Abstract
Objective
To determine the effect of the autophagy inhibitor, 3-methyladenine (3-MA), on cognitive function changes, microglia activity, neuronal apoptosis, and inflammation in rats following radiation-induced brain injury.
Methods
The following groups were established: control, model, and 3-MA. A rat model of radiation-induced brain injury was generated with a medium dose of X-rays. A Morris water maze was used to observe the cognitive function of the rats. H&E staining was used to observe the pathological changes in the hippocampus. The morphological and quantitative changes of neuronal nuclear (NeuN)-positive neurons and Iba-1-positive microglia in the ipsilateral hippocampus were analyzed by immunohistochemistry. Western blot analysis was done to measure the changes of NeuN ionized calcium binding adapter molecule 1(Iba-1) and apoptosis-related proteins. Immunofluorescence staining of Iba-1 and Microtuble-associated protein light chain 3 (LC3) was done to evaluate the changes in microglia autophagy. TUNEL staining was used to detect apoptosis in the hippocampus. Enzyme-Linked Immunosorbent Assay was used to detect the levels of TNF-α and IL-6 as a measure of the inflammatory response in the hippocampus.
Results
After irradiation, the nucleus of the neurons in the hippocampus was constricted, the pyramidal tract structure was disordered, neuronal apoptosis was increased (P < .001), the expression of microglia increased (P < .01), autophagy was increased (P < .05), and conversion of microglia to the M2 type increased (P < .05). After 3-MA administration, the level of autophagy decreased (P < .05), the damage to the hippocampal region was reduced, neuronal apoptosis decreased (P < .01), and the activity of the microglia decreased (P < .01).
Conclusion
Radiation can active the Microglia. 3-MA inhibits autophagy and excessive activity in microglia, and promotes the conversion of microglia from the M1 to the M2 type, thereby promoting the recovery of brain tissue following radiation exposure.
Introduction
At present, there are not many treatment options for brain tumors. Radiotherapy is a common treatment that is applied to the affected location. However, radiotherapy also has significant side effects, especially for the nervous system, in which cognitive dysfunction may occur. Studies show that direct and indirect damage to the hippocampus by radiation can affect cognitive function. The dose of radiation has noticeable effects on the appearance of these complications. There is often an incubation period and continuous damage to the brain that occurs over time after radiotherapy, so cognitive impairment is a severe complication that can destroy a patient’s quality of life.1,2 Radiation may have a direct or indirect influence on blood vessels, glial cells, and surrounding tissues, resulting in persistent, and irreversible damage to the hippocampus, which ultimately causes memory disorders and cognitive dysfunction. The early effects of radiation exposure occur within a few days of exposure and typically include damage to the body’s hematopoiesis. However, its delayed response generally involves damage to the lung, skin, heart, brain, and kidney, and may occur over months or years. With respect to brain injury, radiation can damage neurons, neuroglia, and blood vessels of the brain, whereas small colloidal cells are found throughout the brain tissue, which are innate immune cells of the nervous system that are involved in brain tissue damage and repair.3-5
Autophagy is a metabolic pathway that is widespread in eukaryotic cells involving more than 40 proteins. 6 This unique process functions in eukaryotic cells 7 to eliminate intracellular metabolic waste through the lysosomal pathway and stabilize the homeostasis of the intracellular environment. However, excessive activation of autophagy can accelerate apoptosis in nerve cells. 8 Thus, the appropriate inhibition of autophagy may delay the progression of some diseases. Some research shows that irradiation increased autophagy flux in GL261 glioma cells as evidenced by increased levels of LC3-II but decreased levels of p62 (SQSTM1). Indicating that autophagy plays a cytoprotective role in glioma cell survival. 9 According to this study, changes in autophagy are closely associated with the onset and development of Alzheimer disease (AD), Parkinson’s disease (PD), and other diseases. Regulating autophagy can effectively remove abnormally accumulated proteins in the nervous system and alleviate disease progression at the cellular level. Radiotherapy produces an inflammatory response in the brain following acute injury, with microglia and autophagy involved. Type 2 A lysosomal associated membrane protein (LAMP2A) is a marker of molecular chaperone-mediated autophagy (CMA). During the repair of brain injury, LAMP2A is significantly increased in the microglia, indicating that CMA is associated with microglial activation. 10 This results in the production of diversified factors, including IL-6 and IL-1β, which promote inflammation and inhibit the replenishment of hippocampal neurons. Moreover, it inhibits the synthesis of neurons, regulates the expression of the Arc gene in the hippocampal region, and mediates the emergence of cognitive dysfunction. 11 Some scholars promote the transformation of M1 type to M2 type by interfering with the activity of microglia, which can promote the recovery of inflammation and promote the recovery of brain tissue. 12
Currently, studies on microglia autophagy outnumber those on the acute phase of radiation brain injury, and its underlying mechanism is unclear. Accordingly, we established a rat model of radiation-induced brain injury using medium dose X-ray exposure. Rats were injected with 600 nmol of 3-MA into the lateral ventricle. A Morris water maze experiment was used to test the effects of 3-MA on cognitive dysfunction, such as spatial learning and memory in RBI rats. H&E staining was used to observe the pathological changes in the ipsilateral hippocampus. The morphological and quantitative changes of positive neuronal nuclear (NeuN) neurons and Iba-1 microglia in the ipsilateral hippocampus were evaluated by immunohistochemistry. Immunoblot analysis was used to observe the changes of NeuN, Iba-1, Arg-1, and the apoptosis-related proteins, Bcl-2 and Bax. The changes in autophagy of microglia were evaluated by double fluorescence staining with Iba-1 and LC3. The inflammatory response was evaluated in the hippocampus by measuring the levels of TNF-α and IL-6 by Enzyme-Linked Immunosorbent Assay (ELISA). Apoptosis of the hippocampal neurons was detected by TUNEL. Overall, this study was aimed to explore the effect of 3-MA on RBI damage and its potential mechanism.
Methods
The rats were adaptively fed for 1 week before the experiment and were randomly divided into three groups: a control group, model group, and 3-methyladenine (3-MA) group, with 12 animals in each group. The RBI model established by administering single whole brain irradiation of 20 Gy. All rats were irradiated after fasting for 12 hours and were anesthetized with 5% chloral hydrate at 6 mL/kg and placed on the radiotherapy table for less than 5 min. The rats in the Sham group were fasted and given anesthesia, but no irradiation. Following the completion of irradiation, the rats were immediately fixed on a stereotaxic instrument, their scalps were cut open, skulls exposed, the surface tissue was digested with H2O2, and the coordinates of the lateral ventricle of the rats were located with the aid of stereotactic instrument. In the model group, 5 μL of .9% NaCl was injected with a microinjector. For the 3-MA group, 600 nmol of 3-MA was injected (.9% NaCl diluted to 5 μL in volume), the scalps of rats were cut open. No drugs were injected into the rats of the control group. We continued feeding the rats for 8 weeks.
Analysis of learning and memory ability were tested using a Morris water maze. 13 The temperature in the laboratory was 22°C ± 2°C, the water temperature was 22°C, and the roof and walls were smooth and tidy. The maze was covered with a black shading cloth to avoid light, and the lighting equipment was located directly above the water maze and consisted of soft lighting. Rats in all groups were trained for 4 days prior to irradiation. (1) Positioning navigation experiment: The researcher placed the rats facing the pool wall in the middle of the 3 quadrants outside the platform and recorded the movement trajectory of the rats and the time they required to climb onto the platform within 120 s (escape latency). After climbing the platform, the rats were on their feet for 30 s. Then, the rats were returned to the cage for 60 s. If the rats did not find the platform within 120 s, they were towed to it where they remained for 30 s. The escape latency was recorded as 120 s and the rats were put back into the cage to rest for 60 s. The rats were trained 4 times a day in the morning and afternoon for 4 days; (2) Space exploration experiment: The next day after the positioning navigation experiment was done, the platform was removed, and the number of rats passing through the platform within 120 seconds was recorded. All rats were subjected to the water maze test again before 8 weeks of treatment. The conditions and operation were the same as above, and the learning and memory abilities of the rats were recorded.
Results
Morris Water Maze
Eight weeks after RBI, the trajectories of the rats in each group were recorded (Figure 1). Table 1 contains the data analysis instructions for the positioning navigation test for each group. Eight weeks after RBI in each group, the Morris water maze positioning navigation swimming trajectory is shown. A Sham 组 B Model 组 C 3-MA 组. Comparison of escape latency of positioning navigation test in four days in each group. Table lvs Sham. aP <.05. bP <.01; vs Model *P < . 05. cP < .01.

For D1 and D2, compared with the sham group, there was no statistical difference (P > .05), therefore, radiation did not have a relevant effect on memory function in rats. For D3, the escape latency of the rats in the model and 3-MA groups was significantly (P < .05) longer compared with that of the Sham group. However, in the 3-MA group, it was shorter compared with that of the Model group and there was a statistically significant difference (P < .01). After 8 weeks of RBI, the incubation period of the rats searching for the underwater platform was significantly prolonged, suggesting a certain learning and memory dysfunction after radiotherapy. After administration of 3-MA, the escape latency of the rats was significantly shorter compared with the Model group and the learning and memory ability was restored to some extent. The results showed that cognitive and memory impairment started on the third day.
Space Exploration Test
Comparison of the number of crossing platforms in space exploration experiments of rats in each group.

Table 2vs Sham.
aP < .05.
bP < .01 vs Model.
cP < .05.
dP < .01.
H & E Staining
The model group showed more obvious nuclear pyknosis and pyramidal tract disorder. The change in the 3-MA group was smaller compared with that of the model group. After the rats experienced radiation injury, there was evident damage appearing in the hippocampus and nuclear pyknosis was observed in the nerve cells, which was alleviated by 3-MA treatment (Figure 2). H & E staining results for the ipsilateral hippocampus (×200).
TUNEL Staining
TUNEL staining was used to evaluate apoptosis in the injured hippocampus. Compared with the Sham group, there were morphological changes and a large number of positive TUNEL cells in the model group. The positive TUNEL cells of the 3-MA group were decreased significantly compared with the model group. Therefore, after suffering radiation injury, the hippocampus of the rats was clearly damaged, which caused neuronal apoptosis, and 3-MA treatment reduced the level of apoptosis (Figure 3). Terminal deoxyribonucleotidyl transferase-mediated deoxyuridine 50-triphosphate nick-end labeling (TUNEL) staining of the ipsilateral hippocampal region. Arrows indicate TUNEL-positive cells. (Magnification ×400) Mean SD. n = 3. ***P < .001 , **P < .01 vs. Model group.
Immunohistochemistry
After radiation, neurons may be damaged directly or through the activity of microglia. The level of NeuN decreased significantly, whereas the level of Iba-1 increased. After 3-MA treatment, the level of NeuN increased significantly, whereas the level of Iba-1 decreased. Therefore, radiation activated microglia and damaged neurons simultaneously, whereas 3-MA inhibited the excessive activation of microglia and promoted the recovery of neurons (Figure 4). IHC staining of NeuN and Iba-18 weeks after RBI in the 3 groups (×400) **P < .01 ***P < .001 (Magnification ×400).
Immunofluorescence Staining
Some positive LC3 cells were observed in the Sham group by immunofluorescence, suggesting autophagy activity in the microglia of normal brain tissue. Eight weeks after RBI, LC3 expression in the model group was significantly up-regulated compared with that in the Sham group. The expression of LC3 in the 3-MA group was lower compared with that in the model group. RBI activated microglia autophagy, whereas 3-MA treatment significantly reduced the level of microglia autophagy, thus inhibiting the activity of the microglia (Figure 5). Immunohistochemical staining of ionized calcium-binding adapter molecule 1 (Iba-1) in the ipsilateral hippocampus. **P < .01; ***P < .001 (Magnification ×400).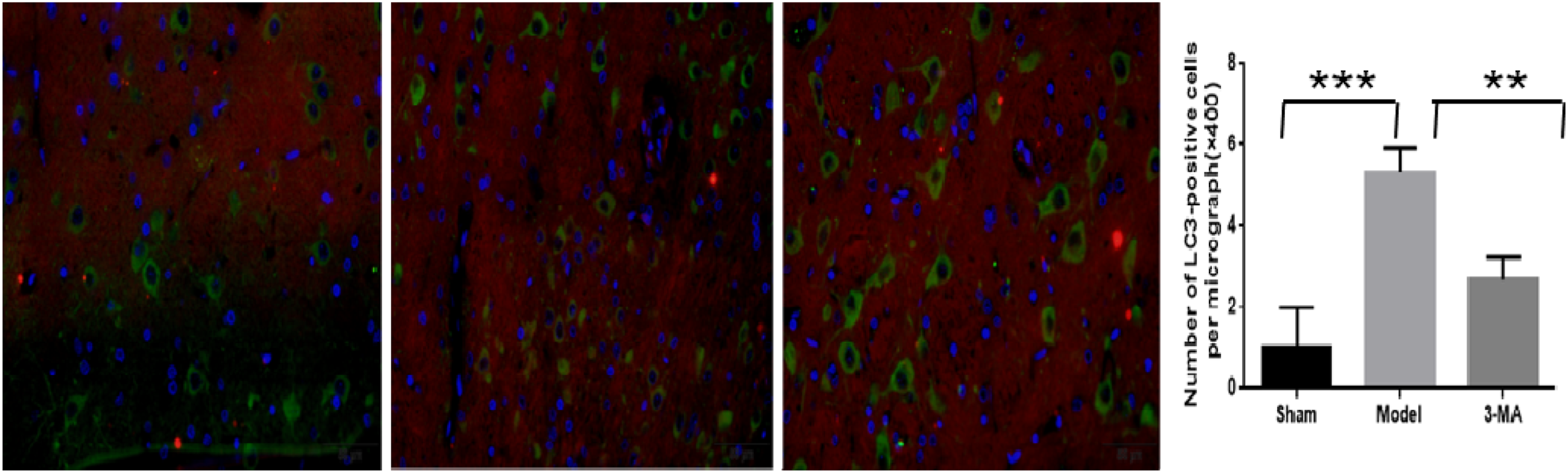
Western Blot Analysis
We measured inflammatory factors and a marker of microglia, then evaluated the effect of 3-MA on the activity of microglia and apoptosis in neurons using western blot analysis. Radiation can activate microglia, whereas 3-MA can promote the recovery of brain tissue (Figure 6). Western blot of NeuN, Bcl-2, Bax, Iba-1, and Arg-1 of the ipsilateral hippocampus. Data in the bar graph represent the mean ± SE. β-Actin was used as a loading control. *P < .05 **P < .01 ***P < .001.
Next, we determined the molecular mechanism of RT-induced cell apoptosis. We measured the classical mitochondria-associated pro- and anti-apoptotic proteins, Bax and Bcl-2, involved in RD-induced cell apoptosis. In the radiation exposed group, there were higher levels of Iba-1and Bax, whereas there were lower levels of NeuN and Bcl-2. Radiation activated the microglia and promoted inflammation. In the 3-MA group, 3-MA inhibited the expression of Bax and Iba-1, promoted the repair of neurons, and decreased the activity of microglia. These results suggest that 3-MA can protect the brain tissue after radiation by attenuating the activity of microglia (Figure 6).
Microglia may be divided into an M1 and M2 type. The M2 type can protect brain tissue. Arg-1 is a marker of M2 type microglia. Radiation can increase the expression of Arg-1, thus microglia can protect themselves in a harsh environment. The 3-MA group exhibited a higher level of Arg-1, indicating that 3-MA can promote the transformation of microglia into the M2 type (Figure 6).
ELISA
TNF-α and interleukin are important signaling molecules involved in the inflammatory response and they are significantly increased in the early stage of brain injury. To determine the changes in the inflammatory response, we measured the levels of TNF-α and IL-6. After 8 weeks of radiation, although the expression of TNF-α and IL-6 increased, there was no significant difference (P > .05). Although the expression of these factors decreased in the 3-MA group, it was also not significant (P > .05) (Figure 7). Therefore, the inflammatory response in the brain tissue disappeared after 8 weeks. Enzyme-linked immunosorbent assay of (A) tumor necrosis factor-a (TNF-a) and (B) interleukin-6 (IL-6) in the ipsilateral hippocampus. P > .05.
Discussion
In this study, we established a rat brain injury model by exposure to medium dose radiation. After radiation, the microglia was activated and autophagy in the microglia increased. The 3-MA-treated group showed a significant decrease in microglia activity, promoted microglia to M2 differentiation, and inhibited neuronal apoptosis. These data indicate that the inhibition of microglia autophagy may be a new treatment strategy for brain injury caused by radiation.
Establishment of RBI Model and Selection of Radiation Dose
The establishment of various rat models of radiation-induced brain injury has been reported, and 10–30 Gy is often used at different exposures. The dose is primarily associated with the variety, body weight, and research purpose of the rats. In addition, there are lots of radiation sources, such as X-rays and γ-rays. As early as 1991, Delapaz et al 14 established a rat model of radiation-induced brain injury using X-rays for the first time and observed corresponding changes in the brain tissue. Based on the literature, we irradiated the whole brain with X-rays of 10 Gy, 20 Gy, and 30 Gy, the total dose was offered in 2 minutes. The rats were fasted and provided intraperitoneal anesthesia before irradiation, covering the eyes to avoid damage and blindness. After irradiation, the weight, and hair of the rats were recorded. One week later, H&E staining was performed in the hippocampus of the brain. We found that there was no obvious damage in brain tissue in the 10 Gy irradiation group. There was no significant change in all of the brain tissues. There was obvious cell edema and hippocampal pyramidal tract disorder in both the 20 Gy and 30 Gy groups, followed by weight loss, loss of appetite, vomiting, and mental malaise. The symptoms of the rats in the 30 Gy group were more serious and the number of deaths increased, which seriously affected the statistical analysis of the data. Comparatively, we determined that the appropriate experimental radiation dose was 20 Gy and the irradiation mode was single whole brain irradiation.
Autophagy and Tissue Damage
Autophagy is a widespread metabolic pathway in eukaryotic cells, which mainly degrades macromolecular substances and organelles in the cytoplasm through the lysosomal pathway. LC3 is the gold standard for detecting autophagy. Under normal circumstances, autophagy remains stable with the function of regulatory pathways. However, under some pathological conditions, autophagy is overactivated, or inhibited, which aggravates injury. Autophagy can be highly induced under different conditions, such as nutritional deprivation, oxidative stress, hypoxia, and chemotherapeutic drugs exposure.15-18 Mitochondrial autophagy is a type of autophagy that is critical for mitochondrial homeostasis. Some studies have indicated19-21 that the deficiency and excess of mitochondrial autophagy promotes cell damage and death. Jin et al 22 found that, through the study of the changes of autophagy after traumatic brain injury in rats, double fluorescence staining of the hippocampus was positive for the autophagy-associated proteins, LC3 and Beclin-1. The expression was significantly different from the microglial marker, Iba-1. The positive cells in the hippocampus increased significantly in the radiation group, indicating that autophagy of the microglia in the damaged brain was significantly up-regulated. Autophagy inhibitors further aggravated the original damage and its underlying mechanism may be the result of PI3K/Akt/mTOR pathway activation by radiation. 23 In a study of Parkinson’s disease, Redmann et al 24 found that an autophagy activator increased autophagy as well as LC3-II levels, which ameliorated Parkinson’s disease. This suggests that the symptoms of Parkinson’s disease may be related to the reduction of autophagy and abnormal protein accumulation. New evidence suggests that autophagy plays an important role in the pathogenesis of amyotrophic lateral sclerosis (ALS).25-27 Xiaojie et al 28 found that the autophagy protein marker, LC3-II, was increased significantly and specifically in spinal motor neurons of ALS mice and autophagy vacuoles accumulated significantly in the dystrophic axons of spinal motor neurons of ALS mice. After the classical autophagy activator rapamycin was used to treat transgenic mice with ALS, disease progression occurred, and promoted the death of the mice. It is evident that autophagy stability is an important physiological process needed to maintain the homeostasis of the intracellular environment, and excessive activation aggravates the damage to the original tissue and cells. In contrast, inhibition prevents the excretion of damaged organelles and metabolites, thus causing chronic and persistent damage to tissues and cells.
Autophagy and Microglia Activity
Most studies on cognitive impairment and brain damage are focused on the relationship between autophagy and neurons; however, recent studies have found that autophagy can also regulate the function of glial cells, thus regulating acute and chronic brain injury.29,30 Some studies indicate a role for the autophagy-associated protein, BECN-1, in microglia phagocytosis of the β-amyloid protein (Aβ). The down-regulation of BECN-1 decreased the phagocytosis of Aβ by microglia, suggesting that BECN-1 may be necessary for microglia-mediated phagocytosis. 31 Some studies have suggested that autophagy can also regulate the inflammatory response of microglia. 32 The mTOR pathway may be a bridge between autophagy and microglia activity. The activation of class I phosphatidylinositol-30-kinase leads to the activation of the Akt-mTOR signal transduction cascade and suppresses autophagy.33,34 In this study, the autophagy inhibitor, 3-MA, was used to specifically block the transformation of LC3-I to LC3-II35,36 by inhibiting phosphatidylinositol-3-kinase. Double fluorescence staining showed that the expression of LC3 in microglia decreased significantly, as did Iba-1, a specific marker of microglia. Thus, autophagy can have an effect on the activity of microglia. Whereas mTOR can significantly affect the microglia, Erlich et al 37 demonstrated that the inactivation of the mTOR pathway inhibited the activation of microglia, increased the number of neurons in the injured brain tissue, and improved brain injury. Finally, there are a few studies on the effect of autophagy on microglia flux; however, more data is needed to confirm the relationship between autophagy and microglia activity.
Radiation Brain Injury and Microglia
Microglia are innate immune cells of the nervous system. In a healthy adult brain, microglia constantly detect changes in the surrounding environment. Any slight change may cause the activation of microglia, including changes in morphology and the regulation of inflammatory factors.38,39 In studies of radiation-induced brain injury, radiation exposure was found to be associated with an immune response,40,41 as well as cell necrosis and apoptosis. 42 Bálentová et al 43 found that when studying the changes in cognitive function following radiotherapy, microglia were significantly activated after radiotherapy; however, the activity of the microglia gradually returned to normal over time. In an experiment on rhesus monkeys after whole brain radiotherapy, Andrews et al 44 found that the white matter was damaged after radiotherapy and microglia were activated. The microglia could mediate neuroinflammatory reactions and aggravate the brain tissue injury. Similarly, using moderate doses of radiation in rats, Lia et al found that the inflammasome NLRP3 (NLR pyrin domain containing 3) and caspase-1 in microglia were activated, but not in neurons. 45 Microglia are involved in the repair of brain tissue, but they may release inflammatory factors after over-activation to mediate brain tissue damage.
In the present study, radiation caused brain damage to rats. We found that the expression of Iba-1 was significantly increased and double fluorescence staining showed that autophagy was increased in microglia, which indicates that after 8 weeks of radiation injury, microglia are activated and the brain could not recover in a timely manner. After treating the rats with 3-MA, the expression of Iba-1 was significantly down-regulated and autophagy was decreased in the microglia. The detection of apoptosis-related indicators and the neuron-specific marker NeuN revealed that in the group in which autophagy was inhibited, the number of necrotic neurons decreased and the expression of apoptosis-related proteins improved, which resulted in a certain degree of recovery from brain injury. However, because of time limitations, the mechanism of microglia activation (i.e., mechanism of autophagy) was not fully examined, although it warrants further investigation.
Conclusion
This study evaluated the pro-survival and anti-apoptotic effects of 3-methyladenine on radiation-induced brain injury. Through the detection of apoptosis-related proteins and microglia markers, we found that 3-MA regulates autophagy and affects the activity of microglia. This further promotes the repair of brain tissue, which may represent a potential treatment for brain injury. However, the detailed molecular mechanisms still require further study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Provincial Government Funded Clinical Medicine Excellent Talents Training Projective in 2014, Hebei Provincial Health Department [grant number H2013209253].
