Abstract
Objective
To assess whether there is a relation between allopurinol use and the probability of type 2 diabetes mellitus (T2DM) in persons with gout and/or hyperuricemia.
Methods
According to the PRISMA 2020 guidelines, a meta-analysis was performed by searching literature published from 2000 to 2021 in two electronic databases (Ebscohost and PubMed). The end point was set as a new diagnosis ofT2DM between people with the use of allopurinol and people with non-use of allopurinol. The random-effects model was performed to evaluate the pooled hazard ratio (HR) with 95% confidence interval (CI) for T2DM associated with allopurinol use.
Results
Three cohort studies could meet the inclusion criteria in the meta-analysis. There was a high heterogeneity of the outcome between studies (I2 = 99%). The research duration ranged from 13 to 16 years. The subject number in each work ranged from 1114 to 138,652. A meta-analysis disclosed that there was not an association between allopurinol use and the risk of developing T2DM (pooled HR = 1.01 and 95%CI = .55–1.84).
Conclusions
The meta-analysis shows that no correlation is detected between allopurinol use and the risk of T2DM in individuals with gout and/or hyperuricemia. Because there are not enough eligible studies, the strength of evidence in our meta-analysis is weak. More cohort studies are needed to determine an association between use of allopurinol and the probability of T2DM for individuals with gout and/or hyperuricemia.
Significance
(1) This is the first meta-analysis revealing that no correlation can be detected between allopurinol use and the risk of T2DM among individuals with gout and/or hyperuricemia. (2) Because there are not enough eligible studies, the strength of evidence in our meta-analysis is weak. (3) More cohort studies are needed to determine an association between use of allopurinol and the probability of T2DM in individuals with gout and/or hyperuricemia.
Introduction
Hyperuricemia is usually defined as the serum levels of uric acid >6.8 mg/dl. 1 In addition to causing gout flares, people with hyperuricemia often have comorbidities including chronic kidney disease, diabetes mellitus, dyslipidemia, hypertension and metabolic syndrome.2,3 One cross-sectional study reported that hyperuricemia correlated with increased insulin resistance and impaired insulin secretion. 4 One 2002–2011 cohort study in US reported that a cumulative incidence of type 2 diabetes mellitus (T2DM) was 19% for the levels of serum uric acid ≦7 mg/dl, 23% for 7-9 mg/dl, and 27% for >9 mg/dl. 5 One meta-analysis including 11 cohort studies reported that the increase of serum uric acid by 1 mg/dl will increase the risk of T2DM by 17% (relative risk = 1.17 and 95% confidence interval (CI) = 1.09–1.25). 6
Based on the aforementioned reviews, we hypothesize that lowering serum uric acid may help prevent T2DM. Allopurinol is a urate-lowering drug mediated by the inhibition of xanthine oxidase. Allopurinol is commonly used to treat gout and/or hyperuricemia. An animal model showed that allopurinol use might have an ameliorative effect on vascular complications of insulin resistance. 7 One study showed that allopurinol use for 3 months was associated with improvement of insulin resistance in people with asymptomatic hyperuricemia. 8 One cohort study showed a reduced hazard ratio (HR) for T2DM associated with allopurinol use (HR = .57), 9 but the other cohort studies showed an increased HR for T2DM associated with allopurinol use (HR ranging from 1.17 to 1.70).10,11 There are different results about an association between allopurinol use and the probability of developing T2DM. Therefore, a meta-analysis was conducted to comprehensively understand the impact of allopurinol use on the risk of T2DM for individuals with gout and/or hyperuricemia.
Methods
Searching Strategy
Based on the PRISMA 2020 guidelines, 12 a meta-analysis was conducted through searching literature published from January 2000 to June 2021 in two electronic databases (Ebscohost and PubMed). We used the following keywords to search literature of interest: “diabetes,” “gout,” “hyperuricemia,” “uric acid,” “allopurinol,” “urate-lowering,” and “uric acid-lowering.” The different keywords were combined together according to the following rules: diabetes (title) AND gout (title), diabetes (title) AND hyperuricemia (title), diabetes (title) AND uric acid (title), diabetes (title) AND allopurinol (title), diabetes (title) AND urate-lowering (title), as well as diabetes (title) AND uric acid-lowering (title). The following inclusion criteria were applied: (1) cohort studies including subjects with a baseline diagnosis of gout and/or hyperuricemia; (2) cohort studies assessing the use of allopurinol and no use of allopurinol; and (3) cohort studies using T2DM as an end point.
Data Extraction
The qualification of all searched articles was evaluated by two authors based on the aforementioned inclusion criteria (KFL and YHK). The following variables were extracted: family name of the first author, published time, data source, research design, study period, sample size, male percentage, mean age of study subjects, the incident users of allopurinol, the primary endpoint of each study, adjusted hazard ratio with 95% CI, bias, and study limitation. Disagreement of all searched articles was resolved through the discussion of the two authors (BFH and CSL).
Risk of Bias Assessment
A Newcastle-Ottawa Scale (NOS) system was applied to examine the quality of the included works. 13 A NOS score ≥8 was considered as a high-quality of research.
Publication Bias
Because only 3 studies were eligible for analysis, it indicated that those studies without statistical significance might not be published. Therefore, the publication bias should exist and it was inevitable.
Statistical Analysis
The random-effects model was applied to assess the pooled HR with 95% CI for T2DM associated with allopurinol use. We used the I2 and Q statistics to determine the outcome heterogeneity between these works.
14
I2 > 50% suggests a significant heterogeneity between these works. All of statistical analyses were carried out by using Cochrane’s Review Manager 5.3 software (Copenhagen: The Nordic Cochrane Center, The Cochrane Collaboration, 2014). A
Results
Literature Selection
Figure 1 presents a process of literature selection. Totally, 23 articles were identified through searching two electronic databases (Ebscohost and PubMed) based on the keywords. There were 4 articles excluded due to duplication. There were 16 articles excluded due to topic not interested. Finally, 3 articles were eligible for a meta-analysis. Flow diagram of literature selection.
Study Characteristics
Characteristics of eligible studies included in a met-analysis.
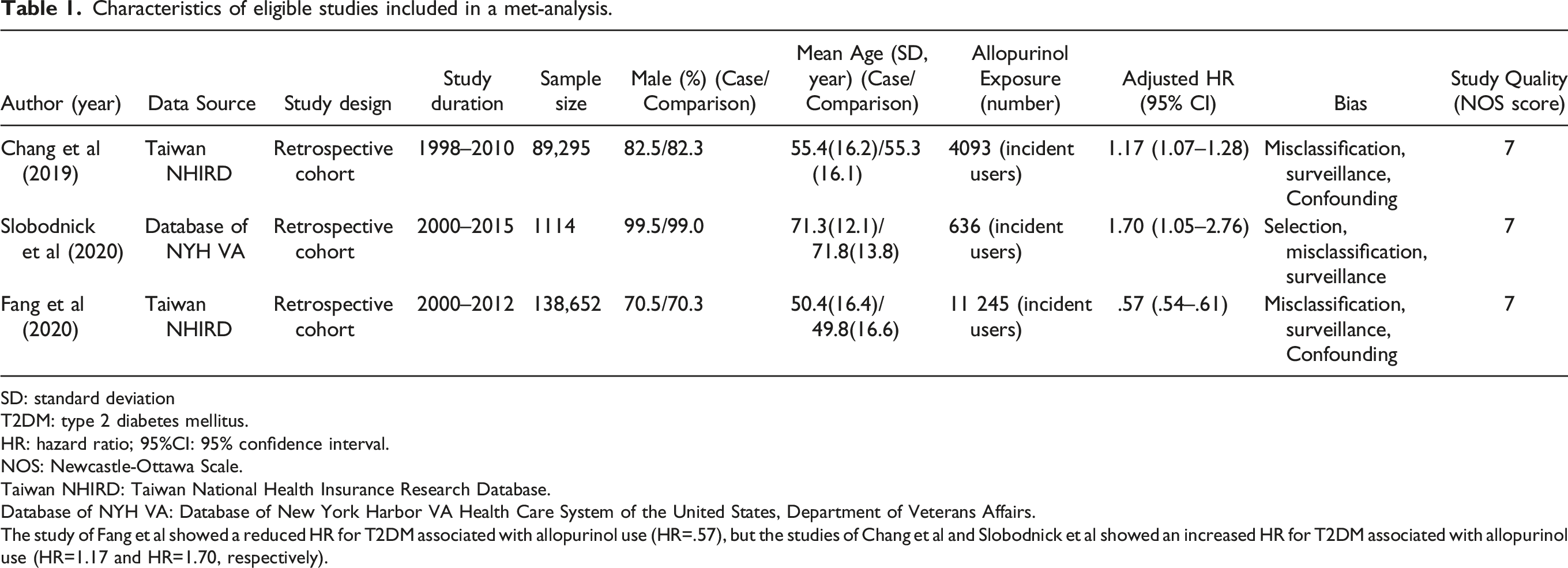
SD: standard deviation
T2DM: type 2 diabetes mellitus.
HR: hazard ratio; 95%CI: 95% confidence interval.
NOS: Newcastle-Ottawa Scale.
Taiwan NHIRD: Taiwan National Health Insurance Research Database.
Database of NYH VA: Database of New York Harbor VA Health Care System of the United States, Department of Veterans Affairs.
The study of Fang et al showed a reduced HR for T2DM associated with allopurinol use (HR=.57), but the studies of Chang et al and Slobodnick et al showed an increased HR for T2DM associated with allopurinol use (HR=1.17 and HR=1.70, respectively).
The study of Fang et al showed a reduced HR for T2DM associated with allopurinol use (HR=.57), 9 but the studies of Chang et al and Slobodnick et al revealed an increased HR for T2DM associated with allopurinol use (HR = 1.17 and HR = 1.70, respectively).10,11
The NOS score of study quality was 7 for the 3 cohort studies.
Pooled Hazard Ratio of T2DM and Allopurinol Use
In Figure 2, the pooled HR of T2DM associated with allopurinol use was 1.01 (95% CI = .55–1.84, and P = .98). It did not have statistical significance. There was a high heterogeneity of the outcome between studies, with a I2 > 50%. (Cochran’s Q = 177.39, I2 = 99% and P < .001). Random-effects model of the pooled hazard ratio of type 2 diabetes mellitus associated with allopurinol use.
Sensitivity Analysis
One study by Wang et al showed an increased HR for T2DM associated with all urate-lowing medications (HR = 3.66, 95% CI = 1.51–8.86), 15 but the authors only reported that allopurinol accounted for 93.6% of all urate-lowing medications. The specific HR associated with allopurinol use was not noted. So the study of Wang et al was not included in our meta-analysis. When including the Wang et al’s study for a sensitivity analysis, the pooled HR of T2DM associated with allopurinol use was 1.26 (95% CI = .72–2.21 and P = .42). It still did not have statistical significance.
Discussion
The meta-analysis disclosed that there was not a significant association between allopurinol use and the risk of T2DM for individuals with gout and/or hyperuricemia. Theoretically people with gout always have hyperuricemia, but the majority of people with hyperuricemia will not have gout over a long time. 16 Therefore, we included study subjects with gout as well as study subjects with hyperuricemia for a meta-analysis based on the same underlying disorder “hyperuricemia.”
A new theory of uric acid-associated dysfunction of the pancreatic beta-cells has been proposed to explain the link between hyperuricemia and the risk of T2DM.17-19 A high uric acid level will induce oxidative stress and inflammation within the pancreatic beta-cells, which further could lead to apoptosis of the pancreatic beta-cells and a decrease of insulin secretion.17-19 In addition, an elevated level of uric acid is related to an increase of insulin resistance.20-22 Subsequently, hyperglycemia occurs and then T2DM develops later. If the relation between hyperuricemia and T2DM is causal, a urate-lowering effect may be beneficial to prevent T2DM. The animal models disclosed that allopurinol could ameliorate insulin resistance in rats and mice.23-26 Previous clinical research also showed allopurinol could ameliorate insulin resistance in humans. 8 The study of Fang et al included in our meta-analysis disclosed a reduced HR for T2DM associated with allopurinol use. 9 But the other 2 studies included in our meta-analysis disclosed an increased HR for T2DM associated with allopurinol use.10,11 Based on the above discussion, there seems to be a discrepancy between experimental research and clinical research. We think that the causes of T2DM are multi-factorial. Only lowering serum uric acid but without ameliorating other risk factors is not sufficient to prevent T2DM. For example, body weight loss and increased physical activity are effective to prevent T2DM.18,27
Because of different research populations and different research approach, there was a high degree of the outcome heterogeneity between included works. Some bias should be mentioned. First, people with non-allopurinol exposure were defined as people with gout who did not take allopurinol in Slobodnick et al’s study,
11
but the other urate-lowering medications could be used in these people and thus the urate-lowering effects might be still present. The efficacy of allopurinol alone could not be correctly evaluated in Slobodnick et al’s study. Therefore, selection bias could not be excluded from Slobodnick et al’s study. However, people with non-allopurinol exposure found in the studies of Fang et al and Chang et al were defined as those people who did not have the gout history.9,10 Thus those who did not have the gout history did not need to take allopurinol or the other urate-lowering medications. The efficacy of allopurinol alone could be correctly evaluated in the studies of Fang et al and Chang et al. We think that the different selection of the comparison group between the 3 cohort studies made their research results different. Second, people who took allopurinol needed to have their blood checked regularly. These people had more chances to be found with T2DM
The limitations of our meta-analysis should be mentioned. First, males and older people were predominant in Slobodnick et al’s study. 11 Their results could not be generalized to the other populations. Second, because the three included studies did not provide the detailed HR by sex distribution, we were not able to measure the HR of T2DM stratified by sex. Whether there could be different results between sex needs to be further studied. Third, unmeasured variables, such as physical activity, life style, smoking habit and drinking alcohol, could have the impacts on the health outcome in the three included studies. Fourth, only three studies were eligible for the meta-analysis, so the publication bias should be present. Fifth, the NOS score of research quality was 7 for the three cohort studies. It means that the quality of the 3 cohort studies was not high. More high-quality research is needed to overcome these limitations.
Conclusion
The meta-analysis shows that no correlation is detected between use of allopurinol and the risk of T2DM for individuals with gout and/or hyperuricemia. Because there are not enough eligible studies, the strength of evidence in our meta-analysis is weak. More cohort studies are required to determine an association between use of allopurinol and the probability of T2DM.
Supplemental Material
Supplemental material - Allopurinol Use and the Risk of Type 2 Diabetes Mellitus: A Meta-Analysis of Cohort Studies
Supplemental material for Allopurinol Use and the Risk of Type 2 Diabetes Mellitus: A Meta-Analysis of Cohort Studies by Shih-Wei Lai MD, Bing-Fang Hwang PhD, Yu-Hung Kuo MS, Chiu-Shong Liu MD and MS, and Kuan-Fu Liao MD and PhD in Dose-Response
Footnotes
Author contributions
Shih-Wei Lai contributed to the conception of the article, initiated the draft of the article, and has approved the final draft submitted. Yu-Hung Kuo and Kuan-Fu Liao conducted data analysis. Bing-Fang Hwang and Chiu-Shong Liu interpreted the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was waived because the study was a meta-analysis of published data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
