Abstract
Radiation-induced intestinal injury (RIII) restricts the therapeutic efficacy of radiotherapy in abdominal or pelvic malignancies. Also, intestinal injury is a major cause of death following exposure to high doses of radiation in nuclear accidents. No safe and effective prophylactics or therapeutics for RIII are currently available. Here, we reported that the apigenin, a natural dietary flavone, prolonged the survival in c57 mice after lethal irradiation. Apigenin pretreatment brought about accelerated restoration of crypt-villus structure, including enhanced regenerated crypts, more differentiated epithelium cells, and increased villus length. In addition, intestinal crypt cells in the apigenin-treated group exhibited more proliferation and less apoptosis. Furthermore, apigenin increased the expression of Nrf2 and its downstream target gene HO-1, and decreased oxidative stress after irradiation. In conclusion, our findings demonstrate the radioprotective efficacy of apigenin. Apigenin has the potential to be used as a radioprotectant in cancer therapy and nuclear accidents.
Introduction
The small intestine is one of the most radiosensitive organs. Radiation-induced intestinal injury (RIII) manifests after a dose of more than 6 Gy total-body or abdominal irradiation. Ionizing radiation compromises epithelial integrity and impair intestinal epithelium renewal, which further result in fluid loss, electrolyte imbalance, sepsis, and even death.1,2 RIII is a dose-limiting factor for radiotherapy of abdominal or pelvic malignancies that impairs the efficiency of therapy and reducing the patient’s quality of life. Moreover, a large number of victims emerge in a mass casualty setting from nuclear accidents or terrorism. RIII, one of the main types of acute radiation-induced injury, limits the survival of these victims. Unlike hematopoietic failure, which can be rescued with bone marrow transplantation and supportive care, currently there are no approved specific drugs for protecting or mitigating against RIII. 3 Amifostine (WR2721) has been approved for xerostomia associated with radiotherapy. However, Amifostine cannot be used for the treatment of RIII due to its serve side effects include hypertension, nausea, and vomiting. 4 Therefore, highly effective countermeasure with fewer side effects that accelerates injured intestinal tissue regeneration and improves survival is urgently needed.
It is generally believed that the cellular injury of normal tissue after radiation exposure is caused by reactive oxygen species (ROS) including OH•, O2 •−, H2O2 generated by the radiolysis of water. These ROS are known to cause oxidative stress which deposit energy and mediate damage to macromolecules 5 (DNA, lipid, and proteins). Nuclear factor erythroid 2-related factor 2 (Nrf2) is a key transcription factor thought to be the master regulator of cellular redox homeostasis. Nrf2 binds to antioxidant response elements (AREs), which promote the production of proteins involved in reducing oxidative stress. 6 Therefore, Nrf2 may be a target for new therapeutic approaches to treat RIII. Existing studies have shown that activation of Nrf2 and its target gene HO-1, NOQ-1 enhances the survival of intestinal stem cell and promote epithelium regeneration against ionizing radiation.7,8
The potential use of natural antioxidants that are widely sourced and low in toxicity as radioprotectors has received increasing attention. 9 Apigenin (4',5,7-trihydroxyflavone) is a flavonoid compound widely found in fruits and vegetables. Accumulating evidence has shown that apigenin has various biological activities, such as antioxidant, anti-inflammatory, and anti-tumor properties.10-13 Apigenin shows radioprotective effect both in vitro and in vivo studies. Apigenin was observed to alleviate radiation-induced chromosomal damage and cytogenetic alteration in cultured human lymphocytes.14,15 The research of Begum et al. 16 highlighted the radioprotective effect of apigenin against γ-radiation-induced oxidative stress and hematopoietic injury in a mouse model. Moreover, apigenin was reported to promote Nrf2 into the nucleus, thereby activating Nrf2 pathway and enhancing the expression of the anti-oxidative stress related genes.17,18 However, the function and mechanism of apigenin as a radioprotector against RIII has not yet been investigated. The aim of this research is to determine whether apigenin can attenuate the intestinal injury and promote intestinal regeneration and functional repair in a mouse model.
Materials and Methods
Mice
Male C57BL/6J mice aged 8–10 weeks were obtained from the SPF (Beijing) Biotechnology Co., Ltd, China. The mice were kept under controlled conditions, including constant temperature, and were allowed free access to regular chow and sterile water. All animal experimental procedures were approved by the ethics committee of the PLA Rocket Force Characteristic Medical Center and were performed in accordance with the Guide for the Care and Use of Laboratory Animals.
Apigenin Preparation and Administration
Apigenin (Apigenin, A-320001, Innochem, China) dissolved in 5% dimethyl sulfoxide (DMSO), 5% Tween-80, and 90% saline was administered intraperitoneally to mice 12 hours and 2 hours prior to total body irradiation (TBI) or total abdomen irradiation (TAI) at 2 dose of 200 mg/kg. The animals were randomly assigned to different treatment groups. For survival experiments, at least 10 mice/groups were used and at least 4 mice/groups were used for other experiments.
Radiation Protocols
X-rays produced by an XCELL 225 high energy x-ray irradiator (KUBTEC USA) in the laboratory of the PLA Rocket Force Characteristic Medical Center (Beijing, China) were used as radiation sources. For total body irradiation (TBI), mice were irradiated at a dose rate of .72 Gy/min (225 kV, 13.2 mA). For total abdomen irradiation (TAI), mice were anesthetized and exposed abdomens fully, lead chamber was used to protect other parts of body from radiation.
Histology and Immunohistochemistry
Intestinal tissues were freshly isolated and fixed with 4% paraformaldehyde before paraffin embedding. After deparaffination and rehydration, the sections were stained with hematoxylin and eosin (H&E). For immunohistochemistry, sections were repaired by sodium citrate solution or EDTA antigen repair solution, blocking at room temperature with goat serum, incubated overnight at 4°C with primary antibody, and then sections were immunostained using the streptavidin peroxidase method. Following antibodies were used: anti-BrdU (ab6326, Abcam, UK); anti-Cleave Caspase-3 (9664S, CST, USA); anti-Villin (16488-1-AP, Proteintech, USA); anti-Chromogranin A (10529-1-AP. Proteintech, USA); anti-Muc2 (27675-1-AP, Proteintech, USA); and anti-Lysozyme (ab108508, Abcam, UK). For 5bromo-2'-deoxyuridine (BrdU), BrdU (B5002-5GSigma-Aldrich, USA) was given to mice by intraperitoneal injection at a dose of 120 mg/kg 2 hours before execution. The viability of the surviving crypt was confirmed by admixing BrdU into five or more epithelial cells within each regenerating crypt. Five intestinal circumferences were randomly selected from each mouse to count and positive cells were counted in 5 crypts in each intestinal circumference.
Terminal Deoxynucleotidyl Transferase dUTP Nick-end Labeling Assay
The apoptosis at 6 hours and 24 hours post-10 Gy TBI in the intestinal tissues was determined using a Deoxynucleotidyl Transferase dUTP Nick-end Labeling (TUNEL) apoptosis assay kit (1098, Beyotime, China) according to the manufacturer’s protocols. Five intestinal circumferences were randomly selected from each mouse to count and positive cells were counted in 5 crypts in each intestinal circumference.
Western Blot
Proteins were extracted from intestinal tissues using ice-cold lysis buffer (Solarbio, China). Protein concentrations were quantified using a bicinchoninic acid (BCA) protein assay kit (Beyotime, China), and equal amounts of protein were separated by SDS-PAGE gels. The blocked membranes were incubated overnight at 4°C using anti-HO-1 (27281-1-AP, Proteintech, USA), anti-Nrf2 (L6396-1-AP, Proteintech, USA), and anti-Tubulin (80762-1-RR, Proteintech, USA). The membranes were then incubated with the appropriate horseradish peroxide-coupled secondary antibodies for 1 hour at room temperature. Chemiluminescent substrates are used to detect proteins. Integrated density was measured by Image J.
Malondialdehyde Assay
Mice intestinal tissue was collected 6 hours after 10 Gy irradiation and homogenized in precooled PBS and the supernatant was centrifuged to obtain samples for the assay. The Lipid Peroxidation Malondialdehyde (MDA) Assay Kit (S0131-M, Beyotime, China) was used according to the manufacturer’s instructions.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 8.0 (San Diego, CA, USA) software. All data were obtained from at least 3 repetitions and were presented as the mean ± standard error mean (SEM) unless otherwise specified. The statistical significance was calculated with unpaired Student’s t-test. Log-rank analysis was performed for survival analysis. Asterisks indicate P values as follows: *P < .05, **P < .01, ***P < .001, and ****P < .0001. P < .05 was considered statistically significant.
Results
Apigenin Improves the Survival and Alleviated Intestinal Injury Induced by Radiation in Mice
To evaluate the radioprotective effect of apigenin in vivo, 200 mg/kg compound was intraperitoneally administered twice before (12 hours and 2 hours) radiation, with a schedule chosen experimentally for optimal efficiency of apigenin. (Supplemental Figure 1A and B). The median survival time of mice treated with apigenin was prolonged from 4 d to 7 d. Compared with the vehicle group, apigenin treatment attenuated the weight loss in mice (Figure 1A and B). To exclude the death caused by radiation-induced acute hematopoietic injury, we used the total abdominal irradiation (TAI) model in which mice were exposed to 12 Gy (Supplemental Figure 2A and D). The results showed that all the mice treated with vehicle died within 10 d, while 50% of the apigenin-treated mice survived for more than 30 d (Figure 1C and D). We further determined the maximum exposure dose that apigenin can protect against RIII. Surviving crypt decreased sharply when mice were exposed to escalating doses of radiation. In the apigenin-treated group, surviving crypts were increased by 177% (8 Gy), 185% (10 Gy), and 158% (12 Gy), respectively. Apigenin had no significant radioprotective effect on RIII at radiation dose above 14 Gy (Figure 1E and F). These results suggest that apigenin protects against RIII in mice exposed to a wide range of irradiation doses. A dose of 10 Gy was selected in the subsequent experiment. Apigenin protects against deadly intestinal injury caused by irradiation. (A-B) Survival curves of vehicle or API-treated C57BL/6J mice after 10 Gy TBI and relative body weight at 5 d. (C-D) Survival curves of mice treated with vehicle or API after 12 Gy TAI and relative body weight at 5 d (n = 10). (E-F) Representative images of H&E-stained intestinal sections 3.5 days after 8, 10, 12 and 14 Gy TBI and quantitative analysis of surviving crypt (n = 4). Values are mean ± SEM; Scale bar = 200 μm; *P < .05, **P < .01, ****P < .0001, vehicle vs apigenin treatment by unpaired two-tailed Student’s t-test. TAI, total abdominal irradiation; TBI, total body irradiation.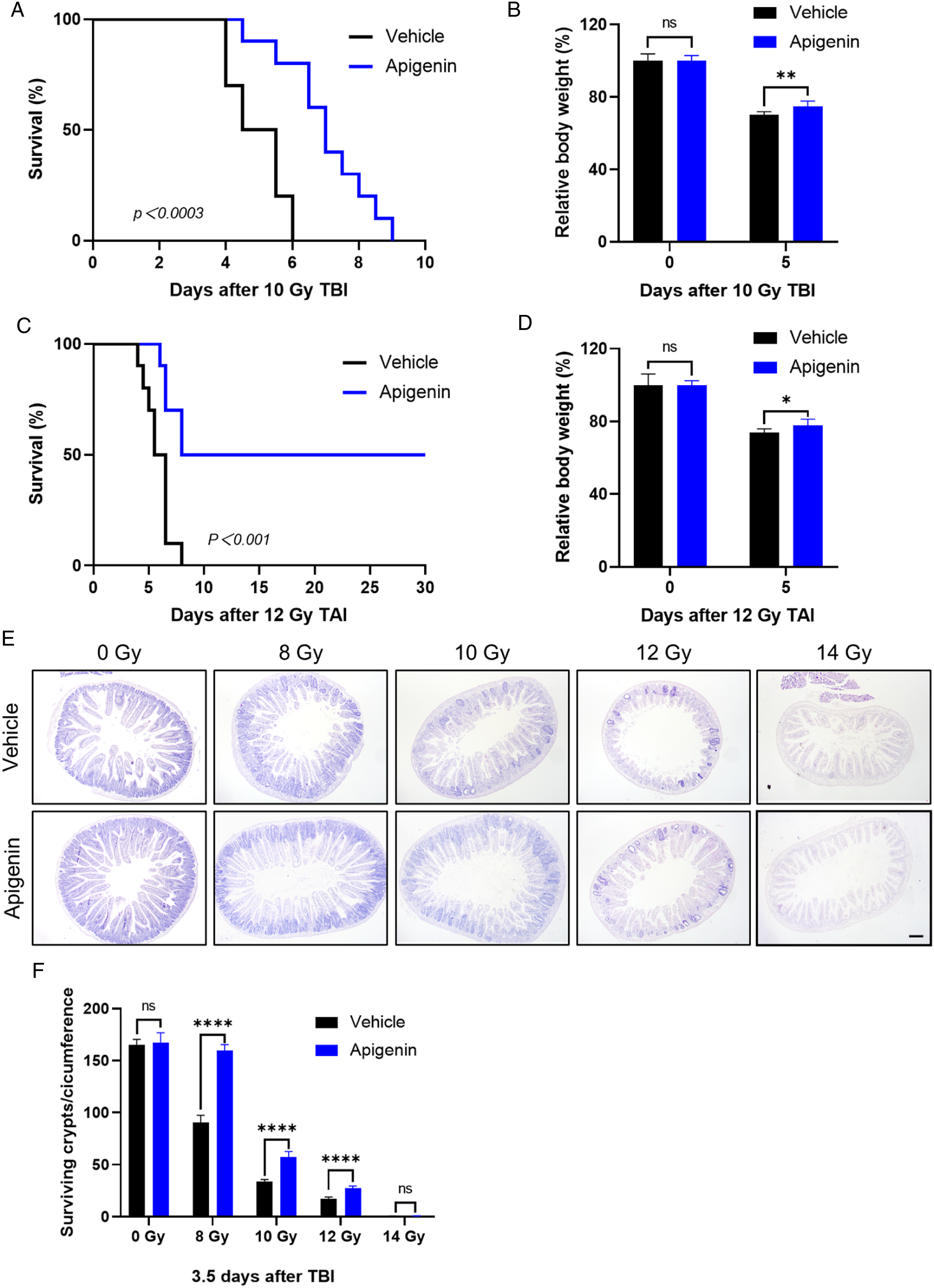
Apigenin Preserves Intestinal Crypt-Villus Structure After Irradiation
We further assessed the radioprotective effect of apigenin on RIII histologically 96 hours post-IR, the key time point for estimation the reconstruction of crypt-villus structure.
19
The results indicated that the mean villus height in irradiated group was 377 μm, shortening to 61.5% of that of unirradiated villus, while pretreatment with apigenin increased the mean villus height to 81.9% (Figure 2A and B). To further explore the beneficial role of apigenin in repairing intestinal epithelial cells post-radiation, we analyzed differentiated intestinal epithelial cells, including Paneth, goblet, and enteroendocrine cells. The results showed that the number of Muc2+ goblet cells, lysozyme+ Paneth cells, and chromogranin A+ (Chga+) enteroendocrine cells were markedly increased on the intestinal sections of apigenin-treated mice compared with the vehicle group (Figure 2C and H). These data suggest that apigenin maintains intestinal crypt-villus homeostasis after radiation-induced injury. Apigenin promotes the recovery of small intestinal structures after 10 Gy TBI. (A-B) Representative images of Villin-IHC intestinal sections 96 hours after 10 Gy TBI and villi length quantitative analysis. (C-H) Representative images of Lysozyme-, Muc2-, Chga- IHC intestinal sections and the quantification of the positive cells at 96 hours after 10 Gy TBI. Scale bar = 100 μm. Values are mean ± SEM; n = 4 mice per group. ****P < .0001, vehicle vs apigenin treatment by unpaired two-tailed Student’s t-test. Chag: Chromogranin A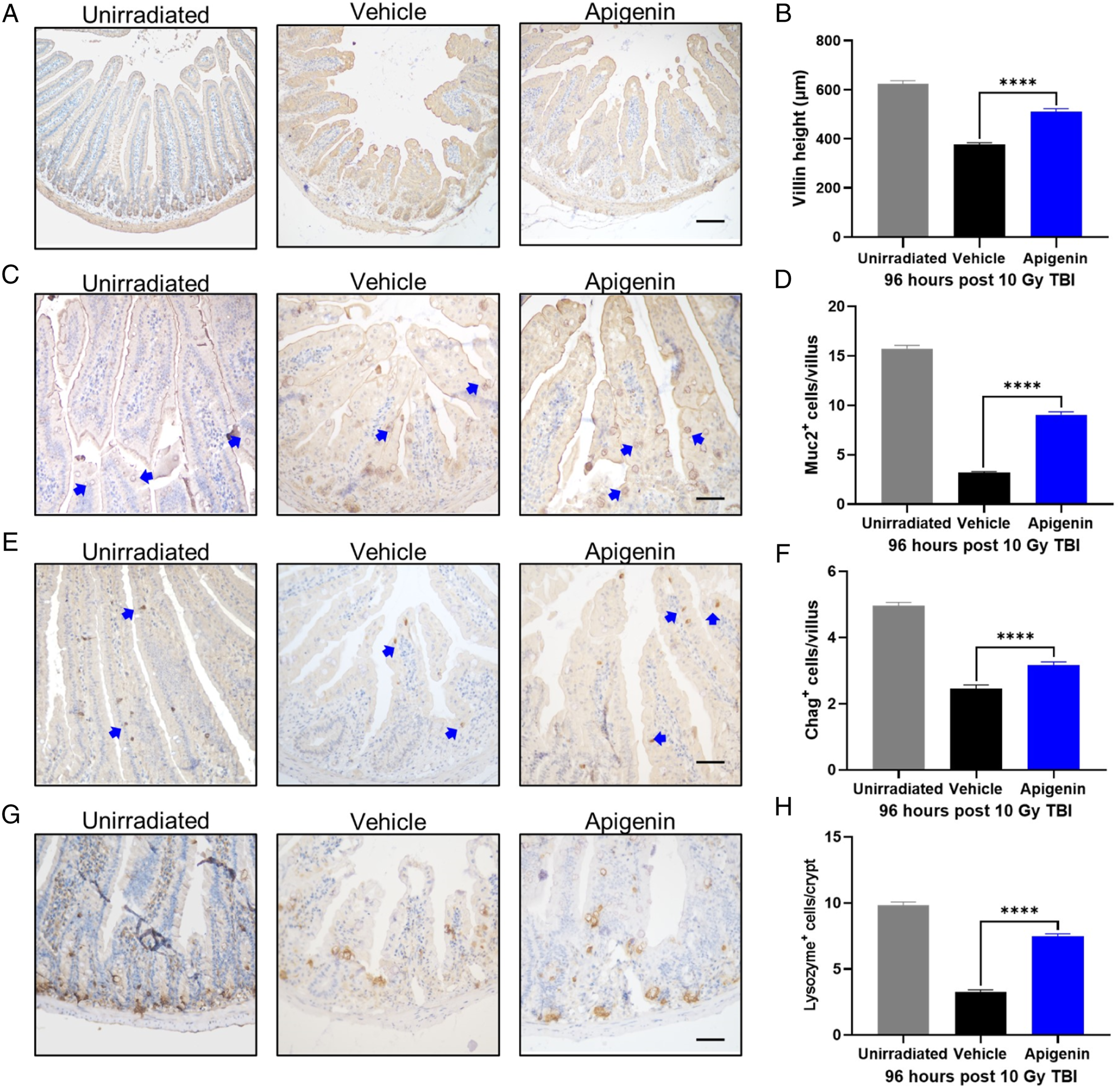
Apigenin Promotes Crypt Regeneration in Mice After Irradiation
Exposure to high doses of radiation triggers acute and fatal intestinal crypt damage.20,21 To further observe the effect of apigenin on the intestinal pathology of irradiated mice, we measured the crypt depth and found that apigenin administration significantly increased the intestinal crypt depth compared to the vehicle group (Figure 3A and B). In addition, the number of regenerated crypts increased by 312% in the apigenin-treated group 3.5 d after 10 Gy TBI (Figure 3C and D). We proceeded to explore the crypt regeneration from 0 hours to 96 hours post-TBI using the BrdU binding assay, which captures S-phase entry and DNA replication (Figure 3E). The number of proliferative crypt cells in vehicle-treated mice gradually decreased from 6 h after TBI, reached to the lowest value at 48 hours, and returned to the similar level to unirradiated group until 96 h post-IR. Compared to vehicle-treated mice, apigenin-treated mice intestinal crypt cells exhibited potent proliferation each time point, with the average number increasing by 1.28 times (6 hours), 1.60 times (24 hours), 2.24 times (48 hours), and 2.19 times (96 hours), respectively (Figure 3F). These data suggest that apigenin can promote crypt regeneration after TBI. Apigenin enhances crypt regeneration after irradiation. (A-B) Representative images of H&E-stained intestinal sections 3.5 d after 10 Gy TBI and quantitative analysis of crypt depth. (C-D) Representative images of BrdU IHC intestinal sections at 3.5 d after 10 Gy TBI and quantitative analysis of regenerated crypts per circumference. Scale bar = 100 μm. (E-F) Representative images of BrdU IHC intestinal sections at the indicated times after 10 Gy TBI and quantitative analysis of BrdU+ cells in each crypt. Scale bar = 50 μm. Values are mean ± SEM; n = 4 mice per group. ****P < .0001, vehicle treated with apigenin by unpaired two-tailed Student’s t-test. BrdU,5-Bromo-2′-deoxyuridine; IHC, immunohistochemistry.
Apigenin Reduces Apoptosis of Crypt Cells After Irradiation
We examined crypt cell apoptosis by TUNEL and cleaved caspase-3 staining at 6 hours and 24 hours after 10 Gy TBI. TUNEL staining results showed apoptosis in the crypt increased greatly with time in the vehicle-treated group at 6 hours and 24 hours post-IR. This trend was reversed in the apigenin-treated group, which reduced the total number of apoptotic cells by 40% compared to the vehicle group at 6 hours post-irradiation and reduced by 50% at 24 hours (Figure 4A and B). Similarly, the cleaved caspase-3 cells in crypts were decreased in apigenin-treated mice compared with radiation control (Figure 4C and D). Apigenin decrease radiation-induced apoptosis in the intestine after TBI. (A-B) Representative images of TUNEL staining and quantitative analysis of positive cells. (C-D) Representative images of cleaved caspase-3 IHC and quantitative analysis of positive cells. Scale bar = 5 μm. Values are mean ± SEM; n = 4 mice per group. ****P < .0001, vehicle vs apigenin treatment by unpaired two-tailed Student’s t-test.
Apigenin Activates Nrf2/HO-1 Pathway and Decreased Oxidative Stress After Irradiation
To assess the effect of apigenin on oxidative stress, we measured the lipid peroxidase (MDA) levels in mice intestinal tissue 6 hours post-IR, the apigenin treatment significantly reduced the MDA levels in the intestine of irradiated mice. (Figure 5A). Nrf2 plays a critical role in maintaining endogenous redox homeostasis.
22
We isolated intestinal tissues after irradiation and analyzed the expression of Nrf2 and its downstream target protein, HO-1, by immunoblotting. At 6 hours after irradiation, the protein expression levels of Nrf2 and HO-1 were significantly higher in the apigenin-treated group than in the vehicle group. (Figure 5B). Collectively, it can be postulated that the radioprotective effect of apigenin on RIII is related to Nrf2. Apigenin attenuates oxidative stress injury after TBI. (A) Measurement of malondialdehyde (MDA) levels in intestinal tissues at 6 hours after TBI. (B) Western blotting detection of Nrf2 and HO-1 protein expression of small intestinal tissues. (C) Quantitative protein analysis. Values are mean ± SEM; n = 3–4 mice per group. **P < .01, vehicle vs apigenin treatment by unpaired two-tailed Student’s t test.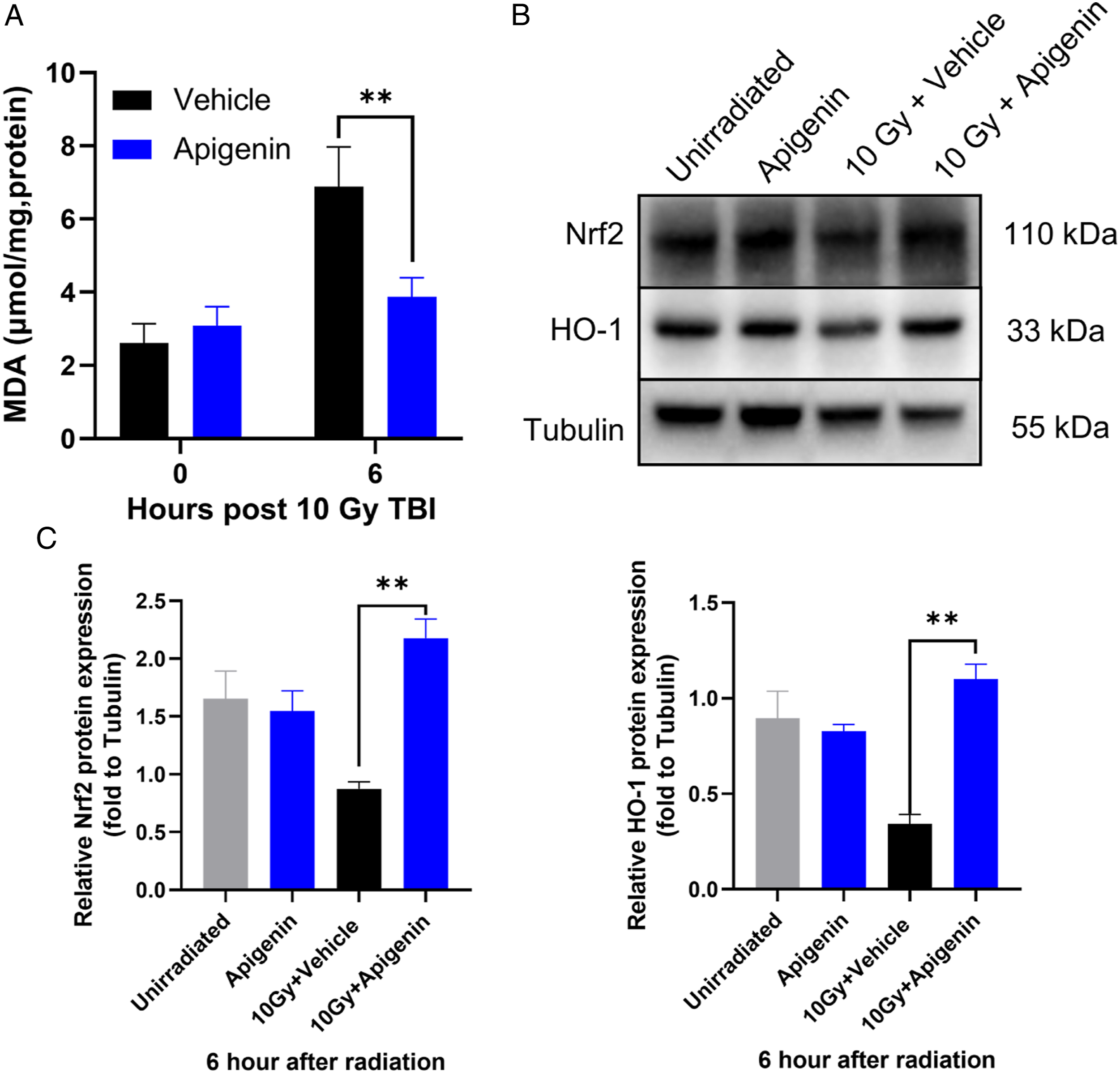
Discussion
The intestinal tract is susceptible to planned radiation exposure during pelvic and abdominal radiotherapy in cancer treatment and accidental radiation exposure during nuclear accidents. However, no safe and effective prophylactics or therapeutics have been approved at present for RIII. 3 In the present study, we provided in vivo evidence that the apigenin, a natural dietary flavone, confers a protective effect on intestinal injury induced by radiation. Our data revealed that apigenin pretreatment reduced mortality and alleviated weight loss in C57BL/6J mice exposed to lethal radiation. In recent years, apigenin has gained increasing interests because it is abundant in common fruits, vegetables, and herbs, exerts various biological activities but hardly promotes adverse metabolic reactions in vivo 12 Apigenin was reported to render protection against 7 Gy radiation-induced hematopoietic injury when administered intraperitoneally at a dosage of 15 mg/kg for 7 consecutive days. 16 In this study, we found that the apigenin dosage lower than 50 mg/kg has no effect on RIII (data not shown). To increase the plasma concentration of apigenin in response to higher radiation exposure, we increased the administration dose to 200 mg/kg, which showed no observable toxicity. Apigenin is characterized by poor systemic availability due to its low solubility and short half-life, which will severely limit its pharmacological efficacy and clinical use. 10 Li et al 23 prepared apigenin-loaded solid lipid nanoparticle and used it to attenuate diabetic nephropathy. Similarly, new technologies or formulations can be used to improve the bioavailability of apigenin for the development of radioprotector. The anti-tumor effects of apigenin have recently been reported in multiple studies, 12 and further study will be needed to determine whether apigenin treatment is also protective against conventionally fractionated radiotherapy, especially when used for spontaneously growing abdominal and pelvic tumors.
Intestinal epithelial cells originate from intestinal crypts harboring intestinal stem cells. Moreover, intestinal crypts are the most radiosensitive compartment of the intestinal epithelium, and intestinal crypt regeneration is required for efficient reconstruction of normal crypt-villus structure after radiation injury. Our data shows that apigenin significantly increased regenerated crypts post 8-12 X-irradiation, indicating apigenin radioprotects intestinal stem cell. However, whether apigenin directly or indirectly protects intestinal stem cells requires further research. Four main differentiated cell types mediate the functions of the intestinal epithelium: absorptive (enterocytes), enteroendocrine, mucosecreting (goblet cells), and Paneth cells. 24 We observed more of these differentiated epitheliums after apigenin treatment in RIII mouse model. Enterocytes are responsible for the absorption of nutrients, goblet cells and enteroendocrine cells control gut physiology by secreting a variety of hormones, and Paneth cells play an essential role in the control of the microenvironment of intestinal stem cell. 24 Taken together, apigenin preserves crypt-villus structure after radiation injury, which further maintains intestine homeostasis and enhances regeneration after radiation.
Reactive oxygen species induced by photon irradiation including X and gamma ray is the main cause of damage to normal cells and tissues. 25 Photon irradiation, including X and gamma rays, generates a large amount of ROS, which exceeds the scavenging capacity of the cellular antioxidant system, leading to the peroxidation of DNA, lipids, and proteins, and ultimately induces cell death. Our data show that apigenin decreased lipid peroxide contents (MDA) and inhibited apoptosis of intestinal crypt cells post-radiation, indicating apigenin can effectively reduce ROS. Transcriptional activator Nrf2, a master regulator of cellular redox homeostasis, induces the expression of multiple cytoprotective genes in response to oxidative stress. 26 Researches have shown that activation of Nrf2 signaling attenuates radiation-induced crypt injury.7,8 We observed that the expression of Nrf2 and its downstream gene HO-1 were significantly increased after pretreatment with apigenin. These findings were consistent with previous reports, in which the endogenous mRNA and protein levels of Nrf2 and HO-1 were increased by apigenin.17,18 Nrf2 may be required for apigenin preventing radiation-induced oxidative stress in intestine, the mechanism of apigenin for preventing RIII needs further study.
Conclusion
The present study demonstrates the protective effects of apigenin against radiation-induced intestinal injury and mortality of mice. These effects may be due to activation of the Nrf2/HO-1 pathway to reduce oxidative stress. Apigenin is an attractive candidate to be used as a radioprotector, and its mechanism remains to be further studied.
Supplemental Material
Supplemental Material - The Protective Effects of Apigenin Against Radiation‐Induced Intestinal Injury
Supplemental Material for The Protective Effects of Apigenin Against Radiation‐Induced Intestinal Injury by Danjie Liu, Renjun Peng, Zhongmin Chen, Huijie Yu, Sinian Wang, Suhe Dong, Wei Li, Wen Shao, Jing Dai, Fengsheng Li, Qisheng Jiang and Wanjun Sun in Dose-Response
Footnotes
Acknowledgments
The authors thank Dr Qingwen Yuan, Dr Zeze Huang, and Dr Shipeng Liu for their help with this experiment.
Author Contributions
W.J.S., Q.S.J. and R.J.P. designed the study. D.J.L., R.J.P., Z.M.C., H.L.Y., S.N.W., Z.M.W., S.H.D., W.L., W.S., J.D., F.S.L., performed laboratory experiments. D.J.L and R.J.P., analyzed the data and prepared the manuscript. W.J.S. and Q.S.J. directed final version of all contents. All authors reviewed and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Military Research Program (AEP17J001).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
