Abstract
Background
The vascular endothelium regulates vascular inflammatory responses essential for controlling cardiovascular disease-induced complications. Moreover, regulating intercellular adhesion molecule-1 (ICAM-1) expression in response to pro-inflammatory stimuli is critical for controlling cardiovascular inflammation. Although bakuchiol is an established natural anti-inflammatory agent, its effects on intravascular inflammation remain elusive.
Objectives
This study aimed to assess bakuchiol’s inhibitory effects on vascular inflammatory responses by inducing adhesion molecule expression using tumor necrosis factor-α (TNF-α).
Materials and Methods
Vascular inflammation was stimulated with TNF-α by culturing human primary endothelial cells (ECs). Nuclear factor kappa B (NF-κB) signaling and adhesion molecule expression due to TNF-α stimulation were confirmed by Western blotting.
Results
ICAM-1 and vascular cell adhesion molecule-1 (VCAM-1) expression in human umbilical vein endothelial cells (HUVECs) were significantly increased by TNF-α but decreased by bakuchiol. Bakuchiol inhibited monocyte adhesion by suppressing TNF-α-induced adhesion molecule expression and reduced NF-κB p65 subunit acetylation and phosphorylation.
Conclusion
These results demonstrate the potential of bakuchiol in preventing TNF-α-induced vascular inflammation and cardiovascular diseases. Bakuchiol may be used to prevent and treat vascular diseases, including hypertension, systemic inflammation, and oxidative stress.
Keywords
Introduction
Inflammation occurs as a physiological process in response to infection or injury. Vascular inflammation, mediated by several factors, including cytokines, immune cells, blood, smooth muscle cells, perivascular adipocytes, and fibroblasts, triggers a cascade of events. This response involves redness and heat-induced vasodilation, which facilitate increased blood flow and direct endothelial cell (EC) damage and vascular permeability through mediators (Peng et al., 2019). In addition, increased extravascular osmotic pressure, vasodilation, and fluid exudation precipitate vascular congestion, wherein blood flow decelerates. Mechanisms of EC injury encompass direct injury induced by cytokines, such as tumor necrosis factor-α (TNF-α), as well as mechanisms that mediate neutrophil adhesion and infiltration through the expression of intercellular adhesion molecule-1 (ICAM-1) on the cell surface (Lentsch & Ward, 2000). The endothelium plays an essential role in regulating vascular permeability, which is essential for controlling or preventing the infiltration of specific molecules and cells into organs such as the brain. TNF-α induces the expression of adhesion molecules, such as vascular cell adhesion molecule-1 (VCAM-1) and ICAM-1, on the endothelium. These molecules facilitate leukocyte adhesion to the endothelium and their subsequent migration to peripheral tissues (Fingar et al., 1997).
The mechanism underlying the involvement of nuclear factor kappa B subunit p65 (NF-κB p65) in vascular inflammation is well-established. P300-mediated acetylation of NF-κB p65 is recognized as a key process that upregulates the expression of vascular adhesion molecules and various chemokines in vascular inflammation. Moreover, this mechanism is implicated in vascular senescence through the mediation of oxidative stress (Di Pietrantonio et al., 2023).
The extracted natural substances exhibit anti-cancer, anti-inflammatory, anti-viral, and antioxidant properties. It has been scientifically established that it has medicinal properties. Among them, catechin [epigallocatechin gallate [EGCG)] contained in green tea (Townsend et al., 2004), contains cordycepin and allicin (Chang et al., 2008), which are representative components of garlic (Morihara et al., 2006), and useful components of plants, include lycopene of tomatoes (Yue et al., 2012), resveratrol of grapes (Shen et al., 2012), and various types of polyphenols and flavonoids. These substances are representative of natural compounds that research findings indicate are effective in the suppression and treatment of vascular diseases.
As a natural substance, bakuchiol is a natural meroterpene primarily found in Psoralea corylifolia, Psoralea glandulosa, and Ulmus davidiana (Choi et al., 2010; Madrid Villegas et al., 2015; Wang et al., 2020), with established anti-cancer, anti-inflammatory, and anti-aging effects (Dhaliwal et al., 2019; Kumar et al., 2021; Miao et al., 2018). Bakuchiol has also been demonstrated to inhibit lipopolysaccharide-stimulated neuroinflammation in activated microglia through the p38 mitogen-activated protein kinase and extracellular signal-regulated kinase signaling pathways (Lim et al., 2019). Furthermore, at a concentration of 50 µM, bakuchiol exerted notable effects, such as neutrophil degranulation inhibition, cell migration reduction, myeloperoxidase activity suppression, and nitric acid synthase expression inhibition. As a natural anti-inflammatory agent, bakuchiol exhibits the ability to control leukocytic functions (Ferrándiz et al., 1996). However, its effects on intravascular inflammation remain unexplored. Therefore, this study was conducted to investigate the effects and signaling mechanisms of bakuchiol on mediators expressed during vascular inflammation in human umbilical vein endothelial cells (HUVECs). The results of this study are poised to provide valuable insights to support future endeavors in preventing the development of cardiovascular diseases.
Materials and Methods
Materials and Cell Culture
Bakuchiol was isolated from U. davidiana according to a previously reported method. To summarize, U. davidiana var. japonica specimens were collected in Gyeongnam, South Korea, and the dried plant material was extracted with 80% ethanol and concentrated under a vacuum. After dividing this extract into fractions, 5 g of the butanol fraction residue was separated into four fractions via silica column chromatography. Subfraction IV was further purified using silica gel to obtain a compound (Choi et al., 2010). HUVECs were sourced from PromoCell (Heidelberg, Germany,
Western Blotting
Western blotting was conducted using a previously described method (Ahn et al., 2004). Briefly, protein samples extracted from the cells were mixed with 5× sample buffer; each cell lysate was boiled in sample buffer at 95°C for 7 min and electrophoretically separated via sodium dodecyl sulfate-polyacrylamide gel electrophoresis using mini-PROTEAN TGX-Free Precast gels (Bio-Rad Laboratories, Hercules, CA, USA). Subsequently, the lysates were transferred onto polyvinylidene fluoride membranes and incubated with primary and secondary antibodies. After rinsing the membranes thrice with Tris-buffered saline with Tween (20 mM Tris, pH 7.5, 150 mM NaCl, 0.5% Tween 20) for 10 min, protein bands were visualized by chemiluminescent detection using GE Healthcare ECL Start Western Blotting Detection Reagent (Cytiva, Marlborough, MA, USA) and ChemiDoc XRS Imaging System (Bio-Rad Laboratories). NF-κB p65 and β-actin were used for the comparison of protein expression.
Monocyte Adhesion Assay
For the adhesion assay, THP-1 cells (American Type Culture Collection, Manassas, VA, USA) were maintained as monocytes by culturing in the RPMI 1640 medium. THP-1 cells were labeled with a fluorescent tracer, calcein acetoxymethyl (calcein AM, 2 µg/mL; Invitrogen, Carlsbad, CA, USA), for 15 min at 37°C to serve as a cell tracker (Ahn et al., 2004). Calcein AM undergoes rapid hydrolysis inside the cells to form calcein, which is fluorescent and easily detected using a fluorescence plate reader (Ex/Em, 494/517 nm). This process increases the intracellular concentration of calcein AM, thereby increasing fluorescence. In this study, calcein AM was used as a marker for monocyte fluorescence. HUVECs were pre-treated with bakuchiol (50 µM) for 30 min, stimulated with TNF-α (10 ng/mL), and finally treated with bakuchiol (50 µM) for 4 h. Subsequently, a total of 1 million fluorescently labeled THP-1 cells were spread onto HUVEC monolayers and allowed to adhere for 2 h. The fluorescence emitted by the adherent THP-1 cells was measured using a fluorescence microscope (Olympus IX71; Olympus, Japan) and quantified using ImageJ software (version 1.53, NIH, USA). Adhesion was validated by counting all monocytes attached to a single HUVEC based on individual cells and was quantified by measuring the fluorescence intensity.
Statistical Analysis
Data are expressed as mean ± standard deviation (SD). A one-way analysis of variance was used to compare the means between the two groups. Differences between means were analyzed using Tukey’s post hoc test, with statistical significance set at p < .05.
Results
Cell Viability of Bakuchiol-treated HUVECs
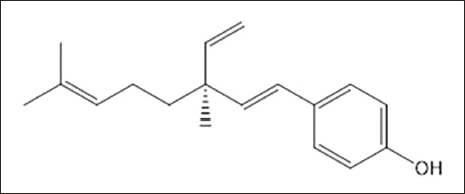
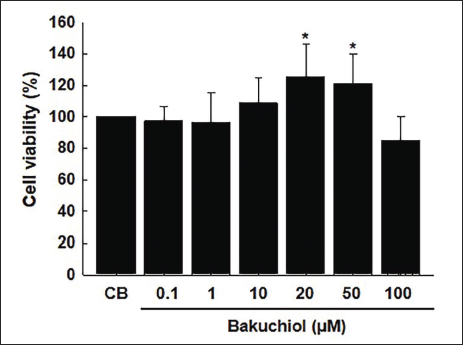
The chemical structure of bakuchiol is depicted in Figure 1. Cell viability was analyzed by measuring the physiological activity of cells following bakuchiol treatment using the CCK-8 assay. The results indicated no cytotoxicity following bakuchiol exposure, with a tendency toward increased cell survival rates depending on the concentration of bakuchiol (1–50 µM). Although slight toxicity was observed at 100 µM, it was not statistically significant (Figure 2).
Chemical Structure of Bakuchiol.
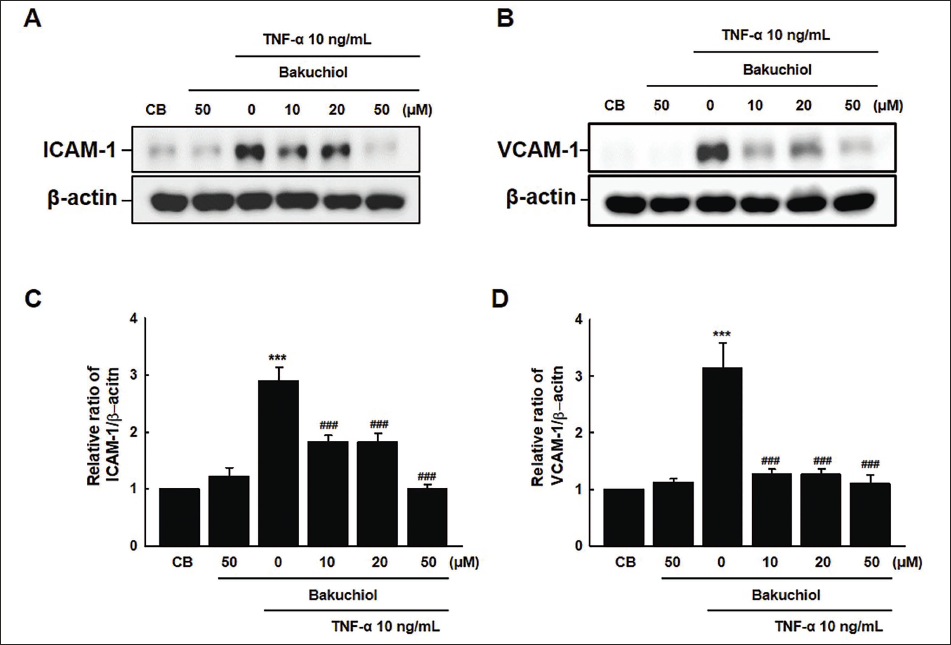
Bakuchiol Attenuated TNF-α-induced Expression of Adhesion Molecule Proteins in HUVECs
A study was conducted using HUVECs to investigate the effects of bakuchiol on the expression of adhesion molecules, such as ICAM-1 and VCAM-1. In this study, HUVECs were pre-treated with bakuchiol at various concentrations (10, 20, and 50 µM) for 30 min, followed by stimulation with TNF-α for 6 h to induce inflammation. The results showed that bakuchiol at a concentration of 50 µM significantly reduced the expression of ICAM-1 compared to that at lower concentrations of 10 and 20 µM. Additionally, bakuchiol treatment resulted in a substantial decrease in VCAM-1 expression, which was evident even at the lowest concentration of 10 µM. This reduction continued to be significant at the 50 µM concentration (Figure 3A and 3B). These findings suggest that bakuchiol effectively attenuates TNF-α-induced expression of adhesion molecules, potentially contributing to its anti-inflammatory properties.
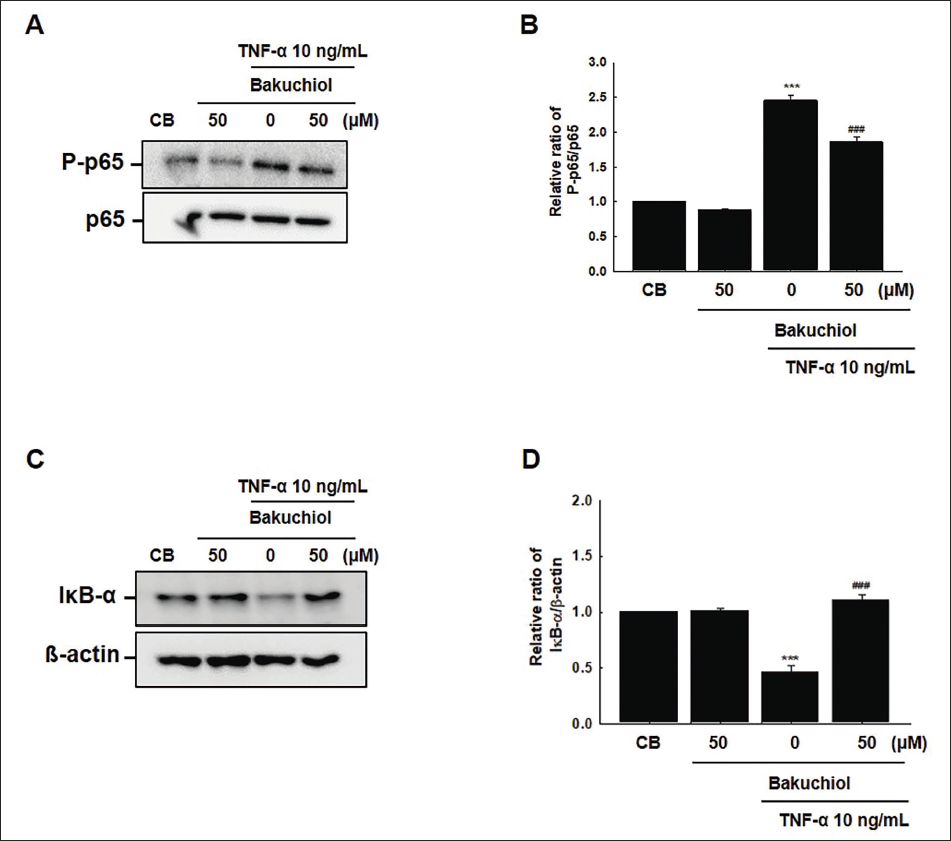
Bakuchiol Inhibited TNF-α-induced NF-κB p65 Acetylation and Phosphorylation
To assess the potential effect of bakuchiol on NF-κB signaling, we evaluated the changes in the acetylation and phosphorylation states of the NF-κB p65 subunit. Treatment with TNF-α resulted in approximately 2.2-fold and 2.4-fold increases in the acetylation and phosphorylation of the p65 subunit of NF-κB, respectively, compared to the control group (CB; vehicle treatment alone). However, this TNF-α-induced effect was significantly attenuated by pre-treatment with bakuchiol at a concentration of 50 µM (Figure 4A). Quantitative analysis of protein levels revealed the relative expression ratios, as illustrated in Figure 4B and 4D. Additionally, the degradation of the IκB-α, a process stimulated by inflammatory cytokines (Mathes et al., 2008), was notably observed under TNF-α stimulation; this degradation was effectively reversed by bakuchiol treatment at 50 µM (Figure 4C and 4D). Although bakuchiol at 10 µM showed a reduction in ICAM-1 and VCAM-1 expression, this reduction, to some extent, was limited due to the degradation of IκB-α (data not shown). However, at a 50 µM concentration, bakuchiol clearly reversed IκB-α degradation and simultaneously inhibited the acetylation and phosphorylation of p65. These findings suggest that bakuchiol suppresses TNF-α-induced expression of ICAM-1 and VCAM-1 by inhibiting the acetylation and phosphorylation of NF-κB p65, as well as preventing the degradation of IκB-α. This indicates a multifaceted inhibitory mechanism of bakuchiol on NF-κB signaling, contributing to its anti-inflammatory properties.
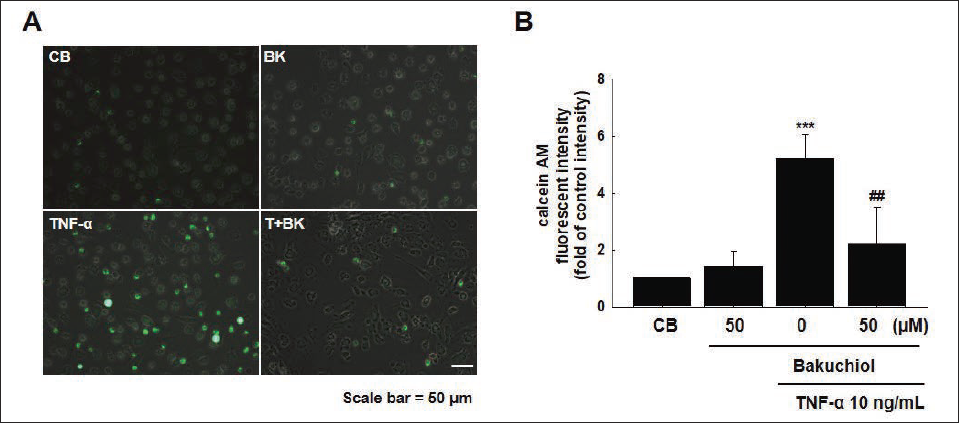
Bakuchiol Inhibited Monocyte Adhesion to HUVEC Monolayers
Fluorescent labeling was conducted to identify the recruitment of monocytes through the expression and interactions of cell surface adhesion molecules. HUVECs stimulated with TNF-α (10 ng/mL) exhibited an approximately 5.1-fold increase in monocyte adhesion compared to the control group. However, treatment with bakuchiol (50 µM) resulted in a significant 61% decrease in monocyte adhesion (Figure 5A and 5B). This suggests that bakuchiol effectively reduces the adhesion of monocytes to ECs, potentially through the inhibition of adhesion molecule expression induced by TNF-α.
Discussion
Elevated plasma levels of several inflammatory mediators correlate with cardiovascular risk and various clinical outcomes. These markers include cell-adhesion molecules, cytokines, and pro-atherogenic enzymes (Blake & Ridker, 2002). Despite recent advances in research on associated factors, preventive measures against vascular disease development remain insufficient. Bakuchiol is a natural meroterpene mainly derived from P. corylifolia and U. davidiana. In the present study, bakuchiol inhibited intravascular inflammation by suppressing TNF-α-induced ICAM-1 and VCAM-1 expression in HUVECs. These findings suggest that bakuchiol attenuates pro-inflammatory cytokine production or activity and impedes immune cell migration into tissues. Future endeavors aiming to determine whether bakuchiol influences the intracellular signaling mechanism through cytokines should incorporate an initial assessment of the mRNA levels of ICAM-1, VCAM-1, and E-selectin. Established anti-inflammatory agents encompass flavonoid natural compounds, including quercetin, hesperidin, and luteolin, which are recognized for their ability to inhibit mediators associated with inflammatory cytokines and for their vascular protective properties. Bakuchiol, which exhibited a protective effect on vascular ECs in this study, has been validated as an effective natural anti-inflammatory agent.
The evolutionarily ancient transcription factor NF-κB holds important regulatory functions in immune and stress responses, cell apoptosis, growth, survival, and differentiation. Among the NF-κB subunits, p65 and p50 have been identified to be reversibly acetylated by p300, with a lysine residue in p65 serving as the receptor site for p300 acetylation (Ishinaga et al., 2007). Our results revealed that bakuchiol regulated NF-κB p65 acetylation and phosphorylation, as well as IκB-α degradation, indicating that bakuchiol directly affects the NF-κB signaling pathway involved in the overall immune system.
Leukocyte migration from the blood to tissues is a crucial aspect of the inflammatory response, orchestrated through several steps mediated by adhesion molecules and chemoattractant. Inflammation is the initial step in the development of several diseases, with the pioneering mechanism controlling leukocyte migration representing a therapeutic target (Rossi et al., 2011). Bakuchiol inhibited leukocyte adhesion to adjacent ECs by reducing ICAM-1 and VCAM-1 expression during inflammation. These results indicate that bakuchiol impedes the involvement of neutrophils, which play a predominant role in the early stages of inflammation and initiate tissue damage repair through macrophage activity (Butterfield et al., 2006). Bakuchiol has been demonstrated to exert its effects by altering the cell surface properties of Candida, which resulted in decreased adhesion and subsequent reduction in biofilm formation and virulence (Nordin et al., 2015). Our results indicate that bakuchiol inhibited NF-κB p65 signaling and reduced the expression of adhesion molecules during the inflammatory response of TNF-α-stimulated ECs. Notably, this effect did not stem from direct interaction with receptors on the cell membrane. Ultimately, this outcome demonstrates the anti-vascular inflammatory effect of bakuchiol by reducing the release of cytokines from immune cells with increased infiltration into the inflammatory site, thereby preventing monocyte adhesion. Future research examining the effect of bakuchiol in regulating ECs and infiltrated immune cells sequentially would be instrumental in analyzing the effect of CD34, a molecule expressed in EC precursors and known for its involvement in the migration and differentiation of blood cells.
Bakuchiol has been reported to demonstrate anti-cancer activity in preclinical models, attributed to its structural similarity to resveratrol (Chen et al., 2010). Additionally, a rat study suggested that bakuchiol and extracts of the herbal plant P. corylifolia may prevent bone loss (Wang et al., 2015). Despite lacking structural similarity to retinol, bakuchiol has exhibited similar functionality through retinol-like regulation of gene expression (Chaudhuri & Bojanowski, 2014). In a clinical study, bakuchiol exhibited efficacy comparable to that of retinol in improving photoaging (wrinkles, hyperpigmentation) and was notably better tolerated by the skin (Dhaliwal et al., 2019). Bakuchiol is primarily used in skin care cosmetics, but it is not commonly found in foods. This is because bakuchiol was developed as a cosmetic ingredient and is not used as a food additive or does not exist naturally in many foods. However, in this study, the direct vascular inflammation-protective effect of bakuchiol was demonstrated by blocking monocyte adhesion to vascular ECs. These intracellular findings, as well as known animal and clinical findings, provide ample support for the intravascular anti-inflammatory effects of bakuchiol, and we anticipate observing in vivo effects in animal models with vascular systemic inflammation in future research.
Conclusion
In summary, bakuchiol inhibits the early-stage vascular inflammatory process by inhibiting the expression of adhesion molecules and leukocyte adhesion. These results demonstrate bakuchiol’s ability to prevent vascular inflammation and its protective potential against various inflammatory diseases, comparable to known natural anti-inflammatory agents that inhibit vascular inflammation. Bakuchiol, used independently or in combination with other compounds, may elicit synergistic effects and may play a pivotal role in the early management of vascular diseases such as hypertension, systemic inflammation, and oxidative stress.
Footnotes
Abbreviations
ICAM-1: Intercellular adhesion molecule-1; NF-κB: Nuclear factor kappa B; VCAM-1: Vascular cell adhesion protein-1.
Authors Contribution
Experimental research: ASL; Data analysis: ASL, JYH; Manuscript writing: ASL, SYC; Study design and supervision: SYC. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Korea Food Research Institute [Grant number E0210300] and the National Research Foundation of Korea (NRF), funded by the Korean government (MIST) [Grant numbers GA224600 and 2022M3H9A1084670].
