gengec and Kobya in 2013. 19 Industrial effluents are a major source of environmental toxicity. The presence of phenolic compounds, betaine, melanoids, caramel, and furfural components contributes to the dark brown color of yeast effluent and makes its processing wastewater difficult to treat. 20 Using GC-MS, 5 compounds were identified compared to NIST library with domination of furfural followed by furfuryl ethyl ether and 1-pentanol. Furfural is a product of the dehydration of sugars, as occurs in a variety of agricultural and industrial by-products. 21 Furfural derivatives can be used as alternative fuels or fertilizers reducing the amount of waste produced. 21 One of these derivatives is furfuryl ethyl ether which is researched as potential fuel additives and considered even as a potential replacer of petroleum diesel.22,23 1-pentanol is a natural by-product of microbial fermentations from amino acid substrates with a potential application as biofuels. 24 Acetic acid is a physiological product of yeast alcoholic fermentation, and it is a well-known food preservative due to its antimicrobial action. 25 Our results confirm those found by España-Gamboa et al. 20
The effect of cream on barley and pea seed germination for 5 days of treatment showed a GI of 190 ± 17% and 150 ± 14% for barley and pea, respectively, at a low dose of 2.5 mg/g and then gradually decreases. Phosphorus has been shown to be an effective way of promoting early seedling growth. Usage of phosphorus-treated seeds improves assimilation of mineral nutrient, avoiding contamination of the environment. 26 However, high doses of nitrogen significantly suppressed seedling. 27 Our results are in an agreement with those obtained by Perveen et al. 28 Soaking barley seeds in strong alkalinity as NaOH medium does not have any effect in germination compared to control. 28 It seems that using neutral pH medium enhances barley seedling enzyme activity.
Irrigation with diluted cream at .62 mg/g increases plant length average to reach 52 ± 4 cm and 45 ± 1.4 cm for barley and pea, respectively, compared to the control. These results are consistent with those reported by Rana et al in their review dealing with the use of urban wastewater for growth and germination stimulation of maize, rice, and sorghum 29 and confirm those found by Sharma et al dealing with pea germination and growth promotion by paper industry's effluent. 30
To hypothesize the mode of action of baker’s yeast effluent cream on phytohormones, auxin in treated pea plants was characterized by GC-MS analysis. A correlation between auxin levels and plant length enhancement was noted in these plants comparing to control with levels of 10.60 ± .81 and 8.16 ± .43 ng/gFW, respectively. Similar results were found in our previous study dealing with the effect of wool wax from washing effluent on olive plant growth improvement by auxin induction. 9 A dose-response correlation was noted when pea plants were irrigated by doses of furfural as a major compound of cream. Auxin was enhanced in plants treated by .31 mg/g of furfural, to reach 9.89 ± .56 ng/gFW. For our best knowledge and literature survey, this is the first report on indole-3-acetic acid (auxin) enhancement in Pisum sativum by baker yeast effluent and its main compound, furfural. Indole-3-acetic acid (IAA) is a key plant growth hormone, involved in diverse processes such as branching, seed development, germination, and gravitropism. 31 IAA is synthesized mainly from tryptophan, via 1 intermediate, indole-3-pyruvic acid. 32 Volatile compounds, like aldehydes as plant growth modulating agents, were studied. 33 España-Gamboa et al reported that baker yeast effluent is rich on furfural, a highly volatile aldehyde and a precursor for cytokine hormones.20,34 This can give insight on the mode of action of baker yeast effluent and its major compound furfural on pea growth by auxin production induction. Further studies dealing with the effect on seed size and number are needed for a commercial point of view.
Conclusion
Baker yeast industries release unpleased liquid waste. Our scientific work deals with the valorization of this liquid in agriculture. Cream of liquid waste enhanced germination and growth of barley and pea. Besides, being rich in furfural, it improved auxin production in pea at a dose of .62 mg/g. This presents new tools to valorize industrial wastes in agriculture.
Footnotes
Author Contributions
AISSANI N ensured the preparation of the experimental protocol and its realization, involved in all the analyses carried out, wrote the manuscript, and interpreted the results obtained. AISSANI R participated in the experiment realization. GHIDAOUI M supplied instruments and space for germination and fertigation assay. ZOUIDI F participated in the improvement of the manuscript and English correction. SEBAI H participated in revision and correction of the manuscript. All authors have reviewed and approved the submission of this manuscript version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author extends his appreciation to the Deanship of Scientific Research at King Khalid University for funding this work through the General Research Project under Grant number (RGP.2/83/44).

