Abstract
Objective
In this study, we investigated the protective effect of Metformin on fibrosis of trabecular meshwork cells induced by TGFβ2.
Methods
Transformed and primary human trabecular meshwork cells (HTMCs) were treated with TGFβ2 or Metformin alone or combination, western blotting and immunofluorescence staining assays to detect autophagy activity and fibrotic proteins expression levels. TGFβ2 or Metformin alone or combination were injected into the anterior chamber of mouse eye. Mouse intraocular pressure (IOP) was measured every week, mouse eye sections were conducted immunofluorescence staining to analyze Col1 and Col3 expression. pSmad3 level and localization to evaluate TGFβ/Smad3 pathway activity. Chloroquine phosphate was used to block autophagy-lysosome pathway.
Results
Metformin activates autophagy of HTMCs in a dose dependent manner and efficiently ameliorates TMCs fibrosis induced by TGFβ2 in vitro and in mouse model, and decreased elevated IOP caused by TGFβ2. Metformin promotes fibrotic proteins degradation through the autophagy-lysosome pathway.
Conclusion
Our study found Metformin could alleviates fibrosis of HTMCs induced by TGFβ2 and decreased elevated IOP in mouse model.
Introduction
Glaucoma is the first leading cause of irreversible blindness worldwide, affecting more than 70 million people, with primary open angle glaucoma (POAG) accounting for 74% of cases.1,2 Glaucoma are a group of conditions leading to irreversible sight loss and characterised by progressive loss of retinal ganglion cells (RGCs). 3 It is more common in the older population, with a global prevalence of 3.5% in people over 40 years old and an adjusted odds ratio of 1.73 per decade age increment. Glaucoma may increase to over 110 million by the year 2040 as a result of longer life expectancy and a subsequently aging population. 4
Aqueous humor is produced by ciliary body and flow through trabecular meshwork and maintain the intraocular pressure (IOP). Elevated IOP is a major risk factor of glaucoma, lowering IOP has been proven to be an effective measure in managing glaucoma. 5 Fibrosis of trabecular meshwork cells and extracellular matrix deposition cause function defects of trabecular meshwork cells, which is the major reason of decreased outflow of aqueous humor and elevated IOP. 6
Metformin, a famous drug that commonly used in patients with type 2 diabetes. While, increasing amount of evidence find that Metformin also has anti-aging and anti-fibrosis effects in many organs.7-9 Its use in a diabetic population with reduced incidence of POAG.10,11 The molecular mechanism through which Metformin is acting to prevent glaucoma is not yet clear but potentially involves effects on the pathogenesis of fibrosis and mitochondrial bioenergetic dysfunction in glaucoma.12,13
Autophagy is an indispensable self-degradative process for intracellular quality control and homeostasis, which promotes degradation of organelles and misfolded or aggregated proteins under stress. 14 When autophagy is activated, autophagosomes encompass damaged organelles and aggregated proteins and fuse with lysosomes to form autolysosome, then organelles and aggregated proteins digested and degraded by enzymes in autolysosome. Activation of autophagy was found could ameliorate fibrosis of trabecular meshwork cells and other cell types.15,16
Metformin was reported could activate autophagy and alleviates fibrosis in various animal disease models.17,18 Whether Metformin could activate autophagy, and ameliorate fibrosis induced by TGFβ2 on HTMCs is still unclear. In the present study, we found Metformin induces autophagy activation in HTMCs in a dose dependent manner and efficiently ameliorates TM cells fibrosis induced by TGFβ2 in vitro and in mouse model, which has no effect on TGFβ/Smad3 pathway activity, while the protective effect can be weakened by autophagy-lysosome pathway inhibitor Chloroquine phosphate.
Materials and Methods
Human Trabecular Meshwork Cells Culture
Primary HTMCs were extracted and verified in accordance with our earlier research, and transformed HTMCs were acquired from iCell Bioscience Inc. (Shanghai, China).15,19-21 Primary HTMCs were obtained from two different donors, which from two eye trauma patients who have to do eye enucleation surgery, and both two patients were male (45 and 51 years old) and did not have other eye diseases. Primary HTMCs within five to eight passages were employed in all experiments. The Henan Eye Hospital’s Medical Ethical Committee gave its consent to collections of human eye materials (approval no. HNEECKY-2022[18]). DMEM/F12 media (CD0001, SparkJade, China) supplemented with 15% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 mg/mL streptomycin was used to maintain both transformed and primary HTMCs.
We used two primary HTMCs to do the in vitro experiments to confirm our results, and used one primary HTMCs to do three times replications and do the quantification.
Pharmacological Agents
Recombinant human TGF-β2 protein (10 ng/mL, HZ-1092, Proteintech, China) or Metformin (HY-B0627, MedChemExpress, China) and chloroquine phosphate (10 μM, HY-17589, MedChemExpress, China) were administered to cells for 48 hours. Physiological saline solution (PSS) was used to dissolve the stocks of every agent. Equal volumes of PSS were administered to the control groups.
Western Blotting
The cells underwent centrifugation (14,000 g × 10 min at 4°C) after being lysed with RIPA buffer (Beyotime, P0013 B, China) and supplemented with a protease inhibitor cocktail (Beyotime, P1049, China). The BCA technique was used to determine the protein contents in cell lysates, and 10 g of total/sample was electrophoresed on 10% SDS-PAGE gels (ET15010Gel, FuturePAGE™, ACE, China). Following transfer to nitrocellulose membranes, Ponceau S (Kermey, ML5001, China) staining was used to confirm protein loading and transfer. The membranes were then incubated with fast blocking buffer (Kermey, MF0501, China) for 15 minutes at room temperature. Thereafter, membranes were incubated overnight at 4°C with diluted primary antibodies(antibody dilution bufer bought from Kermey, MX0402, China), washed three times with TBST for 20 min, then incubated with HRP-conjugated secondary antibodies(Goat Anti-Rabbit IgG, SparkJade, EF0002) in TBST for 1h at room temperature. After further washing with TBST for 15min, the antibody decorated bands were visualized using a chemiluminescent substrate (Epizyme ECL PicoLight Substrate, SQ202-1, China) and images recorded using a ChemiDoc MP system (BioRad). The following primary antibodies were used: GAPDH (Abways, AB0037, China, 1:3000), Smad3 (Proteintech, China, 66516-1-Ig, 1:1000), pSmad3 (Cell signaling technology, USA, 9520S, 1:1000), Fibronectin (HUABIO, China, ET1702-25, 1:5000), COL1 (Proteintech, China, 14695-1-AP, 1:2000), COL3 (Abways, China, DY1299, 1:1000), αSMA (Proteintech, China, 14395-1-AP, 1:2000), p62 (Proteintech, China, 18420-1-AP, 1:3000), LC3 (Proteintech, China, 14600-1-AP, 1:500)).
Immunofluorescence Staining for Cultured Cells
Transformed and primary HTMCs were allowed to attach to glass coverslips for 24h before applying the indicated treatments, then washed with PBS before 15 min fixation with 4% PFA in PBS. Thereafter, the samples were washed in PBS buffer, permeabilized with 0.5% Triton X-100 in PBS for 15min, further washed with PBS before blocking with 4% BSA in PBS (0.22 μm filtered) for 60min at room temperature. The coverslips were then successively incubated with diluted primary antibodies overnight at 4°C and secondary antibodies Donkey anti Rabbit IgG (H + L) 488 (Jackson ImmunoResearch Inc., 711-545-152, USA, 1:1000) for 60 min at room temperature, both diluted in blocking solution. After nuclear counter staining with DAPI solution (MedChemExpress, HY-D1396, China) for 5 min, the coverslips were mounted onto glass slides and images captured using a Leica DMi8 confocal microscope. The following primary antibodies were used: Fibronectin (HUABIO, ET1702-25, China; 1:1000), COL1 (Proteintech, 146951-1-AP, China, 1:500), COL3 (Abways, China, DY1299, 1:500), αSMA (Proteintech, China, 14395-1-AP, 1:500), Phospho-Smad3 (Cell signaling technology, USA, 9520S, 1:500).
Animal Experiments
Female C57BL/6J mice (8 weeks old) were purchased from GemPharmatech (Nanjing, China) 32G needles were used to deliver drugs into one eye’s anterior chamber. Mice were anesthetized by small animal gas anesthesia machine before and during injection (Shenzhen RWD Life Technology Co., Ltd, China). Mice in the model group received an injection of TGFβ2 (1 µl 10 ng/mL TGF TGFβ2 dissolved in physiological saline solution (PSS), while mice in the therapy group received a co-injection of TGFβ2 (10 ng/mL) and Metformin (1 µl 300 mM dissolved in PSS). Mice in the control group received an injection of either Metformin alone or equivalent PSS, with 10 mice in each group. IOP was monitored in the morning using a TonoLab rebound tonometer (Icare Finland Type: TV02) and injections were performed once a week. Mice were humanely killed four weeks later, and the removed eyeballs were preserved for paraffin embedding using FAS eye fixation buffer (Servicebio, China, G1109-100 mL). Animal experiments complied with the Association for Research in Vision and Ophthalmology Statement. All animal experiments were approved by Animal Ethics Committee of Zhengzhou University.
Paraffin Section and Immunofluorescence Staining of Paraffin Sections
Previous articles have detailed the procedures for paraffin sectioning and immunofluorescence staining tissue sections. 19
Statistical Analysis
GraphPad Prism version 7.0 was used for all analyses. The findings of at least three separate trials are shown as means±SDs for each result. One-way analysis of variance (ANOVA; three or more groups) was used to compare the data. P < 0.05 was seen as an indicator of statistical significance.
Results
Metformin Activates Autophagy of HTMCs in a Dose Dependent Manner
It is reported that Metformin could activate autophagy in many cell types,22,23 but whether it could activate autophagy in HTMCs is not clear. Firstly, we used different dose of Metformin (100 μM, 200 μM, 400 μM, 600 μM, 800 μM) to treat transformed human HTMCs for 48h, and detected autophagy markers p62 (SQSTM1) and LCII levels by western blotting. Our data showed p62 levels significantly decreased with 400 μM, 600 μM and 800 μM Metformin treatment, LCI and LCII levels also decreased after 400 μM, 600 μM and 800 μM Metformin treatment in transformed HTMCs (Figure 1A–D). We also used different dose of Metformin to treat primary HTMCs, western blotting data also showed p62, LCI and LCII levels significantly decreased with 400 μM, 600 μM and 800 μM Metformin treatment (Figure 1E–H). These data indicated that 400 μM, 600 μM and 800 μM Metformin treatment activates autophagy in transformed and primary HTMCs. 800 μM Metformin didn’t affect cell growth and viability of transformed and primary HTMCs (data not show). Metformin Induces Autophagy Activation in HTMCs in a Dose Dependent Manner. (A, E) Autophagy Markers (p62, LC3I and LC3II) Were Detected after 48h Treatment With Metformin at Indicated Dose in Transformed HTMCs (A) and Primary HTMCs (C). GAPDH Served as Loading Control. (B-D) Quantification of p62, LC3I and LC3II Protein Bands Intensities in Transformed HTMCs. (F-H) Quantification of p62, LC3I and LC3II Protein Bands Intensities in Primary HTMCs. Three Independent Experiments, Data Were Showed as mean ± SD; and Data Were Determined by One-Way Analysis of Variance (ANOVA), ***P < .001.
Metformin Alleviates Fibrosis Induced by TGFβ2 in HTMCs
We found 400 μM Metformin activates autophagy in HTMCs, and autophagy activation could ameliorate fibrosis, so we wonder whether Metformin could alleviate fibrosis of HTMCs. TGFβ2 was usually used to induce fibrosis of HTMCs in vitro, and we treated transformed HTMCs with 400 μM Metformin, 10 ng/mL TGFβ2, or 400 μM Metformin+10 ng/mL TGFβ2 for 48 h. Western blotting data showed fibrotic proteins levels, including FN, Col1, Col3 and αSMA, were significantly up-regulated after TGFβ2 treatment, while 400 μM Metformin co-treatment significantly decreased fibrotic proteins expression levels compared with TGFβ2 treatment. 400 μM Metformin treatment alone didn’t affect fibrotic proteins expression levels (Figure 2A and B). We also used 400 μM Metformin, 10 ng/mL TGFβ2, or 400 μM Metformin+10 ng/mL TGFβ2 to treat primary HTMCs for 48h, western blotting data also showed fibrotic proteins levels were significantly up-regulated after TGFβ2 treatment, while 400 μM Metformin co-treatment significantly decreased fibrotic proteins expression levels compared with TGFβ2 treatment (Figure 2C and D). Metformin Down-Regulates Elevated Fibrotic Proteins Levels Induced by TGFβ2 in TM Cells. (A, C) Fibrotic Proteins (FN, Col1, Col3 and aSMA) Levels of Transformed HTMCs (A) and Primary HTMCs (C) after Treated With Vehicle, 400 μM Metformin, 10 ng/mL TGFβ2 or Co-treated With 400 μM Metformin. (B, D) Quantification of FN, Col1, Col3 and aSMA Proteins Bands Intensities in Transformed HTMCs (B) and Primary HTMCs (D). GAPDH Served as Loading Control. Data Were Showed as mean ± SD; and Data Were Determined by One-Way Analysis of Variance (ANOVA), *P < .05, **P < .01, ***P < .001, Three Independent Experiments.
To confirm whether 400 μM Metformin could reduce fibrosis of HTMCs induced by TGFβ2, we used immunofluorescence staining assay to detect FN, Col1, Col3 and αSMA levels in transformed HTMCs. Confocal images and fluorescence quantification data showed that TGFβ2 strongly increased FN, Col1, Col3 and αSMA fluorescence intensities, while 400 μM Metformin co-treatment significantly repressed fibrotic proteins fluorescence intensities compared with TGFβ2 treatment alone (Figure 3A–E). Metformin Rescues Fibrosis Induced by TGFβ2 in Transformed TM Cells. (A–D) Immunofluorescence Staining of Col1 (A, Green), Col3 (B, Green), FN (C, Green) and aSMA (D, Red) in Transformed HTMCs after Treated With Vehicle, 400 μM Metformin, 10 ng/mL TGFβ2 or Co-treated With 400 μM Metformin. Cell Nuclei Were Decorated With DAPI (Blue). (E) Quantification of Col1, Col3, FN and aSMA Intensities in A-D. Three Independent Experiments, Each Experiment 15 Cells Were Quantified per Group, Data Were Showed as mean ± SD; One-Way Analysis of Variance (ANOVA), ***P < .001.
We also used 400 μM Metformin, 10 ng/mL TGFβ2, or 400 μM Metformin+10 ng/mL TGFβ2 to treat primary HTMCs for 48 h, immunofluorescence staining images and quantification data also showed fibrotic proteins intensities were significantly up-regulated after TGFβ2 treatment, while 400 μM Metformin co-treatment significantly repressed fibrotic proteins intensities compared with TGFβ2 treatment (Figure 4A–E). Metformin Rescues Fibrosis Induced by TGFβ2 in Primary TM Cells. (A–D) Immunofluorescence Staining of Col1 (A, Green), Col3 (B, Green), FN (C, Green) and aSMA (D, Red) in Primary HTMCs after Treated With Vehicle, 400 μM Metformin, 10 ng/mL TGFβ2 or Co-treated With 400 μM Metformin. Cell Nuclei Were Decorated With DAPI (Blue). (E) Quantification of Col1, Col3, FN and aSMA Intensities in A-D. Three Independent Experiments, Each Experiment 15 Cells Were Quantified per Group, Data Were Showed as mean ± SD; One-Way Analysis of Variance (ANOVA), ***P < .001.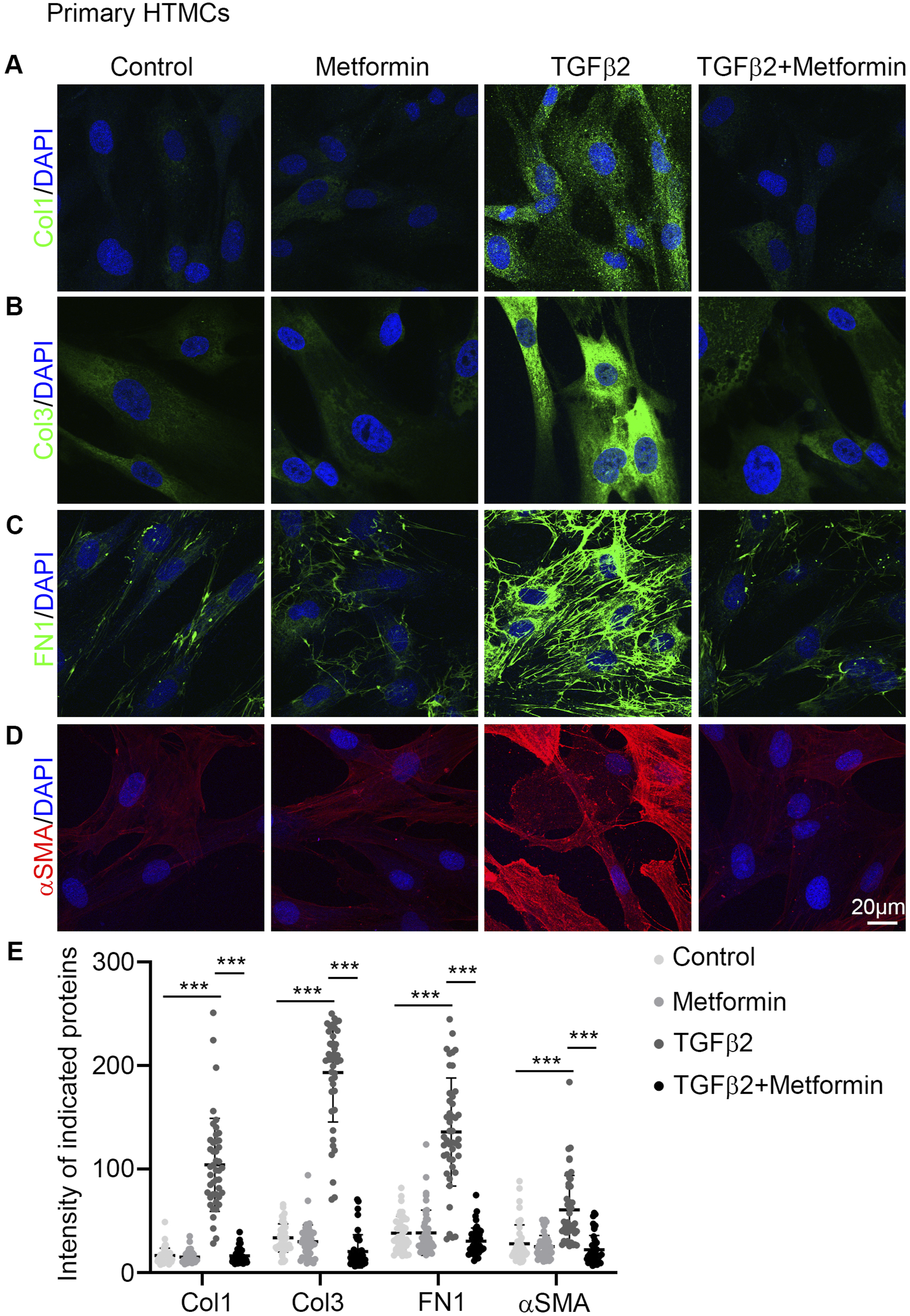
Metformin Reduces Elevated IOP and Ameliorates Fibrosis of Trabecular Meshwork Tissues Induced by TGFβ2 in Mouse Model
Our western blotting and immunofluorescence staining data indicated that 400 μM Metformin could activate autophagy and alleviate fibrosis of TM cells induced by TGFβ2 in vitro. Then we plan to figure out whether Metformin could mitigate elevated IOP and TM tissue fibrosis induces by TGFβ2 in mouse model.
We established strategies as described in Figure 5A using pharmacological drugs in the mouse model. Anterior chamber of mouse eyes was injected with Metformin, TGFβ2 alone or in combination, and vehicle group mice were injected with physiological salt solution (Figure 5A). IOP measurements showed TGFβ2 injection caused IOP elevation, while Metformin could reduce elevated IOP after TGFβ2-treated, and Metformin injection alone didn’t significantly affect IOP of mice (Figure 5B). Metformin Reduces Elevated IOP Induced by TGFβ2 in Mouse Model. (A) In Vivo Mouse Model was Established by Injecting Metformin, TGFβ2 Alone or Co-injection With Metformin Into the Anterior Chamber of One Eye once a Week as Shown in (A). (B) IOP of Mice (n = 10 per Group) Were Measured Every Week. Data Were Showed as mean ± SD, One-Way Analysis of Variance (ANOVA), ***P < .001.
Furthermore, immunostaining of eye sections and quantification data showed that TGFβ2 injection increased intensity of Col1 and Col3 in TM tissues of mice, while the elevated intensity of Col1 and Col3 in TM tissues of TGFβ2-treated mice was reduced after Metformin injection (Figure 6A–D). These data indicated Metformin could reduce elevated IOP and ameliorate fibrosis of TM tissues induced by TGFβ2 in mouse model. Metformin Ameliorates Fibrosis of TM Cells Induced by TGFβ2 in Mouse Model. (A, B) Immunofluorescence Staining of Col1 (A, Green) and Col3 (B, Green) in Eye Sections after Treated With Vehicle, Metformin, TGFβ2 or Co-treated With Metformin. Cell Nuclei Were Decorated With DAPI (Blue) and the Boxed Area Indicating the TM. (C, D) Intensities of Col1 and Col3 in TM Tissues Were Quantified (n = 5 per Group). Data Were Showed as mean ± SD, One-Way Analysis of Variance (ANOVA), ***P < .001.
Metformin has no Effect on TGFβ/Smad3 Pathway
TGFβ2 induces fibrosis of HTMCs mainly depending on Smad3 pathway,
24
so we ask a question that whether Metformin attenuates fibrosis induced by TGFβ2 through inhibiting TGFβ2/Smad3 pathway. Transformed HTMCs and primary HTMCs were treated with 400 μM Metformin, 10 ng/mL TGFβ2 or 400 μM Metformin+10 ng/mL TGFβ2 for 48h. Western blotting results and quantification data showed TGFβ2 significantly increased pSamd3 levels in transformed HTMCs and primary HTMCs, and co-treated with Metformin had no significant effect on pSmad3 levels (Figure 7A–D). Also, immunofluorescence staining images showed TGFβ2 treatment promoted nuclear localization of pSmad3 in transformed HTMCs, while co-treated with Metformin and TGFβ2 didn’t affect nuclear localization of pSmad3 (Figure 7E), which was consistent our western blotting results (Figure 7A–D). These data indicated that Metformin alleviated fibrosis of HTMCs induced by TGFβ2 not depending on inhibition of pSmad3 activity. Metformin has No Effect on TGFβ/Smad3 Pathway. (A, C) Transformed HTMCs and Primary HTMCs Were Treated With Vehicle, 400 μM Metformin, 10 ng/mL TGFβ2, or Co-treated With 400 μM Metformin, Western Blotting of pSmad3 and Smad3 Were Conducted in Transformed HTMCs (A) and Primary HTMCs (C). (B, D) pSmad3/Smad3 Ratio Were Quantified in Transformed HTMCs (B) and Primary HTMCs (D). (E) Immunofluorescence Staining of pSmad3 (Green) in Transformed HTMCs Treated Same With A, Cell Nuclei Were Decorated With DAPI (Blue). Three Independent Experiments, Data Were Showed as mean ± SD; One-Way Analysis of Variance (ANOVA), ***P < .001, ns Means No Significance.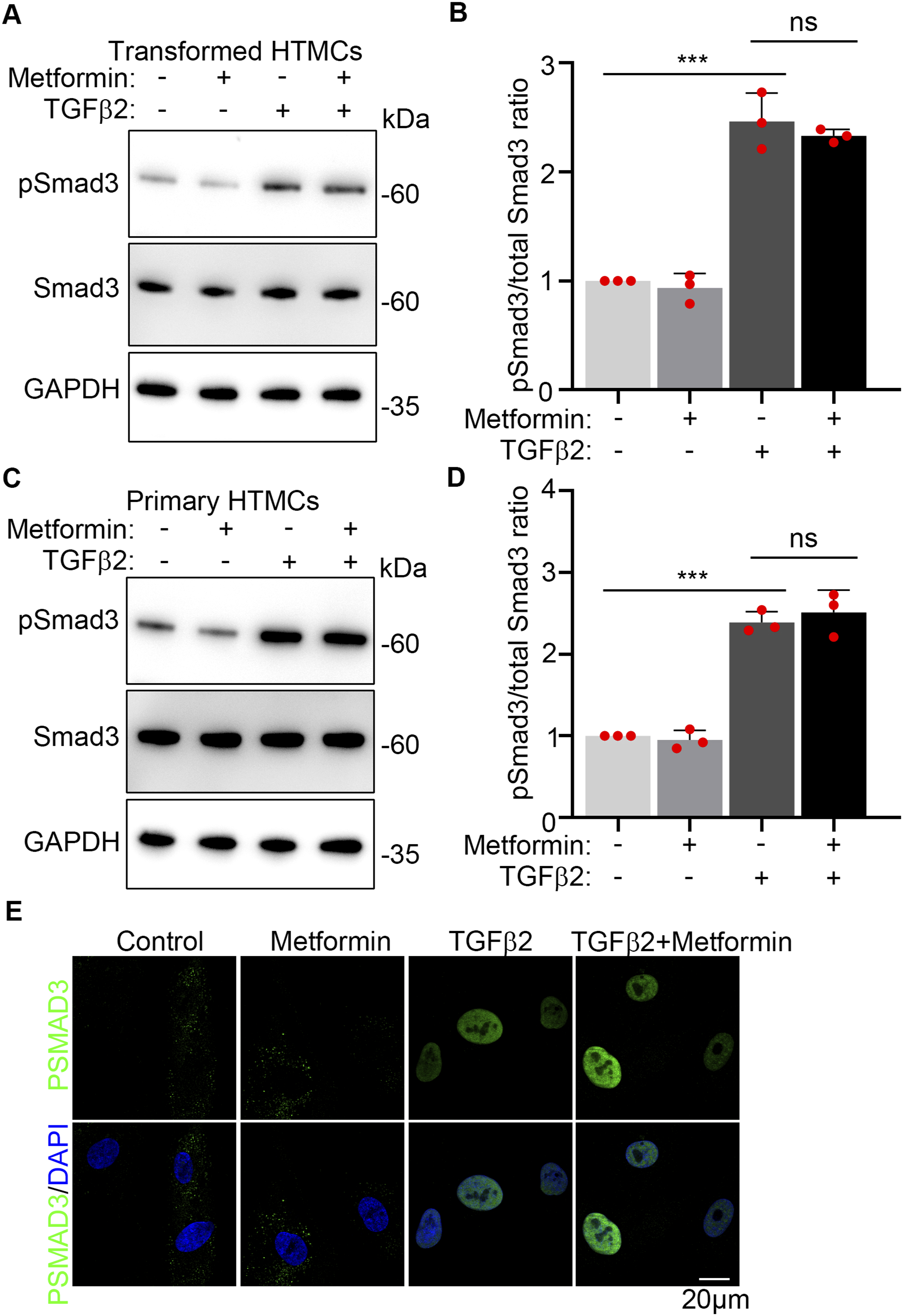
Block Autophagy/Lysosome Pathway Weaken the Protective Effect of Metformin on Fibrosis Induced by TGFβ2 in HTMCs
As protein degradation caused by autophagy activation mainly depends on lysosome pathway, we hypothesized that block lysosome pathway may weaken the protective effect of Metformin on fibrosis induced by TGFβ2 in HTMCs. Chloroquine phosphate is an inhibitor of lysosome pathway. Transformed and primary HTMCs were treated with Vehicle, Metformin (400 μM), or TGFβ2 (10 ng/mL), or treated with TGFβ2 (10 ng/mL) combined with Metformin (400 μM) for 48h. Chloroquine phosphate (10 μM) was added 12h before cells collection to conduct western blotting. Western blotting images and quantification analysis showed co-treated with Chloroquine phosphate increased the fibrotic proteins (FN, Col1, Col3 and αSMA) expression levels compared with Metformin + TGFβ2 group and strongly increased p62 and LC3II levels, indicating Chloroquine phosphate blocked autophagy-lysosome degradation pathway and weakened the protective effect of Metformin on fibrosis induced by TGFβ2 in HTMCs (Figure 8A–D). These data revealed Metformin alleviated fibrosis induced by TGFβ2 through activating autophagy-lysosome degradation of fibrosis related proteins. Autophagy/Lysosome Inhibitor Reduces the Protective Effect of Metformin on Fibrosis Induced by TGFβ2 in TM Cells. (A, C) Fibrotic Proteins Levels (FN, Col1, Col3 and aSMA) and Autophagy Markers (p62 and LC3) of Transformed HTMCs (A) and Primary HTMCs Were Detected by Western Blotting (C) after Treated With Vehicle, 400 μM Metformin, 10 μM Chloroquine Phosphate, 10 ng/mL TGFβ2, Co-treated With 400 μM Metformin, and Tri-treated With 10 μM Chloroquine Phosphate or Not. GAPDH Served as Loading Control. (C, D) Quantification of FN, Col1, Col3, aSMA and p62 Proteins Intensities after Treated in Transformed HTMCs (C) and Primary HTMCs (D). Three Independent Experiments, Data Were Showed as mean ± SD; One-Way Analysis of Variance (ANOVA), *P < .05, **P < .01, ***P < .001.
Discussion
Metformin has protective effect on glaucoma treatment, but the detail mechanism is poorly understood.11,25 Previous study found Metformin protected trabecular meshwork against oxidative injury via activating integrin/ROCK signals in Dexamethasone induced HTMCs damage model. 12 In this study, we found Metformin could activate autophagy of HTMCs and alleviated fibrosis induced by TGFβ2 in vitro and in mouse model. Chloroquine phosphate blocked autophagy-lysosome degradation pathway and weakened the protective effect of Metformin on fibrosis induced by TGFβ2 in HTMCs, indicating Metformin alleviated fibrosis induced by TGFβ2 through promotion of fibrotic proteins degradation via autophagy-lysosome pathway.
While the molecular mechanism of anti-fibrosis effect of Metformin was mainly dependent on AMPK/autophagy pathway,26,27 whether the protective functions of Metformin on HTMCs via AMPK pathway, it needs further study. The other hand, the protective roles of Metformin on RGCs also need more research in the future. Metformin also has anti-aging function,28,29 whether Metformin could inhibit HTMCs aging in human eyes and prevent glaucoma development is another topic worth to be explored.
In this study, we found Metformin could alleviates fibrosis of trabecular meshwork cells induced by TGFβ2 in mouse model, which proves Metformin has a protective effect on HTMCs. While Metformin was injected to eye anterior chamber with needles, it is worth to explore eye drops containing Metformin in mouse model to prevent fibrosis of HTMCs similar with previous reported. 30 Our study proposes a new approach to the treatment of glaucoma.
Limitations
Metformin has multiple protective roles in cells, while we just focus on its anti-fibrosis function on HTMCs, we didn’t explore its functions on other cells in the eye, such as RGCs. We just used TGFβ2 induced HTMCs fibrosis model in this study, Dexamethasone is another fibrosis inducer always used in HTMCs, we don’t test whether Metformin could alleviate fibrosis of HTMCs induced by Dexamethasone.
In the mice experiments, we used 10 mice per group, maybe it is not big enough, the other hand, we injected 1 μL 300 mM Metformin into eye of mouse, this concentration maybe too high, it need more assay to find a relative low and safe concentration of Metformin to do the mouse experiments.
Conclusion
Our study found Metformin induces autophagy activation in transformed and primary HTMCs in a dose dependent manner and efficiently ameliorates HTMCs fibrosis induced by TGFβ2 in vitro and in mouse model, and decreased elevated IOP caused by TGFβ2. Metformin treatment has no effect on TGFβ/Smad3 pathway activity, and its protective effect on HTMCs weaken by autophagy-lysosome pathway inhibitor, indicating Metformin alleviates fibrosis of HTMCs induced by TGFβ2 through the activation of autophagy and promotion of autophagy-lysosome degradation of fibrosis related proteins. Our study proposes a new approach to the treatment of glaucoma.
Supplemental Material
Supplemental Material - Metformin Alleviates Fibrosis of Trabecular Meshwork Cells Induced by TGFβ2 Through the Activation of Autophagy
Supplemental Material for Metformin Alleviates Fibrosis of Trabecular Meshwork Cells Induced by TGFβ2 Through the Activation of Autophagy by Jing Ren, Pengtao Wu, Mengli Liu, Xuan Zhang, Zilu Guo, Yihan Liu, Rumeng Zhao, Gang Wang, Shichao Duan, and Haijun Li in Dose-Response
Footnotes
Acknowledgments
The authors would like to extend their gratitude to the patients fortheir participation in the study.
Ethical Statement
Author Contributions
Haijun Li and Shichao Duan conceived and designed the experiments. Jing Ren, Pengtao Wu, Mengli Liu, Xuan Zhang, Zilu Guo, Yihan Liu, Rumeng Zhao, Gang Wang performed the experiments. Shichao Duan and Gang Wang analyzed the data. Shichao Duan, Haijun Li, and Gang Wang wrote the paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [U1904166]; Henan Medical Science and technology research plan [SBGJ2018072]; Henan province natural science foundation [242300421284].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
