Abstract
Present research work evaluates variation in volatile chemicals profile and biological activities of essential oil (EO) obtained from the leaves of eucalyptus (Eucalyptus camaldulensis Dehnh.) using hydro-distillation (HD) and supercritical fluid extraction (SFE). The yield (1.32%) of volatile oil by HD was higher than the yield (.52%) of the SFE method (P < .05). The results of physical factors like density, color, refractive index, and solubility of the EOs produced by both the methods showed insignificant variations. Gas chromatography - mass spectrometry (GC-MS) compositional analysis showed that eucalyptol (31.10% and 30.43%) and α-pinene (11.02% and 10.35%) were the main constituents detected in SFE and HD extracted Eucalyptus camaldulensis EO, respectively. Antioxidant activity-related parameters, such as reducing ability and DPPH free radical scavenging capability exhibited by EO obtained via SFE were noted to be better than hydro-distilled EO. Supercritical fluid extracted and hydro-distilled essential oils demonstrated a considerable but variable antimicrobial potential against selected bacterial and fungal strains. Interestingly, oil extracted by SFE showed relatively higher hemolytic activity and biofilm inhibition potential. The variation in biological activities of tested EOs can be linked to the difference in the volatile bioactives composition due to different isolation techniques. In conclusion, the EO obtained from Eucalyptus leaves by the SFE method can be explored as a potential antioxidant and antimicrobial agent in the functional food and nutra-pharmaceutical sector.
Keywords
Introduction
A variety of plants are used traditionally as folk medicine to treat various diseases. 1 Such medicinal health functions of plants can be due to the occurrence of certain bioactives that have multiple biological and pharmaceutical activities. 2 Various herbs and spices are also among those plants which have been historically used in the food and flavor industry as well as employed as native medicines in different civilizations.3,4 The essential oils (EOs) produced from vegetable materials such as leaves and flowers are responsible for multiple functions in plant’s body. Plant EOs are composed of various volatile compounds 3 with major part comprising of terpenoids, which are further divided into monoterpene, diterpene, and sesquiterpene. 5
The EOs extraction from plant materials depends upon its rate of diffusion to the upper surface from plant tissues, from where it can be collected by hydro-distillation (HD), steam distillation, 6 use of supercritical CO2 and high or low pressure distillation, etc.4,7 Steam distillation is used as a conventional method for isolation of EO but this method has some limitation especially with regard to the use of accelerated conditions and long preparation time. 8 Some of the other shortcomings include low extraction efficiency and loss of some heat sensitive volatile components. 9 Therefore, the use of an alternative eco-friendly method, that is, SFE, is encouraged for the extraction of EOs and CO2 is highly promising in this regard because of its experimentally affordable critical conditions of pressure and temperature.2,10,11 A number of EOs are isolated/obtained from various medicinal plants by using supercritical fluid extraction (SFE) such as jasmine, basil, purslane, etc.2,12,13 In SFE, liquid CO2 is used as a supercritical liquid/fluid to recover the aromatic components of plants because of its non-toxic nature, inert behavior, and mild values of critical points such as temperature (31-40°C) and pressure (7.38 MPa). The use of SFE involving liquid CO2 is an impressive green extraction technique applicable for the isolation of high-quality and functional food/nutra-pharmaceutical grade extracts and EOs. The SFE process variables such as critical temperature and pressure and concentration of co-solvent can be optimized to improve the efficiency of the method offering better quality and optimal yield of EOs and extract.7,10,11 Furthermore, this technique is also quite applicable to selectively extract high-value vegetable oils from different seeds. 13 However, SFE is especially superior method to extract EOs compared to other conventional techniques because it is more selective, protects the components from thermal decomposition, and offers contamination free healthier and pure volatile mixtures and natural extracts. 14
Essential oils are composed of biologically active components, and thus, they possess antioxidant, bactericidal and fungicidal properties, and can be used for pharmaceutical purposes. 15 Because of their natural antioxidant and biological attributes, they are much popular in the food and preventive medicines. Recent studies reveal that EOs can be used for curing infectious diseases and as therapeutic agents.5,16 The extensive use of EOs in the field of pharmacy, cosmetics, foods, and beverages industries prompts the need to explore and characterize new plant sources of EOs. 17
In Pakistan, a variety of medicinal plants are grown in various areas of the country, including deserts, plains, and hilly areas. 2 Soon valley is famous as a rich source of medicinal plants with wide array of bioactives such as phenolics and other antioxidant components. This valley’s average temperature is in the range of 1°C (January) to 36°C (June), with low annual precipitation. In view of explicit agro-climatic conditions and soil composition, plants in this region are supposed to consist of a unique profile of phytonutrients. 18 The Eucalyptus spp. EO is mainly used in cosmetics, nutrapharmaceutical, and medicinal applications. Because of different medicinal and pharmaceutical applications, various species of Eucalyptus are employed in folk medicines. 19 Some reports suggest that Eucalyptus EO contains bioactive ingredients and thus is capable of showing multiple biological activities, including antiradical and antioxidant activities, 20 antifungal activity, 21 bactericidal activity, 22 and anti-larva activity. 23
Very little research work is reported for the comparative study of oil obtained by HD and SFE from wild eucalyptus plants. So the current research work is planned to appraise and compare the yield variations, physico-chemical and biological attributes of EOs obtained by two different techniques (HD and SFE) from wild Eucalyptus plant leaves grown in the Soon Valley, District Khusab, Pakistan.
Material and Methods
Collection of Samples
The samples of leaves were obtained from the mature eucalyptus (Eucalyptus camaldulensis L.) plants wildly grown in the Soon Valley, Dist. Khushab, Pakistan. These samples were harvested in spring season and further verified and authenticated by a botanist from Dept. of Botany, University of Sargodha. The leaves sample was then rinsed using deionized water to eradicate dirt and then dried under shade to minimize the moisture content (6.5% ± .10). These dried and powdered samples (particle size 7-12 mm) were used for the isolation of EO.
Extraction of EO
For obtaining the essential oil form the sample material, two different methods were used, including HD and SFE.
Hydro-distillation
In this method, a Clevenger-type apparatus was employed for isolation/extraction of EO. The plant material was hydro-distilled for 3 hours, and EO obtained was dried with Na2SO4 (anhydrous). After filtration, the filtered EO was kept at −4°C before used for further analyses.6,20
Supercritical Fluid Extraction
A commercial-scale supercritical fluid extractor (DEVEN supercritical Pvt. Limited, India) was used (Figure 1). A sample of 5 kg was placed in the extractor, and before extraction, it was heated for 60 minutes. The extraction conditions of the instrument were maintained at the temperature of 45°C with 10 mL/min flow rate of liquid carbon dioxide (density = 1.101 g/mL) at a pressure of 100 bar. The time of the extraction for the static method was 90 minutes, and the dynamic method was 30 minutes. The collection of EO was done in a glass vial collector.
24
Supercritical fluid extractor.
Physical Analysis
The EOs were obtained by both the techniques were subjected to solubility test in 70% alcohol, refractive index and density (at 25°C) measurement. 25 A densimeter (digital) (DMA-602, Austria) was used for density determination, while an automatic refractometer (Model RX-7000, Atago, Tokyo, Japan) was used for refractive index determination. Three replicate readings for each analysis were taken, and results averaged.
Essential Oil Composition
Gas Chromatography
A gas chromatographic instrument (PerkinElmer, 8700, USA) coupled with a FID/flame ionization detector was employed for studying the composition of chemical components of eucalyptus EOs. An HP-5 MS capillary column (30 m × .25 mm) was used for separation of chemical volatiles. The temperature of the column was programmed from 80°C to 220°C by the rate of 4°C/minute while maintaining the initial temperature for 3 minutes and final temperature for 10 minutes constant, total time taken in the column was 48 minutes. Helium (He), as carrier gas/mobile phase, was used with a 1.5 mL/min flow rate. The injector temperature was maintained at 220°C, while the detector temperature at 290°C. The sample (1.0 μL) was injected in split mode (1:100, split ratio). Helium (He), as carrier gas/mobile phase, was used with a 1.5 mL/min flow rate. All the final calculations were performed by using software attached to the chromatographic instruments.
GC-MS Analysis
The volatiles profiling of EOs was also checked and authenticated by using GC-MS technique. A gas chromatographic equipment (Agilent, 6890 N) combined with a mass selective detector (MS-5975) and injector (autosampler, 7683-B) was used. An HP-5 MS capillary column (30 m × .25 mm) was utilized. The column chromatographic conditions and mobile phase were the same as described above for GC-FID. For MS detection, 70 eV energy was used for electron ionization mode, and the temperature of the MS transfer line was maintained at 290°C. A 50 and 600 m/z the mass scanning range was used.
Identification of Components
The compounds separated were identified by comparing their RI (retention indices) value relative to n-alkanes (C9–C24), with the data already published and some previous spectra of compounds already authenticated. Further identification and authentication were performed by their mass spectrometric data comparison with the library of NIST mass spectra and previously reported spectra. 26
Antioxidant Activity Assays
DPPH. Scavenging Activity
For DPPH assay, a previously reported method
27
was used. In this test, .5-100 μg/L of oil sample was added into 1 mL of 90 μM solution of DPPH free radical. The solution volume was adjusted up to 4.0 mL by the addition of 95% methanol. The contents were kept for 60 minutes and noted the optical density at 515 nm. A positive control (BHT) was used separately along with investigated sample. The DPPH• inhibition (%) was estimated as given below
Here, As represents the absorbance of tested sample EO, while Ab is the absorbance of control mixture. For calculation of IC50 value (oil concentration that caused 50% of radical scavenging), a plot, that is, obtained equation, between percentage inhibition and sample concentration was used.
Reducing power
The reducing power assay was also used to determine potential antioxidant potential. 2 According to this method, 2.5-10.0 mg of EO was added with 5.0 mL of .2 mol/L Na-phosphate buffer of pH 6.6 along with 5.0 mL of 1.0% K-ferricyanide solution. Incubation was for 20 minutes at 50°C, then 10% trichloroacetic acid solution (5 mL) was added. Centrifugation of the above-prepared content was done at 980 × g for 10 minutes using a refrigerated centrifuge machine (H-200-NR, Japan) at 5°C. Then, 5.0 mL of the clear upper layer was mixed with 5.0 mL of H2O, followed by 1.0 mL of .1% freshly prepared solution of ferric chloride and finally, the absorbance was measured at 700 nm.
Antimicrobial Activity
A group of microorganisms which included four strains of bacteria (S aureus, E coli, P multocida, and B subtilis) and four fungi strains (A alternata, G lucidum, A niger, and A flavus) were used for the evolution of antimicrobial activity. A culture of nutrient agar (Oxoid) for optimum growth of bacteria and potato dextrose agar (Oxoid) for optimum fungal growth was used at 30°C.
Inhibition Zone
The isolated EOs were subjected to the disc diffusion method 28 for assessing the antimicrobial activity. For this purpose, 6 mm diameter discs containing 50 μL of obtained EOs were put on an agar plate where microorganisms were inoculated previously. For positive reference, Streptomycin (30 μg/disc) was used for bacterial and Fluconazole (30 μg/disc) for fungal strain as a standard drug. The same experimental setup was also performed but without a sample or standard drug as a negative control treatment and incubated these Petri dishes for 24 h at 37°C for bacteria and 3 days at 25°C for fungi. The inhibition zone (millimeter) was measured for the calculation of antimicrobial activity.
Minimum inhibitory concentration
The microdilution broth assay was adopted for MIC analysis. 28 In a 96 well microtiter plate, a series of essential oil dilutions were placed along with one sterility control and one growth control. For bacteria, 160 μL of NB and fungi 160 μL of SDB were mixed into plates and a tested solution (20 μL). The plates were inoculated with standard microorganism suspension (20 μL of 5 × 105 CFU/mL). The plates were kept for 48 hours at 28°C for fungal and 24 hours at 37°C for bacterial growth. The standard drugs were the same as the disc diffusion method used as a positive control. A white pellet that appeared at the bottom indicated the growth of the microorganisms.
Biofilm Inhibition
Analysis of biofilm inhibition was done by a previously reported spectrophotometric method
2
using 96 well microtiter plate reader. In this method, the essential oil was added to the microtiter plate with 100 μL of E coli and S aureus cell suspension. The plates were kept for 3 days at 37°C and then the liquid suspension was removed and 100 μL of 1.0% crystal violet aqueous solution was added. After staining for 30 minutes, wells were washed after removing the dye and again incubated for 15 minutes after adding a 95.0% ethanol. Absorbance was measured at 570 nm spectrophotometrically, and the formula given below was used to calculate the percentage biofilm inhibition.
Hemolytic Activity
Hemolysis of the tested EOs was checked by a method previously reported by Ref. 29 on human erythrocytes of O blood group. This activity was performed under the in-vitro condition. The blood samples were obtained from the healthy volunteers. Initially, the blood sample was centrifuged at 5000 rpm for 5 minutes and prepared the erythrocyte solution (2.0%) in phosphate buffer for analysis. The essential oil, concentration ranging from 50 to 500 μg/mL, were assorted with sodium chloride solution (.85%) followed by erythrocytes suspension (2.0%). The mixture was incubated at 25°C for 30 minutes and centrifuged and absorbance was recorded at 540 nm for liberated hemoglobin in the supernatant. A negative control without essential oil but only sterile phosphate buffer solution and a positive control using Triton X-100 (.1%) were used for comparison. The percentage hemolysis was calculated by multiplying the ratio of absorbance of sample and positive control with 100.
Statistical Study
Three samples of eucalyptus plant leaves were obtained to extract essential oil, and each was separately analyzed. Experiments were done in triplicate, and data is statistically analyzed by ANOVA (STATISTICA, Stat Soft Inc. USA). The significant difference was considered at P ≤ .05, and data are given as mean ± SD for triplicate values. 30
Results and Discussion
Yield and Physicochemical Parameters
Percent Yield and Some Physical Attributes of Eucalyptus Essential Oils Recovered by Different Extraction Methods.

The tabulated data is mean of replicate (triplicate measurements) of tested oil. Significant (P < .05) difference of values between the extraction methods are portrayed by different superscripts inside the same column.
A slight difference was found in color intensity of EOs obtained by two different techniques. The color of hydro-distilled EO is slightly lighter than SCF extracted oil, possibly due to the main component’s lower concentration in the EO by HD. 2 The refractive index, at 25°C, of oil extracted by the SFE method (1.4280) is higher than EO obtained by HD (1.3930); however, this difference is not significant. Similarly, at 25°C, the density of EO extracted by SFE is slightly higher (.942 g/mL) than EO obtained by HD (.891 g/mL). Obviously, this difference in the physical parameters might be due to the varying composition of EO obtained by two different techniques. 32
Chemical Compositional Analysis
GC-MS chromatograms relating to the analysis of the chemical constituents of Eucalyptus camaldulensis EO extracted by SFE and HD methods are depicted in Figures 2 and 3, whereas the percentage of each component is given in Table 2. A total of 31 components were detected in the EO extracted by SFE, while 43 components were identified from the EO extracted by HD. Eucalyptol was detected to be the principal component in E camaldulensis EO obtained by both the techniques; SFE gave a slightly higher percentage of eucalyptol (31.10%) as compared to HD (30.43%). The other important chemical components were α-pinene [SFE (11.02%) and HD (10.35%)], Spathulenol [SFE (5.59%) and HD (10.15%)], Globulol [SFE (4.19%) and HD (4.44%)], Aromadendrene [SFE (4.44%) and HD (4.40%)], menthol [SFE (6.34%) and HD (.42%)], trans-Pinocarveol [SFE (−) and HD (4.17%)], and Epiglobulol [SFE (1.21%) and HD (1.58%)]. GC chromatogram of supercritical fluid extraction eucalyptus essential oil. GC chromatogram of hydro-distillation eucalyptus essential oil. Chemical Composition (%) of Eucalyptus Essential Oils Recovered by Different Extraction Methods. The tabulated data is mean of replicate (triplicate measurements) of tested oil. Significant (P < .05) difference of values between the extraction methods are portrayed by different superscripts inside the same row. *Retention index on HP-5MS column.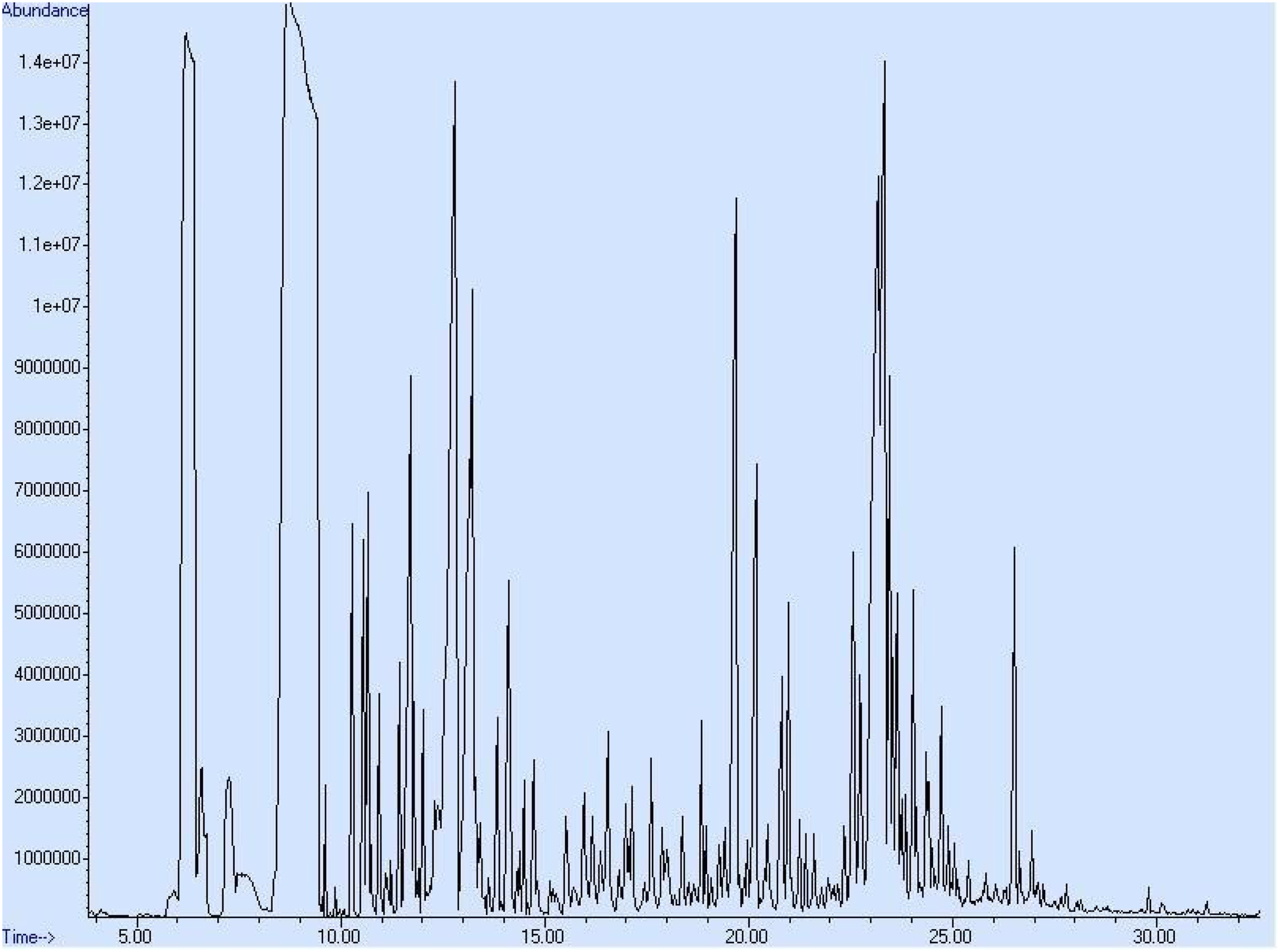

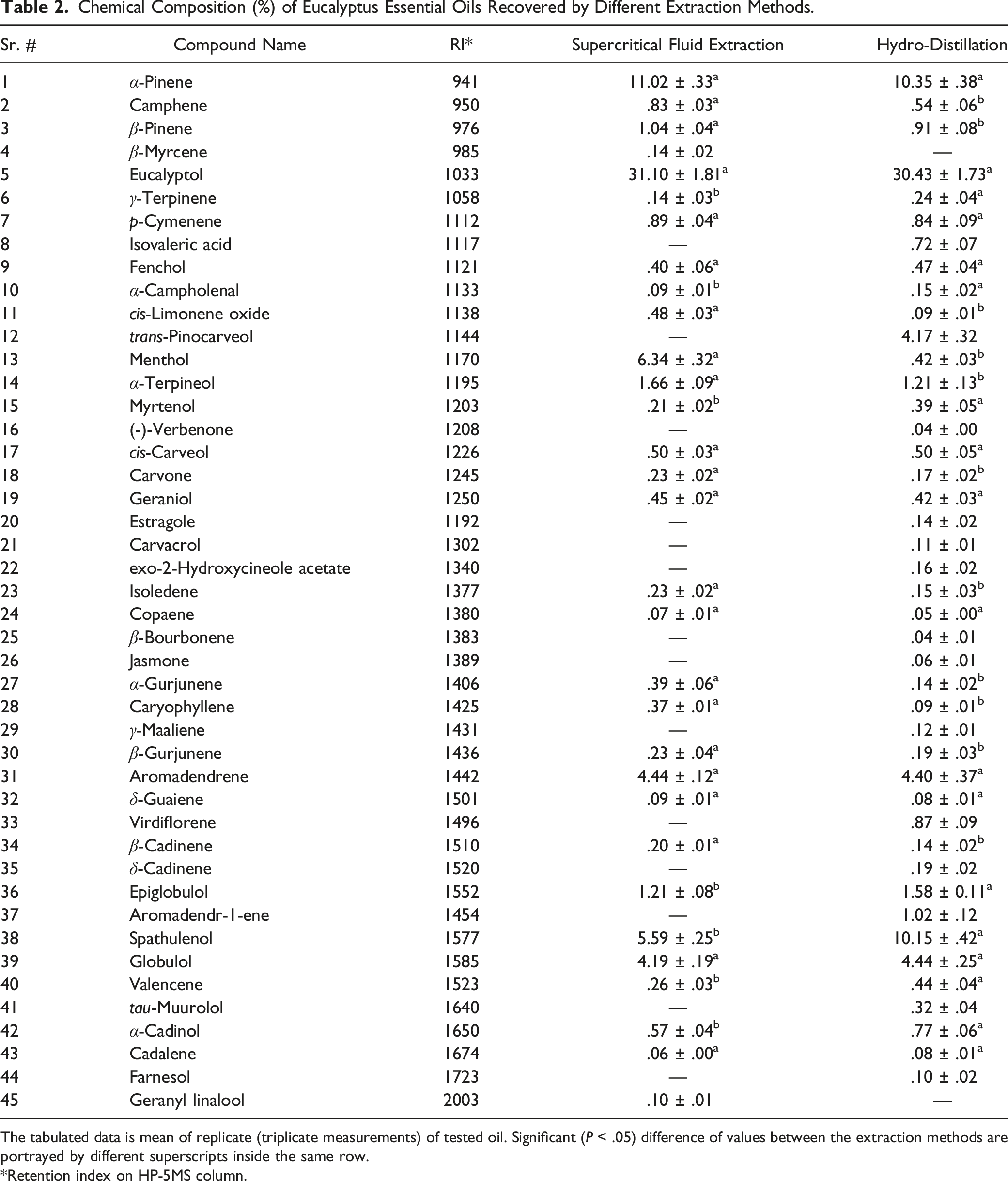
The composition of eucalyptus leaves EO has been reported previously by some researchers23,33,34 who, in well agreement with our present data, reported 1,8-cineole (eucalyptol) as the principal member/component in the oil. Meanwhile, the present results varied compared to a few previous studies that found a different profile for key compounds of essential oil. 35 The difference in the chemical composition can be attributed to various features such as geographical location, the agro-climatic condition of the plant, soil nature, physical state of plant material, that is, fresh or dried, time of collection, age of the plant, and extraction method.2,19 The chemotype and genotype of the plant species might also affect the chemical composition of EOs, as previously investigated by Zouari et al. 36 SCFE method was previously used as extraction technique to obtain volatile components/essential oil from different plant materials, which showed that this technique is a good eco-friendly alternative to conventional methods.13,14
Antioxidant Activities
DPPH radical scavenging activity
Antioxidant Potential of Eucalyptus Essential Oils Recovered by Different Extraction Methods.

The tabulated data is mean of replicate (triplicate measurements) of tested oil. Significant (P < .05) difference of values between the extraction methods are portrayed by different superscripts inside the same column.
Reducing Power
The antioxidant activity of different plant EOs can also be assessed by finding out its reducing ability. 39 For this purpose, the reduction of ferric ions into ferrous ion is made and this process changes the yellow color of the test solution to bluish-green. The intensity of color gives us information regarding the reducing ability of tested EOs. The reducing power of plant EO can be directly correlated to color intensity of the solution, which can easily be assessed colorimetrically. 2 Hence, higher absorption revealed higher reducing power. The results regarding the reducing power of eucalyptus EO are depicted in Table 3, which show a regular increase in the reducing power with an increase in the concentration of EO. At a 10 mg/mL concentration, the maximum absorbance was observed for SF extracted EO (1.84) compared with the EO obtained by HD (1.18). The antioxidant assays outcome furnished that EO from SFE has better antioxidant activity as compared to the EO by HD, so the SFE method is superior for the extraction of high-value bioactives/active constituents in the oil.
Antimicrobial Activity
Antimicrobial Attributes of Eucalyptus Essential Oils Recovered by Different Extraction Methods.
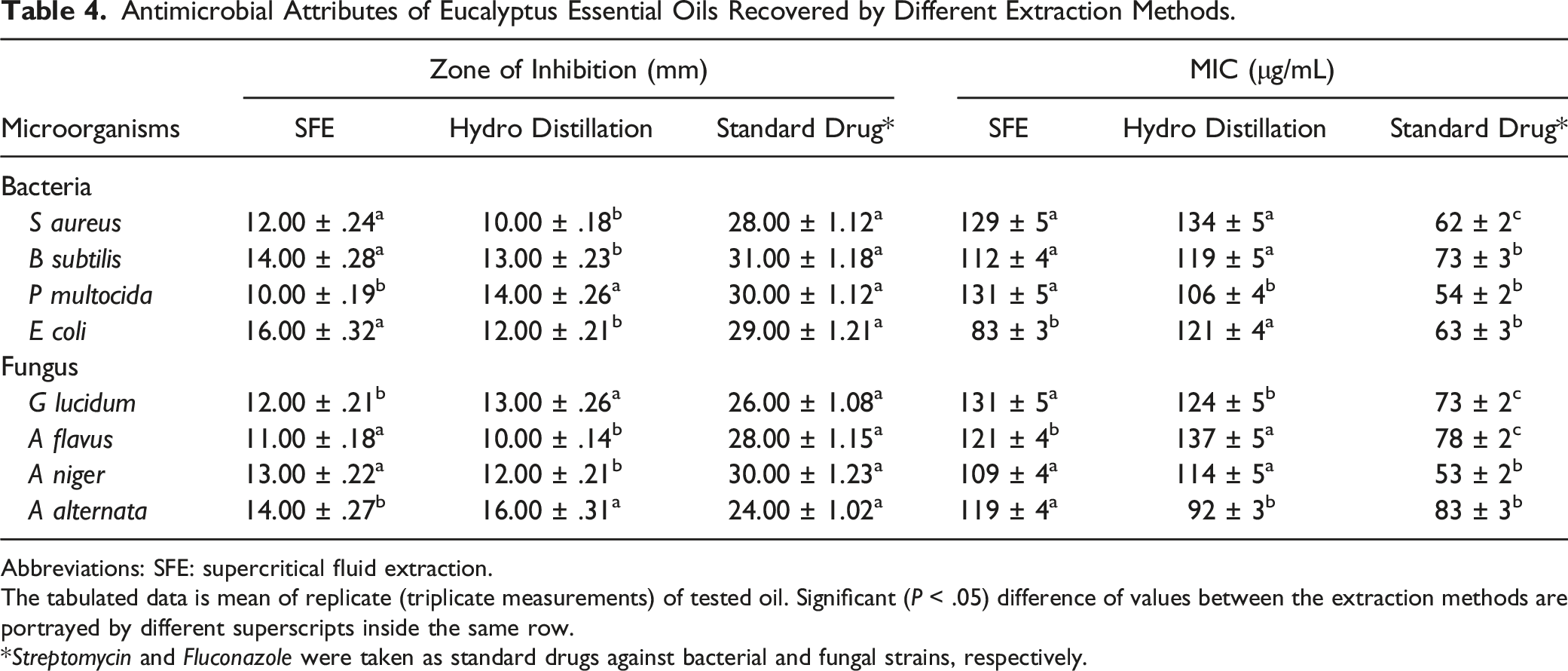
Abbreviations: SFE: supercritical fluid extraction.
The tabulated data is mean of replicate (triplicate measurements) of tested oil. Significant (P < .05) difference of values between the extraction methods are portrayed by different superscripts inside the same row.
*Streptomycin and Fluconazole were taken as standard drugs against bacterial and fungal strains, respectively.
Minimum Inhibitory Concentration
The MIC values (μg/mL) of the tested EOs using the micro dilution broth method are given in Table 4. The SFE extracted EO was a good antibacterial agent (83 μg/mL) against E coli as compared to HD EO (106 μg/mL) against P multocida. The antifungal activity showed that HD based EO was better antifungal agent (92 μg/mL) against A alternata than SFE extracted EO (119 μg/mL) against the same. Overall, the EOs obtained by both the techniques gave a lower antibacterial and antifungal activity as compared to the standard drug, fluconazole, and streptomycin, used in this experiment. In previous research, 40 it is reported that the EO from many plant leaves, including E salubris, E torquata, E woodwardii, and E stricklandii showed an appreciable antimicrobial action against bacterial and fungal strains. Some other reports also showed that eucalyptus is very active against fungal and bacterial strains. 41 Another study 42 investigates the antimicrobial activity of two eucalyptus species and reveals that eucalyptus essential oil is a natural candidate/source of antibacterial and fungal bioactives.
Biofilm Inhibition
Biological Activities of Eucalyptus Essential Oils Recovered by Different 685 Extraction Methods.

The tabulated data is mean of replicate (triplicate measurements) of tested oil. Significant (P < .05) difference of values between the extraction methods are portrayed by different superscripts inside the same row.
EOs can also be used as anti-caries treatments because of their significant inhibition in biofilm formation. 43 EOs have been reported to be good antimicrobial agents, 44 therefore they are very effective against oral microorganisms, thus used in mouthwashes because of their safe nature. 43 It has been previously reported that eucalyptus extracts can help treat tooth decay because of its biofilm inhibition property. Nagata et al, 45 performed a clinical experiment to evaluate the effect of eucalyptus extracts against periodontal diseases.
Hemolytic Activity
The EOs were subjected to assessment of hemolytic activity because some compounds having biological activity may be harmful for hemoglobin and cannot be properly used in pharmacological preparation. This assay may also help to reveal some information regarding the cytotoxicity mechanism. In this research work, 100% hemolysis was obtained using Triton X-100 while a simple buffer was used to get 0% hemolysis. The hemolytic activity results are given in Table 5, which show that the minimum hemolysis (10.80%) is given by SFE extracted EO while the EO by hydro-distillation has higher hemolytic activity (63.65%). So, it can be concluded that the quality of EO obtained using the SFE method46-51 because of its lower hemolysis" as "So, it can be concluded that the EO obtained using the SFE method46-51 is safer since no toxicity was observed in hemolytic assay because of its lower hemolysis. No such work is reported on the hemolytic activity of eucalyptus EO to compare our present research data.
Conclusions
Volatiles chemical profiling and biological potentialities of EOs of E camaldulensis leaves using hydro-distillation and supercritical fluid extraction were investigated. GC-MS analysis revealed eucalyptol and α-pinene as the major chemical constituents in SFE extracted and HD essential oil, respectively. SFE extracted essential oil presented potent antioxidant activity compared to hydro-distilled counterpart. Both the SFE extracted as well as HD EOs demonstrated a considerable antimicrobial potential against bacterial and fungal strains. Superior hemolytic and antibiofilm activities were recorded in SFE extracted oil. In conclusion, the EO obtained from eucalyptus leaves by the SFE method might be explored as a prospective antimicrobial and antioxidant agent in the functional food and nutra-pharmaceutical industries.
Footnotes
Acknowledgments
The authors express their gratitude to Princess Nourah bint Abdulrahman University Researchers Supporting Project (Grant No. PNURSP2022R158), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia (PNURSP2022R158).


