Abstract
Introduction
Hepatic inflammation is considered key driver of hepatic tissue impairment.We aimed to explore the interaction of Halamphora coffeaeformis (Amph.) with low dose ionizing γ radiation (γR) exposure against D-galactosamine (D-GaIN)-induced chronic hepatitis in Albino rats.
Methods
Chronic hepatitis was induced with single dose of D-GalN (400 mg/kg BW i.p.). Rats received 400 mg Amph/kg BW daily by gastric gavage concomitant with .25 Gy γ-R. Liver oxidative stress and inflammatory status were assessed. Gene expression levels of signal transducer and activator of transcription 3 (STAT3) and nuclear factor kappa B (NFKB) were estimated by q-PCR. D-Galactosamine injection significantly encouraged hepatic oxidative damage and inflammatory disturbance accompanied with improved intercellular adhesion molecule-1 level (ICAM-1).
Results
messenger RNA gene expression levels of STAT3 and NF-kB were expressively higher in D-GaIN-treated animals. Histopathological examination supported results. Interestingly, Amph treatment with γ-radiation (γ-R) subjection displayed significant improvement of oxidative and inflammatory status along with controlled signaling molecular factors which was supported by amended histological structure of induced liver hepatitis.
Conclusion
Results conclude the efficacious control of liver hepatitis progression by dual collaboration of Amph. with low dose γ-R via control of vital growth signaling factors linked with inflammation thru anti-inflammation, antioxidative and anti-proliferative activities.
Keywords
Introduction
Liver injury is a mutual cause of liver disease, which is considered the key controller of hepatic tissue injury, causing evolution from nonalcoholic fatty liver disease (NAFLD) to severe fibrogenesis and, lastly, hepatocellular carcinoma. Liver diseases generally happen because of chronic hepatocellular injury triggered by the alcohol overuse, hepatitis C virus chronic infections, bile duct impairment and NAFLD. 1
D-Galactosamine (D-GaIN), a discriminating hepatotoxin, leads to liver damage like human viral hepatitis through reduction of uridine nucleotides, leading to diminished formation of RNA and proteins. 2 D-Galactosamine causes reduction of cellular energy load thus leads to cellular death. The steady compensation of energy levels activated by D-GaIN encourages a process which ends in apoptosis. 3 D-Galactosamine prevents hepatocytes synthesis of messenger RNA (mRNA) and protein with hypersensitivity to Kupffer cells released tumor necrosis factor-α (TNF-α), that stimulate cellular death signaling pathways. 4 Reactive oxygen species (ROS) are generated from activated Kupffer cells after D-GaIN treatment, moreover TNF-α that leads to apoptosis was synthesized. Excessive ROS disturbs homeostasis, leading to oxidative stress, that plays a serious part in liver diseases. 5
Plant derived food additives have developed natural antioxidant in order to alternate synthetic antioxidant with several disadvantage. Worthy to remark that algae are able to produce similar effective and valued natural products. Consequently, utilization of algae in the pharmaceutical applications has attracted global attention. 6 The brown micro-algae including Halamphora, could be a source of a diverse class of bioactive compounds, it was found that H. coffeaeformis alga belongs to Bacillariophyta which has high levels of chlorophyll and carotenoids such as fucoxanthin and β-carotene which have various biological activities as antioxidant, anticancer, antimicrobial. 7 Studies stated that phenolics and carotenoids have antioxidant activity against peroxidation and free radicals. H. coffeaeformis algae extract exhibited hopeful scavenging properties to neutralize DNA damage caused by hydrogen peroxide. 8
Although the health effects of ionizing radiation are quite variable, it is generally known that high doses are hurtful and can destroy organisms, but low dosages of radiation are remarkable and beneficial to health. Earlier studies presented that low dose frequency ionizing radiation improves body immunity, antitumor, and antioxidant activities and also accelerates oxidative stress recovery triggered by chemotherapy. 9 So, the significance of this study is to evaluate the interaction between low dose gamma radiation (IR) and algae extract to suppress D-GaIN-induced chronic hepatitis in male Albino rats by targeting signaling molecular targets.
Materials and Methods
Chemicals
D-galactosamine was acquired from Sigma Aldrich Limited, United States (Catalog No: 1772038), and all chemicals used of high grade were purchased from Sigma Aldrich Limited, United States.
Preparation of Algal Extract
Gas Chromatography Mass-Spectrometry Activity of H. coffaeiformis Extract.


GC-MS chromatogram represents the separated bioactive constitutes of H coffaeiformis extract.
Charachterization of H. coffeaeformis alga was done by El-Sayeed et al 9 from whom we obtained H. coffeaeformis alga extract in the current study.
Animals
Male Wistar rats (170 ± 30 g) were acquired from Nile Pharmaceutical Co., Cairo, Egypt. Before experiment begins, animals were permitted to acclimatize at the animal’s house of National Center for Radiation Research and Technology for 7 days. Animals exposed to typical laboratory circumstances of 12 hours light/12 hours dark cycle, with temperature 25 ± 2°C and moisture of 60 ± 5%. Rats in cages have free access to food and ad libitum water. Standard laboratory diet was administrated to rats (58% carbohydrates, 3.4% lipid, 20.5% protein, 1.49% minerals, .53% phosphorus, 3.1% cellulose, .98% calcium, and 12.0% moisture). The current study protocol follows the Guide for the Care and Use of Laboratory Animals (NIH Publications No. 85-23, revised 1996) and was supervised by the Research Ethics Committee of the National Center for Radiation Research and Technology, Atomic Energy Authority (Ref. no. 202/21).
Radiation Facility
Rat’s body was exposed to gamma irradiation at Atomic Energy Agency’s National Center for Radiation Research and Technology using a Cesium-137 source (gamma cell 40), Atomic Energy of Canada Limited (Chalk River, ON, Canada). As reported by Kandil et al, rats received a single dose of .25 Gy at a dose rate of .423 Gy/min. 10
Median Lethal Dose of Algal Extract (LD50)
Algae extract’s LD50 was determined via rats according to Akhila et al. 11 Rats were administrated high dose (500-5000 mg/kg b.wt.) of algae extract and death was documented 24 hours after treatment. Animals were fed through gavage with 10% (LD10) of LD50 (400 mg/kg b.wt.).
Experimental Design
This experimental study was conducted at the animal’s house of National Center for Radiation Research and Technology. After 7 days acclimatization period, rats were randomly separated to 8 groups (8 rats each). 1. Group I (control): rats received 1.0 mL of physiological saline solution/day by gastric gavage and were left till the end of the experiment (1 month) then they were sacrificed. 2. Group II (Gal group): rats were injected with D-GalN (Sigma Aldrich) intraperitoneally in a single dose of 400 mg/kg body weight
12
for 1 month. 3. Group III (Amp group): rats received daily extract of Halamphora Coffeaeformis (400 mg/kg b.wt) for 1 month by gastric gavage. 4. Group IV (IR): rats were subjected to single gamma radiation (IR) dose .25 Gy
10
and left for 1 month (end of experiment). 5. Group V (Gal + IR): rats received D-GalN injection like group II, then after 48 hours; rats were subjected to low dose gamma radiation as in group IV. 6. Group VI (IR + Amp): rats received H. coffeaeformis extract daily for 1 month by gastric gavage then subjected to low dose gamma radiation as in group IV 7. Group VII (Gal + Amp): rats received D-GalN intraperitoneal injection like group II then after 48 hours; rats received H. coffeaeformis extract as in group III 8. Group VIII (Gal + IR + Amp): rats received D-GalN injection like group II, then after 48 hours rats received H. coffeaeformis extract as in group III, followed by introduction to low dose gamma radiation like group IV.
Samples Collection
Blood Sampling
By the end of experiment duration (1 month), rats were anesthetized using urethane and blood samples were acquired via cardiac puncture and gathered in plain vacutainer tubes. Then samples were left for coagulation and sera were obtained after centrifugation at 4000 rpm for 10 minutes for biochemical analysis.
Tissue Sampling
Livers of rats were dissected out and separated into 2 portions, 1 portion was stored in 10% formalin for histological studies and the other portion was homogenized (10% w/v) in phosphate buffered saline (.02 M sodium phosphate buffer with .15 M sodium chloride, pH7.4). Liver homogenates were established using a portion of liver homogenized in a tissue glass homogenizer with a Teflon pestle.
Biochemical Investigations
Liver Function Markers and Some Related Parameters in Serum
The serum levels of liver enzymes (aspartate aminotransferase (AST) and alanine aminotransferase (ALT)) were assessed colorimetrically. 13 Activities expressed as IU/L. Serum level of albumin was determined using the bromocresol green method. 14 Levels of serum total proteins were estimated by coloremetric method using Biuret reagent. 15
Inflammatory Markers
The levels of proinflammatory cytokines including, IL6 (Catalog No: MBS175908) and TNF-α (Catalog No: MBS2507393), ICAM (Catalog No: MBS267983) were assessed in liver tissue by ELISA kits purchased from MyBiosource (United States).
Determination of Nuclear Factor Kappa B and Signal Transducer and Activator of Transcription 3 (STAT3) mRNA Gene Expression by Quantitative Real-Time PCR in Liver Tissues
Extraction of RNA and Reverse Transcription
mRNA extraction from liver tissues was done using the constructer’s instructions of TRIzol reagent (Life Technologies, USA). Complementary DNA (cDNA) synthesis was done with reverse transcriptase (Invitrogen) where 1 μg of total RNA was used as template.
Quantitative Real-Time Polymerase Chain Reaction
PCR Primers.

Western Blotting of c-Jun N-Terminal Kinase Protein
Tissue liver proteins were isolated using TRIzol reagent (Invitrogen) as stated by Chomczynski, 17 and concentration of proteins were measured as stated by Bradford. 18 Proteins (20 μg/lane) were separated on 10% SDS-PAGE then moved onto PVDF films. The films incubated with rabbit IgG Jun N-Terminal Kinase (JNK) antibody (Cell Signaling Technologies, USA), and β-actin antibody (Proteintech, USA) as loading control. After washing films incubated at room temperature with HRP-conjugated goat anti-rabbit IgG (Cell Signaling Technologies, USA). The films were established and pictured by Amersham detection chemiluminescence kit along with the constructor’s instructions and then subjected to X-ray. c-jun proteins concentrations were analyzed using a scanning laser densitometer (Biomed Instrument Inc., USA).
Determination of Liver Tissue Oxidative Stress Markers
Lipid peroxidative products; malondialdehyde (MDA) were detected clorimetrically in 10% liver homogenates via the thiobarbituric acid test as stated by Satoh. 19 The pink color intensity was spectrophotometrically detected at 535 nm.
Reduced glutathione (GSH) amount were detected according to Ahmed et al method, 20 the yellow color formed of liver homogenate (.2 ml) was measured spectrophotometrically against the blank at 412 nm.
While the oxidized glutathione (GSSG) was measured in Liver homogenate as stated by Hissin and Hilf. 21 Reaction was followed kinetically for 2 min, at a wavelength of 415 nm, GSSG was measured after GSH derivatization by 2-vinylpyridine.
The Paraoxonase-1 (PON-1) activity was kinetically detected in liver homogenate as stated by the method of Kumar. 22 The rate of hydrolysis of paraoxon was followed spectrophotometrically by measuring the liberation of p-nitrophenol at 405 nm at 37°C over 50 sec after 1 min lag time.
Histopathological Study
Liver Specimens of all studied rats were cleansed, dehydrated in ethanol, cleaned in xylene, and fixed in paraffin wax. Liver was sliced (5-6 μm thick), deparaffinized then Hematoxylin and Eosin (H & E) tainted for microscopic investigation. 23
Statistical Analyses
Data was analyzed using SPSS programme (version 20) through one-way analysis of variance (ANOVA) followed by Duncan’s multiple Range test (DMRT). Data were expressed as mean ± standard deviation (SD). P values < .05 is significant.
Results
Biochemical Studies
Effect of H. caffeaeformis and/or Low Dose IR on Liver Function Among Groups
Liver Function Test Parameters Among all Studied Groups.
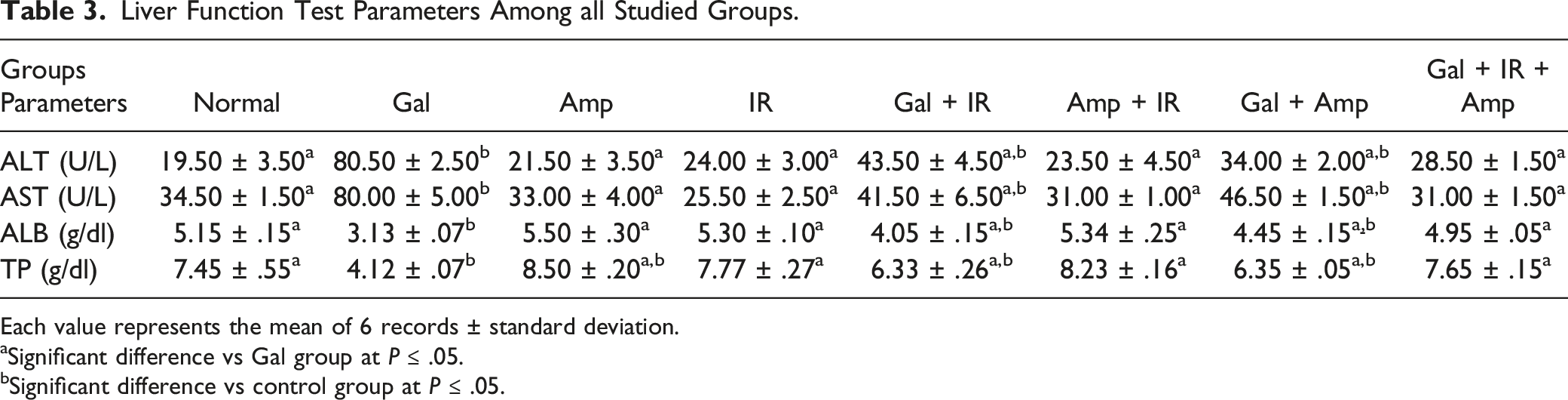
Each value represents the mean of 6 records ± standard deviation.
aSignificant difference vs Gal group at P ≤ .05.
bSignificant difference vs control group at P ≤ .05.
Effect of H. caffeaeformis and/or Low Dose Ionizing Radiation on Inflammatory Markers Among Groups
Levels of (TNF-α), (IL6), and (ICAM-1) were significantly improved in Gal group related to normal group. Meanwhile administration of H. caffeaeformis and/or introduction to low ionizing radiation dose suppress these elevations in (Gal + Amp), (Gal + IR) and (Gal + IR + Amp) groups compared to Gal group (See Figure 2). Protein levels of inflammatory markers (A) TNF-α (B) IL-6 (C) ICAM. Values with different superscripts are significantly different at P < .05.
Gene Expression Level of Inflammation Markers Among Groups
Gene expressions of NFκB and STAT3 using RT-PCR are illustrated in Figure 3. NFκB and STAT3 expression in Gal group displayed a substantial increase in Gal group related to normal group; however, treatment with H. caffeaeformis only or combined with low gamma radiation dose resulted in significant decrease in (Gal + Amp), (Gal + IR), and (Gal + IR + Amp) groups compared with Gal group. Gene expression level of (A) NFκB (B) STAT3 among groups. Values with different superscripts are significantly different at P < .05.
Effect of H. caffeaeformis and/or Low Dose Ionizing Radiation on c-Jun Expression
c-jun protein immunoblot of different treated rats were detected in liver tissue (Figure 4). The hepatic c-jun protein level was expressively increased with Gal toxicity compared to control group, though the dual collaboration between H. caffeaeformis and γ-R pointedly repressed its expression after Gal toxicity. Western blot analysis of proteins JNK relative to β-actin. The bottom panels represented quantification of the immunoblot by densitometry. Values are expressed as mean ± S.E. * P < .05 compared to control.
Effect of H. caffeaeformis and/or Low Ionizing Radiation Dose on Oxidative Stress Markers in Liver Homogenate
Level of Oxidative Stress Markers Among Groups.

Each value represents the mean of 6 records ± standard deviation.
asignificant difference vs Gal group at P ≤ .05.
bSignificant difference vs control group at P ≤ .05.
Histopathological Results
Livers of control group that treated by saline showed normal hepatic lobules (Figure 5A). While livers of albino rats treated by D galactosamine revealed hepatic cords disorganization with hepatic necrosis. Hyperplasia of Kupffer cells with lymphocytes and macrophages infiltration were also observed (Figure 5B). Photomicrograph of hepatic tissue of rats stained with hematoxylin and eosin and examined with a light microscope at ×200 magnification. (A) Normal group section showing normal hepatic lobules which made up of radiating plate’s or strands of polygonal cells arrow, (B) Gal group showing disorganization of hepatic cords and multifocal area of hepatic coagulative necrosis arrow, (C) IR group showing mild ballooning of hepatocyte with clear faint stain cytoplasm arrow (D) Amp group showing normal histological structure of hepatic lobules with organized hepatic cords arrow, (E) Amp + IR group showing mild swelling of hepatocytes and narrowing of hepatic sinusoids, (F) Gal + IR group showing disorganization of hepatic cords, swelling of hepatocytes with granularity of its cytoplasm arrow, (G) Gal + Amp showing ballooning of hepatocytes and narrowing of sinusoids with hyperplasia of Kupffer cells arrow, (H) Gal + Amp + IR showing normal histological structure of hepatic lobule arrow.
Animals subjected to low dose gamma radiation showed mild ballooning of hepatocyte in some of hepatic lobules with narrowing of hepatic sinusoids (Figure 5C). Moreover, rats treated by Halamphora caffeaefomis displayed organized hepatic cords made up of polygonal cells with prominent round nuclei and eosinophilic cytoplasm. Hepatic lobules have no significant pathological alterations (Figure 5D).
Animals exposed to H. caffeaefomis and low dose gamma radiation showed organized hepatic cords of polygonal cells. Mild swelling of hepatocytes with narrowing of hepatic sinusoids were seen (Figure 5E). On the other side, animals exposed to D galactosamine and low dose gamma radiation exhibited disorganized hepatic cords and swelling of hepatocytes with granularity of its cytoplasm. Also, narrowing of hepatic sinusoids and hyperplasia of Kupffer cells were seen (Figure 5F). Hepatic tissue of animals group exposed to D galactosamine and H. caffeaefomis, showed ballooning of hepatocytes and narrowing of sinusoids with hyperplasia of Kupffer cells (Figure 5G). While animals exposed to D galactosamine and H. caffeaefomis and low dose gamma radiation showed near normal histological structure of hepatic lobules made up of cords of polygonal cells with prominent round nuclei and eosinophilic cytoplasm with noticed hyperplasia of Kupffer cells (Figure 5H).
Discussion
Liver regulates many vital roles in our body; thus, it causes grave sickness once diseased or wounded. Liver diseases could be caused from medications, toxins, or alcohol abuse. Because of deficiency of reliable hepatoprotective drugs in current treatment, there is a vital need for the progression of powerful in addition to safe hepatoprotective agents for the hepatitis treatment. 24 To the best of our knowledge, there were no information or experimental researches concerning H. caffeaeformis hepatoprotective effect against hepatotoxicity induced by D-GalN-. It was stated that low-dose radiation decreases inflammation (i.e., hepatitis) in experimental animals and induces antioxidant defenses.25,26
Low IR dose (<1 Gy) improves cell defense thru stimulation of AKT/NF-KB pathway, which controls cell death and cell propagation. It was found that mitogen-activated protein kinase (MAPK) superfamily (i.e., c-(JNK)) is controlled by IR thus affecting cell existence or passing. 27 Lall et al 28 stated that regular human cells dealing with low IR dose enhances metabolic switch from oxidative phosphorylation to aerobic glycolysis accompanied by improved radiation resistance. This switch exhibited up-regulation of enzymes encoding genes of glycolysis and glucose transportation in the direction of pentose phosphate oxidative pathway thus introduction to low IR dose enhances cell fight against oxidation stress.
In the current study, changes linked with Gal—induced hepatic injury are like that of viral hepatitis. 29 Hereafter, D-GalN induced hepatic-toxicity was selected as perfect model of investigation. D-galactosamine changes antioxidant status making certain organs much vulnerable to oxidative stress. Numerous researches have revealed that Gal causes variation in hepatic-enzymes. 30 Liver impairment induced by Gal indicates metabolism troubles in hepatic cell leading to distinctive variations in serum enzyme activities. 31
In the current study, the elevation in ALT and AST activities could be interpreted due to liver cell destruction or membrane absorptivity change both are distinguishing enzymes of liver impairment; so, their serum discharge proves GaI induction of liver injury. On other hand, Gal produced a substantial decline in level of albumin in serum as a hepatotoxicity marker, consistent with Wong et al study. 32 Moreover, serum dropping of albumin level is because of mRNA down expression of albumin. Also, the decrease of total liver proteins evidently is due to liver injury 33 which confirms our results. However, our research team showed that Halamphora and/or IR exposure efficiently amended the integrity of parenchymal hepatocyte structural and ameliorates these biochemical parameters of liver injury, verifying the ameliorative effect of Halamphora/and IR against liver inflammation. However, a limitation of our study is the small sample size; therefore, the results of this experimental study should be viewed as indicative and should be validated in a larger cohort.
Inflammation is a closely related biological event involved in hepatitis pathogenesis. 34 Preceding studies have confirmed that hepatitis made by Gal was associated by the discharge of many proinflammatory cytokines, counting TNF-α, and IL-6, from liver cells. The current study stated that Halamphora caffeaefomis efficiently decreased the discharge of serum TNF-α, and IL-6 in hepatitis rat model.
ICAM-1 is a cell surface glycoprotein and an adhesion receptor that is best known for regulating leukocyte recruitment from circulation to sites of inflammation. However, in addition to vascular endothelial cells, ICAM-1 expression is also robustly induced on epithelial and immune cells in response to inflammatory stimulation. 35 Increased ICAM-1 likely reflects the persistence of inflammation 36 which agree with our histopathological results. ICAM-1 is an important early marker of response to inflammatory mediators and immune activation released from a variety of cells including hepatocytes. 37
ICAM-1, that activated strongly in several cell types by proinflammatory cytokines, has a critical role in immune cell enrolment. ICAM-1 offers adhesion sites between lymphocytes and neutrophils with activating release of inflammatory mediators by vascular endothelial cells. 38 Elevated levels of ICAM-1 in Gal group agreed with a previous study indicated that TNF-α is a powerful activator of expression of ICAM-1. 39 Elevated levels of TNF-α and NF-κB agreed with a previous study reported increased TNFα levels in liver tissue as a direct result of increased NFκB. After TNF-α binds to type 1 TNF-α receptors, NF-κB is phosphorylated and activated, thus generating a vicious cycle, and increasing the degree of injury. 40
The NF-.B/IL-6/STAT3 pathway shows an essential role in hepatic defense and renewal in a diversity model of liver injury, counting hepatotoxins induced noxious injury with ischemic liver injury, and Fas-mediated injury. STAT3 displays a vital part in stopping cell death by the activation of antioxidant (i.e., Ref-1) and antiapoptotic proteins. It was reported that STAT3 binds NF-κB and increases induction of NF-κB -dependent gene. NF-κB is vital for the initiation of mRNA expression of IL-6, also ROS activates NF-κB as a result of TNF-α in immune cells. 41 In our study, the NF-κB and STAT3 expression showed significant increase in Gal group.
Jun N-Terminal Kinase pathway is the main path of cell apoptosis and inflammation induction. 42 It was established that JNK signals are inspired by numerous exterior aspects like stress (high pressure infiltration, UV radiation, ischemia/reperfusion damage), cytokines (TNF-α, IL-1), and growth factors. p38 and JNK are stimulated by liver injury, and the regulatory effect is the result of their cooperation. In vitro researches in numerous types of cells as hepatic cell have shown that long-term stimulation of JNK signaling pathway is the mechanism of TNF-induced cell death. 43 Our results confirmed that treatment with the Halamphora and/or IR amends liver injury induced by D-GalN in rat.
Moreover, TNF-α is controlled in immune cells by NF-κB throughout the inflammatory reply. 44 Agreeing with Gao, 45 the raise of IL-6 level could be the protection policy to restore the damaged liver and control inflammation. IL-6 stimulates STAT3 then induce variation of hepatoprotective genes in hepatocytes. 45
The current results propose that useful effect of Halamphora and/or IR against Gal induced liver toxicity may somewhat be accredited to anti-inflammatory effects of Halamphora and/or IR that protect the hepatic cell through proinflammatory cascade mediated by toxins. Treatment with algae extract or/and IR significantly reduced NF-κB, TNF-α, ICAM, and IL6 levels in Gal induced rats compared to normal group suggesting their anti-inflammatory therapeutic effect. One of the main compounds found in algae extract is hespertin (Table 1). Hesperetin was found to inhibit NFκB by down regulating STAT3 signaling axis. 46
Oxidative stress is considered as one of the pathological mechanisms leading to the development and progression of many liver diseases, such as chronic viral hepatitis. 47 The present study showed that MDA levels, an indicator of lipid peroxidation, were significantly increased in liver homogenates accompanied with depleted activity of paraxonase-1 enzyme and imbalance in the ratio between GSSG/GSH of D-GalN treated rats. These results were supported by the finding of previous study, 48 that stated the vital role of GSH in detoxification and cell protection and that loss in GSH level indicates oxidant protection mechanism.
Alternatively, the intake of algal extract or/and IR depressed lipid peroxidation and improved antioxidants levels significantly when related to group treated with D-GalN. This was matching with improvement in liver function test telling that H coffaeiformis extract or/and low radiation dose have oxidants defense activities and tissues protection from lipid peroxidation in D-GalN induced hepatotoxicity group. This agreed with Boukhris et al 49 who specified that effective antioxidant efficiency of Halamphora sp. extract is displayed owing to its flavonoids, phenols and active polyphenolic ingredients high content.
H. coffeaeformis alga displayed hopeful scavenging properties of DNA lesions induced by hydrogen peroxide. The effectiveness of coffeaeformis alga to prevent the oxidative stress is attributed to many vital phyto-constituents existence counting phenolics, carotenoids, and chlorophylls additionally to existence of pyrogallol, flavonoids, and catechin. 9
Likewise, the highlighted antioxidant activity of H. coffeaeformis depended on occurrence of carotenoids, β-carotene and fucoxanthin that have numerous efficient groups as epoxy, hydroxyl, carbonyl, and carboxyl moieties which are in charge of the antioxidant effect. 49
Conclusion
In conclusion, therapeutic herbs that have anti-inflammatory and antioxidant activity have turn out to be respectable candidates for study to improve tissue injury. Moreover, our outcomes offer powerful molecular suggestion in support of the hepto-ameliorative effect of Halamphora and/or IR against D-GalN induced hepatotoxicity through modulating signaling molecular targets associated with antioxidant, anti-inflammatory, and anti-proliferative activities. Our conclusions were well valued with histopathological results.
Footnotes
Acknowledgments
The authors would like to thank Dr Ahmed Othman (Professor of Histopathology, Faculty of Medicine, Cairo University) for carrying out blinded histopathological examination in this work.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by all authors. The first draft of the manuscript was written by Elsaman and all authors commented on subsequent versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All Animal procedures were performed according to the international ethical guidelines laid down in Helsinki declaration and are in accordance with the recommendations of the Ethical Committee of the National Research Center for Radiation Research and Technology (NCRRT), Atomic Energy Authority, Cairo, Egypt, conformed to “Guide for the care and use of Laboratory Animals” for the use and welfare of experimental animals, published by the US National Institutes of Health (NIH publication No. 85-23, 1996).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
