Abstract
Purpose
Nuclear factor erythroid 2-related factor 2 (Nrf2) is a crucial cytoprotective protein that shields cells from electrophilic and oxidative stress. Mice lacking Nrf2 exhibit heightened susceptibility to myelosuppression due to impaired hematopoietic reconstitution. In this study, we examined the altered sensitivity to ionizing radiation (IR) and 7,12-dimethylbenz(a)anthracene (DMBA) in Nrf2−/− mice separately.
Materials and Methods
Irradiate Nrf2−/− or wild-type mice with a dose of 4 Gy to observe changes in body weight, survival rate, and blood routine at 12 months. DMBA was used to treat Nrf2−/− and wild-type mice, and the body weight and survival rate of the mice were measured. The changes of heme oxygenase-1(HO1) and NAD(P)H: quinone oxidoreductase 1(NQO1) in mice treated with IR or DMBA were detected by RT-qPCR and western blotting.
Results
Our results indicate that Nrf2 deficiency leads to more severe blood and immune system injury in mice exposed to IR or DMBA. Additionally, long-term monitoring revealed that Nrf2 deletion resulted in more severe myelosuppression, leukemia-like symptoms, and higher cancer rates. At the mRNA and protein levels, there was no significant increase in HO1 and NQO1 levels in the Nrf2−/− mice treated with IR or DMBA. These adverse effects might be attributed to the inhibited protein levels of HO1 and NQO1 and significant DNA damage in hematopoietic stem and progenitor cells (HSPCs).
Conclusions
We demonstrate that the genetic deficiency of Nrf2 in mice leads to reduced antioxidant capacity and suppression of hematopoietic and immune system function, resulting in increased sensitivity to IR or DMBA.

Introduction
Ionizing radiation (IR), characterized by its capacity to ionize atoms and molecules, is extensively employed across a spectrum of applications, including military, industrial, and medical sectors. While it has brought progress and convenience to mankind, it has also brought threats to human health. Reports show that IR exposure is associated with carcinogenesis, circulatory diseases and metabolic diseases.1,2 In particular, irradiation with IR has been shown to stimulate the synthesis of reactive oxygen species (ROS), which can cause DNA double-strand breaks or result in cellular oxidative stress. 3 Additionally, 7,12-dimethylbenz(a)anthracene (DMBA), a polycyclic aromatic hydrocarbon present in cigarette smoke, is also a known carcinogen. DMBA mediated carcinogenic effects involve cellular DNA damage and dysregulation of genes important for cell proliferation and survival. 4
Previous studies have shown that leukemia is usually formed from genetic and epigenetic changes of hematopoietic stem cells (HSCs), which maintains the homeostasis of hematopoietic system. 5 Both IR and DMBA are myelosuppressive agents, and they cause DNA damage and genomic instability in HSCs.6,7 Therefore, in view of the wide application of IR or DMBA, the leukemia caused by it needs to attract people’s attention, and the specific mechanism of leukemia formation needs to be further studied.
Nuclear factor erythroid 2-related factor 2 (Nrf2) is a member of the cap 'n' collar (CNC) family, characterized by its leucine zipper (bZIP) domain, which functions to facilitate its role as a transcription factor. Under physiological conditions, Nrf2 and its cytosolic inhibitor, KEAP1, form a complex within the cytoplasm. As a potent transcription factor, Nrf2 orchestrates the expression of a suite of cytoprotective proteins, including heme oxygenase-1 (HO-1) and NAD(P)H: quinone oxidoreductase 1 (NQO1), among others. 8 Due to its extensive transcriptional regulatory role, Nrf2 influences diverse cellular processes, including drug metabolism and redox homeostasis, among others.9,10 Recent studies have explored the roles of Nrf2 in the hallmarks of cancer, highlighting its dual role in both the prevention of carcinogenesis and the promotion of tumor progression.
In our study, we found that Nrf2 was essential for organ protection in mice exposed to IR and DMBA. Nrf2 affected cancer rates and long-term survival in these mice and the incidence and mortality of leukemia-like symptoms significantly increased in Nrf2−/− mice. This phenomenon was attributed to more severe cell damage, genomic instability, and delayed recovery in the hematopoietic system of Nrf2−/− mice caused by IR and DMBA. Overall, our study established a link between Nrf2 and IR/DMBA-induced leukemia-like symptoms in mice.
Material and Methods
Animals
All mice utilized in this investigation were housed in a specific-pathogen-free facility, with environmental conditions meticulously maintained at a temperature of 22 ± 2°C. The mice in the study were fed for 14 months, including DMBA or IR treatment. For cancer type evaluation, we used the classical method of microscopic observation of cell morphology to determine various cancer types, for example, pathological examination of mouse bone marrow, spleen, and liver, and proportion of immature cells in bone marrow to determine leukemia.
Long-Term and Short-Term Treatment of Mice with IR
Four groups of mice were divided, including: WT,
Long-term treatment: There were 34 male and 32 female mice aged 6 weeks. The mice were exposed to 4 Gy Total body irradiation (TBI), and the 12-month survival rate was calculated.
Short-term treatment: 12 male mice were used to do blood routine examination (6 each); 10 male mice were used to do Comet assay (5 each).
Long-Term and Short-Term Treatment of Mice with DMBA
Four groups of mice were divided, including: WT,
Long-term treatment: DMBA was dissolved in soybean oil at a concentration of 5 mg/mL. The 6 weeks mice were treated with 200 μL DMBA or soybean oil by intragastric administration once a week (84 male mice). The DMBA or soybean oil was administered five times. The mice were then observed and recorded weekly.
Short-term treatment: The 8 weeks mice were treated with 200 μL DMBA (5 mg/mL) or soybean oil by intragastric administration 3 times a week. The mice were euthanized and dissected one week later. 12 male mice were used to do Blood routine and immune organ index analysis, and 12 male mice were used to extract HPSC and perform comet experiments.
Ionizing Radiation Treatment
Cellular and murine specimens were subjected to a standardized exposure regimen involving 137Cs γ-radiation, delivered at a precise dose rate of 0.890 Gy/min. This irradiation was facilitated by the Nordion Gammacell 40 whole-body irradiator, a device renowned for its precision in dosimetry and widely utilized in radiobiological research (BEST Theratronics Ltd., Ottawa, Canada).
Determination of Thymus and Spleen Index
For a quantitative evaluation of the immune response, the spleen and thymus indices (Sx) are calculated as follows:
Sx = Weight of the experimental organ (mg)/Weight of the experimental animal (g).
Antibodies, Kits and Reagents
Antibodies: NRF2 (Proteintech, 16396-AP), HO1 (Abcam, ab137749), NQO1(Abcam, ab28947), Tubulin (Proteintech, 66240-1-Ig), Actin (Proteintech, 66009-1-Ig).
Kits and reagents: EasySep™ Mouse SCA1 Positive Selection Kit (Stem cell technologies, #18756), Human Epidermal Growth Factor (hEGF) (Cell Signaling Technology, #8916), B-27™ Supplement (50X) (Gibco, 17504044).
Apoptosis Detection
The assay was performed with the One Step TUNEL Apoptosis Assay Kit, Fluorescein (Beyotime, Shanghai, China), which employs fluorescein-conjugated dUTP to label free 3′-OH ends of DNA fragments. Following the manufacturer’s guidelines, the TUNEL detection solution was applied, and the tablets were incubated in the dark at 37°C. After rinsing with PBS, the tablets were sealed with an anti-fluorescence quenching solution and examined under a fluor.
Quantitative RT-PCR
Total intracellular RNA was extracted using TRIzol reagent (catalog number 15596018; Invitrogen, Carlsbad, CA, USA). Subsequently, complementary DNA (cDNA) was generated following the manufacturer’s protocol. Quantitative real-time polymerase chain reaction (RT-qPCR) was performed using BlasTaq™ 2X qPCR MasterMix (catalog number G891; Applied Biological Materials Inc., Richmond, BC, Canada), in accordance with the manufacturer’s instructions. Data normalization was carried out by employing GAPDH as a housekeeping gene for internal control.
Colony Formation Experiment of Bone Marrow Cells
Mouse bone marrow Hematopoietic stem and progenitor cells were sorted using the RoboSep™ Mouse SCA1 Positive Selection Kit Protocol (STEMCELL Technologies, Vancouver, Canada). Suspend a total of 2 × 104 BM cells from each mouse in 100 µL of fresh culture medium and mix with 1 mL of methylcellulose medium. The mixture was then seeded into six-well plates and grown at 37°C and 5% CO2. Using an inverted microscope to observe and count colonies.
Hematopoietic Stem and Progenitor Cells (HSPCs) Isolation
Remove the femur of the mouse and remove the muscles as much as possible, and rinse thoroughly with PBS buffer. Soak the femur in PBS buffer for 5 minutes, then cut open at both joints and repeatedly rinse the bone marrow cavity with PBS to obtain a suspension of bone marrow cells. Using ACK lysis buffer (A1049201, Thermo Fisher Scientific) to remove red blood cells (Incubate on ice for 5 minutes). Label SCA1+ HSPCs and screen using flow cytometry.
Comet Assay/ Single-Cell Gel Electrophoresis (SCGE) Assay
The mice were completely exposed to 2 Gy γ-rays, and their peripheral blood was taken at 0.5, 1, 2, and 4 hours after irradiation. The Mouse peripheral blood mononuclear cell Isolation Kit (TBD2011 M, Tianjin, Hao Yang Biological Co., LTD) was used to collect peripheral blood mononuclear cells, which were then adjusted to a concentration of 105-106 cells/ml with PBS. DNA breaks in cells were measured using the comet assay as previously described. 11 Data is analyzed and generate chart in Graphpad Prism 8.
Whole-Cell Lysate Collection and Western Blotting
To prepare whole-cell lysates, cells were initially collected and rinsed with phosphate-buffered saline (PBS). Subsequently, they were resuspended in pre-chilled RIPA lysis buffer (catalog number R0010; Solarbio, Beijing, China) supplemented with 0.1% protease and phosphatase inhibitors. The suspension was incubated on ice for 30 minutes, with regular vortexing to ensure thorough cell lysis. Then, the lysates were supplemented with 4× protein loading buffer (catalog number P1016; Solarbio, Beijing, China) and subjected to boiling in a metal bath at 100°C for 10 minutes to denature the proteins.
Statistical Analyses
Each experimental procedure was conducted three times to ensure reproducibility, with results expressed as the mean ± standard deviation (SD). Statistical comparisons between groups were performed using T test, as implemented in either GraphPad Prism version 8 or SPSS software. Significance was determined at the threshold of
Results
Nrf2 is Essential for Survival of Mice Exposed to IR
Previous research has demonstrated that Nrf2 plays a crucial role in enhancing the survival of mammalian cells subjected to ionizing radiation by accelerating the repair of DNA damage and facilitating the detoxification process of superoxide radicals.
12
Sang Bum Kim et al also showed that synthetic triterpenoids activating Nrf2 improves overall survival in mice exposed to a lethal dose of whole-body irradiation. In our study, we exposed male and female mice to 4 Gy of IR, which did not cause mortality in a short-term, and monitored them for a long-term. Furthermore, 20% of WT mice and 80% of Survival Status Analysis of Mice in Irradiated Models and Pathological Analysis of Major Organs of them. (A) Male Mice were Exposed to 4 Gy TBI, and the 12-Month Survival Rate was calculated. (B) Female Mice were Exposed to 4 Gy TBI, and the 13-Month Survival Rate was calculated. (C) The Body weight of Male Mice was Calculated within 12 months after Receiving 4 Gy TBI. (D) Representative Pathologic Photomicrographs of the Spleens in 
Nrf2 Protects Hematological System from IR by Reducing Damage, Maintaining Genomic Stability and Promoting Functional Recovery
The hematological system is highly sensitive to IR. Whole-body doses of 2 Gy or higher can cause bone marrow depression and reduction in lymphocytes.
13
IR promotes the development of hematopoietic malignancies. Given that
To test this hypothesis, we conducted blood tests to observe how Nrf2 affected the number of different cell types in the blood after 4 Gy TBI (Figure S2A). No statistically significant differences in red blood cell (RBC) counts were observed between the two mouse cohorts (Figure S2C). Although platelet count was significantly higher in Nrf2 Protects Hematological System From IR by Reducing Damage, Maintaining Genomic Stability and Promoting Functional recovery. (A) Statistical Chart of Changes in the Number of WBC. Peripheral blood WBC Count of Mice before IR and 3, 9 and 14 days after 4 Gy IR. (B) Statistical Chart of bone Marrow Cell Colony Formation. Bone Marrow Cells were Isolated From Unirradiated Mice and Mice at 24 h after 2 Gy IR. After Colony Formation Culture, the Total Number of Granulocyte and Macrophage Colonies (CFU-G, CFU-M, CFU-GM) was Counted. (C-E): Comet Assay of Mouse Peripheral blood Mononuclear Cells. Peripheral blood Monocytes were Isolated From Unirradiated Mice and Mice at 0.5, 1, 2 and 4 h after 2 Gy IR. Comet Experiments Represent Pictures (C). Tail DNA Content Statistics (D). Olive Tail Moment Value Statistics (E). (F-G): Micronucleus Test of Polychromatic Erythrocytes in Mouse bone Marrow. Bone Marrow Cells were Isolated From Unirradiated Mice and Mice at 24 h after 2 Gy IR. The Micronucleus was Observed after Staining with Giamsa Solution. The black Arrow Indicates Micronucleus (F). Micronucleus Number Statistics (G). H: Mouse bone Marrow Cell Chromosome. Mice were not Irradiated or 24 hours after Receiving 2 Gy IR. Black Arrows Indicate Chromosome Fragments. (*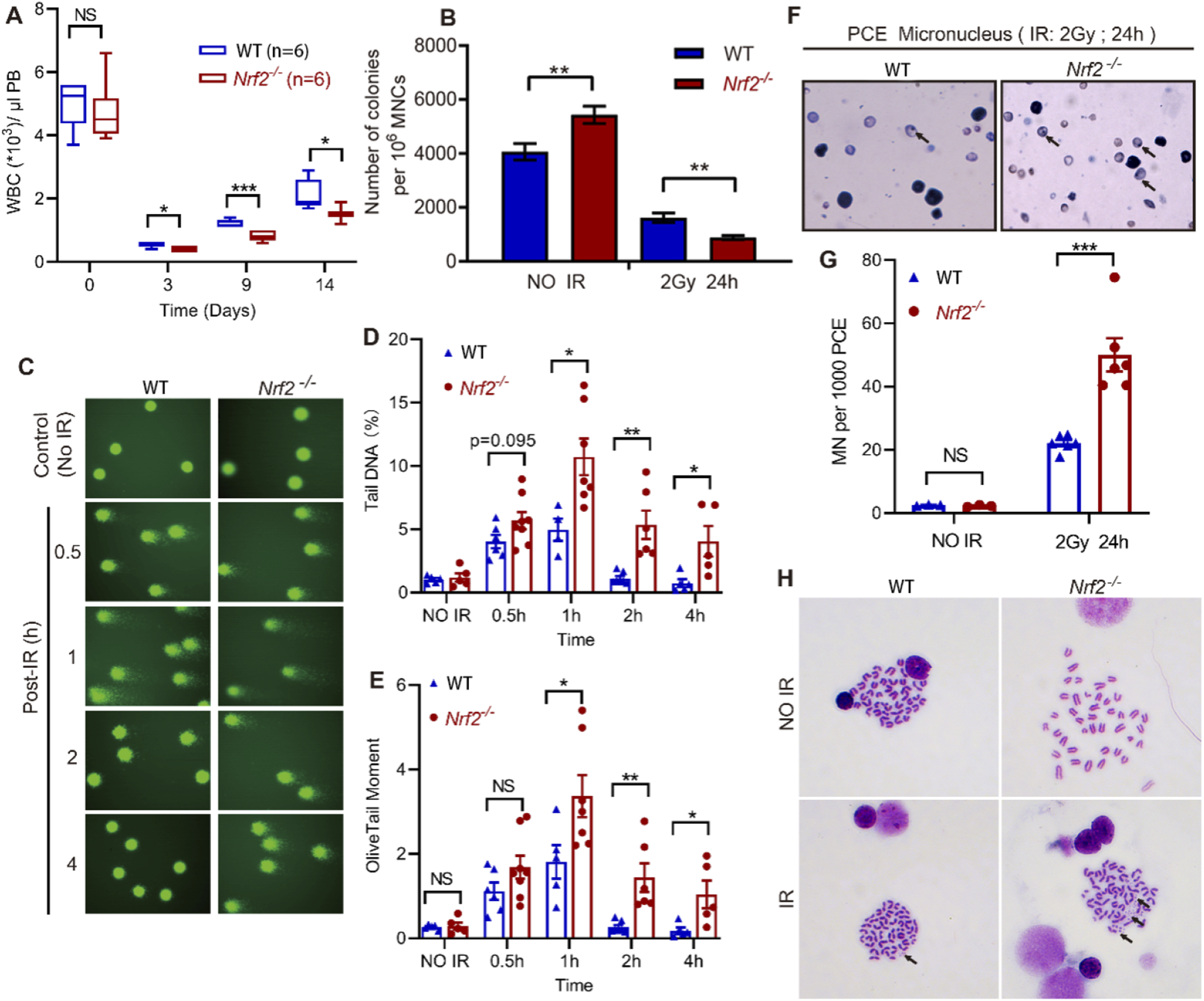
Peripheral blood mononuclear cells (PBMCS) were extracted from mice exposed to 2 Gy IR (Figure S4A). Comet experiments with them showed that tail DNA content and olive tail moment of
Nrf2 Protects Spleen and Thymus from IR
Studies showed that spleen and thymus are also sensitive organs to IR.
14
Thus, we examined these organs and notice that Nrf2 Protects Spleen and Thymus From IR. (A) The Changes of Thymus Index, after Mice Exposed to 4 Gy IR. (B) The Changes of Spleen Index, after Mice Exposed to 4 Gy IR. (C) Pictures of Spleen, after Mice Exposed to 4 Gy IR. (D) Representative Pathologic Photomicrographs of Thymus, after Mice Exposed to 4 Gy IR 48h. White Scale bar, 100 μm. (E) Representative Pathologic Photomicrographs of Spleens, after Mice Exposed to 4 Gy IR 48h. White Scale bar, 100 μm. (F) The Apoptosis of Spleen Cells. TUNEL Detection Method was Used for Detection, after Mice Exposed to 4 Gy IR 24h. White Scale bar, 100 μm.
Nrf2 Absence Leads to DMBA-Induced Cancer Predispositions, Especially Leukemia
Since Nrf2 was associated with IR-induced leukemia, we wanted to test whether it affects other drug-induced leukemia. DMBA has been reported to induce acute bone marrow cytotoxic and leukemia in mice and rat.15,16 Thus, we administered intragastric DMBA treatment to the mice for five weeks and monitored them for cancer development. The results showed that DMBA caused death in mice within 45 weeks post-administration, with the mortality rate of Survival Status Analysis of Mice in DMBA-Induced Cancer Models, Type and Incidence of Cancer, and Pathological Analysis of Major Organs of them. (A) Mice Gavage with DMBA for 5 weeks, and the 45-week Survival Rate was calculated. (B) The Body weight of Mice was Calculated within 38 weeks after Receiving DMBA Gavage for 5 weeks. (C) Statistical Chart of Cancer Types and Incidence in Dead mice. (D) Statistical Chart of blood Routine Analysis in Peripheral blood of Leukemia mice. (E) Representative Pathologic Photomicrographs of the Livers in DMBA-Induced Leukemia mice. White Scale bar, 100 μm. (F) Representative Pathologic Photomicrographs of the Spleens in DMBA-Induced Leukemia mice. White Scale bar, 100 μm. (G) Representative Pathologic Photomicrographs of the bone Marrows in DMBA-Induced Leukemia mice. White Scale bar, 100 μm.
Nrf2 Absence Made Major Organs More Sensitive to DMBA
To compare the responses of WT and Nrf2 Protects Immune System From DMBA damage. (A) Statistical Chart of Changes in the Number of WBCs in Peripheral blood of Mice after Intragastric Administration of DMBA for 3 times in One week. (B) Representative Pathologic Photomicrographs of the bone Marrows in Mice after DMBA Gavage Treatment. White Scale bar, 100 μm. (C) Representative Pathologic Photomicrographs of the Spleens in Mice after DMBA Gavage Treatment. White Scale bar, 100 μm. (D) Representative Pathologic Photomicrographs of the Livers in Mice after DMBA Gavage Treatment. White Scale bar, 100 μm. (*
HSPCs Lacked Stress Response and Were More Prone to DNA Damage for Nrf2 Deficiency
Hematopoietic stem and progenitor cells (HSPCs) are the source of hematopoietic homeostasis, and their damage and genomic instability are important causes of leukemia.17,18 Thus, we suspect that the higher rate of leukemia-like symptoms formation in HSPCs Lack Nrf2 More Prone to DNA damage. (A) Quantification of mRNA Expression for NQO1 in Mouse HSPCs 6 hours after 4 Gy IR. (B) Quantification of mRNA Expression for HO1 in Mouse HSPCs 6 hours after 4 Gy IR. (C) Western Blot Analysis of HO1 and NQO1 Levels in HSPCs 6 hours after 4 Gy IR. (D) Quantification of mRNA Expression for HO1 in Mouse HSPCs Treated with DMBA. (E) Quantification of mRNA Expression for NQO1 in Mouse HSPCs Treated with DMBA. (F) Western Blot Analysis of HO1 and NQO1 Levels in HSPCs Treated with DMBA. (G) Statistical Plot of the Olive Tail Moment Values Measured by Comet Assay on Mouse HSPCs after IR. (H) Statistical Plot of the Olive Tail Moment Values Measured by Comet Assay on Mouse HSPCs after DMBA treatment. (I) Western Blot Analysis of Phosphorylation Levels of ATR and CHK1 in HSPCs after IR. (*
Discussion
Nrf2 plays important roles in the formation of various solid tumors. This regulation of Nrf2 impacts various cellular processes such as redox homeostasis, metabolism, apoptosis, proliferation, proteasome degradation, DNA repair, and mitochondrial physiology, all of which are critical for cancer prevention and treatment strategies.
10
Due to the tumor model, tumor type and tumor progression, the role of Nrf2 in different reports is also different. Overexpression of Nrf2 has been linked to excessive tumor cell proliferation, resistance to radiotherapy and chemotherapy, adaptation to the tumor microenvironment, tumor metastasis, and poorer clinical outcomes.19,20 To investigate the role of Nrf2 in leukemia-like symptoms formation, we used IR and DMBA to establish mouse model, and monitored the occurrence of cancer in
Furthermore, a multitude of studies have underscored the pivotal role of Nrf2 in cellular defense mechanisms. Specifically, Wang et al. have demonstrated that the activation of Nrf2 signaling confers protection against immunosuppression in the spleen and thymus. 21 In Nrf2−/− mice, histological examination of spleen sections revealed a significant disruption of the normal follicular architecture, characterized by the loss of distinct white and red pulp regions. This was attributed to the infiltration of granulocytes and megakaryocytes following treatment with TBI and DMBA. Studies have shown that Nrf2 is critical in controlling the expression of antioxidants in the bone marrow matrix and the sensitivity of these cells to oxidative and electrophilic stress. 22 Our results provide direct evidence that Nrf2−/− mice are highly susceptible to develop myelosuppressive disease. Histological section of decalcified femur from Nrf2−/− mice showed hypocellular bone marrow and the increased apoptosis in myeloid cells was associated with the myeloid hypoplasia in irradiated and DMBA-treated Nrf2−/− mice (Figure 4). This conclusion is further substantiated by previous studies that have demonstrated the hematopoietic system’s heightened sensitivity to irradiation, and the capacity of Nrf2 activators to ameliorate myelosuppression and reduce mortality. 23 Therefore, the apoptosis resulted in increased incidence of bone marrow hematopoietic failure in Nrf2−/− mice.
HSPC transplantation is recognized as an effective therapeutic approach for the treatment of various malignant hematological malignancies, including acute myeloid leukemia (AML), acute lymphoblastic leukemia (ALL), among others. Despite achieving good clinical results, recurrence after allogeneic HSPCs transplantation remains the main challenge at present. 24 Therefore, it is necessary to further improve its therapeutic effect to reduce the recurrence rate after transplantation. HSPCs have the potential for multi-directional differentiation and self-renewal. 25 HSPCs can be affected by both internal regulatory signals and external microenvironment. 26 There are reports that reactive oxygen species (ROS) can affect the biological activity of HSPCs. Under different cell cycle states of HSPCs, ROS levels affect their proliferation, differentiation, and regeneration potential.27,28 Nrf2, as an important oxidative stress regulatory factor, plays an important role in sensing ROS balance. After DMBA and IR treatment, the antioxidant related proteins HO1 and NQO1 of HSPCs in the WT mice were significantly increased to resist the stress microenvironment (Figure 6). However, the deletion of Nrf2 led to the maintenance of HO1 and NQO1 at a low level, which may lead to a poor outcome of HSPCs under stress stimulation (Figure 6). Moreover, the DNA damage response related proteins in HSPCs of Nrf2−/− mice also showed a significant decrease in post radiation stress (Figure 6). These evidences all suggest that NRF2 may maintain the ROS balance of HSPCs by regulating antioxidant related proteins. This may be the same outcome as the ROS balancing effect of β3-adrenergic receptors in hematopoietic stem cell transplantation. 28 Specifically, which specific functions of HSPCs are affected by Nrf2 need further exploration. Although low physiological levels of ROS are necessary for maintaining the function of hematopoietic stem cells, high ROS levels are also a risk factor for poor bone marrow transplantation in experimental and clinical settings. Developing relevant antioxidants can help improve the efficacy of bone marrow transplantation, and Nrf2 may serve as a target for corresponding small molecule agonists.
The research identifies two significant constraints that could be targeted for improvement in future investigative work. Firstly, we preliminarily established a link between Nrf2 and IR/DMBA-induced leukemia-like symptoms in mouse models, further research is needed to thoroughly analyze leukemia types and specific characteristics. Secondly, a larger sample size is needed to validate and extend our results with more and different forms of experiments, excluding factors such as differences in facility environments.
Conclusion
In this study, we observed that Nrf2 deficiency led to more severe damage to the blood and immune systems in mice exposed to IR or DMBA. This increased susceptibility was associated with reduced levels of HO1 and NQO1 proteins and significant DNA damage in HSPCs. Our findings indicate that Nrf2−/− mice are more vulnerable to environmental mutagens such as IR and DMBA, resulting in a higher risk of developing leukemia.
Supplemental Material
Supplemental Material - Nrf2 Deficiency Brings About Increased Sensitive to IR and 7,12-dimethylbenz(a)anthracene and Leukemia Predisposition
Supplemental Material for Nrf2 Deficiency Brings About Increased Sensitive to IR and 7,12-dimethylbenz(a)anthracene and Leukemia Predisposition by Mingxin Dong, Ping Yang, Xinyu Zhang, Shasha Nie, and Xiaohui Sun in Dose-Response
Footnotes
Ethical Statement
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (32201015, 32171239, 82273580), the Natural Science Foundation of Shandong Province (ZR2022QC260).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
