Abstract
Although the adult human brain has a small number of neural stem cells, they are insufficient to repair the damaged brain to achieve significant functional recovery for neurodegenerative diseases and stroke. Stem cell therapy, by either enhancing endogenous neurogenesis, or transplanting stem cells, has been regarded as a promising solution. However, the harsh environment of the diseased brain posts a severe threat to the survival and correct differentiation of those new stem cells. Hormesis (or preconditioning, stress adaptation) is an adaptation mechanism by which cells or organisms are potentiated to survive an otherwise lethal condition, such as the harsh oxidative stress in the stroke brain. Stem cells treated by low levels of chemical, physical, or pharmacological stimuli have been shown to survive better in the neurodegenerative brain. Thus combining hormesis and stem cell therapy might improve the outcome for treatment of these diseases. In addition, since the cell death patterns and their underlying molecular mechanism may vary in different neurodegenerative diseases, even in different progression stages of the same disease, it is essential to design a suitable and optimum hormetic strategy that is tailored to the individual patient.
Keywords
INTRODUCTION
Regenerative medicine is an emerging field of treatments based on the concept of producing new cells to replace malfunctioning or damaged ones as a vehicle to treat disease and injury (Steindler 2007; Hess and Borlongan 2008). It could aid in easing symptoms or even reverse disease progression of diseases where pharmacological interventions and other treatment modalities are no longer sufficient or not available (Gogel et al. 2011). This is especially true for diseases associated with aging such as neurodegenerative diseases, type II diabetes, and heart failure (Haas et al. 2005; Chang et al. 2007; Caspi et al. 2007). This is also true for medical conditions resulting from damage to cells due to acute disease, such as trauma, infarction and burns (Li et al. 2008a; Chang et al. 2007).
Although stem or progenitor cells are presumed to survive various stresses, the harsh insults from the diseased proteins, inflammatory cytokines, and cytotoxic cells in the areas of tissue damage and regeneration may significantly hamper their viability, especially during the first few days after transplantation (Norgaard et al. 2006; Bliss et al. 2006; Kelly et al. 2004; Hicks et al. 2009).
A term initially used in toxicology, hormesis has been defined as a dose-response relationship in which there is an inhibitory response at higher doses but a stimulatory response at lower doses, resulting in a U-shaped or inverted U-shaped dose response (Fig. 1) (Mattson 2008; Calabrese et al. 2007; Kendig et al. 2010). When given at a small quantity, a harmful substance gives rise to beneficial effects in living organisms. A good examples is alcohol consumption, in which light drinking helps prevent heart disease and stroke (Cook and Calabrese 2006). Other examples of hormesis include physical exercise, ischemia preconditioning, and chemical treatment of cells (Calabrese et al. 2007; Mattson 2008; Calabrese et al. 2008a; Wang et al. 2012; Wang et al. 2011).

The U-shaped dose response curve of Hormesis. From Calabrese and Baldwin (Calabrese and Baldwin 2001), used with permission.
Hormesis is believed to be a common paradigm throughout nature and is the foundation of evolution (Calabrese et al. 2007; Norgaard et al. 2006; Rattan 2004; Mattson 2008). Over the years, life on earth has went through harsh environment in which cells and organisms were often exposed to adverse stresses, including free radicals and toxic substances. Only those organisms that developed a complex mechanism to adapt to or cope with the changing environment have survived (Mattson 2008). Several studies have reported that a mild stress produces stress adaptation in cells that potentiates them to endure a much severe condition (reduction of cell death) (Norgaard et al. 2006; Geraci et al. 2006; Kraft et al. 2006; Madhavan et al. 2006; Mattson 2008; Kendig et al. 2010; Wang et al. 2011).
In this article, we will discuss the potential application of hormesis in regenerative medicine for neurodegenerative diseases and stroke. Since cell death/survival is crucial for regenerative medicine, and the cell death patterns and their underlying molecular mechanism may vary in different neurodegenerative diseases, even in different progression stages of the same disease, it is essential to design a suitable and optimum hormetic strategy that is tailored to the individual patient, we will first briefly review the common cell death pathways related to neural stem/progenitor cells, namely apoptosis, autophagy, and necrosis, which could be potentially averted by hormesis.
CELL DEATH PATHWAYS
Apoptosis - the programmed cell death
Apoptosis is an important part of life for multicellular organisms (see reviews (Lockshin and Zakeri 2004; Jin and El-Deiry 2005; Rufini and Melino 2011)). It plays an essential role in development and homeostasis. During early development of many organs, for example the brain and the fingers, cells are produced in excess which eventually undergo apoptosis and thereby contribute to organ and tissue morphogenesis. Features of apoptosis include condensed nuclei, cell shrinkage, cell membrane blebbing, and DNA fragmentation. Apoptotic cell death is considered to be a major cell death pathway for transplanted stem cells, including neural progenitor cells (NPCs), mesenchymal stem cells (MSCs), and bone marrow stem cells (BMSCs) (Theus et al. 2008; Wang et al. 2011; Geng 2003). For example, It has been reported that only less than 1% of the injected BMSCs survive past one week (Toma et al. 2002).
Apoptotic cell death is usually categorized into two distinct pathways: caspase-dependent and caspase-independent (Jin and El-Deiry 2005; Rufini and Melino 2011; Lockshin and Zakeri 2004) (Fig. 2). In the caspase-dependent pathway, a series of specific cysteine proteases, named caspases, are activated in a cascade of proteolytic maturation steps in response to environmental or physiological cell death signals (Fig. 3) (Kroemer et al. 2009; Galluzzi et al. 2012). The initiator caspases, caspase 8 and caspase 9, are derived from procaspase 8 and procaspase 9, which will activate the effector caspases, such as caspase 3, and caspase 7 (Sivananthan et al. 2010). The activated effector caspases then cleave a subset of more than 100 cellular proteins which are important for cell structure, cellular repair, and cell cycle related proteins (Degterev et al. 2003), causing arrest of vital cellular functions as well as lethal catabolic reactions (Martins et al. 2011; Cheng et al. 2010; Tadokoro et al. 2010; Boatright et al. 2003). In mammals, the caspase-dependent apoptosis is further subcategorized into extrinsic pathway (death receptor pathway) and intrinsic pathway (mitochondria pathway) depending upon the source of the stress signals (Fig. 3). The extrinsic pathway is activated by extrinsic stimuli such as binding of ligands to cell surface death receptors, which include Fas, tumor necrosis factor receptor, and Trail receptors. On the other hand, the intrinsic pathway is initiated by diverse cellular apoptotic stimuli, such as DNA damage and virus infection, which converge at the mitochondria (Baliga and Kumar 2003). Release of cytochrome c from the mitochondria to the cytoplasm activates the caspase cascade. Cytosolic cytochrome c (cytoC) associates with apoptosis protease- activating-factor 1 (Apaf-1) and procaspase 9, generating an intracellular cell death inducing-complex known as “apoptosome”. Within the apoptosome, caspase 9 is activated, leading to the cleavage of procaspase 3 to form the activated caspase 3. The extrinsic and intrinsic pathway then converge on the caspase 3 and subsequently on other proteases and nucleases that drive the cleavage of chromosome DNA and other terminal events of apoptosis (Baliga and Kumar 2003; Kroemer et al. 2007). However, caspase inhibition rarely prevents apoptosis completely, indicating that there are caspase-independent pathways (Kroemer et al. 2007; Galluzzi et al. 2012; Mailleux et al. 2007; Cordeiro et al. 2010; Vandenabeele et al. 2010; Broker et al. 2005). Apoptosis inducing factor (AIF) and cofilin operate as death-effectors in the complete absence of caspase activation (Fig. 2) (Patterson et al. 2000; Wabnitz et al. 2010; Lee et al. 2010). The key regulators of caspase-independent apoptosis are located on the mitochondria membranes, whose integrity (mitochondrial outer membrane permeabilization (MOMP)) is regulated by multiple proteins and processes, including Bcl-2 family members (Esposti 2010), BH3-only proteins (Cabon et al. 2012), tumor suppressor protein P53 (Galluzzi et al. 2008a; Galluzzi et al. 2011), Protein kinase Cs (Allen-Petersen et al. 2010; Paoletti et al. 2010), epigenetic perturbations (Yelamanchili et al. 2010; Tenedini et al. 2010; Sancho-Pelluz et al. 2010; Lei et al. 2010; Lanzillotta et al. 2010), and cell cycle perturbations (Fig. 2) (Mitchell et al. 2010).

Schematic presentation of the caspase dependent and independent apoptotic pathway.

Schematic presentation of the intrinsic and extrinsic pathways of the caspase dependent apoptotic pathways.
Autophagy - A physiological hormesis case
The autophagic cell death is initially considered a “Type II” cell death pathway in that it requires activation of the lysosomal compartment, in which parts of the cytoplasm are enwrapped in double-memberaned vesicles (called autophagosomes) and degraded by lysosome hydrolases (Lockshin and Zakeri 2004; Madeo et al. 2010; Criollo et al. 2010). Autophagy is a normal physiological process that functions to turn over abnormal proteins and organelles (Lockshin and Zakeri 2004) and is closely related to apoptosis (Martins et al. 2011; Galluzzi et al. 2008b; Rufini and Melino 2011; Agostini et al. 2011). Autophagy can counteract or sustain apoptosis or vise versa and exhibit a typical hormesis phenotype. Low levels of autophagy is pro-survival, as it allows for the removal of the damaged proteins and helps maintain metabolic homeostasis (Eisenberg-Lerner et al. 2009). Conversely, a high level of autophagy leads to cell death and cooperates with apoptosis because of the extensive cellular damage (Galluzzi et al. 2008b; Eisenberg-Lerner et al. 2009). Autophagy and apoptosis exhibit cross talk on the molecular level, too. For example, the apoptosis-inducing BH3 proteins have been shown to liberate Beclin 1 from its interaction with Bcl-2-like proteins, thereby leading to autophagy (Buttner et al. 2011; Juhasova et al. 2011).
Necrosis -
Necrosis is historically considered a “type III” cell death pathway (Golstein and Kroemer 2007; Kroemer et al. 2009; Krysko et al. 2008a). It is originally defined as a type of cell death that lacks the features of apoptosis and autophagy (Golstein and Kroemer 2007; Syntichaki and Tavernarakis 2002; Nicotera and Melino 2004; Chiong et al. 2011). Cells suffer necrotic cell death when exposed to extreme environmental or physiological conditions (Syntichaki and Tavernarakis 2002). Necrosis was often considered chaotic and uncontrolled, leading to local inflammation, and being connected to unwanted cell loss in many types of human pathologies (Golstein and Kroemer 2007; Festjens et al. 2006; Yuan 2006). However, recent advances have slowly overturned this grim prospect and suggested that the occurrence and course of necrosis might be tightly regulated (Syntichaki and Tavernarakis 2002; Golstein and Kroemer 2007; Nicotera and Melino 2004). Increasing evidence demonstrates that regulated necrosis, referred to as necroptosis (Vanlangenakker et al. 2012; Nicotera and Melino 2004), is not anymore an isolated observation of a particular cell line or in certain conditions, but is also recognized as an in vivo cellular defense mechanism against viral infections and as being critically involved in ischemia-reperfusion damage(Degterev et al. 2005; Galluzzi et al. 2012; Vanlangenakker et al. 2012). Breakthroughs come from the discovery that the kinase activities of RIPK1/ RIPK3 are crucial for necroptosis (Degterev et al. 2008; He et al. 2009; Zhang et al. 2009; Vandenabeele et al. 2010). Despite these advances, there are still no specific positive biochemical biomarkers for the in situ detection of necrosis in vitro and in vivo (Vanlangenakker et al. 2012). The release of intracellular proteins such as high-mobility group box 1 protein (HMGB1) (Scaffidi et al. 2002) and cyclophilin A (CypA) (Christofferson and Yuan 2010) has been proposed as a candidate necrotic biomarker. However, HMGB1 and CypA can also be passively released from cells dying by secondary necrosis following apoptosis (Scaffidi et al. 2002) or actively secreted from activated immune cells or cells dying from pyroptosis, a term introduced to describe necrosis-like cell death that is dependent on caspase-1 activity (Fink and Cookson 2005). The ratio between caspase-cleaved cytokeratin-18 released from apoptotic cells and intact cytokeratin-18 released from cells dying from other causes, including necrosis, has also been proposed as a marker to determine qualitatively and quantitatively the extent of both types of cell death (Kramer et al. 2004), but should again be taken with caution (Vanlangenakker et al. 2012). So far, the mostly used method to determine necrosis is to combine morphology with immunofluorescence studies (Vanlangenakker et al. 2012; Krysko et al. 2008a; Krysko et al. 2008b). Necrotic cells typically exhibit intact extracellular nuclei in electron microscopy images, and are Tdt-mediated dUTP nick end labeling (TUNEL) positive, but caspase 3 negative in immunofluorescence images (Krysko et al. 2008a; Krysko et al. 2008b; Vanlangenakker et al. 2012; Golstein and Kroemer 2007; Kroemer et al. 2009).
MECHANISMS OF HORMESIS IN NEURAL CELLS
Hormesis stimulates multiple bioprotective mechanism, including induction of cytoprotective pathways (such as molecular chaperones), antioxidative stress, DNA repair, and immune system activation to protect neural cells from various forms of cell death (Calabrese et al. 2008a; Calabrese et al. 2008b; Kendig et al. 2010). Here, we will briefly discuss the induction of anti-apoptotic molecular chaperones and antioxidative stress proteins by hormesis.
Expression of molecular chaperones (usually called heat shock proteins (Hsps)) is an important intracellular defense mechanism against stressful conditions (Homma et al. 2007; Wang et al. 2001, 2003, 2004). Hsps are indispensible for protein homeostasis in normal physiological conditions (Calabrese et al. 2010; Terasawa et al. 2005; Whitesell and Lindquist 2005; Trepel et al. 2010). They also serve a vital role of protein protection and cell protection against cell death during adverse situations (Arumugam et al. 2006). The importance of Hsps in protection of neural cells against neurodegenerative diseases is increasingly recognized. It has been found that levels of Hsp70 and GRP78 are increased in neurons subjected to low level stressors, which serve to protect the neurons against otherwise lethal levels of oxidative or metabolic stresses (Guo et al. 2001; Arumugam et al. 2006).
Low level of free radicals generated by mild oxidative stresses can activate signaling pathways that increase resistance of neurons to more severe stresses (Otani 2004; Juhaszova et al. 2004). This hormesis mechanism involves activation of the N-Methyl-D-Aspartate (NMDA) receptor and brain- derived nerve factor (BDNF) (Marini and Novelli 1991; Marini et al. 2008; Arumugam et al. 2006). Multiple antioxidative systems are triggered or enhanced in response to mild oxidative stresses, including antioxidative enzymes, glutathione and proteins involved in stabilization of mitochondrial membranes (Liu et al. 2002; Arthur et al. 2004).
STEM CELL THERAPY OF NEURODEGENERATIVE DISEASES AND STROKE
Chronic neurodegenerative diseases, such as Alzheimer's disease and Parkinson's disease create a considerable burden to individuals, families, and the society. In addition, the catastrophic impact of stroke to quality of life is severe. Thus, it is of pivotal importance to develop innovative methods to treat or prevent these diseases. Regenerative medicine has emerged to be one of the most promising approaches.
Two sets of strategies have been proposed to regenerate cells in order to repair the damaged brain tissues. The first strategy is through donor cell transplantation (from either autologous or allogenic cell sources). Over the last decade, credible data have demonstrated the safety and feasibility of injected stem cells in inducing neural cell regeneration and functional recovery in animal models of stroke, Parkinson's disease, Huntington's disease, and Alzheimer's disease (Gogel et al. 2011; Francis and Wei 2010; Haider and Ashraf 2010; Herrmann et al. 2010; Savitz et al. 2011). Human neural stem cells (HNSCs) transplanted into aged rat brains differentiated into neural cells and significantly improved the cognitive functions of the animals (Sugaya 2005; Sugaya et al. 2006; Roitberg et al. 2006; Qu et al. 2001; Winkler 2001). Cells from different sources have been examined and proved effective, including neural stem cells (NSC) derived from embryonic stem (ES) cells or induced pluripotent stem (iPS) cells, and fetal or adult neural stem cells (Zeng et al. 2010; Gogel et al. 2011; Haider and Ashraf 2010; Wang et al. 2011; Haas et al. 2005; Wang et al. 2009; Theus et al. 2008; Wei et al. 2012; Kelly et al. 2004; Bliss et al. 2006; Horie et al. 2011). Pluripotent stem cells are no longer seen as a safe cell sources as its transplantation leads to teratoma formation (Gogel et al. 2011). Therefore, direct differentiation and purification of ESCs into defined neural subtypes is preferred.
Another cell source for transplantation is MSCs (Haider and Ashraf 2010; Wang et al. 2009; Wei et al. 2012; Geng 2003; Toma et al. 2002). MSCs can be derived from a variety of tissues such as bone marrow and cord blood. As they seem to penetrate the blood brain barrier, no invasive intracerebral surgery is required and instead a peripheral infusion can be performed to deliver these cells to the brain. However, questions still remain that how many of these cells travel to the brain and to what extent they differentiate into different neural cell types (Gogel et al. 2011).
The second strategy is to enhance endogenous neurogenesis and thus boost endogenous repair mechanisms of the brain (Savitz et al. 2011). Much evidence has been found that the brain is capable of regenerating neurons after their maturation (Sugaya 2005; Sugaya et al. 2006; Qu et al. 2001; Eriksson et al. 1998; Allen et al. 2001; Aimone et al. 2006; Jagasia et al. 2006), and stroke can stimulate neurogenesis migration of neural stem/progenitor cells into damaged brain region (Greenberg 2007; Wiltrout et al. 2007; Chopp et al. 2007; Liu et al. 2007). But the extent to which this neurogenesis can replace lost neural cells and contribute to functional improvement is not clear (Gogel et al. 2011). It is believed that protection, and/or enhancement of proliferation, of the endogenously-newly-generated neural cells by mechanisms of hormesis could be a viable approach, by which a sufficient amount of cells generated to promote the functional recovery of the patients (Gogel et al. 2011; Hess and Borlongan 2008; Marini et al. 2008).
HORMESIS FOR REGENERATIVE MEDICINE
Hormesis has been proven to reduce cell death and aid in survival in both of these two scenarios. For the strategy to transplant donor stem cells, the following hormetic methods have been found to enhance stem cell survival and/or mobilization: mild hypoxia (Lian and Tse 2010; Kamota et al. 2009; Wei et al. 2012; Theus et al. 2008), the antibiotic minocycline (Sakata et al. 2012), an endogenous peptide apelin (Zeng et al. 2012; Wang et al. 2012), and others (Wisel et al. 2009; Salloum et al. 2012; Hoke et al. 2012; Kwon et al. 2008). Our group has found that treatment with a low dose Hsp90 inhibitor 17-N-Allylamino-17-demethoxygel-danamycin (17-AAG) protects NPCs from naturally-occurring and stress induced apoptosis, with a typical reversed U-shaped hormetic dose-response relationship (Wang et al. 2011) (Fig. 4). Mechanistically, low dose 17-AAG activates the stress response pathway, MAP kinase pathway, and PI3K/Akt pathways. This is of profound clinical relevance, since many Hsp90 inhibitors, including 17-AAG, are in clinical trials against multiple malignancies, and have overcome the toxicity and solubility problems (Kummar et al. 2010; Trepel et al. 2010; Waza et al. 2005, 2006).
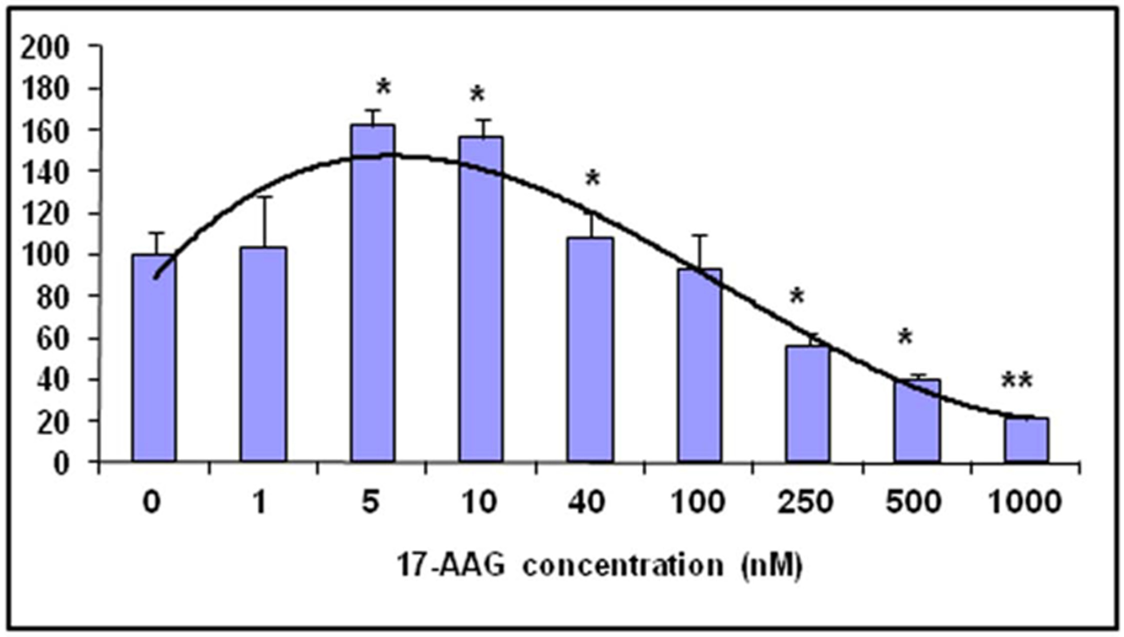
The Hsp90 inhibitor 17-AAG induces an inverted Ushaped cell survival/death dose-response curve in NPCs (Wang et al. 2011).
Hormesis is also a promising strategy to protect endogenous neurons and enhance endogenous neurogenesis (Gogel et al. 2011; Marini et al. 2008). One example of hormetic protection of neurons is glutamate, the major excitatory amino acid that mediates physiological excitation. Glutamate is involved in both acute and chronic neurodegenerative disorders (Dobrek and Thor 2011; Lau and Tymianski 2010). High level activation of the ionotrophic glutamate receptor leads to neuronal cell death. However, low level activation of glutamate receptors exerts neuron protection (Marini et al. 2008; Marini and Novelli 1991; Jonas et al. 2001; Calabrese et al. 2007). Another interesting proposal is to utilize the cytoprotective effect of autophagy to protect neurons from the hostile disease environment. Since mild autophagy can lead to the removal of damaged and potentially dangerous mitochondria, it increases the threshold for cell death induction by MOMP-inducing agents or stressors (Blagosklonny 2010; Bennett et al. 2010; Deng et al. 2010; Degli Esposti et al. 2011). Thus mild autophagy may be a promising strategy to reduce the propensity of neurons to undergo apoptosis or necrosis (Martins et al. 2011).
We have mentioned that chronic or acute brain damage induces neurogenesis, although their beneficial effect is not clear. Supporting these newly generated cells by infusion of neurotrophic factors or other measures that enhance neurogenesis, or boost the survival of the newly generated stem/progenitor cells, could help rebuild a sufficient amount of the lost circuitry, hence improving functional recovery of the patients (Gogel et al. 2011; Hess and Borlongan 2008; Li et al. 2008b; Misumi et al. 2008). Many types of stimulation methods have been tested and proven beneficial, including ischemia preconditioning, exercise, and peripheral stimulation (Li et al. 2008b; Andoh et al. 2002; Dawson 2002). Our group found that injection of 17-AAG increased the number of newly generated neural cells, in both the subventricular zone and the subgranular zone, by about 60%, accompanied by triggering phosphorylation of cytoprotective kinases Akt and GSK-3b in the brain (Wang et al. 2011).
CONCLUSIONS AND PERSPECTIVES
Stem cells delivered into the brain (or newly regenerated) can support a certain amount of neural function by secreting trophic factor vascular endothelial growth factor (Bliss et al. 2006; Horie et al. 2011) However, the injected cells do not infiltrate and survive the damaged region, let alone reconstruction of the lost neural circuit, undermining the idea and full potential of regenerative medicine (Bliss et al. 2006; Kelly et al. 2004; Horie et al. 2011). Therefore it is vital to optimize the treatment procedure so that the newly generated (or transplanted) cells survive and correctly differentiate into desired cell types in the brain. Many studies have found that hormesis potentiates stem cells to a state in which they can endure much harsher environment and improve functional recovery in animal models of neurodegenerative disease. Hence it is rational to promote the combination of stem cell therapy with hormesis to achieve a more desirable outcome, and encouraging more research in the related field.
Cell death is a very complex process and our understanding of its mechanism is advancing in an unprecedented pace. More importantly, since the cell death patterns and their underlying molecular mechanism may vary in different diseases and even in different progression stages of the same disease, it is essential to design a suitable and optimum hormetic strategy that is tailored to the individual patient. Thus it calls for comprehensive approaches to achieve protection of the newly generated (or transplanted) cells so that they are more suited for regenerative medicine.
Footnotes
ACKNOWLEDGEMENT
This work is supported by a grant from American Heart Association and an intramural grant from Georgia Health Sciences University to GW. I am thankful to Dr Erhard Bieberich for critical reading and valuable suggestions. I am also thankful for the support by the Institute of Molecular Medicine of Genetics (under the directorship of Dr. Lin Mei).
