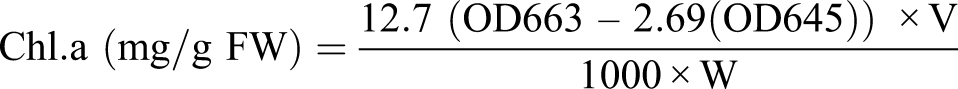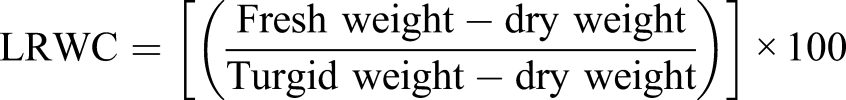Abstract
Choline chloride (CC) application enhanced the tolerance of cluster bean (Cyamopsis tetragonoloba L.) against salinity stress. The aim of the study was to determine the protective role of CC on plant growth, photosynthesis, and biochemical indicators of oxidative stress. The seeds of BR-99 (tolerant) and BR-2017 (sensitive) were surface sterilized and sown in plastic pots containing river sandy soil. The design of the experiments was completely randomized with 4 replicates per treatment. Three weeks after germination, salinity (150 mM) was imposed. Then plants were sprayed with different concentrations of CC (3, 5, and 10 mM), while normal plants were sprayed with distilled water. Salinity decreased growth attributes, relative water contents, photosynthetic attributes, total soluble proteins, total free amino acids, phenolic, flavonoids, ascorbic acid, proline, and glycine betaine and increased the levels of oxidative stress indicators. However, the application of CC (particularly 5 mM) improved growth attributes, photosynthetic pigments, and activities of antioxidant compounds by reducing the levels of H2O2, malondialdehyde in salt-stressed plants in both cluster bean varieties. BR-99 variety showed more tolerance to salinity stress than that of BR-2017 in the form of greater oxidative defense and osmotic adjustment and clear from greater plant dry masses. Thus, our results showed that the application of CC (5 mM) is an efficient strategy for field use in the areas, where salt stress soils limit agriculture production.
Introduction

Abiotic stress is envisaged as a major threat to the world agricultural system. Salinity is a severe problem in the world’s arid and semi-arid regions, notably impeding crop value and production, often occurring together with alkaline stress.1,2 Besides, salinity stress individually or in combination is a significant environmental factor that hampers the normal plant growth production capacity. 3 According to estimates, the salinity of soil affects 6-10% of the total world’s area approximately, over 800 million hectares of land are harmed by salinity.4-6 According to another estimate, the saline-alkaline problem affects 10% of the world’s total fertile land area, causing a considerable number of irrigated lands to fallow.7,8 Some studies show that about 77% of major saline land is found in Asia’s arid and semi-arid areas particularly Pakistan, where about 10 million hectors (12.9%) of the total area is categorized as saline.9-11 The salinity inhibits the growth at various development stages from germination to reproductive stages,12-14 results up to 70% economic crop loss depending upon types of plant.15-17 Thus reduction in their total yield is making the agriculture sector handicapped. 18
Saline salts inhibit the growth, photosynthetic potential, and ion balance in maize, 19 canola, 20 fenugreek, 21 and sunflower. 22 Numerous research studies have recognized the underlying plant defensive responses to salt, leading to the formation of agricultural plants with improved salinity tolerance.23,24 The soil salinity disturbs the root’s functioning for minerals transport from soil atmosphere to plant transpiration stream with excessive damage, 25 and disturbs ionic homeostasis. 26 Furthermore, this will result in the increased photorespiration and decreased photosynthesis, impaired chlorophyll synthesis,27,28 and loss of thylakoid membrane’s integrity. 29 Plants under salinity stress produce a significant amount of reactive oxygen generation (ROS) that induces progressive lipid peroxidation 30 and alteration in enzymatic antioxidant activities. 31 Oxidative stress due to the peroxidation of lipids evokes stress responses in plants, triggering enzymatic and non-enzymatic antioxidants to cope with the oxidative injury. 32 In response to environmental stresses, plants reprogram some defense responses. The accumulation of osmoregulatory compounds and selective ions absorption are necessary for defensive responses in plants against ions’ excess toxicity and osmotic stress. 33 Plants under salinity stress often encounter osmotic stress and ion toxicity. Though plants have various defense mechanisms to acclimate these conditions, 34 and accumulation of osmoprotectants such as proline, soluble sugars, and glycine betaine (GB) to maintain steady water uptake in plants.35,36
Choline is a water-soluble vitamin 37 recognized as an osmoprotectant for its defensive role against multiple stress.38,39 It is an essential component of membrane phospholipids40,41 that ensures the membrane stability, fluidity, and integrity of Mn-cluster and peripheral proteins of photosystems, particularly in cyanobacteria and many plant species42,43 under stress conditions. 44 Furthermore, choline is a vital metabolite for plant growth and development.45,46 Exogenous application of choline in the form of choline chloride (CC) is a novel approach to enhance salt tolerance in plants, cyanobacteria, and their nutritional value39,46. It is also used to accelerate the growth of animal-like chicken and in vitro improved the growth of grasses and morphogenesis of many other plant species.47-49
Cluster bean (Cyamopsis tetragonoloba L.) is a multipurpose leguminous crop, widely grown in the arid and semi-arid regions of India (80%), Pakistan (15%), Sudan, USA, and SA (5%).50-52 In Pakistan, around 15% of total worldwide cluster bean production generally, and more than .181 million hectares area for cluster bean production is cultivated mostly in Punjab, and Sindh province53-55. It has wide application from the food industry to textile, medicine, dyes or varnish, cosmetics, explosives, and petroleum industry.56-58 The crop is being exploited for its active phytochemical compound in the cure of cancer, ulcer, diabetic, anti-asthmatic, and anti-inflammatory properties.59-61 Therefore in the future, there is a need to manage the previously mentioned problem and its impacts in the production of this important crop. Our main objective was to assess the potential of CC to regulate the ROS metabolism and osmotic adjustment in cluster bean under saline conditions and to quantify the proper dose of CC that can effectively mitigate the toxic effect of salt stress on young cluster bean plants.
Methods
Plant Material and Growth Conditions
The present study was conducted at the research site of the Botany Department, Government College University Faisalabad, Pakistan, during the summer season (July 2018). Seeds of 2 cluster bean varieties having the differential salt tolerance capacity namely BR-99 tolerant 62 and BR-2017 intolerant 63 were obtained from Arid Zone Research Institute Bhakkar, Pakistan. During the experiment, the maximum and minimum temperature 38.5 + 2°C and 26 + 2°C with an average 69.0% to 80.1% relative humidity, and photosynthetically active radiation (PAR; 1262 ± 339 μmol (photon) m−2 s−1) was observed. Sodium chloride (NaCl) was used as the source for salt stress. Seeds were surface sterilized with .5% sodium hypochlorite for 5 minutes before use. Afterward, 10 seeds were sown in each plastic pot (22 cm height × 30 cm diameter) containing 10 kg pure dry river sand. Prior to use, river sand was thoroughly washed with distilled water and then oven-dried. After germination, plants were thinned to 4 plants per pot; each pot representing 1 replicate. Plants were fertilized on alternative days in half-strength Hoagland’s nutrient solution 64 to retain moisture contents to avoid drought stress. After 20 days after germination, plants of cluster bean were subjected to salinity stress (150 mM). For this purpose, solution-containing salt (50 mM) was prepared in half-strength Hoagland’s nutrient solution. An increment of 50 mM salt solution was used to avoid any osmotic shock to the plants each day until the desired salinity level (150 mM) was attained. Choline chloride was dissolved in distilled water containing few drops of dimethyl sulfoxide. Saline conditions were maintained for 3 weeks. Foliar spray of CC (Merck, pH 5.5) solution having different levels (3, 5, and 10 mM) 65 with .1% tween-20 as a surfactant was applied at 8-9 am for 2 weeks after the imposition of salinity stress. After 3 days of recovery, the plants were sprayed until thoroughly wet for two weeks. Aluminum foil was wrapped around the base of the plant to prevent any possible mix-up of CC solution in the soil. The average volume of CC applied per plant was 200 mL. For comparison, plants were also sprayed with distilled water. 2 weeks after foliar application of CC, 3 plants were harvested from each pot for recording the growth attributes. The penultimate leaf of the fourth plant was harvested to measure the physiological and biochemical attributes. The experiment was conducted in a completely randomized design (CRD) with 4 replications for each treatment.
Growth Attributes
Plants were harvested, thoroughly rinsed in deionized water, and blotted dry with a paper or towel before recording root and shoot fresh weight (SFW). The dry weight of those samples was estimated that reached a constant weight after oven drying at 80°C for 1 week. The leaf area of plants was measured from the method of grid counting. Leaves were placed on a millimeter graph paper (1 cm grid paper), and the leaf outline was drawn on a millimeter graph paper, and then the occupied block within the outlined area was counted. Plant leaf area was measured by using the following equation
66
Photosynthetic Pigments
Chlorophyll (a, b) and carotenoids were measured from the fresh leaf with the following method of Lichtenthaler.
67
Fresh leaf (.15 g) was homogenized in 10 mL aqueous acetone (80%). The mixture was centrifuged at 4000 r/min for 10 minutes. The absorbance of the supernatant was read at 663, 645, and 480 nm on a UV-Visible (Hitachi U −2910, Tokyo, JP) spectrophotometer. The calculation of chlorophyll (a, b) molecules was performed with the help of the following formula
Leaf Relative Water Content
Leaf discs (1.0 cm in diameter) of penultimate leaves were sampled with a cork borer. The fresh weight of ten leaf discs was calculated. Turgid weight was determined by immersing them in distilled water overnight at 4°C in darkness. Then leaf discs were dried in an oven for 48 hours at 70°C for measuring dry weight. Relative water content was assessed following the equation proposed by Cornic.
68
Hydrogen Peroxide Content
Fresh leaf tissue (.15 g) was crushed in 2.5 mL of .1% trichloroacetic acid (TCA) to extract H2O2. The resultant homogenate was centrifuged at 12 000g for 15 minutes. The supernatant (.5 mL) was reacted with KI (1.0 M) and .5 mL potassium phosphate buffer (50 mM; pH 7.5). The absorbance of the mixture was read at 390 nm wavelength using a UV-Visible spectrophotometer. 69
Malondialdehyde Content
Malondialdehyde content was measured according to the procedure of De Vos et al. 70 Fresh leaf (.15 g) was crushed in 1.5 mL TCA solution (10%). After that, the samples were centrifuged at 15 000g for 15 minutes to collect the supernatant. The supernatant (2 mL) was mixed with .6% thiobarbituric acid (TBA-2 mL). The reaction mixtures were heated at 100°C in a water bath. The samples were removed from the water bath and allowed to cool down to room temperature before taking absorbance at the suggested wavelength (600 and 532 nm).
Proline Contents
Fresh leaf tissue (.25 g) was homogenized in 5 mL sulfosalicylic acid (3%). The homogenate was centrifuged at 10 000g for 5 minutes to obtain the supernatant. The supernatant was used for estimating proline content. The reaction mixture consists of 2 mL of the supernatant, 2 mL of acid-ninhydrin (2.5%), and 2 mL of glacial acetic acid. The reaction mixture was boiled at 100°C for 25 minutes. Then the mixture was cooled at room temperature and extracted with 4 mL of toluene. The supernatant was used to read the absorbance at 520 nm with a spectrophotometer. 71 Proline was calculated using a standard curve and expressed in the form of μmol g−1 fresh weight.
Ascorbic Acid Contents
Fresh leaf (.15 g) was homogenized in 5% TCA solution and filtered. 2 mL filtrate was reacted with 2% 2, 4 dinitrophenyl hydrazine (1.0 mL) and 1 drop of thiourea (10%) dissolved in ethanol. The reaction mixture was incubated in the water bath for 20 minutes at 98°C. After that, the samples were removed from the water bath and allowed to cool down to 25°C followed by the addition of 80% H2SO4 (2.5 mL) at 4°C. The absorbance of the reaction mixture was read at 530 nm using a spectrophotometer. 72
Total Flavonoids Contents
Flavonoids were determined according to the procedure of Zhishen et al. 73 Fresh leaf (.15 g) was ground in aqueous ethanol (80%) and the resulting mixture was centrifuged to obtain the supernatant. 1 mL of extract was reacted with AlCl3 and NaNO2 (300 μL each). After that 2 mL NaOH (1 M) was added to the mixture followed by the 5 minutes incubation at room temperature. The volume of the mixture was made to 10 mL with distilled water. The absorbance of the samples was taken at 510 nm using a spectrophotometer.
Total Phenolic Contents
Julkunen-Tiitto 74 method was used to estimate the total phenolic contents from fresh leaf. Aqueous ethanol (80%) was used to homogenate the fresh leaf sample (.15 g) and centrifuged. Then 100 μL supernatant was reacted with 2 mL folin-ciocalteu phenol reagent and 5 mL of 20% Na2CO3. The volume of the reaction solution was made to 10 mL using distilled water. The absorbance of the mixture was measured at the advised wavelength (750 nm).
Total Free Amino Acids
Ninhydrin assay described by Hamilton and Van Slyke 75 was used to estimate the total free amino acid (TFAA) contents. Fresh leaf material (.15 g) homogenized in 5 mL of 80% ethanol. The homogenate was centrifuged at 15 000g for 20 minutes. .1 mL supernatant reacted with 1 mL of ninhydrin reagents (2% ninhydrin) and 1 mL pyridine (10%). The reaction mixture was shaken vigorously and then incubated in the water bath at 92°C for 30 minutes. After cooling, the absorbance was noted at 570 nm using a spectrophotometer. Ethanol (80%) was used as a blank. Total free amino acids were calculated using a standard curve and expressed in the form of mg g−1 fresh weight.
Total Soluble Protein
Fresh leaf tissues (.15 g) were homogenized in 10 mL chilled phosphate buffer (50 mM; pH 7.5). The supernatant was obtained after centrifugation at 12 000g for 20 minutes at 4°C. The supernatant (enzyme extract) was mixed with Bradford reagent and absorbance was taken at 595 nm for protein estimation. 76
Anthocyanin Content
Hodges and Nozzolillo
77
method was used for the estimation of total anthocyanin contents. Fresh leaves (.15 g) were grinded in 2 mL of acidified methanol with 1% HCl then the material was centrifuged at 12 000g. The resulting supernatant was used to measure the absorbance at 530 and 657 nm. Anthocyanin content was calculated following the equation proposed by Mita et al.
78
Total Soluble Sugar
Total soluble sugar (TSS) was measured with the procedure of Yemm and Willis. 79 Fresh leaf material (.15 g) extracted in 80% aqueous ethanol. 3 mL anthrone reagent (prepared in 75% sulfuric acid) mixed with 100 μL of the extract. The reaction mixture was carried out in a water bath at 95°C for 10 minutes. After incubation, test tubes were cooled down at 25°C, and absorbance was measured at 625 nm. Total soluble sugars were calculated with the help of a standard curve made from the solution of known concentration and expressed in the form of mg g−1 fresh weight.
Reducing Sugar
Reducing sugar (RS) was estimated with the procedure of Nelson. 80 Fresh leaf material (.15 g) extracted in 80% aqueous ethanol. 5 mL of O-toluidine (6%) mixed with 1 mL of aqueous ethanol extract. Then reaction mixture was heated in a water bath at 95°C for 20 minutes. After cooling the reaction mixture, the absorbance of RS was measured at 630 nm. Reducing sugars were calculated with the help of a standard curve. The data of RSs was expressed as mg g−1 fresh weight.
Antioxidant Enzymes Assay
Fresh leaf (.5 g) was powdered in liquid nitrogen, and homogenized in 50 mM potassium phosphate buffer (50 mM; pH 7.5). The supernatant was obtained after centrifugation at 12 000g for 20 minutes at 4°C. The supernatant (enzyme extract) was used for the measurement of enzyme activities. The reaction solution for catalase (CAT) activity contained 1.9 mL potassium phosphate buffer (50 mM; pH 7.0), 1 mL H2O2 (5.9 mM), and .1 mL enzyme extract. The reaction mixture was illuminated and the change in absorbance was read at 240 nm with a spectrophotometer. 81 The peroxidase (POD) activity was also estimated using the method of Chance and Maehly. 81 The enzyme extract (100 μL) was mixed with H2O2 (40 mM) and guaiacol (20 mM). The guaiacol oxidation rate in H2O2 presence read at 470 nm indicates the enzyme activity. Superoxide dismutase (SOD) activity was measured by the method of Gong et al. 82 The reaction solution (3 mL) contained 100 μL enzyme extract, 50 mM potassium phosphate buffer, 56 mM NBT (nitroblue tetrazolium), 1.17 mM riboflavin, and 10 mM methionine. The reaction mixture was illuminated under a light bulb for 10 minutes. The absorbance was recorded at 560 nm. The specific activity of each enzyme was expressed as units of enzyme/mg protein.
Statistical Analysis
The experiment was performed in a CRD in a factorial arrangement. Each treatment had 4 replications. The STATISTICS software (version 8.1) was used to calculate the analysis of variance of the data. The least significant difference was used to compare means at P ≤ .05. The different correlations in the data were drawn using RStudio.
Results
Growth Attributes
Mean Sum of Squares from ANOVA of Data for Growth, Physiological and Biochemical Attributes of Cluster Bean Varieties Treated with Choline Chloride (CC) Under Salt Stress.
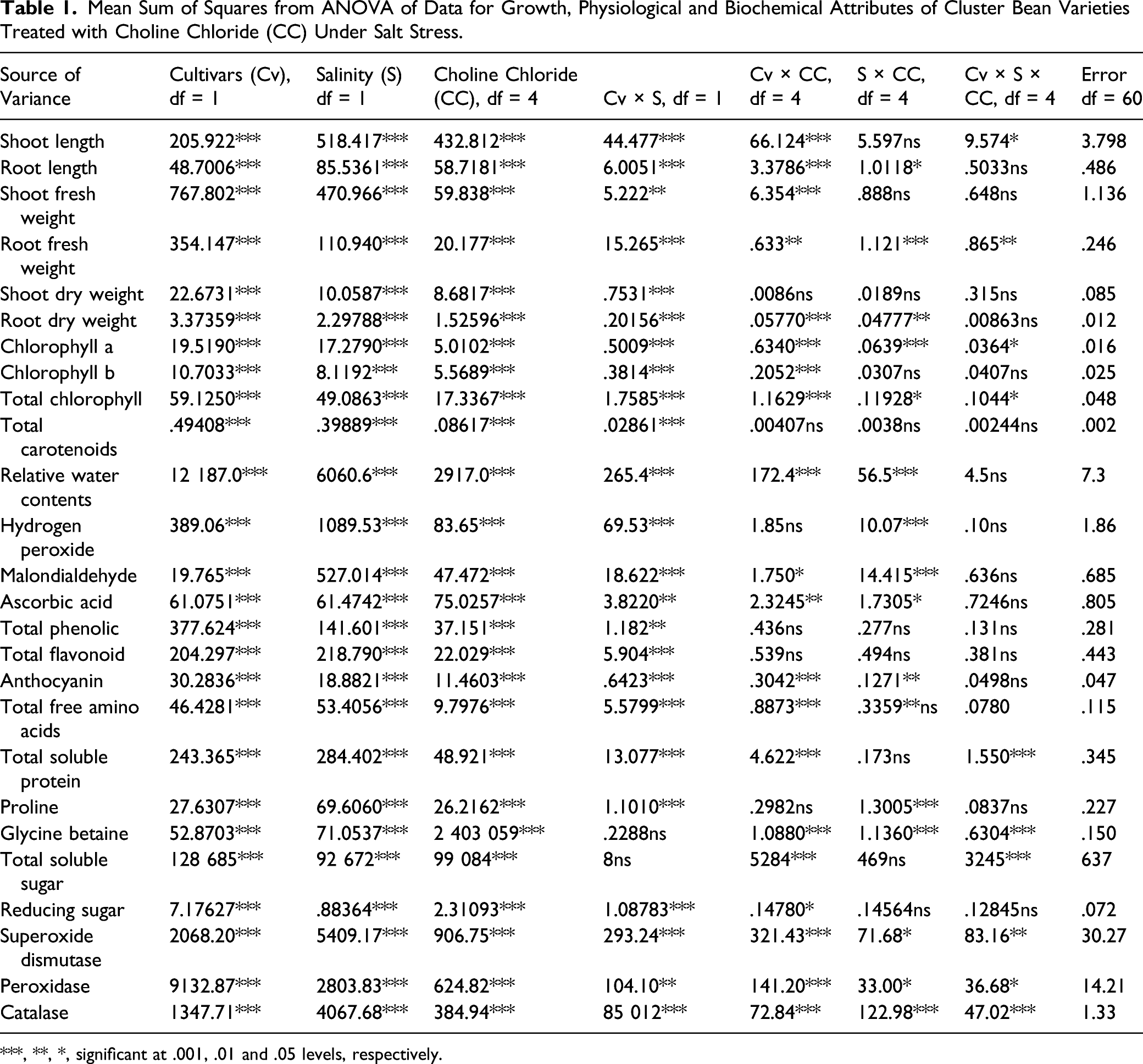
***, **, *, significant at .001, .01 and .05 levels, respectively.
Effect of Exogenous Choline Chloride (CC) on Growth Related Attributes of 2 Cluster Bean Varieties Grown Under Normal and Salt Stress Conditions.

Means are followed by standard errors. Means were compared with least significance difference (LSD ≤ .05) and different letters indicate that means are different at 95% confidence level. Abbreviations: LA, leaf area; RDW, root dry weight; RFW, root fresh weight; RL, root length; SDW, shoot dry weight; SFW, shoot fresh weight; SL, shoot length.
Photosynthetic Pigments and Relative Water Contents
Effect of Exogenous Choline Chloride (CC) on Photosynthetic Pigments and Relative Water Contents of 2 Cluster Bean Varieties Grown Under Normal and Salt Stress Conditions.

Means are followed by standard errors. Means were compared with least significance difference (LSD ≤ .05) and different letters indicate that means are different at 95% confidence level. Abbreviations: Chl. a, chlorophyll a; Chl. b, chlorophyll b; RWC, relative water content; Total Car., total carotenoids; Total Chl., total chlorophyll.
Relative water contents decreased significantly (P ≤ .001) in cluster bean under salt stress conditions (0, and 150 mM NaCl). The decrease in relative water content (RWC) was greatest in BR-2017 (50%) as compared to BR-99 (37%) under salinity (150 mM). Water spray improved the water contents in the plants of both BR-99 (14% and 24%) and BR-2017 (35% and 63%) under both conditions, respectively. Plants given CC had significantly (P ≤ .001) enhanced the RWC in both varieties under control as well as stress conditions. Among CC levels, 5 mM CC spray greater increased the RWC (40% and 59%) in BR-99 and (72% and 151%) in BR-2017 under control and stress conditions, respectively. Although, 10 mM CC reduced this variable in both cluster bean varieties (Table 3).
Oxidative Stress Indicators
Plants exposed to salt stress significantly increased (P ≤ .001) the MDA contents (Table 1). Plants exposed to 150 mM NaCl showed significantly higher MDA levels in both BR-99 (176%) and BR-2017 (190%) than control plants. Water spray decreased the MDA levels in both BR-99 (2% and 18%) and BR-2017 (23% and 11%) under normal and stress conditions, respectively. Under the saline condition, plants given CC (3 and 5 mM) showed a significant (P ≤ .001) reduction in MDA levels in both varieties (Table 1). In addition, the maximum reduction in MDA levels was evident in plants of both BR-99 (38% and 62%) and BR-2017 (65% and 54%) with 5 mM CC administration under normal and stress conditions, respectively. Plant-given CC (10 mM) increased the minimal levels of MDA in both varieties, greatly in BR-2017 under stress conditions (Figure 1). Changes in malondialdehyde (MDA), and hydrogen peroxide (H2O2) in 2 cluster beans varieties sprayed with different regimes of choline chloride (CC) under salt stress (n = 4 ± SE). Bars expressed with different letters are significantly different according to using least significant difference (LSD) at P ≤ .05.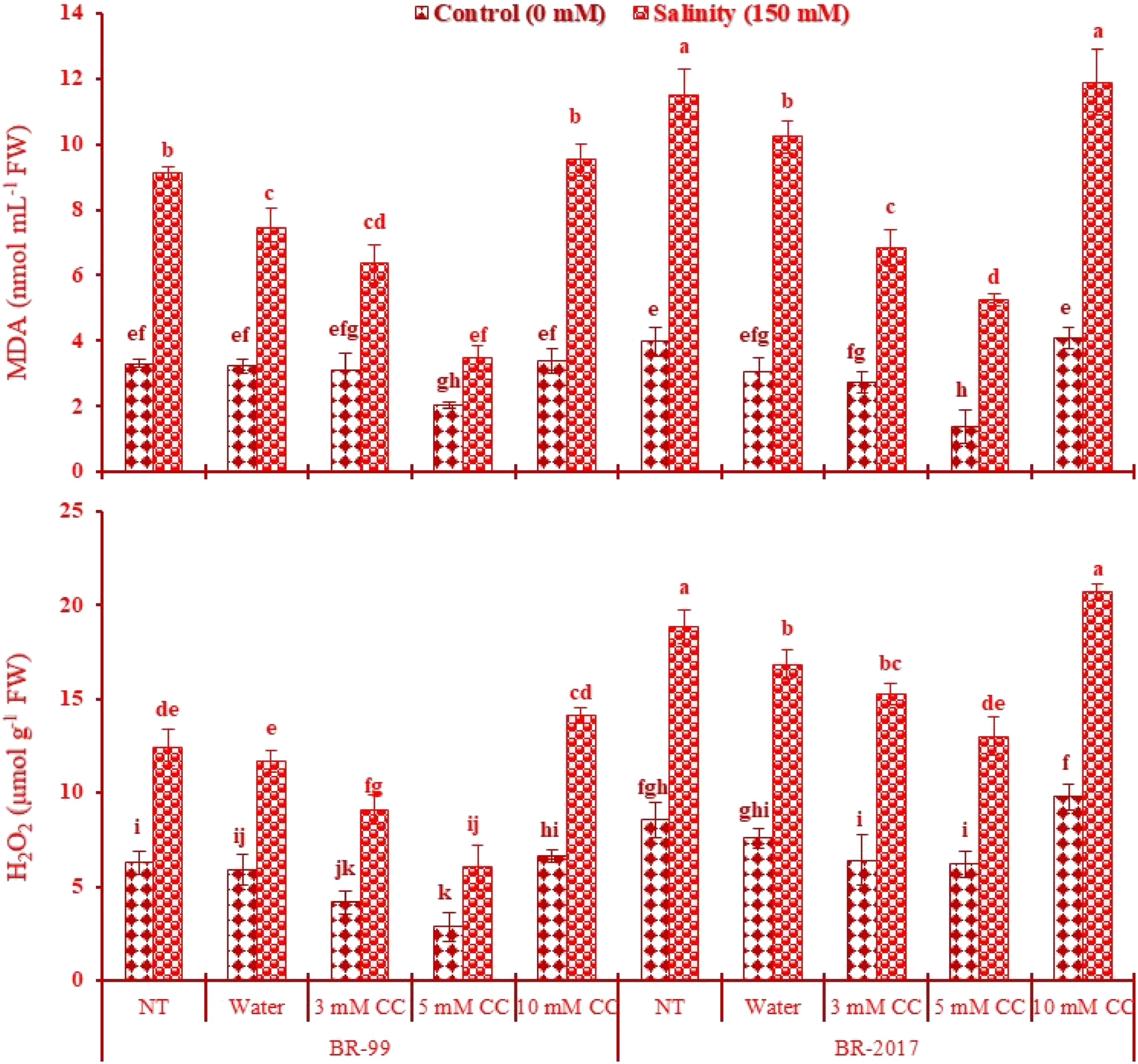
Hydrogen peroxide (H2O2) levels increased significantly (P ≤ .001) in cluster bean plants under salt stress (Table 1). H2O2 values were higher in BR-2017 (121%) than BR-99 plants (98%) when exposed to 150 mM NaCl. Moreover, water and CC (3 and 5 mM) treatments resulted in a significant (P ≤ .001) reduction in this variable under salt stress in both varieties (Table 1). Likewise, minimal H2O2 levels were evident in both BR-99 (55% and 51%) and BR-2017 (28% and 31%) varieties with 5 mM CC administration under normal and salt stress conditions, respectively. By contrast, plants given 10 mM CC raised the H2O2 levels in both varieties, more evident in the BR-2017 variety (Figure 1).
Cytosolutes
Effect of Exogenous Choline Chloride (CC) on Biochemical Attributes of 2 Cluster Bean Varieties Grown Under Normal and Salt Stress Conditions.
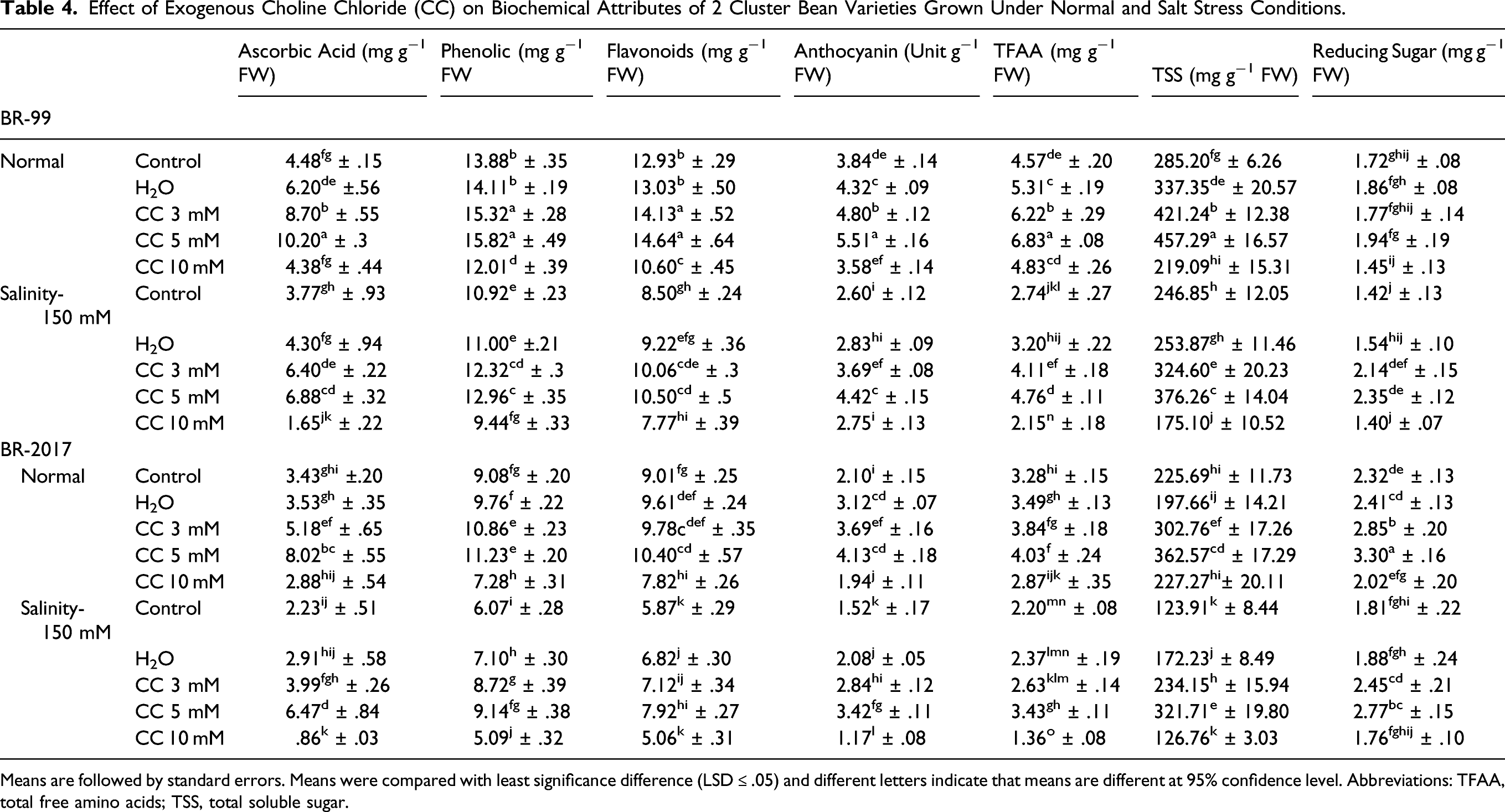
Means are followed by standard errors. Means were compared with least significance difference (LSD ≤ .05) and different letters indicate that means are different at 95% confidence level. Abbreviations: TFAA, total free amino acids; TSS, total soluble sugar.
Total soluble proteins (TSPs) decreased significantly (P ≤ .001) in cluster bean plants under salt stress conditions (0 and 150 mM NaCl) as shown in Table 1. Water spray increased the TSP contents in BR-99 (5% and 10%) and BR-2017 (10% and 20%) under both conditions, respectively. Plants given CC (3 and 5 mM) had even more (P ≤ .001) TSP in both BR-99 and BR-2017, a higher increase was noted in both BR-99 and Br-2017 at 5 mM CC under normal (28% and 27%) and stressed (44% and 104%) conditions, respectively (Figure 2). Changes in total soluble protein (TSP), proline contents and glycine betaine (GB) contents in 2 cluster beans varieties sprayed with different regimes of choline chloride (CC) under salt stress (n = 4 ± SE). Bars expressed with different letters are significantly different according to using least significant difference (LSD) at P ≤ .05.
Cluster bean plants had significantly (P ≤ .001) increased the TFAAs on exposure to stress conditions (Table 1). Decrease in TFAA levels was evident in both BR-99 (40%) and BR-2017 (33%) variety with 150 mM NaCl. In stressed cluster bean plants, CC (3 mM and 5 mM) substantially (P ≤ .001) improved the TFAA levels (Table 1). Plants given 5 mM CC accumulated more TFAA in BR-99 (50% and 73%) and BR-2017 (23% and 56%) under control and stress conditions, respectively. Exogenous CC (10 mM) reduced the TFAA contents in both varieties, greatly reduced in BR-2017 (38%) than BR-99 (22%) variety (Table 4).
Proline accumulation was substantial (P ≤ .001) in cluster bean plants under salt stress conditions (Table 1). Plants grown at 150 mM NaCl level had the greatest increase (82% and 71%) in proline compared with control plants in both BR-99 and BR-2017, respectively. Besides, CC (3, 5, and 10 mM) remarkably enhanced (P ≤ .001) the proline accumulation in plants under stress conditions (Table 1). Accumulation of proline was the maximum in BR-99 (139% and 148%), and BR-2017 (109% and 134%) varieties administered 10 mM CC under both control, and stress conditions, respectively (Figure 2).
Exposure of cluster bean plants to salt stress significantly (P ≤ .001) reduced GB. Plants exposed to 150 mM NaCl had a more drop-in GB content in BR-99 (43%) and BR-2017 (57%). CC (3 and 5 mM) remarkably (P ≤ .001) improved the GB in both varieties under stress conditions, maximum increase was noted in both BR-99 (54% and 101%) and BR-2017 (78% and 117%) at 5 mM CC under normal and stress conditions, respectively. Plants given 10 mM CC reduced the GB in both varieties under both conditions (Figure 2; Table 1).
Non-Enzymatic Antioxidants
Cluster bean plants had significantly (P ≤ .001) decreased the Ascorbic acid (ASA) contents when exposed to NaCl stress (Table 1). The lower values for this variable were seen in plants grown during salt stress (150 mM) in BR-99 (16%) and BR-2017 (35%). Moreover, water spray, and CC (3 and 5 mM) administration produced a further increase in ASA content under stress conditions. Under stress, plants fed 5 mM CC indicated a substantial increase in this attribute in BR-99 (127% and 82%) and BR-2017 (133% and 189%) under both conditions, respectively. Furthermore, exogenous 10 mM CC had a significant decrease in ASA levels in BR-99 (56%) and BR-2017 (62%) under stress conditions (Table 4).
Salt stress also caused a significant increase (P ≤ .001) in anthocyanins in cluster bean plants (Table 1). Plants grown at 150 mM NaCl levels had a maximal decreased in anthocyanin contents in BR-99 (32%) and BR-2017 (28%) compared with control plants. Under salt stress conditions, and CC (3 and 5) treatments to plants raised anthocyanin levels substantially (P ≤ .001) in both BR-99 and BR-2017 (Table 1). Plants given 5 mM CC accumulated more TFAA when stressed in BR-99 (44% and 70%) and BR-2017 (97% and 125%) under control and stress conditions, respectively (Table 4). Furthermore, exogenous CC (10 mM) reduced the anthocyanin contents in BR-2017 under control (7%) and stress conditions (23%), respectively (Table 4).
A substantial decrease (P ≤ .001) in phenolic was seen, when cluster bean plants were exposed to NaCl stress (Table 1). In comparison to control plants, plants exposed to 150 mM NaCl level showed less phenolic contents in BR-99 (21%) and BR-2017 (33%). Furthermore, plants given CC (3 and 5 mM) showed greater (P ≤ .001) phenolic levels under salt stress (Table 1). Under stress conditions, plants fed 5 mM CC showed more phenolic in both BR-99 (19%) and BR-2017 (51%) than control plants. Plants given 10 mM CC reduced the phenolic contents in BR-2017 (20% and 16%) than BR-99 (13% and 14%) under normal and stress conditions (Table 4). Flavonoids were significantly (P ≤ .001) decreased in cluster bean plants under salt stress. Plants of both varieties exposed to 150 mM NaCl levels showed the same values for flavonoids. Furthermore, CC (3 and 5 mM) administration improved (P ≤ .001) flavonoids by several times under stress conditions. Exogenous CC (10 mM) had significantly reduced the flavonoid levels in BR-99 (18% and 9%) and BR-2017 (13% and 14%) under normal and stress conditions, respectively (Table 4).
Antioxidant Enzymes
Salt stress conditions showed an important (P ≤ .001) increase in the activity of SOD in cluster bean plants. The increase in SOD was visible in BR-99 (87%) and BR-2017 (40%) at 150 mM NaCl. Exogenous application of CC) (3, 5 mM) to plants of both varieties, remarkably (P ≤ .001) improved the SOD activity under salt stress conditions. Plants that have been given 5 mM CC had more improved SOD activity in BR-99 (81% and 58%), and BR-2017 (25% and 16%) under both normal and salt stress conditions, respectively. Exogenous CC (10 mM) had reduced the SOD contents in BR-99 (9%) and BR-2017 (11%) under stress (Figure 3; Table 1). Changes in antioxidant enzymes (SOD, POD and CAT) in 2 cluster beans varieties sprayed with different regimes of choline chloride (CC) under salt stress (n = 4 ± SE). Bars expressed with different letters are significantly different according to using least significant difference (LSD) at P ≤ .05.
Both varieties of cluster beans showed a substantial (P ≤ .001) increase in the POD activity when exposed to salt stress. The POD activity of both BR-99 (208%) and BR-2017 (107%) varieties increased under 150 mM NaCl. POD activity rises considerably in both varieties after CC administration (3 and 5 mM). The rise in POD activity was significant in both BR-99 (248% and 55%) and BR-2017 (114% and 78%), when given 5 mM CC under normal and stress conditions. Plants given 10 mM CC increased the POD activity in BR-99 and reduced in BR-2017 under control and salt stress conditions, respectively (Figure 3; Table 1).
Cluster bean displayed a significant (P ≤ .001) increase in CAT activity when subjected to salt stress. CAT activity was substantially enhanced in plants of both BR-99 (88%) and BR-2017 (71%) grown at 15 mM NaCl. Under stress, CC spray (3 and 5 mM) resulted in a substantial increase in the CAT activity in both BR-99 (39% and 53%) and BR-2017 (70% and 100%), respectively. In this regard, plants given 10 mM CC showed less CAT activity in BR-2017 (15% and 9%) under normal and salt stress conditions, respectively (Figure 3; Table 1).
Principal Component Analysis
The loading plot of the principal component analysis (PCA) was performed to assess the relationship between treatments (control, salt stress, CC, and CC+ salt stress), growth, and physio-biochemical attributes in cluster bean grown in sandy river soil under salinity stress with exogenous applied CC (Figure 4). Among the principal components, components 1 and 2, referred to as Dim1 and Dim2 revealed maximum variation by 71.2% and 13.4%, respectively, with 84.6% of the whole dataset. The first component (PC1) with which the following parameters was positively correlated including growth-related-attributes, chlorophyll contents (Chl.), TFAAs, TSP, GB, POD, SOD, and CAT. A significantly negative correlation of parameters falling in PC1 was observed with the parameters of PC2 such as MDA content (MDA) and hydrogen peroxide (H2O2) (Figure 4). The principal component analysis showing the negative association of oxidative stress markers with the growth attributes and enzymatic antioxidants. Abbreviations: CAT, catalase; Chlb, chlorophyll b; Flavo, flavonoids; GB, glycine betaine; H2O2, hydrogen peroxide; MDA, malondialdehyde; POD, peroxidase; PRO, proline; RFW, root fresh weight; RS, reducing sugar; SDW, shoot dry weight; SL, shoot length; SOD, superoxide dismutase; TFAA, total free amino acids; TSP, total soluble protein.
Correlation Matrix
A correlation matrix (Pearson) graph was made to study positive and negative correlations between the studied parameters under the influence of applied treatments and to determine the existing relationships among studied parameters (Figure 5). Salinity treatment was positively correlated with MDA, H2O2, Proline, SOD, and CAT content, while negatively correlated with growth attributes, anthocyanin contents, TSP content, chlorophyll (a, b) contents, total chlorophyll, and total carotenoids contents. This association showed a close correlation between antioxidant enzymes and growth in cluster bean (Figure 5). The correlation among different physio-chemical and growth characteristics in cluster bean under salt stress. Abbreviations: Antho, anthocyanin; ASA, ascorbic acid; CAT, catalase; Car, carotenoids; Chl.a, chlorophyll a; Chl.b, chlorophyll b; Flavo, flavonoids; GB, glycine betaine; H2O2, hydrogen peroxide; LA, leaf area; MDA, malondialdehyde; POD, peroxidase; PRO, proline; RDW, root dry weight; RFW, root fresh weight; RL, root length; RS, reducing sugar; RWC, relative water contents; SDW, shoot dry weight; SFW, shoot fresh weight; SL, shoot length; SOD, superoxide dismutase; T.Chl, total chlorophyll; TFAA, total free amino acids; TSP, total soluble protein; TSS, total soluble sugar; TPC, total phenolic contents.
Discussion
Salinity becomes detrimental to abiotic stress, leading to crop reduction worldwide.6,83 The inhibition in plant growth under saline conditions is mainly attributed to Na+ toxicity, leads to the failure of normal physio-chemical properties of plants. 84 As a result, plants with weak roots could not absorb water and nutrients, inhibiting plant development in saline conditions,25,85 results in the accumulation of highly reactive oxides in various cells parts, and disturbed the ionic imbalance under saline conditions.26,86 The present research results indicated a significant decline in plant growth characteristics under saline conditions more obvious in BR-2017 than BR-99 on growth variables (Table 2). Usually, salinity above the threshold level of plants lowers the water and beneficial minerals like sodium and calcium uptake leads to plant growth reduction.87,88 Exogenous CC (5 mM) resulted in a significant improvement in plant growth characteristics, and photosynthetic pigments in variety BR-2017 as compared to BR-99 (Tables 2 and 3). This counteraction was due to the maintenance of chloroplast membrane,89-91 the integrity of thylakoid membrane in photosystem-II,92,93 and stability of extrinsic proteins or enzyme-like Rubisco, 94 under low leaf water potential or high salt concentration. Our findings are consistent with those of Salama et al, 65 who found a substantial increase in photosynthetic pigments in wheat under salt stress. Choline chloride is an efficient ROS scavenger with the ability to abridge lipid peroxidation and protect membranes. Choline chloride also improves plant growth and development. 46 Our results demonstrated that a CC-mediated strengthened oxidative defense system efficiently scavenged ROS in both cluster bean varieties, but the most prominent result was seen in the intolerant BR-2017 variety (Fig 1). Choline chloride is also protecting plants from excess ions toxicity and oxidative injury. 95 Moreover, our study showed that oxidative damage, as assessed by MDA and H2O2, greatly hampered plant growth (Figure 4). Furthermore, growth-related attributes (SL, RL, SFW, RFW, SDW, RDW, and LA), physiological attributes (Chl. a Chl. b, total Chl. total carotenoid, and RWC), and biochemical attributes (anthocyanin, TFAA, TSP, TSS, RS, flavonoid, phenolics, and GB) were negatively correlated with oxidative stress markers such as H2O2, and MDA levels (Figure 5). Choline is the precursor of phosphatidylcholine, which ensures protection for membrane fluidity and permeability in the membrane transport system. 95 Our results coincide with the findings of Sakamoto and Murata 96 and Salama et al, 65 who found a significant decrease in lipid peroxidation. Salt stress induces significant injury to membranes, as evident in the form of higher electrolyte leakage (EL) and greater lipid peroxidation. 97 As a result, the decrease in RWC and enhancement in EL is estimated as plasma membranes cannot efficiently regulate the movement of substances. 98 The decrease in RWC was also seen in Amaranthus tricolor plants under salt stress. 99 Odjegba and Chukwunwike 100 reported a decrease in RWC contents in Amaranthus hybridus plants under saline conditions. The inorganic ions and organic metabolites, including TFAAs, TSS, and RS are involved in osmoregulation. 6 Choline chloride improved ROS detoxification; results in a decrease in lipid peroxidation (MDA) and relative membrane permeability. 95 Moreover, plants treated with CC in saline conditions showed higher osmotic adjustment, as seen by improvement in several compatible solutes accumulation in cytoplasm, which might have increased the RWC in plants. 101
Chlorophyll pigment is a potential indicator to determine the photosynthetic capacity of plants under saline conditions. 102 Salt stress hampered the chlorophyll synthesis and enhanced the pigment degradation, in addition to more chloroplast structural decline. 103 The deterioration in chlorophyll pigments led to reduced plant biomass under saline conditions. 19 This study also revealed that plants with the maximum chlorophyll molecule breakdown in saline conditions showed minimal plant biomass (Table 2). The breakdown of chlorophyll molecules is also greater in salt stress conditions due to the greater activity of chlorophyllase. 104 Rasheed et al 22 indicated a significant degradation of chlorophyll molecules in sunflower plants under salt stress conditions. Plants have given CC-mediated better ROS detoxification and dropped the breakdown of chlorophyll under stress conditions. 38
The increased production of osmolytes like TFAA, TSS, TSP, proline, and GB remarkably reduced the osmotic potential of the cell. The decrease in osmotic potential is helpful for water intake and maintenance of cell turgidity. Therefore, greater levels of osmolytes also defended the plants from ions excess toxicity under saline conditions. 22 The results of this study indicated that there was a significant increase in osmolytes such as proline, proteins, and GB in cluster bean plants when subjected to salt stress conditions (Figure 2). Rasheed et al 22 also reported same results in sunflowers under salt stress. Total soluble sugars and GB contents showed a positive association with the growth of cluster bean plants under saline conditions (Figure 5). Choline chloride-mediated enhancement in these compatible solutes in the cytoplasm results in considerably increased plant growth under salt stress conditions. This GB production was likened to the metabolism of CC into GB.65,105, Phenolic compounds are strong antioxidants that efficiently hunt the ROS, and reduce the lipids peroxidation in Phaseolus vulgaris plants, consequently, phenolic protects the plants against oxidative injury. 106 Higher accumulation of phenolic shows a greater stress tolerance level.107,108 Under salt stress, CC significantly increases the phenolic contents in cluster bean (Table 4). Our findings are in line with the results of similar responses reported in potato. 109 Likewise, anthocyanins not only detoxify the free radicals but nevertheless, anthocyanins also inhibit them from generating in stressed plants. Results of this study showed that plants treated with CC produced a significant rise in anthocyanins contents under salt stress conditions (Table 4). In previous literature, exogenous CC improved the growth of radish plants with an increase in phenylalanine, anthocyanin. 109 Flavonoids are antioxidants that help in the regulation of enzyme activity and the production of primary metabolites. Flavonoids accumulate in plant tissues and have significant potential to scavenge the free radicals species.110,111 Flavonoids defend the plants from oxidative damage, 112 and plants with lesser oxidative impairment depict greater plant growth under salt stress conditions. 113 Plants treated with CC showed higher flavonoids that improved the salt stress tolerance in cluster bean plants (Table 4).
Ascorbic acid and TSP accumulate in the tissues of plants and have a strong potential to scavenge the free radicals.114,115 Several studies in the literature have shown that stability of protein and its assemblage under stress is necessary to maintain membrane integrity and cellular persistence in tomato, 116 Vicia faba, 117 and maize 118 though the decline in protein accumulation in plants under salinity is also reported. 119 In this study, our results are consistent with those of Khurana et al, 90 who found a substantial decrease in TSP under salt stress. Similarly, ascorbic acid contents were reduced under salt stress (Table 4).
Salt stress causes a substantial change in antioxidant enzyme activities. The higher antioxidant enzyme activities powerfully protected the plants from oxidative damage reflected as insignificant levels for MDA, and H2O2, contents in plants.120,121 In this study, a significant rise in oxidative damage revealed a greater free radicals generation, loss of membrane integrity, and enhanced lipid peroxidation under salt stress conditions (Figure 1) Furthermore, we observed that H2O2 showed a positive correlation with CAT and SOD activity. By contrast, POD had a substantial negative association with H2O2, and reducing sugar (Figure 5). Choline chloride supplementation in plants enhanced the antioxidant compounds and antioxidant enzyme activities that lessened the oxidative damage reflected as lesser MDA and H2O2 contents (Figure 3).
Conclusions
Salt stress considerably decreased the growth characteristics, and photosynthetic pigments, in cluster bean plants. In addition, MDA and H2O2 production were several folds greater in plants under salt stress. Variety BR-2017 was found to be sensitive, contained a higher concentration of oxidative stress markers (H2O2 and MDA), under salt stress. Choline chloride supplementation resulted in a substantial increase in growth-related attributes, chlorophyll pigments, and antioxidants potential. Choline chloride enhanced free radical detoxification and substantially protected the plants from oxidative impairment under salt stress. Furthermore, CC protected the plants from osmotic stress under salt stress conditions. Choline chloride (5 mM) has shown a significant potential to increase more tolerance to salt stress by regulating the growth, physiological and biochemical, and antioxidant attributes in cluster bean. Thus, this study showed that the CC application (5 mM) is an efficient strategy for field use in the areas, where salt stress soils limit agriculture production.
Footnotes
Acknowledgments
The presented data is a part of PhD dissertation of MS. Saima Riaz and financially supported by GCUF-RSP (4325-4359) and Project Code: 48-BOT-7.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.