Abstract
Cancer stem cells (CSCs) are implicated in the genesis, development, and recurrence of lung cancer (LC) with great resistance to radiation and chemotherapy. The aim of this study is to assess the inhibitory potential of ethanol extract of Withania somnifera (WS); 500 mg/kg body-weight/day and 8 Gy of ionizing radiation (IR) could inhibit the stemness gene and confer the radiosensitizing effect of W. somnifera extract in the female rat LC model. Compared to IR or WS, the in vitro assay showed that WS + IR potentiates proliferation-inhibition and cell death of the A-549 cell line and suppresses sphere formation. The Hedgehog (Hh) signaling associated with the expression levels of lung CSC markers, octamer-binding transcription factor-4 (OCT4), SRY-box 2 (SOX2), CD133, ATP Binding Cassette Subfamily G Member 2 (ABCG2), and NANOG was upregulated with stimulated epithelial-to-mesenchymal transition (EMT) indicators α-smooth muscle actin (α-SMA), Drosophila embryonic protein (SNAIL-1), Vimentin, and E-cadherin in the LC rat model. The W. somnifera extract plus IR inhibits Hh activation factors, which has resulted in the suppression of CSC gene markers and EMT factors. W. somnifera extract may be a significant adjuvant in the course of radiotherapy, contributing to the termination of tumor progression, and thus providing cure insights into the molecular mechanisms of lung CSCs intervention.
Introduction
A cancer cell is characterized by the 6 major hallmarks of cancer: self-sufficiency in growth signals, resisting cell death, induction of angiogenesis, insensitivity to growth suppressors, limitless replicative potential, and invasion and metastasis. Further, it has become clear that not only the cancer cell alone but also the surrounding tumor microenvironment is contributing to the hallmark properties. 1 Radiotherapy provides local control and cure for many tumor types by using high-energy rays. Under aerobic conditions, radiation-induced DNA oxidation resulting in permanent DNA damage and strand breaks. Under hypoxic conditions, the lack of oxygen enables the DNA radicals to be reduced to their original form, which that hampers the generation of strand breaks. 2 Cancer stem cells also known as tumor-initiating cells are a distinct subpopulation within a tumor that play a pivotal role in tumor development. CSCs have greater resistance to radiation and chemotherapy and thus determine the fate of the tumor and the effectiveness of radio/chemotherapy. 3 The formation and augmentation of CSCs is based on the orchestration of multiple essential transcription factors such as NANOG and the signature genes OCT4, SOX2, and proto-oncogene protein C-Myc, which are linked with the progress of CSCs. 4 It has been documented that lung cancer cells that have undergone ionizing radiation treatment show complex phenotypes, including CSCs and EMT factors such as cytoskeletal proteins α-SMA and Vimentin, cell-surface proteins E-cadherin and transcription SNAIL1. 5 Another study found that liver cancer cells exhibit a greater capacity for self-renewal and promote radio-resistance in the HepG2 cell line. 6 Earlier, studies highlighted that Hedgehog signals mediating the transfer and invasion of cancer epithelial cells to the mesenchyme have an essential role in CSCs growth. The hedgehog pathway is essential for embryogenesis and tissue renewal in vertebrate development, homeostasis, and tumorigenesis. 7
W. somnifera, commonly known as ashwagandha, belonging to the family Solanaceae, is one of the enormously interesting medicinal plants with pharmaceutical applications. Previous research has found it to have anti-ulcer, anti-inflammatory, anti-hepatotoxic, antioxidant, antitumor, and immunomodulatory properties.8-10 The anticancer activity of W. somnifera relates to its multiple functions, as it may increase the overall effectiveness of cancer treatment. Besides that, the anticancer activity is probably due to the action of its main constituents, namely, Withaferin-A and withanolide-D, which inhibit RNA-dependent protein kinase, which leads to increased cancer cell death. 11 Considering the need for the introduction of new drugs into the market, plants have an added advantage when compared with synthetic ones, as their molecular diversity and consequent changes in biological function are more complex. Concerning research and development of new potential herbal medications, screening and evaluation of their phytopharmacological effects are essential. 12
Several experiments are being carried out to develop new radiosensitizers, in which the low toxicity and low cost of natural drugs have drawn considerable interest. In a search for an agent that inhibits proliferation and invasion of the lung cancer CSCs, it was found that an ethanolic extract derived from an Asian herb, W. somnifera, which is a nightshade medicinal plant that contains active components to treat a variety of ailments, including cancer, and reduce various effects of metastasis. 13 However, the role of W. somnifera extract in radio-sensitivity and its molecular mechanism is unclear. Therefore, the present study aims to investigate whether W. somnifera extract can overcome the resistance to IR through inhibiting hedgehog signaling-driven stemness by regulating the expression of OCT-4 and C-Myc.
Materials and methods
Material and Kits
Unless stated otherwise, all chemicals used in these studies were obtained from Sigma Chemical (St Louis, MO). The rat ELISA kits were purchased from CUSABIO Technology, LLC, Houston, TX, USA, for the estimation of Vimentin, E-cadherin, C-Myc, BAX, and BCL-2 according to the protocol provided by the manufacturer. TGF-β levels were measured using an ELISA kit purchased from BD Biosciences (San Jose, CA) and carried out according to the manufacturer’s instructions. The sample extinction values were determined using an ELISA plate reader (Biotek, USA) at 450 nm.
Methods
Preparation of the ethanolic and water extracts of W. somnifera
The W. somnifera herb was collected from the Agriculture Research Center, Egypt. The herb was pulverized in an electric grinder. Water and ethanol were used to prepare the plant extract. 10 g of fresh herb was kept in a conical flask and 50 mL of distilled water or 50 mL of ethanol (80%) were added, kept in a shaker for 25 min for continuous agitation. Then, extracts were filtered by using muslin cloth followed by Whatman No. 1 filter paper.
Determination of the active compounds in the ethanolic and water extracts of W. somnifera
The content of total phenols in the ethanolic extract of W. somnifera was measured by a spectrophotometric determination using a modified Folin–Ciocalteu method described by Singleton et al. 14 Total flavonoid was determined by an aluminum chloride colorimetric method based on the method developed by Chang et al. 15 The content of total alkaloid and total saponin was measured according to the methods described by Shamsa et al. 16 and Hiai et al., 17 respectively.
Determination of the antioxidant capacity of the ethanolic extract of W. somnifera
The antioxidant capability of ethanolic extract of W. somnifera was studied through the evaluation of the free radical scavenging effect on the DPPH radical based on the method proposed by Ferreira et al. 18 Also, a ferric reducing antioxidant power assay (FRAP) was carried out using the method of Benzie and Strain. 19
Fractionation of phenols by HPLC
High performance liquid chromatography (HPLC) analysis was performed using 4 different gradients of mobile phase (The graphics in the supplementary material). Elution of 2 solvents was used: solvent A (methanol) and solvent B (acetic acid in water, 1:25). 20
Extraction and fractionation of alkaloids for HPLC analysis
One gram of the herb sample was accurately weighed and dissolved in 50 mL methanol HPLC grade. Further, 1 mL of this solution was diluted to 50 mL using methanol HPLC grade. 20 μl of standard and sample solutions were injected into HPLC. The content of alkaloids was calculated in comparison with the standard HPLC estimation of Withaferin-A performed on the Shimdzu 10 as HPLC system, equipped with UV detector 21 (the graphics in the supplementary material).
In Vitro Study
Cellular toxicity study
A human non-small cell lung cancer (NSCLC-A-549) obtained from the Vacsera Vaccination Centers, Cairo, Egypt, was used to determine the cytotoxic effect of W. somnifera extract according to Vijayan et al. 22
Subculture cell line and culture media
To verify the degree of confluence and to confirm the absence of contaminants (such as bacterial and fungal), the cultures were viewed using an inverted microscope (CKX41; Olympus, Japan). Briefly, the cell monolayer was washed with phosphate-buffered saline (PBS) free of Ca2+/Mg2+, using a volume equivalent to half the volume of the culture medium. Then, trypsin/EDTA was added to the washed cell monolayer using 1 mL/25 cm2 of surface area, and the flask was rotated to merge the monolayer with Trypsin/EDTA. Then, the flask was left for 10 minutes in the incubator. After that, the cell monolayer was examined using an inverted microscope to ensure that all the cells were detached and floated. The cultured cells were kept in a humid environment at 37°C and 5% CO2 in RPMI1640 medium supplemented with penicillin (100 IU/mL), streptomycin (100 μg/mL), and 10% fetal bovine serum (FBS). To maintain a culture media enriched with a high glucose concentration, Dulbecco’s minimum essential medium eagle (DMEM) was supplemented with 10 mM glucose, 1 mM glutamine, 10% heat-inactivated dialyzed fetal calf serum (56°C, 30 min), 100 U/mL penicillin G, 100 μg/mL streptomycin, and fungizone (250 μg/L).
Cell viability assay
To investigate the cytotoxic effect of ethanol extract of W. somnifera on A-549 NSCLC human cell line, the crystal violet assay was performed as earlier described. 23 A-549 cells were plated onto 96-well culture plate and exposed to various concentrations of W. somnifera extract (3.9, 7.8, 15.6, 31.25, 62.5, 125, 250, and 350 μg/mL), in addition to untreated cells of A-549 cell line that served as a control. The plate was incubated for 24 hours in a humidified incubator at a temperature of 37°C and 5% CO2 atmosphere. The medium was then aspirated at the end of the incubation period. The cells were washed using 100 μL of PBS. The MTT (3-[4,5-dimethylthiazol-2-yl]-2.5-diphenyl tetrazolium bromide) reagent was added (10 μL/well) and the culture plate was incubated for 4 hours allowing for the intracellular reduction of the soluble yellow MTT into the insoluble purple formazan crystals. Finally, the dye was extracted from the cells by the addition of the iso-propanol/HCl mixture was prepared (2 mL of .1 N HCl in 23 mL isopropanol) with each well. The resulting-colored solution was quantified by measuring the absorbance at 490 nm in triplicates using an ELISA reader BioTekTM EonTM microplate reader, (Winooski, TV, USA). The cytotoxicity was assessed as the end point of crystal violet reduction and the quantitative external morphology of exposed cells in relation to control cells was assessed by an inverted microscope. The percentage of cell survival was calculated as the percentage of the ratio of the optical density (OD) as shown by the following equation: cell survival % = ODtreated/ODcontrol × 100.
Radiation exposure in vitro
First, the cultured A-549 cells line was irradiated with doses of 4, 6, and 8 Gray (Gy) to choose the optimum dose. A constant source-to-surface distance of 40 cm was employed with a fixed exposure area. Irradiation was carried out at the National Center for Radiation Research and Technology (NCRRT), Atomic Energy Authority, Egypt using a Gamma Cell 220 high dose rate laboratory irradiator (Atomic Energy of Canada Limited, Mississauga, Ontario, Canada) with an Indian 60 Co cell at a dose rate of 1.144 K Gy/h.
The A-549 cultured cells were divided into 4 groups: Group 1 (A-549): untreated A-549 cells, Group 2 (A-549 + W. somnifera extract): A-549 cells treated with W. somnifera extract (350 μg/mL dissolved in DMSO at a final concentration of .1%), Group 3 (A-549 + IR): A-549 cells exposed to 8 Gy, and Group 4 (A-549 + W. somnifera extract + IR): A-549 cells treated with W. somnifera extract and exposed to 8 Gy.
Colony survival assay
The formation of colonies was determined according to the method of Weigel et al. 24 To assess the surviving fraction of A-549 cells incubated with W. somnifera extract and/or IR, the cells (3000 cells/mL) were suspended in 20 mL of culture medium and treated with vehicle (DMSO) or W. somnifera extract (350 μg) for 24 h and then were exposed to the indicated doses of IR (4, 6, and 8 Gy) or integration of both at 37°C in a humidified atmosphere of 5% CO2 for 2 weeks to allow the formation of macroscopic colonies on polymerized bactoagar plates. After the incubation period, colonies were stained with .5% crystal violet in methanol/acetic acid (7:1) and the colonies were counted microscopically. The numbers of colonies containing ≥30 cells were determined. The surviving fraction of A-549 cells was determined by the clonogenic assay and calculated relative to the non-irradiated control. Experiments were performed in at least triplicate, and the mean and standard error were calculated.
Spheroid-formation assay
The formation of a spheroid was determined according to the method of Ivanov. 25 Cells were suspended in the cultured medium to a density of 2.5 × 104 cells/mL on plates to evaluate the ability of human A-549 cells in non-adherent environments. The cells were subjected to varying doses of IR (4, 6, 8 Gy) or incubated with W. somnifera extract (350 μg), or incubated with W. somnifera extract (350 μg) 48 hrs and then subjected to the varying doses of IR (WS+IR). The cells were washed with PBS, trypsinized, and 5 × 104 cells were then cultured in β-mercaptoethanol .1 mM and insulin transferrin selenium-ethanolamine of 1/100 v/v. The drop hanging system contained 20 μL of volume, and the base of the plates contained 10 mL of PBS. Then, the plates were incubated for 2 days at 37°C with 5% CO2. The formed spheres were transferred to an uncoated plate, and images of spheroid progress were examined by light microscopy and counted.
The proliferation assay
The cell proliferation of the (A-549) cell line in all groups was estimated using the MTT cell proliferation kit from Trevigen Inc., Gaithersburg, MD, USA, as per the manufacturer’s protocol. Briefly, (5 × 103) cells were seeded in a 96-well microplate in the absence or presence of IR (8 Gy) and/or W. somnifera extract (350 µg) in a total volume of 100 μL of cell culture medium and were then allowed to attach overnight. To start the coloring reaction, the stock solution was diluted to a final concentration of .5 mg MTT/mL. The MTT reagent was added (10 µL/well) and the culture plate was incubated for 4 hours, allowing for the intracellular reduction of the soluble yellow MTT into the insoluble purple formazan crystals. The iso-propanol/HCl mixture was prepared (2 mL of .1 N HCl in 23 mL isopropanol) was added to each well to solubilize the formazan crystals and produce a colored solution. The intensity of the formazan solution was determined by measurement of the absorbance at 550-600 nm using a microplate reader (BioTekTM EonTM microplate reader, Winooski, VT, USA). 23 Six wells were used for each group. Cell proliferation was assessed as the percentage of cell proliferation compared to the untreated A-549 cells as a control.
In Vivo Study
Animal’s study
Fifty Wister female rats, 5 weeks old and weighing 160 ± 10 g collected from the animal house in NCRRT, Cairo, Egypt, were used as experimental animals. The animals were kept under standard laboratory conditions of light and dark cycles (12 × 12 hrs.), temperature (25 ± 2°C), and humidity (60 ± 5%). The rats were housed in cages with free access to food and drinking water ad libitum. All animal procedures were carried out in accordance with the Ethics Committee of the National Center and conformed to the “Guide for the Care and Use of Laboratory Animals” published by the US National Institute of Health (NIH publication No. 85-23, 1996), with the use of laboratory animals approved by the Research Ethics Committee (REC) for animal experimental studies at the NCRRT-Egyptian Atomic Energy Authority, Cairo (serial no. of the study is 193/3/20).
Lung cancer induction
Lung cancer was provoked by injection of freshly prepared ethyl carbamate (urethane) by dissolving 1.5 mg/kg in .9% saline by intraperitoneal injection following the method of Forkert. 26 Urethane was administered in 2 doses to each animal and observed for 18 weeks.
Determination of the LD50 of W. somnifera extract in experimental animals
According to OECD guideline 423, the acute oral toxicity method set out in this guideline is a stepwise procedure with the use of 3 animals of single-sex per step. Depending on the mortality and/or the moribund status of the animals, on average, 2–4 steps may be necessary to allow judgment on the acute toxicity of the test substance. 27 The method allows for the determination of an LD50 value when at least 2 doses result in mortality higher than 0% and lower than 100%. We have performed the screening of the W. somnifera extract. LD50 is typically an early step in the calculation and estimation of a substance’s toxic property.
Radiation exposure in vivo
The animals were exposed to irradiation, at a dose of 8 Gy fractionated in 4 doses (2 Gy/week), used an indoor-shielded was performed at the National Center for Radiation Research and Technology of the Atomic Energy Authority (NCRRT, Cairo, Egypt) using an indoor shielded Canadian gamma cell-40 (Cesium-137) at a dose rate of .67 Gy min−1.
Experimental design
After 18 weeks, the establishment of the rats' of lung cancer was confirmed by histology examination and degree, rats were randomly divided into 5 groups (n = 10): Group 1 (Control): Health rats, Group 2 (LC): Rats were used as a positive control and were administered urethane as described above to induce lung cancer in rats. Group 3 (LC + WS): LC rats were orally gavage with W. somnifera ethanol herb extract (500 mg/kg body weight/day), for 8 weeks. Group 4 (LC + IR): LC rats were subjected to repeated doses of ionizing radiation (2 Gy/week up to 8 Gy) once a week for the first 4 weeks, of the experimental period of 8 weeks. Group 5 (LC + WS + IR): LC rats were administered with W. somnifera ethanol herb extract one hour before the first fraction of IR and continued daily for 8 weeks. At the end of the study, rats of each group were fasted overnight before the time of sacrifice by cervical dislocation, and the blood was collected by retro-orbital sinus puncture. Part of the lung tissue was collected for molecular and biochemical analysis.
Detection of rat stemness genes OCT4, SOX2, CD133, ABCG2, NANOG, Hedgehog pathway genes expression, Sonic Hedgehog (SHH), Patched homolog-1 (PTCH-1), Smoothened (SMO), glioma-associated oncogene-1 (GLI1), and the expressions of EMT markers (SNAIL1, α-SMA) using quantitative real-time-polymerase chain reaction (qRT-PCR)
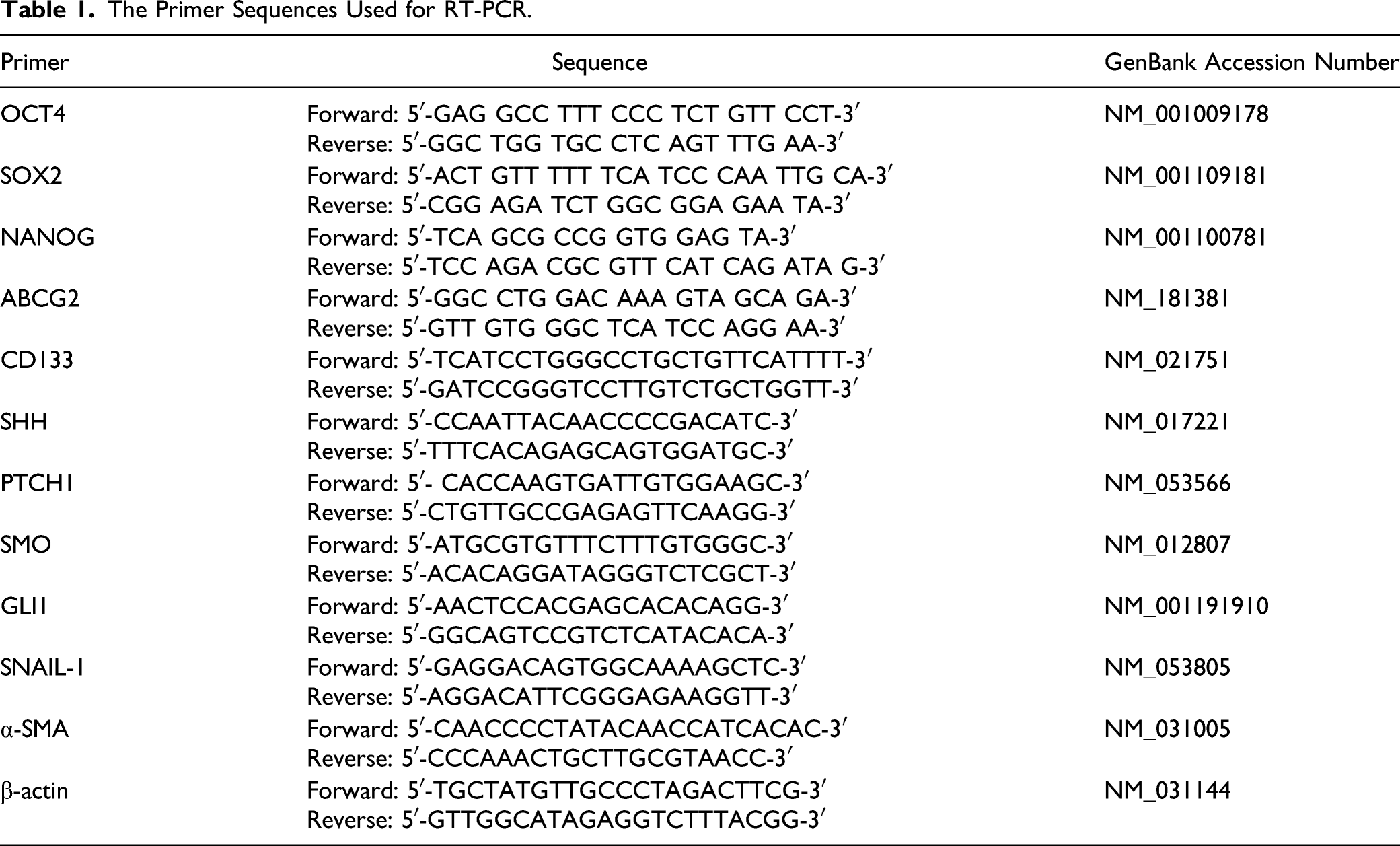
The Primer Sequences Used for RT-PCR.
Histological examination
Lung tissue was collected from all rat groups and excised, rinsed in saline solution, and cut into suitable pieces which were fixed in neutral buffered formalin (10%) for 24 h. The fixed specimens were trimmed, washed, and dehydrated in ascending grades of alcohol, cleared in xylene, embedded in paraffin, sectioned at 4–6 μm thickness, stained with hematoxylin and eosin according to, and examined by light microscope for histopathological investigation as described by Bancroft et al., 29 and examined by light microscope for histopathological investigation. The response of tumor mass to treatment was divided into the following: Grade I marginal or no regression; Grade I b, morphologic evidence of therapy-induced changes but >10% residual tumor; Grade II a, extensive response but with residual tumor ≤10%; and Grade II b, pathologic complete response. 30
Statistical Analysis
The data were statistically analyzed using SPSS computer Program (v. 17). The results were presented as Mean ± SE. The differences between mean values were analyzed by one-way analysis of variance (ANOVA test) followed by LSD as a post hoc test for multiple comparisons. All statements of significance were based on probability of P ≤ .05.
Results
In Vitro Studies
Active compounds in W. somnifera
Active Compounds in Withania somnifera.

Fractionation of phenols and alkaloids in the ethanol extract of Withania somnifera
Fractionation of Phenols in the Ethanol Extract of Withania somnifera Using HPLC Analysis.

Fractionation of Alkaloids in the Ethanol Extract of Withania somnifera Using HPLC Analysis.

The W. somnifera extract inhibits the cellular proliferation, colony survival, and sphere-forming in A-549 cancer cells
The antitumor activity of ethanol extract of W. somnifera using the MTT assay was evaluated using human A-549 cells' capability. The herb ethanolic extract was applied at different concentrations and the results are presented in Figure 1A. The cytotoxic effects against A-549 cells were detected under these experimental conditions with IC50 = 350 ± 9.1 μg/mL. Effect of ionizing radiation (IR); ethanolic extract of Withania somnifera (WS); and WS + IR on A-549 cells proliferation: (A) Cell viability % of human A-549 cells using different concentrations of WS (B) Clonogenic assay for Human A-549 cells treated with IR (4, 6, and 8 Gy), WS (350 μg/mL); and WS + IR (4, 6, and 8 Gy), (C) Colony surviving fractions of A-549 cells exposed to IR (4, 6, and 8 Gy), WS, and WS + IR (4, 6, and 8 Gy). * P < 0.05, ** P < 0.01, and *** P < 0.001, compared to untreated A-549 cells. (D) The sphere formation assay in serum-free medium showing the spherogenic potential inhibition of IR (4, 6, and 8 Gy), WS, and WS + IR (4, 6, and 8 Gy), compared to A-549 cells, 50 m scale bars. (E) The number of spheres formed in A-549 cells treated with IR (4, 6, and 8 Gy), WS, and WS + IR (4, 6, and 8 Gy), vs A-549 cells. (F) WS or/IR (8 Gy), effect on the cell survival of A-549 cells. A-549 (untreated A-549 cells), IR (A-549 cells exposed to 8 Gy), WS (A-549 cells treated with WS), WS + IR (8 Gy) A-549 cells treated with WS and exposed to 8 Gy. The sphere formation and cell proliferation assay results are expressed as mean ± standard error (n = 3). Columns with different superscripts are significantly different at P < 0.05 by one-way ANOVA.
To estimate whether extract of W. somnifera increases cellular sensitivity to IR, A-549 cells were incubated with W. somnifera extract (350 μg/mL) for 24 h before being exposed to 3 different doses of IR (4, 6, and 8 Gy). We found that W. somnifera extract inhibits the proliferation and colony survival of A-549 cells (Figure 1B). Furthermore, sphere-forming capacity was greatly diminished in WS + 8 Gy treated A-549 cell lines (Figure 1D and 1E). Also, we found that, W. somnifera extract radiosensitizes A-549 cells, and the combination of W. somnifera extract and IR was superior to each treatment alone (Figure 1F). These records point to W. somnifera extract potently represses proliferation and colony survival of A-549 cells alone and in combination with IR (Figure 1B, 1C, and 1F).
In Vivo Studies
The combination of W. somnifera extract and IR reduces CSCs in a rat lung cancer model
To assess the potential combined effects of W. somnifera extract and IR on the Hedgehog pathway in NSCLC, analysis of the transcription factors SHH, PTCH1, SMO, and GLI-1 gene expression were performed (Figure 2A: D). The LC group showed a significant upregulation in the gene levels of SHH, SMO, and GLI-1 with respect to the health rat group. Groups receiving W. somnifera extract or IR treatment had lower gene levels of these gene factors compared to the LC group. Remarkably, the combination of W. somnifera extract and IR significant suppressed (P < .05) the expression levels of SHH mRNA, SMO mRNA, and GLI-1mRNA in respect to the LC and LC + IR group. These data indicate that W. somnifera extract may influence radiation to inhibit the SHH pathway in lung cancer rats. The effect of ionizing radiation (IR, 8 Gy), WS, and IR + WS on the regulation of the Hedgehog pathway in a lung cancer (LC) rat model: (A) Relative expression of SHH mRNA, (B) PITCH1 mRNA, (C) SMO mRNA, and (D) GLI-1 mRNA measured by reverse transcription polymerase chain reaction, and normalized to the housekeeping gene actin mRNA expression. The results are presented as Mean ± SE (n = 6 rats). The columns values with different superscripts are significantly different at P < 0.05.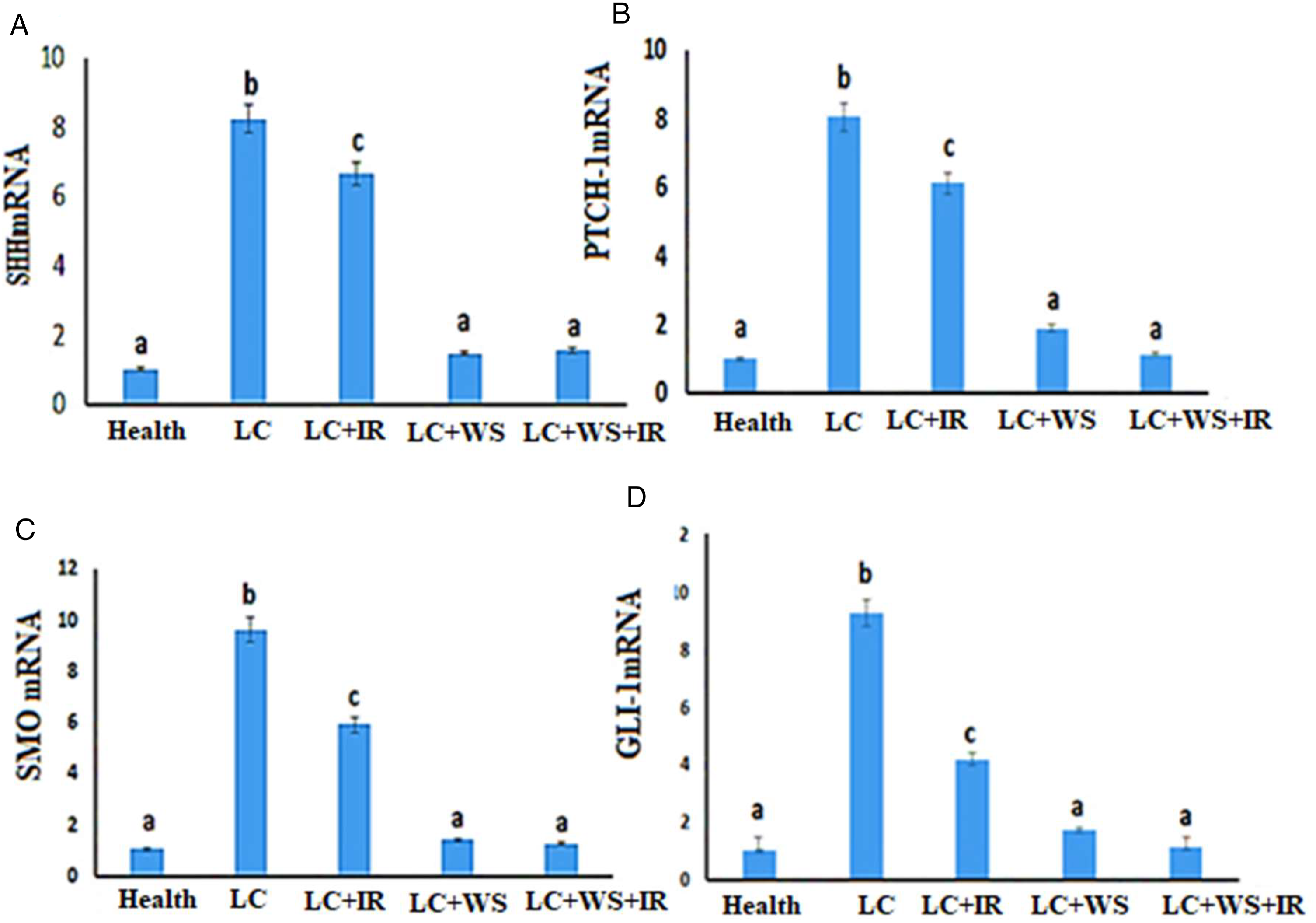
The impact of W. somnifera extract and IR inhibition combined on the CSCs’ gene markers
Radiation promotes cancer cell death induction. In the case of CSCs pro-survival pathways are observed, it seems that these cells shield themselves from cell death induced by radiation.
31
The results in Figure 3A: F, illustrates that the mRNA levels of OCT4, SOX2, CD133, ABCG2, NANOG, which are the lung cancer stem cell markers were elevated significantly in LC, and LC + IR relative to the control group (P < .05). Treatment with W. somnifera extract has significantly attenuated these increases compared to their respective values in the LC and IR groups. CSCs, which may promote stem cells self-renewal and differentiation has been related to the high expression level of pluripotent transcription factors, including OCT-4, SOX2, and C-Myc.
32
To clarify the role of W. somnifera extract and ionizing radiation on CSCs target genes markers in the lung cancer rat model, the present study examined the expression of these genes. As revealed in Figure 3A and 3F, compared with their LC group, the expression levels of OCT-4 and C-Myc were decreased significantly (P < .05) in the IR treated LC model rat. Compared to the LC group, treatment with W. somnifera extract significantly decreased the CSCs gene indicators as compared to IR group. Overall, these data revealed that the synergistic effect of W. somnifera extract with IR could effectively diminish the characteristics of lung CSCs. W. somnifera extract might be a promising adjuvant therapeutic agent for the eradication of stemness lung cancer. The effect of ionizing radiation (IR, 8 Gy), WS, and IR + WS on the expression of cancer stem cells target genes in lung cancer (LC) tumor tissues in vivo: (A) relative expression of OCT4mRNA, (B) SOX2 mRNA, (C) CD133 mRNA, (D) ABCG2 mRNA, and (E) NANOG mRNA and (F) C-Myc mRNA measured by reverse transcription polymerase chain reaction and normalized to the housekeeping gene actin mRNA expression. The results are presented as Mean ± SE (n = 6 rats). The columns with different superscripts are significantly different at P < 0.05.
EMT epithelial cells are known to play vital roles in the progression of cancer. As shown in (Figure 4A: E), the levels of TGF-1β (ng/g tissue) which is the main angiogenesis promoter during lung cancer, vimentin (ng/mg tissue) interstitial marker and α-SMA mRNA were elevated in LC and LC + IR, while the E-Cad (pg/mg tissue) epithelial marker declined significantly (P < .05) in LC and LC + IR relative to the control group. Treatment with Withania somnifera extract has significantly attenuated the increase in TGF-1β, and vimentin levels, and α-SMA mRNA expression (Figure 4A, 4B, and 4E) and the decrease in E-Cad (Figure 4C). Furthermore, the expression of transcription factors SNAIL-1 mRNA marker of EMT in lung tissue was significantly augmented (P < .05) in rats exposed to LC and LC + IR compared to their respective values in the health group. Following these findings, W. somnifera extract treatment with IR diminishes the expression of SNAIL-1 (Figure 4D). The effect of ionizing radiation (IR, 8 Gy), WS, and IR + WS on epithelial-mesenchymal transition markers in lung cancer (LC) tumor tissues in vivo: (A) TGF-β1, (B) Vimentin, and (C) E-Cad levels were measured using ELISA. (D) The relative expression of SNAIL-1 mRNA and (E) α-SMA mRNA was determined using reverse transcription polymerase chain reaction and normalized to the expression of the housekeeping gene actin mRNA. The results are presented as Mean ± SE (n = 6 rats). The columns with different superscripts are significantly different at P < 0.05.
The impact of Withania somnifera extract in combination with IR on apoptosis in the LC rat model
The effect of W. somnifera extract combined with IR on the apoptosis of LC is illustrated in Figure 5A and 5B. Compared to the healthy rat group, the apoptotic BAX level of LC treated by W. somnifera extract or IR significantly increased, associated with a BCL2 decline, and the WS + IR group exhibited a higher apoptotic level. These findings indicate that W. somnifera extract induces LC apoptosis and that combined treatment increases apoptotic levels, implying that W. somnifera extract enhances the cytotoxic effect of radiation on tumor. Regulation of apoptosis-related factors by ionizing radiation (IR, 8 Gy), WS, and IR + WS in a lung cancer (LC) model: (A) BCL2 and (B) BAX levels were assessed by enzyme-linked immunosorbent assay. The results are presented as Mean ± SE (n = 6 rats). The columns with different superscripts are significantly different at P < 0.05.
Histological Examination
Examination of the lung tissues in rats of healthy control groups showed normal lung architecture, thin inter-alveolar septa, folded columnar epithelial cells of bronchiole, clearly seen alveolar sacs, normal pulmonary vessels, and normal fibrous tissues distribution. The alveoli appeared inflated with thin inter-alveolar septa. They were lined mostly by squamous type I pneumocytes and a few large cuboidal type II pneumocytes (Figure 6A). The lung of carcinogenic animals group showed undifferentiated tumor mass typically non-small cell lung carcinoma characterized by moderate nuclear pleomorphism, prominent nucleoli, and a moderate amount of cytoplasm. Central “infarct-like” coagulative necrosis was present with a rim of histiocytes and lymphocytic reaction at the edge of the necrosis. In zones at the edge of the necrosis were macrophages with foamy cytoplasm, cholesterol clefts, and multinucleated giant cells (Figure 6B). The histological picture of lung tissue section of animals group treated by Withania somnifera extract not completely varied from untreated group. The responsibility of cancerous mass Grade I b. The neoplastic area showed mixture of foam cells and giant cells, in addition to infarct-like coagulative necrosis and minimal fibrosis. The histological growth pattern of the carcinomas was unchanged by therapy (Figure 6C). In lung tissue section of animals group treated by IR, alternation was seen in pathologic responders circumscribed area of widespread necrosis is showed with a rim of fibrosis, chronic inflammatory infiltrate. Morphologic indication of treatment-induced changes but >10% residual tumor was seen, Grade I b (Figure 6D). The tumor mass shrinkage of the lung tissue slice of animal groups treated by combination of IR and W. somnifera extract was discovered in contrast to the radiotherapy alone animal groups. The cancerous mass was Grade I b. The neoplastic area showed mixture of foam cells and giant cells, in addition to infarct-like coagulative necrosis and minimal fibrosis (Figure 6E). Photomicrographs of sections of rat lung tissues in all groups (H&E × 200): (A) Health rat group: normal lung architecture, thin inter-alveolar septa, folded columnar epithelial cells of the bronchiole, normal pulmonary vessels. (B) LC group: an undifferentiated tumor mass, most commonly non-small cell lung carcinoma, with moderate nuclear pleomorphism, prominent nucleoli, and a moderate amount of cytoplasm. A central “infarct-like” coagulative necrosis was present with a rim of histiocytes and lymphocytic reaction at the edge of the necrosis. In zones at the edge of the necrosis were macrophages with foamy cytoplasm, cholesterol clefts, and multinucleated giant cells (arrow). (C) LC + WS group: an infarct-like coagulative necrosis and a fibrotic rim (arrow). The cancerous mass (Grade Ia) is responsible for the neoplastic area, which showed a mixture of foam cells and giant cells. The histological growth pattern of the carcinomas was unchanged by therapy. (D) LC + IR group: The alternation of coagulative necrosis of neoplastic areas (arrow) was detected. It showed a circumscribed area of widespread necrosis with a rim of fibrosis and chronic inflammatory infiltrate. The morphologic sign of treatment-induced change was that >10% residual tumor was seen (Grade I b). (E) LC + WS + IR group: reduction of the tumor mass surrounded by a thick layer of fibrous connective tissues and chronic inflammatory cell infiltration (arrow) was observed. A (Grade IIa), extensive response but with a residual tumor <10% was recorded.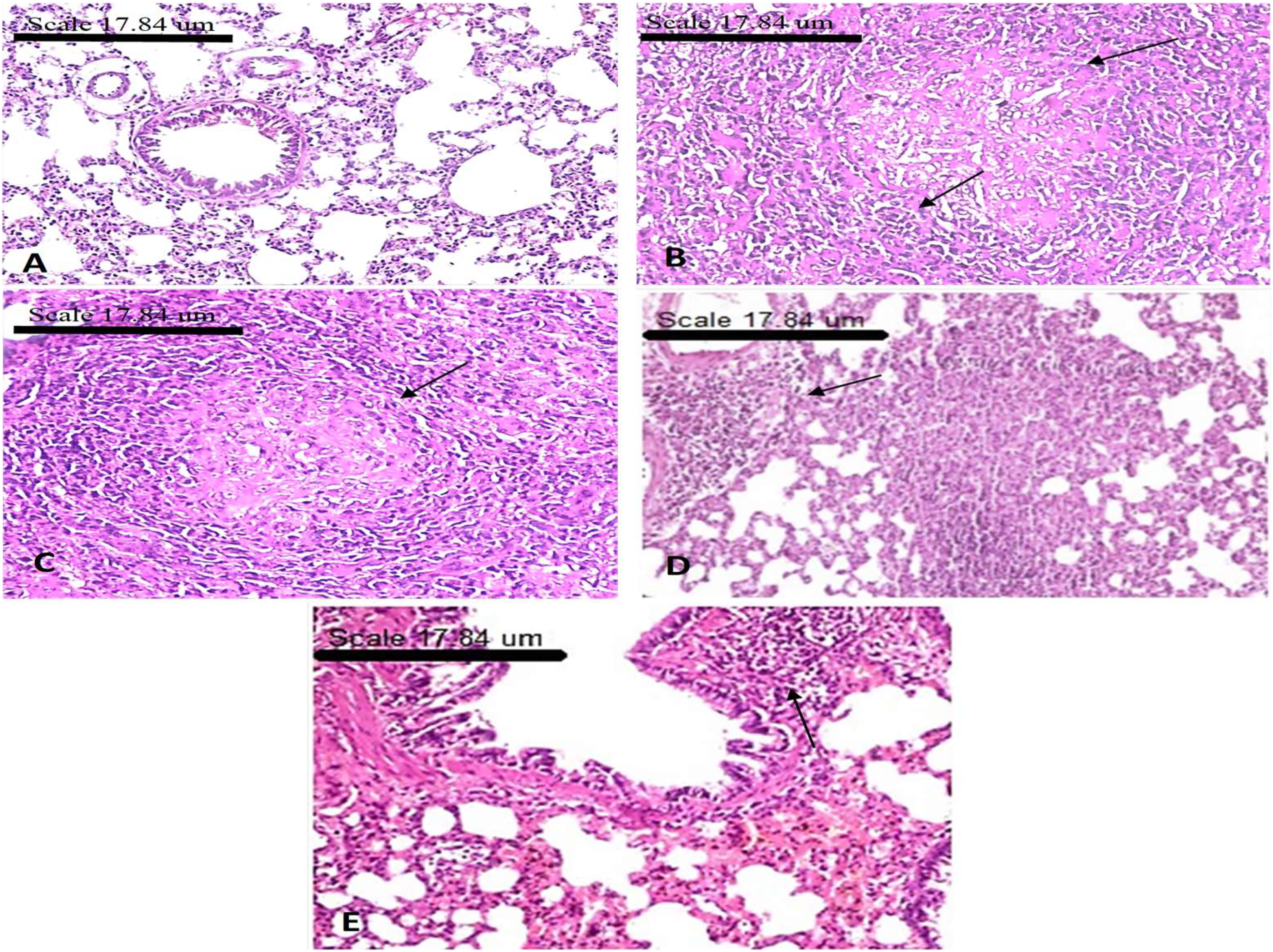
Discussion
Adjunctive therapy such as the application of radiosensitizer is often used to decrease the cell resistance to radiation which is a major cause of the low survival rate in NSCLC patients. Recently, the radiosensitizing potential of multiple drugs has been explored, including synthetic compounds, and natural agents, and molecular targeted drugs. 33 However, there are no clinically validated radiation sensitizers so far. Among those radiosensitizers, natural drugs and their monomers have better application prospects due to their low toxicity, better therapeutic effect, easy accessibility, and low cost. Since the activation of the Hedgehog (Hh) signaling pathway is involved in enhancements of CSCs induced resistance to chemo-radiotherapy, blocking of such a pathway might be helpful to overcome CSCs and radioresistance. Our findings show that Withania somnifera extract can boost the cytotoxic effects of IR in resistant NSCLC in vitro and in vivo via the interconnection of Hh pathway with CSCs signature gene and EMT aggregation.
The hedgehog pathway is vital for cell proliferation, differentiation, apoptosis, and invasion. It was reported that stimulation of Hh is involved in the regulation of self-renewal of CSCs in many cancers, including NSCLC. 34 GLI-1 regulates certain genes that control cell survival, and can activate various downstream proteins such as SHH, PTCH-1, and SMO in addition to CSCs markers including OCT4, SOX2, CD133, ABCG2, NANOG, and proto-oncogeneses C-Myc that regulate cell cycle. 35 The activated Hh cascade is closely related to radiotherapeutic resistance in lung cancer. 36 The hedgehog cascade decreased sensitivity to radio-chemotherapy via regulating the EMT and E-cadherin expression in cancer cells. Further, overexpression of the ATP-binding cassette transporters (ABCG2) that translocate intracellular and exogenous substances outside the cells in many tumor cells is also a leading cause for therapeutic resistance. 37
The present results revealed marked decreases in GLI-1, SHH, PTCH-1, and SMO mRNA expression in the W. somnifera extract plus IR co-treated group with respect to the IR-treated group. These are in accordance with White et al. 38 who found that withanolides from W. somnifera treatment resulted in notable declines in the gene expression levels of SHH in cancer tissue. Similar findings were reported in human pancreatic, prostate, and breast cancer cells. 39 The inhibition of the Hh signaling pathway by W. somnifera extract supports its vital role in enhancing the anticancer effect of IR in NSCLC.
The ethanol extract of W. somnifera (WS) has been used in ayurvedic medicine for its anticancer properties. The diverse effects are attributed to the presence of active steroidal compounds that are called withanolides. Our current data showed that the W. somnifera extract inhibited proliferation and migration of A-549 cells in vitro which is in agreement with Kyakulaga et al. 40 study. This inhibition was greater in combination with exposure to 8 Gy of ionizing radiation than that caused by IR alone as revealed in the colony and formation spheroids assay. The difference in inhibition may be attributed to the fact that W. somnifera extract contains active ingredients that have a synergistic effect that promotes IR cytotoxicity against cancer cells. 41 Previously, it was found that W. somnifera extract increased the percentage of MDA-MB-231 in the sub-G1 phase, indicating that it promotes apoptosis. 42 Withaferin-A, one of the active components in W. somnifera extract, causes G (2)/M cell cycle arrest, as well as modulation of Cyclin B1, p34 (cdc2), PCNA levels, and STAT3 levels. In addition, its phosphorylation and influence on the levels of p53 induce the apoptotic markers BAX, caspase-3, and PARPs. 43
In spite of the marked contribution of the Hh pathway to CSC initiation, growth, metastasis, and relapse, its role is frequently interconnected with other factors. Recently, studies have shown that CSCs are one of the causes of the recurrence of cell carcinoma after radiotherapy. 44
Significant positive correlation was reported between Hh signal and TGF-β1 in breast cancer, where elevated TGF-β1 significantly boosted the expression of SHH/GLI1 axis changes EMT expression markers and revokes cell malignancy including migration and invasion.45,46 In our study, notable elevation in TGF-β level and EMT markers including raises vimentin levels, and α-SMA mRNA expression. Furthermore, the elevation expression of transcription factors SNAIL-1 mRNA marker with decline of E-cadherin expression was detected in LC group in relation with health group while the IR, W. somnifera extract, or their combination lessened markedly its tissue level and normalized the E-cadherin that was confirmed by pathological examination. These results support the anticancer role of W. somnifera extract in controlling IR resistance via regulating Hh pathway and its crosstalk with EMT in lung cancer rat model. The hedgehog-signaling pathway has been also shown to be activated in CSCs in NSCLC. 38 So, we could explain the increased gene expression of CSCs tissue-specific cell surface markers, including ABCG2, OCT4, SOX2, CD133, NANOG, and proto-oncogene C-Myc in LC rat tissues.
In addition, the regulation of apoptotic pathways is a principal target for radio and chemotherapeutic drugs to fight cancer cell cycle progression. 47 During stressful stimuli, BAX protein activation takes place and phosphorylates BCL-2, which showed a marked down regulation in its mRNA expression induced either by W. somnifera extract and IR or their combination in our study, which subsequently led to BCL-2 degradation and release of BAX, allowing the initiation of the apoptosis process. This was agreed with Panjamurthy et al. 48 who confirmed that there was a marked decrease in p53 and BCL2 expression in the rats treated with Withaferin A in cancer tissue. Our current data is consistent with Yang et al. 49
Ethanolic extract of W. somnifera showed dose-dependent inhibition of tumor growth and metastatic lung nodule formation with minimal toxicity to rats. 50 Accordingly, combination therapies of chemo or radiotherapy with Hh inhibitors showed better antineoplastic efficiency and survival in animal models. 51 In the present study, the oral dose of W. somnifera extract used to inhibit tumor metastasis to the lungs was 500 mg/kg/day body weight. This dose was extrapolated from the experiments regarding the effect of W. somnifera extract on rats with lung cancer. W. somnifera extract administered orally inhibited formation and growth of lung cancer in a rat model, indicating that its active ingredients are bioavailable. In addition, W. somnifera extract has inhibited cancer metastasis through suppression of the epithelial-mesenchymal transition and diminished CSCs. Withaferin A treatment inhibited EMT of MCF-10A cells, and in mice, reduced growth tumor, the effects of which were associated with reduced vimentin expression. 52 Overall, these findings showed that combining W. somnifera extract with IR could effectively reduce the characteristics of lung CSCs. A subsequent study using tumor samples from a mammary mouse breast cancer model discovered that withaferin could selectively target CSCs and metastatic CSCs. 53 The pharmacological properties of Withania constituents have pointed to the anticancer effects of Withania constituents against prostate, 49 breast and mammary,54,55 and skin 56 tumor tissues. The anticancer potential of W. somnifera extract and its bioactive withanolides has diverse mechanisms such as cytotoxicity, cell differentiation induction, cancer chemoprevention, cyclooxygenase-2 (COX-2) inhibition, and a potential to inhibit the enzyme quinine reductase. 57 These withanolides are highly oxygenated natural bioactive constituents that are responsible for ashwagandha’s biological properties, including antitumor activity. 58 Considering all the aspects presented, Withaferin-A can be considered as a promising pleiotropic compound in anticancer therapy in vitro and in vivo that guarantees continuous (pre)clinical elaboration. 59 In summary, our study shows that W. somnifera extract could inhibit proliferation and promote apoptosis by decreasing lung cancer stem cells via suppressing the Hh signaling pathway, which inhibits resistance tumor cells to radiotherapy. Yet, our research has some limitations. For instance, investigating more signaling pathways factors related to cancer stem cell markers would have been supportive parameters to describe the mechanisms of action of W. somnifera extract in more detail. We need further studies to explore the fundamental pathways of the alteration that could lead to the advancement of treatment approaches that block the development of prompted CSCs and subsequently enhance the effectiveness of radiation therapy. Conclusion: These results imply that W. somnifera extract may be developed as a radiosensitizer to treat lung cancer. With the vital role of W. somnifera extract in herbal remedies and its impressive actions in current cancer research, it is able to advance as an adjuvant therapeutic strategy.
Supplemental Material
sj-tiff-1-dos-10.1177_15593258221076711 – Supplemental Material for Withania somnifera Modulates Radiation-Induced Generation of Lung Cancer Stem Cells via Restraining the Hedgehog Signaling Factors
Supplemental Material, sj-tiff-1-dos-10.1177_15593258221076711 for Positive Cofactor 4 as a Potential Radiation Biodosimeter for Enas Mahmoud Moustafa, Hemat Sameeh Abdel Salam and Somaya Zakaria Mansour in Dose-Response
Supplemental Material
sj-tiff-2-dos-10.1177_15593258221076711 – Supplemental Material for Withania somnifera Modulates Radiation-Induced Generation of Lung Cancer Stem Cells via Restraining the Hedgehog Signaling Factors
Supplemental Material, sj-tiff-2-dos-10.1177_15593258221076711 for Positive Cofactor 4 as a Potential Radiation Biodosimeter for Enas Mahmoud Moustafa, Hemat Sameeh Abdel Salam and Somaya Zakaria Mansour in Dose-Response
Footnotes
Acknowledgments
For our assistance in the development of histopathology, we thank Prof. Dr Ahmed Osman, professor of Pathology, Faculty of Veterinary Medicine, Cairo University,
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
All the ethical protocols for animal treatment were followed by the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978) and supervised by the animal facilities, National Center for Radiation Research & Technology (NCRRT), Egyptian Atomic Energy Authority (EAEA), Cairo, Egypt.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
