Abstract
Ulcerative colitis (UC) is an inflammatory bowel disease involving chronic and recurring colon inflammation. Current management protocols are limited by adverse effects or short-term symptomatic relief. We aimed to investigate the possible therapeutic prospect of low dose gamma (γ) irradiation or apigenin treatment in acetic acid-induced UC in rats. Induction of UC was carried out by installation of acetic acid intra-rectally. One hour post-induction, rats received a sole dose of γ-radiation (0.5 Gray) or were treated with apigenin (3 mg/kg/day, peroral) for 7 successive days. Antioxidant and anti-inflammatory effects of both agents were assessed via determination of colon malondialdehyde (MDA), reduced glutathione (GSH), total nitrate/nitrite (NOx), mucosal addressin cell adhesion molecule-1 (MAdCAM-1), and interleukin-1beta (IL-1β) contents as well as myeloperoxidase (MPO) activity. Body weight (BW), colon weight/length (W/L) ratio, disease activity index (DAI), and histopathological changes were evaluated. Gamma irradiation and apigenin significantly ameliorated the acetic acid-induced biochemical and histopathological changes. Both therapeutic approaches significantly restored colon contents of the investigated biomarkers. They modulated BW, colon W/L ratio and DAI. This study proposes low dose γ-irradiation as a new therapeutic candidate for the management of UC. We also concluded that apigenin exhibited therapeutic benefits in UC management.
Introduction
Ulcerative colitis (UC) is a bowel disorder associated with chronic inflammation and ulceration of the inner lining of the distal colon and rectum. 1 There is no single collective basic cause for UC; the pathological course is multifactorial, including hereditary, enviromental causes, bacterial flora, and natural body defenses all playing parts.2,3
Due to the restricted comprehension of the exact etiology of UC, management is greatly undetermined depending on targeting the inflammation in preference to any other cause. The majority of the commonly used remedies for UC include administration of 5-aminosalicylic acid, glucocorticoids, and immunosuppressant medication. 4 Despite the fact that numerous kinds of remedies for UC have been suggested and practiced for clinical use, alternative prophylactic and/or curative interventions are required, since many sufferers either show short-term symptomatic relief or experience considerable adverse effects, thus prohibiting the continued use of such remedies.5,6 Considering the crucial role played by exaggerated inflammation and oxidative stress in the UC pathophysiology,7,8 the development of a novel alternative treatment with anti-inflammatory and antioxidant properties has been encountered as an important objective in UC management.
Flavonoids comprise the largest group of secondary metabolic compounds greatly abundant in plants. Numerous reported biological and pharmacological effects have been ascribed to flavonoids including; regulation of cellular proliferation/discrimination along with apoptosis, regulation of inflammation, as well as metastasis progression and angiogenesis.9,10 On account of these actions, flavonoids are regarded as presumptive nutraceuticals that can aid in preventing and/or curing persistent inflammatory cases like inflammatory bowel disease (IBD). 11 The flavone apigenin (4’, 5, 7-trihydroxyflavone) is one of the tea flavonoids abundant in different vegetables, fruits, and herbs. Besides parsley, the greatest genuine sources are rosemary, celery, basil, chamomile, oregano, cloves, thyme, artichokes, liquorice, peppermint, and spinach. 5 Apigenin has gained recognition, owing to its anticarcenogenic 12 and anti-inflammatory 13 actions. In addition, Salehi et al. 14 reviewed other several activities for apigenin including anti-hyperglycemic, 15 antioxidant, 16 and anti-apoptotic in myocardial ischemia. 17 Previous researches have studied the beneficial properties of apigenin against experimental colitis induced by each of dextran sulfate sodium5,18 and acetic acid. 19 Yet, such studies investigated either the protective effects of apigenin against UC or the combined protective and curative effects. Thus, our study aimed to evaluate the potential therapeutic benefits of apigenin rather than its protective ones.
Radiation hormesis is the process by which the biological systems can respond positively, or be activated by biological exposure to low radiation doses that is harmful at high doses. 20 Living organisms respond adaptively to such disturbances in homeostasis induced by low dose levels or dose rates of radiation. They are stimulated to raise their defensive activities by repairing, radioadaptive or protective processes 21 including activation of protein synthesis and DNA repair. 22 The beneficial activities of ionizing radiation have been reported in the management of cancer 23 along with several noncancerous diseases, for example, neurodegenerative diseases, 24 diabetes and diabetic induced complications,25,26 and hypertension. 27
With respect to the gastrointestinal tract, most of the previous radiation literature tended to be dominated by studies of hazardous high doses.28-30 On the other hand, fewer experimental studies provided evidences that low dose radiation is effective against inflammatory diseases along with UC.31-33 As a result, it was of interest to construct our present investigation aiming to assess the possible beneficial actions of low dose gamma irradiation or apigenin as potential therapeutic agents against experimentally-induced UC in rats and comparing each treatment with the commonly used reference drug mesalazine.
Material and Methods
Animals
Adult male albino rats of Wistar strain weighing 200 ± 20 g were purchased from the breeding unit of animals at the National Research Center (Dokki, Giza, Egypt) and kept to adapt for one week before experimental trials at the animal facility of the National Center for Radiation Research and Technology (NCRRT)‐Egyptian Atomic Energy Authority. Rats were retained at 25–28°C with a normal cycle of light and dark. They were provided with a standardized pellet diet (ElNasr chemical company, Abou‐Zaabal, Cairo, Egypt) and allowed free access to water.
Drugs and Chemicals
All drugs and chemicals used were obtained from Sigma‐Aldrich (St Louis, Missouri, USA). Enzyme-linked immunosorbent assay (ELISA) kits specific for rats were used for the determination of mucosal addressin cell adhesion molecule-1 (MAdCAM-1) and interleukin-1beta (IL-1β) and were obtained from Mybiosource®, USA.
Irradiation of Rats
Fully awake rats were exposed to total body gamma radiation at a dose level of 0.5 Gy. 34 Radiation dose was delivered at a 0.43 Gy/min dose rate. Radiation was carried out at the NCRRT by the use of Gamma Cell-40 biological irradiator equipped with a Cesium-137 source, and manufactured by the Atomic Energy of Canada Limited (Sheridan Science and Technology Park, Mississauga, Ontario, Canada).
Induction of Ulcerative Colitis
Twenty four hours before the induction of UC, animals were deprived of food only and allowed free access to water. Subsequently, rats were anesthetized with sodium pentobarbital (30 mg/kg i.p.), 35 followed by gentle insertion of a rubber catheter inside colon (8 cm proximal to the anus). Afterward, slow instillation of 2 mL of acetic acid (3% in normal saline) into colon was carried out. Keeping the animals in a head-down position for 30 seconds was performed to ensure total diffusion of acetic acid in the whole colon and to avoid leakage, then food and water were freely allowed for all of the rats. 36
Experimental Design
Rats were randomly allocated into seven experimental groups; each of 6 rats. Group I (Normal control): rats were given saline for 7 successive days by oral route. Group II (Irradiation): rats were exposed to a single dose of 0.5 Gy gamma radiation. Group III (Apigenin): rats received apigenin (3 mg/kg/day) for 7 successive days by oral route. Rats in groups IV-VII received 2 mL of acetic acid (3% in normal saline) intra-rectally to induce UC, then each group of them was treated individually. Group IV (Colitis) was kept as a positive control group. Group V (Colitis + Irradiation): rats were irradiated at a single dose of 0.5 Gy one hour after induction of UC. Group VI (Colitis + Apigenin): rats were treated orally with apigenin (3 mg/kg/day) for 7 successive days.5,37 Group VII (Colitis + Mesalazine): rats were treated with mesalazine (100 mg/kg/day) for 7 successive days by oral route and served as a reference standard group. 38 Apigenin and mesalazine were dissolved in saline and administered orally, one hour after the induction of UC.
Sample Collection
Sacrifice of rats was performed on day 8 by cervical dislocation under urethane anesthesia. The distal colon was rapidly excised, flushed with ice-cold saline and dried on a filter paper. Then the weight and length of colon were recorded to calculate colon weight/length (W/L) ratio. The tissues of colon were divided into two parts. The initial part was reserved in buffered formalin (10% v/v) for histopathological assessment; the second portion was weighed, split into pieces in a saline bath that is ice-cold and then maintained at – 80°C to be utilized for the evaluation of the different biochemical markers.
Body Weight of Rats
Body weights were recorded daily and the mean body weight (BW) was calculated for each experimental group.
Colon Weight/Length Ratio
The weight and length of colon were measured and the ratio between them (W/L) was calculated. 39
Disease Activity Index
Calculation Criteria of Disease Activity Index.

Biochemical Assessments
Determination of Biomarkers of Oxidative Stress
Colon malondialdehyde (MDA) content was measured spectrophotometrically using a double beam spectrophotometer (Spectro UV-VIS double beam, UVD 2950, Labomed, Inc. USA) at a wavelength of 535 nm, as reported by the method of Mihara and Uchiyama. 41 Glutathione (GSH) content was evaluated in the colon homogenate spectrophotometerically based on the method of Beutler et al, 42 at 412 nm. Total nitrate/nitrite (NOx, a marker for synthesis of nitric oxide) was determined in the colon colorimetrically at 540 nm as stated by Miranda et al. 43
Determination of Colon Myeloperoxidase Activity
Colon activity of myeloperoxidase (MPO) was determined spectrophotometrically at 460 nm in accordance with the method of Bradley et al. 44
Determination of Colon Mucosal Addressin Cell Adhesion Molecule-1 and Interleukin-1beta
Colon content of MAdCAM-1 was measured using ELISA kit (Mybiosource, USA, Catalog number: MBS727604) specific for rats, based on the instructions of the manufacturer. Colon content of IL-1β was measured using ELISA kit (Mybiosource, USA, Catalog number: MBS825017) specific for rats, based on the instructions of the manufacturer.
Histopathological Examination
Samples of tissue were collected from colon and fixed in formalin 10%, stripped, cleaned, and then undergone dehydration by alcohol. The dehydrated samples were afterward cleaned in xylene, implanted in paraffin blocks, and dissected at 4–6 µm thickness. The acquired sections of tissues were deparaffinized via xylol and then Hematoxylin and Eosin (H&E) were used as stains for histopathological examination by the electric light microscope based on the method of Bancroft et al. 45
Statistical Analysis
The values were all expressed as means ± standard error of the mean (SE). Statistical analysis was performed by the use of one-way analysis of variance (ANOVA) then subsequently multiple comparison test of Tukey-Kramer. The significance level for the entire statistical tests was set at p < 0.05. Statistical analysis was accomplished by utilization of GraphPad Prism® software package, version 6 (GraphPad Software Inc., USA).
Results
Body Weight
Inducing UC gave rise to a remarkable decrease in BW of rats by 31%, as compared to control group. Rats irradiated at a single dose of 0.5 Gy, as well as those treated with either apigenin (3 mg/kg) or mesalazine (100 mg/kg) showed almost control values of BW that also amounted to the value of mesalazine-treated group. Meanwhile, the colitis groups treated with low dose radiation, apigenin, and mesalazine showed elevated BW values by 45%, 41%, and 42%, respectively, when compared to colitis group (Figure 1). Effects of low dose gamma irradiation (0.5 Gy), apigenin (3 mg/kg), and mesalazine (100 mg/kg) on body weight (BW) in acetic acid-induced ulcerative colitic rats. Statistical analysis was performed using one-way ANOVA followed by Tukey-Kramer multiple comparisons test. Values are expressed as mean ± SE (n = 6). *Significantly different from control group at P ≤ 0.05. #Significantly different from colitis group at P ≤ 0.05.
Colon Weight/Length Ratio
Using acetic acid to induce UC produced notable increase in colon W/L ratio by 158% compared to control group. Rats irradiated at a single dose of 0.5 Gy, as well as groups of rats treated with either apigenin or mesalazine revealed a pronounced reduction in colon W/L ratio by 60%, 58%, and 58%, respectively, when compared to colitis group. The W/L ratio was also normalized by the three treatment agents. The W/L ratio of groups of rats treated with a single dose of 0.5 Gy or apigenin did not show any change as compared to mesalazine-treated group (Figure 2). Effects of low dose gamma irradiation (0.5 Gy), apigenin (3 mg/kg), and mesalazine (100 mg/kg) on colon weight/colon length (W/L) ratio in acetic acid-induced ulcerative colitic rats. Values are expressed as mean ± SE (n = 6). *Significantly different from control group at P ≤ 0.05. #Significantly different from colitis group at P ≤ 0.05. @Significantly different from colitis + mesalazine group at P ≤ 0.05.
Disease Activity Index
Rats irradiated at a single dose of 0.5 Gy, as well as those treated with either apigenin or mesalazine, all showed normalization of DAI. They also showed significantly reduced DAI by 85%, 73%, and 77%, respectively, as compared to the colitis group. There was no significant difference in the DAI between rats irradiated at a single dose of 0.5 Gy, or those treated with apigenin or mesalazine (Figure 3). Effects of low dose gamma irradiation (0.5 Gy), apigenin (3 mg/kg), and mesalazine (100 mg/kg) on disease activity index (DAI) in acetic acid-induced ulcerative colitic rats. Values are expressed as mean ± SE (n = 6). *Significantly different from control group at P ≤ 0.05. #Significantly different from colitis group at P ≤ 0.05.
Oxidative Stress Biomarkers
Acetic acid administration caused a marked oxidative stress in colon tissue, as revealed by the 4- and 3-fold increase in colon contents of MDA and NOx, respectively, when compared to control group. Furthermore, colon GSH content was markedly reduced by 48%. Irradiation of rats resulted in a significant drop in colon contents of MDA and NOx by 74% and 73%, respectively, while it induced a marked rise in colon GSH content by 90% when compared to colitis group. Treatment with apigenin significantly decreased colon MDA and NOx contents by 51% and 43%, respectively, while it raised colon GSH content by 85%, compared to colitis group. In addition, treatment with mesalazine significantly decreased colon MDA and NOx contents by 62% and 55%, respectively. Moreover, colon GSH content significantly increased by 79% as compared to colitis group. Irradiation induced a marked reduction in colon MDA and NOx contents by 32% and 39%, respectively, when compared to mesalazine-treated group. Furthermore, treatment of colitic rats with apigenin significantly decreased colon MDA and NOx contents by 28% and 26%, respectively, as compared to mesalazine-treated group (Figures 4(A)‐4(C)). Effects of low dose gamma irradiation (0.5 Gy), apigenin (3 mg/kg), and mesalazine (100 mg/kg) on colon malondaialdehyde (MDA); (A), glutathione (GSH); (B), total nitrite/nitrate (NOx); (C) in acetic acid-induced ulcerative colitic rats. Values are expressed as mean ± SE (n = 6). *Significantly different from control group at P ≤ 0.05. #Significantly different from colitis group at P ≤ 0.05. @Significantly different from colitis + mesalazine group at P ≤ 0.05.
Myeloperoxidase Activity
Induction of UC by administration of acetic acid resulted in more than 2-fold rise in MPO activity when compared to control group. Exposure to a single radiation dose of 0.5 Gy, as well as treatment with either apigenin or mesalazine, markedly reduced colon MPO activity by 72%, 54%, and 63%, respectively, when compared to colitis group. Irradiation of rats also led to normalization of MPO activty. Furthermore, it significantly reduced colon MPO activity by 24%, as compared to mesalazine-treated group (Figure 5). Effects of low dose gamma irradiation (0.5 Gy), apigenin (3 mg/kg), and mesalazine (100 mg/kg) on colon myeloperoxidase (MPO) activity in acetic acid-induced ulcerative colitic rats. Values are expressed as mean ± SE (n = 6). *Significantly different from control group at P ≤ 0.05. #Significantly different from colitis group at P ≤ 0.05. @Significantly different from colitis + mesalazine group at P ≤ 0.05.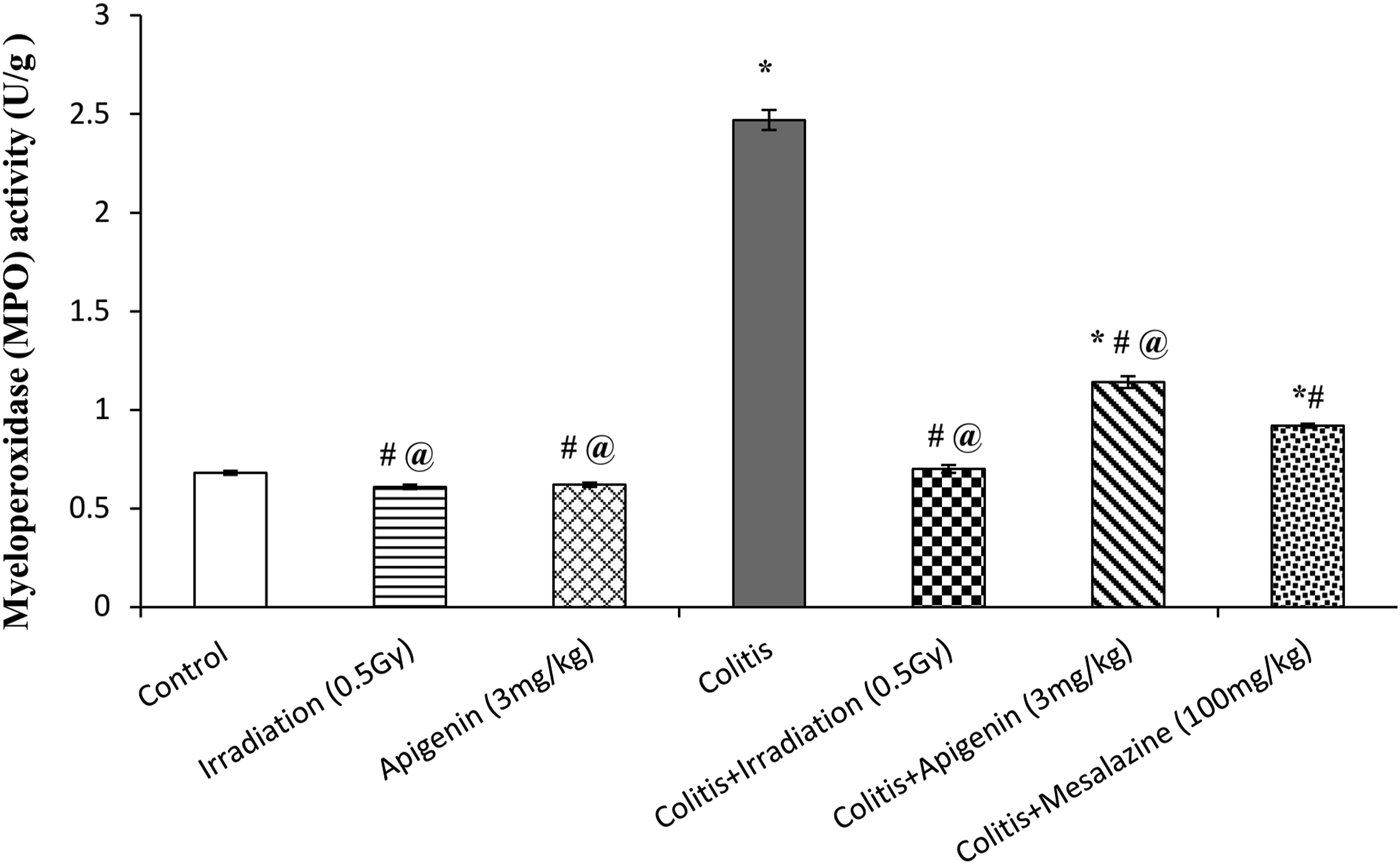
Mucosal Addressin Cell Adhesion Molecule-1
Induction of UC caused a marked rise in colon content of MAdCAM-1 by 141% as compared to control group. Irradiation of rats as well as treatment with either apigenin or mesalazine significantly decreased colon MAdCAM-1content by 50%, 31% and 37%, respectively, when compared to colitis group. Moreover, irradiation of rats significantly decreased colon MAdCAM-1 content by 21%, as compared to mesalazine-treated group (Figure 6). Effects of low dose gamma irradiation (0.5 Gy), apigenin (3 mg/kg), and mesalazine (100 mg/kg) on colon mucosal addressin cell adhesion molecule-1 (MAdCAM-1) content in acetic acid-induced ulcerative colitic rats. Values are expressed as mean ± SE (n = 6). *Significantly different from control group at P ≤ 0.05. #Significantly different from colitis group at P ≤ 0.05. @Significantly different from colitis + mesalazine group at P ≤ 0.05.
Interleukin-1beta
Induction of UC by acetic acid administration produced a pronounced rise in colon content of IL-1β by 206% as compared to control group. Irradiation of rats as well as their treatment with either apigenin or mesalazine induced a marked decrease in colon IL-1β content by 61%, 26%, and 32%, respectively, as compared to colitis group. Furthermore, it significantly decreased colon IL-1β content by 42%, when compared to mesalazine-treated group (Figure 7). Effects of low dose gamma irradiation (0.5 Gy), apigenin (3 mg/kg), and mesalazine (100 mg/kg) on colon interleukin-1β (IL-1β) content in acetic acid-induced ulcerative colitic rats. Values are expressed as mean ± SE (n = 6). *Significantly different from control group at P ≤ 0.05. #Significantly different from colitis group at P ≤ 0.05. @Significantly different from colitis + mesalazine group at P ≤ 0.05.
With the exception of colon GSH, there were no notable differences observed in the majority of the assessed markers between normal (control group) animals and those either irradiated or treated with apigenin. Such findings support the safety presumption of the individual treatment of rats with either apigenin or gamma radiation at a dose of 0.5 Gy.
As for the histopathological findings, the ulcerative damage induced by acetic acid in the colitis group was significantly improved by low dose gamma irradiation as well as treatment with apigenin (Fig. 8). Photomicrograph of colonal mucosa in different experimental groups. (A) Control rats showing normal histological structure of both surface epithelial (arrow head) and glands (arrow). (B) Rats irradiated at a single dose of 0.5 Gy showing normal histological structure of both surface epithelial (arrow head) and glands (arrow). (C) Rats treated with apigenin (3 mg/kg) showing normal histological structure of both surface epithelial (arrow head) and glands (arrow). (D) Colitis group rats showing focal ulcerative area with subepithelial leukocytic infiltration (arrow). (E) Colitis rats irradiated at a single dose of 0.5 Gy showing slight goblet cells depletion and mild edema (arrow). (F) Colitis rats treated with apigenin (3 mg/kg) showing focal epithelial sloughing and inflammatory cells infiltration of muscularis mucosa (arrow). (G) Colitis rats treated with mesalazine (100 mg/kg) showing focal damage of surface epithelium and focal submucosal edema (arrow). (H&E X200).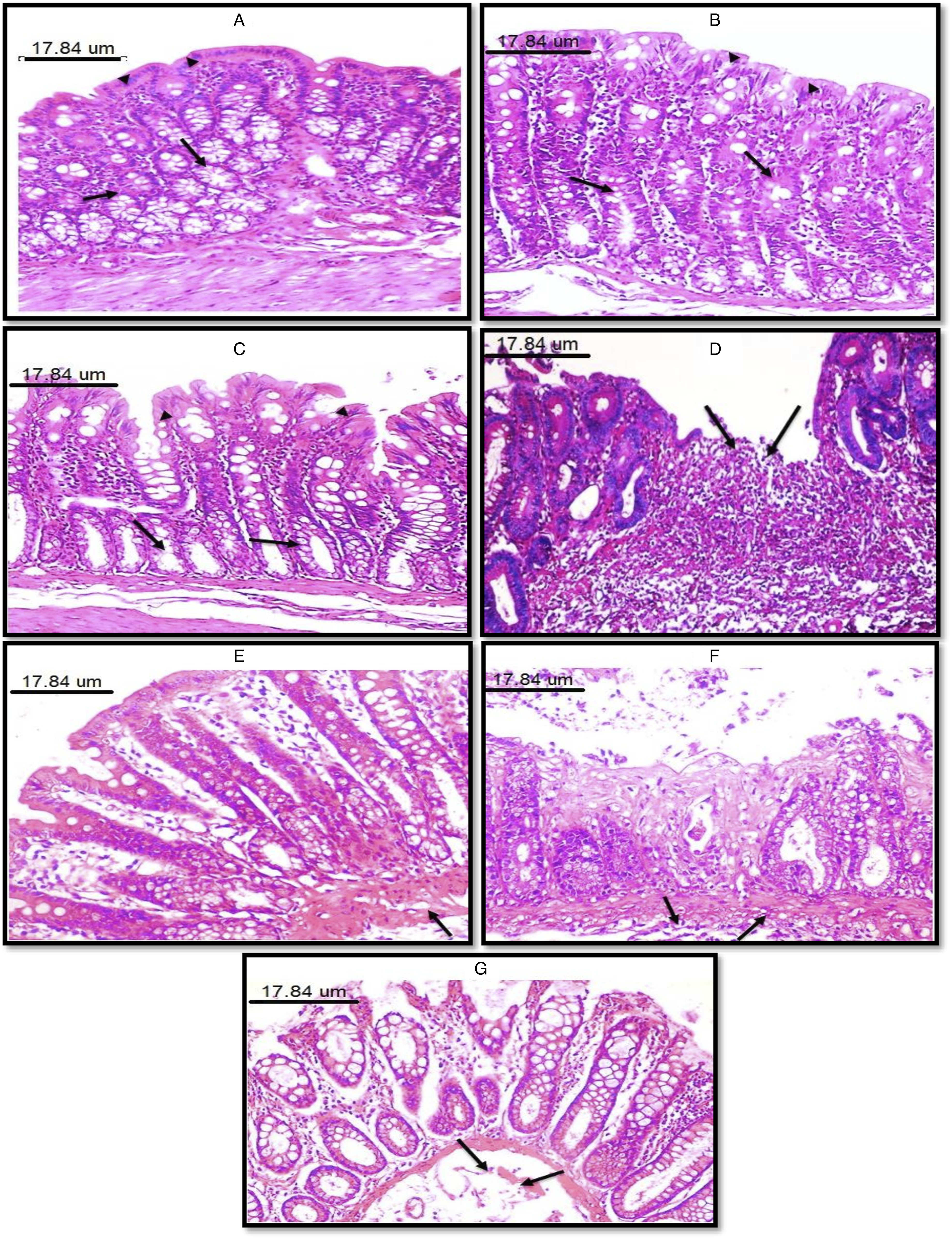
Discussion
Induction of UC by acetic acid has been approved to be an effective UC animal model to investigate the inflammatory process that influences the damage of colon mucosa, rise in inflammatory cytokines and mediators as well as the gross and microscopic histological changes of the colon.36,46 Administration of acetic acid in the present study reduced animals BW, a finding which is in harmony with the previous research of Minaiyan et al. 47 Such loss of weight could be as a consequence of decreased appetite and ingestion of food. Furthermore, the rise in colon W/L and DAI observed in the colitis group animals, are on the same line with the preceding studies of Guerra et al. 39 and Gupta et al. 48
Irradiation of rats or treatment with apigenin induced a considerable rise in BW and a substantial drop in both the colon W/L and DAI of colitic rats. Regarding irradiation, preceding reports have provided appreciable verification from experimental investigations. Nishiyama et al. 49 reported that radon inhalation suppressed the rise in colon DAI and the histological damage induced by experimental UC in mice. In addition, Kojima et al. 33 showed that ingestion of radon-containing water induced stepwise amelioration of UC-induced bleeding in humans. With respect to apigenin, our results are in accordance with those of Jia et al. 50 and Ai et al. 51 that showed apigenin ability to ameliorate weight loss, lower DAI value, and inhibit colon shortening in dextran sodium sulfate-induced UC model.
The current investigation showed a marked oxidative stress induced by UC as demonstrated by the marked rise in colon MDA and NOx contents as well as the significant reduction of colon GSH content. These findings could be attributed to the influence of acetic acid on the redox balance of the colon by causing lipid peroxidation, inhibiting the antioxidant activity of enzymes such as superoxide dismutase and glutathione peroxidase as well as diminishing non-enzymatic antioxidants.52,53 The present work elucidated the antioxidant magnitude of gamma irradiation at low doses. It displayed that low dose gamma irradiation, downgraded the acetic acid-induced oxidative alterations; that is, the promotion of colon contents of MDA and NOx along with the deficiency in colon GSH content. Such observations could be comparable to the earlier study of Avti et al. 34 who pointed out that irradiation of male balb/c mice at a dose of 0.5 Gy was able to induce liver endogenous GSH. The observed rise in colon GSH content might be attributed to the reported rise in mRNA expression for γ-glutamyl cysteine synthase which is a rate limiting enzyme in the production of GSH. 54 Moreover, Kojima et al. 55 reported that, total body gamma-ray irradiation at 0.5 Gy, triggered the immunological processes by inducing GSH in mice splenocytes. Treatment of animals with apigenin significantly reduced colon MDA and NOx while it raised GSH colon content. On the same line, Ganjare et al. 56 previously reported that apigenin ameliorated the production of reactive oxygen species evoked by acetic acid, hindered the free radicals production and restored the redox state of the colonic mucosa. The earlier reported antioxidant, free radical scavenger and metal ion chelating activities of apigenin, might account for such results.16,57
Myeloperoxidase (MPO) is an enzyme excreted copiously in neutrophils. It is a widely known biological marker of diffusion of neutrophils that is used for evaluation of the inflammatory process in colitis.58,59 The current study showed that acetic acid-induced oxidative damage was accompanied by a marked rise in colon MPO activity. The results observed in the present study demonstrated that apigenin had anti- inflammatory effect as evidenced by significantly decreased colon content of IL-1β and colon MPO activity. This is in harmony with the earlier report of Mascaraque et al. 5 who showed that apigenin was capable of reducing colonic MPO activity in colitis models induced by trinitrobenzenesulfonic acid and dextran sulfate sodium in rat. On the same line, the administration of apigenin was reported to effectively attenuate neutrophil infiltration as revealed by the suppression of colonic MPO following induction of colitis by acetic acid 56 and dextran sulfate sodium 51 in rats.
Leukocyte-endothelial interactions taking place in the intestine are essentially relying on MAdCAM-1, which is an endolthelial adhesion molecule conveyed on endothelial cells inside the lymph nodes of the mesentery and the lamina propria of both large and small bowel. MAdCAM-1 is normally expressed in the colon, and its expression is markedly enhanced throughout the process of inflammation.60,61 In the current study, induction of UC resulted in significant rise of colon MAdCAM-1 content. These results are in accordance with preceding studies showing that MAdCAM-1 expression is distinctly amplified in experimental UC.62,63 Our results showed that low dose γ-irradiation induced normalization of the colon content of MAdCAM-1. This finding was previously reviewed by Arenas et al. 32 who reported that decreased expression of adhesion molecules such as E-selectins, vascular cellular adhesion molecule-1 (VCAM-1), intracellular adhesion molecule-1 (ICAM-1), was among the proposed hypotheses to explain the anti-inflammatory action of radiotherapy using low doses. Treatment with apigenin in the current investigation induced a pronounced reduction in colonic content of MAdCAM-1. This observation is consistent with the previous research of Lee et al. 64 that showed apigenin ability to inhibit cyclooxygenase-2 (COX-2) enzyme and adherence of monocytes to the endothelium of human umbilical vein, via down-regulation of cellular adhesion molecules such as ICAM-1 and VCAM-1.
In the current work, a marked rise in colon content of IL-1β in colitis group was observed. This observation is in agreement with the previous experimental reports of Tahan et al. 65 and Bastaki et al. 66 They showed that acetic acid-induced the release of interleukin-1 and other pro-inflammatory cytokines in colonic mucosa of colitic rats. Our results revealed that low dose γ-irradiation exerted a prominent anti-inflammatory action as revealed by bringing the colon content of IL-1β almost to the normal value. This outcome is comparable to the study of Schaue et al. 67 who reported that irradiation of mice at a dose of 0.5 Gy attenuated the inflammatory reactions induced by carrageenan air pouch model as revealed by the marked reduction in exudate content of IL-1β. The ability of apigenin to suppress the induction of NO-synthase and COX-2 enzymes in macrophages, via lipopolysaccharide influence, might account for such anti-inflammatory activity.19,68 The perceived ability of low dose γ-irradiation or apigenin to attenuate the UC-induced by acetic acid was also supported by the histopathological findings. These findings might be ascribed to the anti-inflammatory and antioxidant effects exerted by both apigenin and low dose γ-irradiation.
Conclusion
Based on the above detailed observations, it could be concluded that each of low dose gamma irradiation or apigenin offered promising effectiveness in the management of experimental UC. Both agents showed significant efficacy through modifying the oxidative stress and the inflammatory components of UC pathogenesis.
Footnotes
Acknowledgments
The authors would like to acknowledge Prof. Dr. Ahmed Othman; Professor of Histopathology, Faculty of Veterinary Medicine, Cairo University for his efforts in accomplishing the histopathological tissue examinations of the current study.
Authors’ Contributions
EE Shibrya: Carried out the experiment, data analysis, writing the original draft.
RR Rashed: Data analysis, in addition to revising the manuscript.
MA Abd El Fattah: Helped in supervising the research.
MA El-Ghazaly: Contributed to supervising and directing the research, in addition to revising the manuscript.
SA Kenawy: Supervised the research and contributed to revising the manuscript.
All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
All of the experimental processes were carried out in accordance with the guidelines settled by the European Economic Community (EEC) regulations (revised Directive 86/609/ EEC) and were accredited by the Ethics Committee of the Faculty of Pharmacy, Cairo University, Egypt (permit number PT 2197, 28/5/2018).
