Abstract
The synthesis of new water-soluble N-alkylated derivatives of 1,3,5-triaza-7-phosphaadamantane is presented. Ru(PPh3)2Cl2 has been used to react with 1-(4-nitrobenzyl)-3,5-triaza-1-azonia-7-phosphaadamantane bromide (PTAR). By using elemental analysis, NMR, and IR spectroscopy, the obtained compounds were identified. The UV-visible absorption spectroscopy has been used to monitor the complexation of various transition metal cations. Studies on conductivity have been utilized to validate the complexes’ stoichiometries. Using the disc diffusion method, five bacteria strains were used for the study of the antimicrobial activity of compounds
Keywords
Introduction
Phosphine ligands with sulfonated aryl groups account for the majority of ligands used for water solubilization of organometallic derivatives, but there are other ligands on phosphines that confer water solubility. There is an increasing investigation involving other types of substituents on phosphines which impart water solubility.
1
These include, for example, cationic ammonium groups (Ph2PCH2CH2NMe3+ and Cy2PCH2CH2NMe3+)2-5 and carboxylated aromatic groups (
In addition, the antimicrobial and antioxidant activities of the obtained compounds were studied.
Results and Discussions
Synthesis of N-alkylated PTA Salt
After adding 4-nitrobenzyl chloride to a suspension of PTA in THF, the quaternary N-alkylated PTA compound Synthetic route to compound 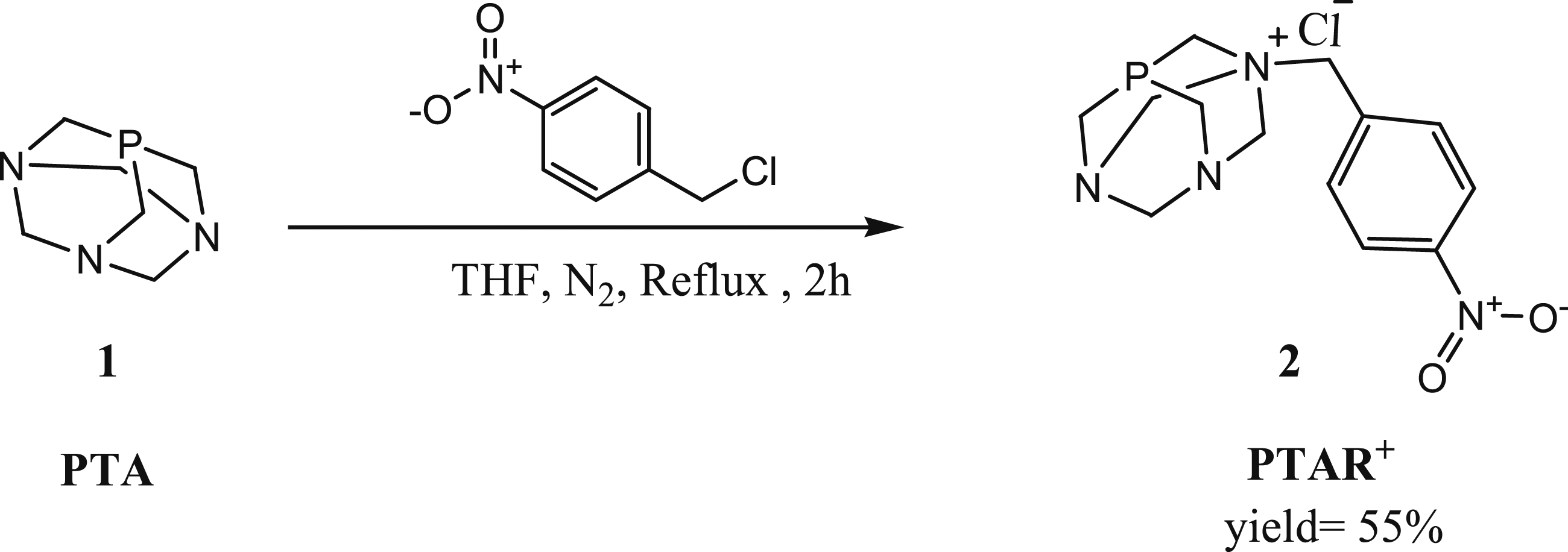
A bialkylated PTA salt was not observed, even after refluxing in MeOH with 2.5 equivalents of 4-nitrobenzylchloride
The 1H NMR spectrum of the water-soluble phosphine
Signals from methylene units between the quaternary and tertiary nitrogens are at δ = 4.90–5.40 ppm. The methylene moiety adjacent to the quarternary nitrogen and the ester group was detected.
The signals of CH2-N of PTA were also found in the same range. The solvent effects seem to play a major role in the chemical shifts in the proton NMR. The proton-phosphorus coupling constants varied from JP,H = 10.5 Hz (NCH2P; 2) to 6.0 Hz (N+CH2P; 2) and 6.5 Hz (N+CH2P; 2). The carbon–phosphorus coupling constants to the two carbons adjacent to the tertiary nitrogen were also found to be similar for 8, 9, and 10 (JP,C = 21.2 Hz). Also the coupling constants to the quarternary nitrogen were in the same range (JP,C = 34.5–35 Hz).
Scheme 2 depicts the reaction of 1-(4-nitrobenzyl)-1,3,5-triaza-7-phosphaadamantan-1-ium (PTAR) derivatives with Ru(PPh3)3Cl precursors to form complex Synthetic route to compound 
Similarly, reaction of 1-(4-nitrobenzyl)-1,3,5-triaza-7-phosphaadamantan-1-ium (PTAR) with CpRu(PPh3)3Cl in a mixture of hot methanol and toluene (7:3) under nitrogen for 2 h results in the formation of rapidly decomposing products.
The complex
The 1H NMR spectra of the water-soluble phosphanes
Consequently, the methylene protons are inequivalent and, in some cases, diastereotopic. The methylene NCH2N protons in all these phosphanes are diastereotopic; although, the methylene protons of the NCH2N + group with one alkylated N atom appear as an AB system. The 31P{1H} NMR spectra display a singlet in the range between δ = −81.7 ppm, downfield from that for free PTA.
Complexation of PTAR+ and Complex 3 in Methanol
In methanol at 25°C, complexation investigations of PTAR+ and complex Complexation of Co2+ by ligand Complexation of Sn2+ by ligand 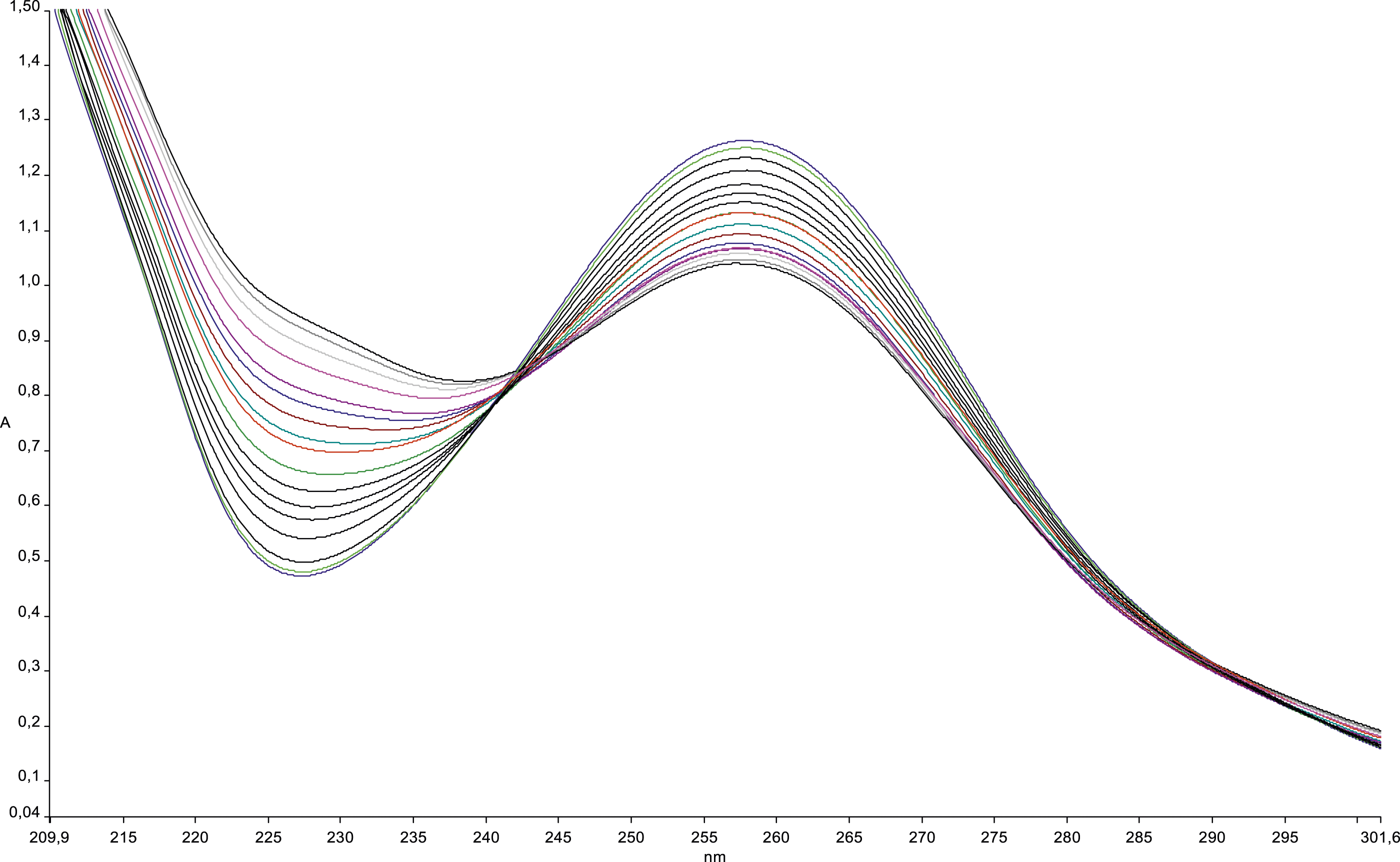

Stability Constants (logβ
xy
± σN-1) of Complexes

Stability Constants (logβxy ± σn−1) of Complexes of Lanthanides, in Methanol at 25°C, I = 10−2 M.

The complex formed by ligand
In the case of the Ru-complex-as-a-ligand Stability profile of complexes M2L of ligand 
In the case of lanthanidesLa3+, Ce3+, and Sm3+, complexes ML are formed with ligand 2 and M2L with ligand
Stoichiometry of some metals cation complexes with ligands Conductometric titration in the case of ligand Conductometric titration in the case of ligand 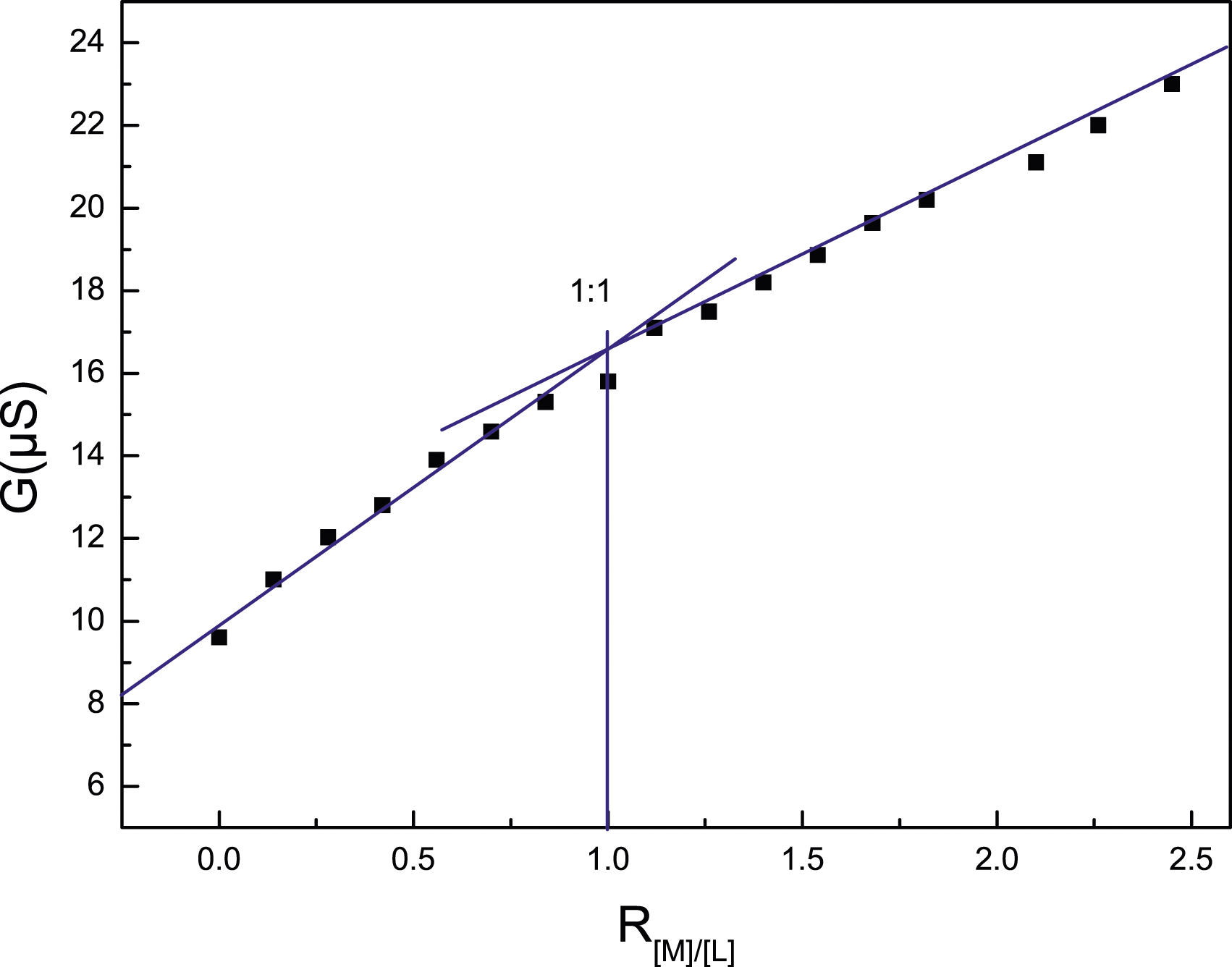

Biological Activities
Antibacterial Inhibition Zones of Compounds

aZone of bacterial inhibition measured in mm.
According to Table 3, compound
Minimum Inhibitory Concentration (MIC) of Synthesized Compounds Against L. monocytogenes ATCC 1911, S. aureus ATCC 6538, and S. typhimurium ATCC. 14028.

Antioxidant Activity
The Antioxidative Activity of Compounds

Conclusions
A new water-soluble N-alkylated PTA derivative
Experimental Section
General Procedures
All manipulations were performed under an atmosphere of dry nitrogen using vacuum-line and standard Schlenk techniques. All reagents were obtained from commercial suppliers and used without further purification. Solvents were dried by standard methods and distilled under nitrogen before use. IR spectra were recorded on a Perkin-Elmer 398 Spectrophotometer. NMR spectra were recorded on a Bruker AC-400 instrument at 400.1 MHz (1H), 161.9 (31P) or 100.6 MHz (13C) using SiMe4 or 85% H3PO4 as standards. DEPT experiments were carried out for all the compounds. Coupling constants J are given in hertz.
Materials and Methods
UV spectra were recorded on a Perkin-Elmer Lambda 11 spectrophotometer. Conductance measurements were recorded on a MeterLab CDM210 conductimeter with cell constant k = .93 cm. Methanol (Riedel-deHaën for HPLC) was commercial and used without further purification. The supporting electrolyte used in the stability constant determinations was Et4NCl4 (Acros Organics). The metal salts chosen were chlorides (Fluka, purum).
Conductimetric of 1-(4-nitrobenzyl)-1,3,5-triaza-7-phosphaadamantan-1-ium (PTAR) and Complex 3 Studies
While complexation by a neutral ligand is not expected to dramatically alter the molar conductivity of a cationic species, the differences can usually be detected and so the measurement of conductance of a solution of a ligand into which a metal ion is added can be a useful rapid means of establishing the stoichiometry of a complex ion species. Thus, this procedure was followed to obtain preliminary estimates of the metal:ligand ratio in the complexes formed by the two ligands
Stability Constant Measurements
Stability constant values were determined by fitting to the changes in absorbance resulting when solutions of the ligands initially at concentrations between 2 × 10−5 and 4 × 10−5mol L−1 were titrated with solutions of the various metal ions up to a final M:L ratio of 20. All measurements were made at 25°C using methanol as solvent, with the ionic strength maintained at .01 mol L−1 using Et4NCl4. Significant changes in absorbance were found in the wavelength range 215–320 nm in most cases and were suitable for analysis using the program “Letagrop.” 52
In principle, multi-step equilibria could have been involved in these reactions, but in all cases where the effects of complexation could be detected, a simple 1:1 model provided a satisfactory fit.
Synthesis of 1-(4-nitrobenzyl)-1,3,5-triaza-7-phosphaadamantan-1-ium (PTAR)
To a solution of PTA (1,3,5-triaza-7-phosphaadamantane (.634 mmol) in THF (20 mL) was added a solution of N-nitrobenzyl chloride (1.585 mmol). Refluxed for 2 hours at rt. The solid was isolated by filtration, washed with THF (3 × 5 mL) and diethyl ether, and dried under reduced pressure. Soluble in polar solvents, slightly soluble in MeOH and EtOH, and insoluble in other medium and low polar solvents. Yield (%) = 80; IR(cm−1): 2935 (CH2), 1063 (C-N), 1510 (C = Carom), 1207 (C-P), 844 (NO2), 1H NMR in D2O (δ): 8.3 (d, 2H, HPh), 7.78 (d, 2H, HPh), 5,14 (q, 4H, NCH2N+), 4,69 (s, 2H, PhCH2N+), 4,3 (s, 2H, PCH2N+), 3,99 (m, 2H, NCH2N), 3.86 (t, 4H, NCH2P) ppm.13 C{1H} NMR in D2O (δ): 149 (s, CNO2), 134 (s, Ar), 132 (s, CPh), 124 (s, Ar), 80 (s, NCH2N+), 70 (s, NCH2N), 65 (d, PhCH2N+), 52 (d, PCH2N+), 45 (s, NCH2P) 31P{1H} NMR in D2O (δ): −81 (s, P)ppm. Anal. Calc. for C13H18PN4O2Cl: C, 47.50%; H, 5.52%; N, 17.04%, Found: C, 47.6; H, 5.6; N, 17.1%.
Synthesis of [ RuCl2(PTAR)2 PPh3]
Solid PTAR (20 mmol) was added to a stirred solution of [RuCl2 (PPh 3) 3 ] (4 mmol) in a mixture of toluene and MeOH. The resulting mixture was refluxed under nitrogen for 30 min. The orange solid obtained was filtered off, washed with toluene (3 × 5 mL) and dried under reduced pressure. Yield: 80%. 31P{1H} NMR in D2O (δ): −31.66 (d, PTAR), −27.98 (d, PTAR), 3.55(t, PPh3), 10.64 (t, PPh3) ppm
Biological activities
Microorganisms, Media, and Growth Conditions
Staphylococcus aureus (S. aureus) ATCC6538, Listeria monocytogenes (L monocytogenes) ATCC 1911, M lutus LB 141107, Salmonella typhimurium (S. typhimurium) ATCC 14028, and B cereus (ATCC 14579) were the bacteria employed as indicators for the antimicrobial determination. All these indicator microorganisms were obtained from International Culture Collections (ATCC). The indicator microorganisms were grown overnight in Luria Bertani (LB) medium (g L−1: peptone 10, yeast extract 5, and sodium chloride (NaCl) 5, pH 7.2) under aerobic condition and constant agitation (200 r/min) at 30°C.
Agar Well Diffusion Method
Agar well diffusion method was employed for the determination of the antimicrobial activity of the synthesized compounds according to Guven et al.65-69
Minimum Inhibitory Concentration
(MIC) of the synthesized compounds and the standards ampicillin (stock solutions at 20 mg mL−1) against the five tested bacteria were determined according to Sellem et al.70-73 The test was performed in sterile 96-well microplates with a final volume in each microplate well of 100 μL. Stock solutions of synthesized compounds and standards were serially diluted with dimethyl sulfoxide (DMSO). To each test well, cell suspension was added to final inoculum concentration of 106 a colony-forming unit (CFU) mL−1 of indicator microorganism. The plates were then incubated at appropriate growth conditions of the corresponding indicator microorganism. The MIC was defined as the lowest concentration of the synthesized compounds and standards at which the microorganism does not demonstrate visible growth after incubation. 25 μl of Thiazolyl Blue Tetrazolium Bromide (MTT) at .5 mg mL−1 were added to the wells and incubated at room temperature for 30 min. The colorless tetrazolium salt acts as an electron acceptor and was reduced to a red-colored formazan product by the indicator microorganisms. When microbial growth was inhibited, the solution in the well remained clear after incubation with MTT.
For the antimicrobial activity determination (inhibition zones and CMIs), each experiment was carried out simultaneously three times under same conditions. The obtained diameters of inhibition zones reported in mm and the MIC values reported in μg mL−1 were quite similar and the reported results are the average of the three experiments.
Antioxidant Activity
Antioxidant activity was expressed as IC50 (the concentration that causes 50% of effect). Antioxidant activity of the synthesized compounds was assessed by three different techniques which are the 2.2-diphenyl-1-picrylhydrazyl (DPPH), the 2.2’-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) radicals scavenging, and the β-carotene linoleic acid bleaching assay. Butylated hydroxytoluene (BHT), known as a strong antioxidant compound, was used as control.
(DPPH) Radical Scavenging Activity
The procedure involves measurement of decrease in absorbance of DPPH at its absorption maxima of 517 nm. This assay determines the scavenging of stable radical species according to the method of Kirby and Schmidt. 74 Briefly, synthesized compounds were dissolved in dimethylsulfoxide (DMSO)/water (1/9; v/v) and diluted with ultrapure water at different concentrations (1, .5, .250, .125, .0625, .03125 mg mL−1). Then, 500 μL of a 4% (w/v) solution of DPPH radical in ethanol was mixed with 500 μL of samples. The mixture was incubated for 30 min in the dark at room temperature. The scavenging capacity was determined spectrophotometrically by monitoring the decrease in absorbance at 517 nm against a blank. The percentage of antiradical activity (% ArA) had been calculated as follows:
% ArA = [(absorbance of control−absorbance of test sample)/absorbance of Control] × 100
(ABTS) Radical Scavenging Activity
This determination was carried out according to Re et al
75
protocol. 20 μL of solution of synthesized compounds or control were mixed with 18 μL of ABTS solution, the whole was mixed vigorously for 30 s. A control consisting of ethanol and ultrapure water was prepared (5:5) v/v to which the previous solution was added. White is ethanol and ultrapure water (5:5) v/v. Samples and controls were incubated for 6 min in the dark, then the OD was measured at 734 nm. The calculation is done according to the following formula:
Ac: absorbance of the control, Ae: absorbance of the sample.
Footnotes
Author Contributions
L. Baklouti conceptualized the project’s primary principles, drafted the analysis methods, conducted the scientific investigation, formal analysis, data curation, and acquired funding. B. Mellah worked on the project's concept, design, and monitoring and evaluation throughout the project. W.S Koko reviewed all the biological part and edited the first draft of the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
