Abstract
The present study aims to evaluate the protective effect of Crataegus azarolus berries decoction extract (CAB-DE) against acetic acid-induced ulcerative colitis as well as the mechanisms implicated in such protection. Adult male Wistar rats were separated into seven groups: Control (H2O), acetic acid (AA), AA + various doses of CAB-DE (100, 200, and 400 mg/kg, b.w.,p.o.), and AA + sulfasalazine (100 mg/kg, b.w.,p.o.) or gallic acid (50 mg/kg, b.w.,p.o.) during 10 days. All rats were kept fasting overnight and ulcerative colitis was induced by rectal infusion of AA (300 mg kg−1, b.w.) (3%, v/v, 5 mL kg−1 b.w), for 30 s. The colon was rapidly excised and macroscopically examined to measure ulcerated surfaces and the ulcer index. In vitro, we found that CAB-DE exhibited a high antioxidant activity against DPPH radical (IC50 = 164.17 ± 4.78 μg/mL). In vivo, pretreatment with CAB-DE significantly protected the colonic mucosa against AA-induced damage by stimulating mucus secretion, reducing ulcer index as well as histopathological changes. Also, CAB-DE limited the oxidative status induced by AA in the colonic mucosa, as assessed by MDA and H2O2 increased levels and the depletion of both enzymatic activities and non-enzymatic levels. In addition, AA intoxication increased iron and calcium levels in colonic mucosa and plasma, while CAB-DE pretreatment regulated all intracellular mediators deregulation and significantly reduced inflammatory markers such as CRP (1.175 ± .04 ─ .734 ± .06 μg/dl) and ALP (161.53 ± 5.02 ─ 98.60 ± 4.21 UI/L) levels. We suggest that CAB-DE protected against AA-induced ulcerative colitis due in part to its antioxidant and anti-inflammatory properties.
Introduction
The last few years have been characterized by a medicine modernization and development in the human health concept. Phytotherapy, or herbalism, is defined as the usage of plants or herbs as medication to treat or prevent diseases in human and animals. 1 The phytochemical studies allowed to identify more than 170,000 bioactive molecules. 2 These compounds are involved in the treatment of multiple disorders and pathologies. 3
Among these plants, Crataegus azarolus L. (Rosaceae family) is considered as a source which contains an inexhaustible quantity of active substances 4 with a wide distribution in Europe and North Africa including Tunisia. 5 It is cultivated for both its fruits and leaves. 6
Azerole also regulates cardiovascular, nervous, and immune systems 7 and has several other therapeutic activities such as antispasmodic, astringent, diuretic, febrifuge, hypotensive, and sedative. 8 Azerole extract was also used for its beneficial effects against digestive and urinary tract pain. 9
Esophagitis, peptic ulcer, ulcerative colitis (UC), and Crohn’s disease (CD) are considered to be the most severe pathologies accompanied by several complications and consequences in gastrointestinal system.10,11 These disorders are accompanied by clinical signs such as diarrhea, abdominal pain, fevers, and weight loss. 12 Further, these inflammatory syndromes can spread the digestive tract and expand to the derma. 13
Cytologically, UC is characterized by colonic mucosa deterioration, destruction of mucus barriers, and decreased defensin synthesis. 14 However, the exposed colonic epithelium activates an auto-immune reaction against the colonic bacteria and damaged tissues to limit and neutralize the propagation of the infectious agent. During this response the organism produces several harmful molecules, 15 as well as activates the cellular recovery process. 16
This inflammatory boil disease provoked several metabolic and enzymatic disorders and even colorectal cancers. 17
It has been described that in cases of acute intestinal inflammation, local inflammatory cells such as mast cells and macrophages were activated. The latter secreted pro-inflammatory chemokines to recruit other innate immune cells. It was previously shown that PNNs are the initial cells in the defensive mechanism in the elimination of the antigen. 18 These cells release several acid and lysozyme hydrolases, lactoferrin, cathepsin G, myeloperoxidase, proteinase 3, chondroitin sulfate, and reactive oxygen species (ROS) production. 19
In the present work, we evaluated the protective action of Crataegus azarolus berries decoction extract against an experimental ulcerative colitis in rats as well as the mechanisms involved in such protection.
Materials and Methods
Reagents and Chemicals
Acetic acid, epinephrine, bovine catalase, 2-thiobarbituric acid (TBA), gallic acid (GA), trichloroacetic acid (TCA), butylated hydroxyltoluene (BHT), hydrochloric acid (HCl), sodium hydroxide (NaOH), acetate sodium (C2H3NaO2), Alcian blue (C56H68Cl4CuN16S4), methanol, magnesium chloride (MgCl2), hydrogen peroxide (H2O2), sulfasalazine, and sodium chloride (NaCl) were from Sigma Chemical Co. (Sigma-Aldrich GmbH, Steinheim, Germany). All other chemicals used were of analytical grade.
Crataegus azarolus berries sampling and decoction preparation (CAB-DE)
Mature Crataegus azarolus berries were collected during September, 2017 from the area of Ain-Draham (north-west of Tunisia). After washing, the edible part of the fruit was dried at 40°C using an air-ventilated oven and then crushed. The Crataegus azarolus berries decoction extract (CAB-DE) was obtained by decoction of 1 g of the berries powder in 10 mL of bi-distilled water (1/10; w/v), for 5 minutes and filtered by Whatman filter paper.
Phytochemical Screening and Anti-Radical Activity
Total Polyphenol Assessment
Total polyphenol contents were established by Folin–Ciocalteu according to the method of Singleton et al. 20 For each test, .5 mL of CAB-DE was mixed with .5 mL of Folin–Cieucalteu reagent (10%) and 1 mL of sodium carbonate (Na2CO3, 7.5%). The reagent mixture was kept in the dark for 1 hour. The absorbance was determined at 760 nm, and the total polyphenol concentrations were expressed as milligrams of gallic acid equivalent per gram of dry matter (mg GAE/g DM).
Total Flavonoid Content
The flavonoid quantification was carried out by aluminum trichloride (AlCl3) method. 21 Briefly, 1 mL of CAB-DE was added to 1 mL of the methanolic AlCl3 (2%) solution. The mixture was incubated in dark for 15 min and the absorbance was determined at 430 nm. The flavonoid content was expressed as milligrams of quercetin equivalent per g of dry matter (mg Qt E/g DM).
Total Tannin Determination
The dosage of total tannins was determined according to the method of Kujala et al. 22 Briefly, .50 mL of decoction extract was added to .5 mL Folin–Ciocalteu (50%) solution and the mixture was kept in dark for 5 min. This latter was added to 1 mL of sodium carbonate (20%), incubated in dark for 15 min, and centrifuged at 1000 × g for 5 min. The supernatant absorbance was measured at 730 nm, and total tannin contents were expressed as milligrams of tannic acid equivalents per gram of dry matter (mg EAT/g DM).
Condensed Tannin Quantification
The condensed tannin quantification was carried out by vanillin method. 23 For each assay we mixed 400 μL of CAB-DE, 3 mL of the methanolic vanillin solution (4%), and 1.5 mL of concentrated HCl. The absorbance was read at 500 nm, and the condensed tannin levels were expressed as milligrams of catechin equivalent per g of dry matter (mg EC/g DM).
Characterization of Phenolic Compounds of CAB-DE by LC-ESI-MS Analysis
Phenolic compounds of CAB-DE have been identified by Liquid Chromatography-Electrospray Ionization-Mass Spectrometry (LC-ESI-MS). Briefly, 20 mg of dry CAB-DE was dissolved in 20 mL of methanol and filtered through a .45 μm pore membrane before being injected into the liquid chromatography system. 24 The LC-Electrospray Ionization-Mass Spectrometry (LC-ESI-MS) analysis was made using the LC-MS-2020 quadruple mass spectrometer system (Shimadzu, Kyoto, Japan) equipped with an electrospray ionization source (ESI) and operated in negative ionization mode. The mobile phase composed of solvent A, H2O/formic acid (.1%, v/v), and solvent B, formic acid/methanol A (.1%, v/v). The mobile phase flow rate was .4 mL/min, and phenolic compounds follow a linear gradient elution: −45 min, 10%–100% B and 45–55 min, 100% B.
The column model used was Aquasil C18 (150 × 3 mm, 3 µm) preceded by an Aquasil C18 guard column (10 × 3 mm, 3 µm) (Thermo Electron, Dreieich, Germany), and its temperature was maintained at 40°C. The injection volume was 5 μL, and re-equilibration time between individual injection was 5 min.
The mass spectrometer was operated in a negative ion mode with a voltage detector of 1.2 V and a capillary voltage of −3.5 V. The dry gas flow rate used was 12 L/min and 1.5 L/min for the clean gas flow rate. The block source temperature was 400°C, dissolution line temperature was 250°C, and full scan spectrum was 50 to 2000 m/z. 24
The phenolic compound identification was established by comparing the retention times and the mass spectra with those of the authentic standards. 24
DPPH-Free Radical Scavenging Activity
The CAB-DE antioxidant activity was determined using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical-scavenging assay, according to the method of Grzegorczyk et al.
25
We firstly prepared various CAB-DE concentrations (10, 25, 50, 100, 150,200, 250, and 300 μg/mL) and each one was mixed with 1 mL of freshly prepared DPPH ethanolic solution (.1 mM). The mixture was incubated at 27° C for 30 min, and the absorbance was measured at 517 nm. Gallic acid was used as a reference antioxidant molecule in a similar concentration range of CAB-DE. The inhibitory concentration 50 (IC50) was defined as the concentration in μg/mL needed to scavenge 50% of DPPH radicals. The radical-scavenging activity (RSA) was determined using the following formula:
Animals and Treatment
The experiments were carried out on 70 adult male Wistar rats (weighing 220–240 g, 15 weeks old, and housed 5 per cage) which were purchased from SIPHAT (Tunisia). Animals were used in accordance with the local ethics committee of Tunis University of the use and care of animals following the NIH recommendation. 26 The protocol has been approved by the “Comited’ Ethique Bio-medicale (CEBM)” (JORT472001) of the “Pasteur Institute of Tunis.” They were provided with standard food (standard diet in Badr-Utique-TN) and water ad libitum and maintained in an animal house at a controlled temperature (22 ± 2°C) with a 12–12 h light–dark cycle. Rats were divided into seven groups of 10 animals each. Groups 1 and 2 served as controls and received bi-distilled water (5 mL kg−1, b.w., p.o.) for 10 days. Groups 3, 4, and 5 were pretreated with various doses of CAB-DE (100, 200, and 400 mg kg −1, b.w., p.o.), while groups 6 and 7 received gallic acid and sulfasalazine at 50 and 100 mg kg −1, b.w., p.o., respectively, for the same period before induction of ulcerative colitis.
Induction of Ulcerative Colitis in Rats
All animals were kept fasting overnight. Ulcerative colitis was induced by the infusion of acetic acid (3%, v/v, 5 mL kg−1, b.w.) for 30 seconds using a polyethylene tube inserted through the rectum into the colon for a distance of 8 cm. 24 h later, rats were anesthetized by intraperitoneal administration of sodium pentobarbital (40 mg kg−1, b.w.) and sacrificed. 27
The colon was rapidly excised, macroscopically examined, and homogenized in phosphate buffer saline for determining the biochemical parameters. The remaining portion of the colon was kept in 10% formalin for a histopathological study. Blood was also collected in heparinized tubes. After centrifugation at 3000 × g for 15 min, plasma was stored at −20°C.
Assessment of Colitis
The distal portions of the colon were cut longitudinally and cleaned with physiological saline (NaCl 0, 9%) to remove all fecal traces. After, they were deployed under a lamp to measure ulcerated surfaces. According to Millar et al., 28 macroscopic inflammation levels were determined by the clinical damage state of the colon on an arbitrary scale from 0 to 4. These values are as follows: no macroscopic changes (number 0), mucosal erythema only (number 1), mild mucosal edema with minor bleeding ulcers or erosions (number 2), moderate edema with minor bleeding ulcers or erosions (number 3), and severe ulceration, edema, and tissue necrosis (number 4). Ulcer indexes were determined as the sum of the lengths of the whole colonic lesions in mm2, and the photos were taken using a Sony model Alpha A7R Mark II camera.
Determination of Adherent Mucus
The determination of colonic mucus content was carried out according to Popov et al. 29 For each assays, a weighed colon fragment was transmitted in Alcian blue (1%) and sucrose (10%) solution and buffered with sodium acetate (pH 5) for 24 h. These sections were washed with a sucrose solution, and the rest of the tincture was fixed by the colonic mucus and was isolated with 10 mL of magnesium chloride (MgCl2, .5 M). Finally, the homogenate was collected and centrifuged at 4000 × g for 15 min. The supernatant absorbance was read at 580 nm. The mucus content was determined in μg/g of wet colonic tissue using a standard curve of Alcian blue concentrations.
Histopathological Study
Immediately after sacrifice, small pieces of distal colon were gathered and washed with a solution of NaCl (.9%). Tissue fragments were then fixed in a 10% neutral buffered formalin solution, embedded in paraffin, and used for histopathological examination. 5 μm thick sections were cut, deparaffinized, hydrated, and stained with hematoxylin and eosin (HE). The colonic sections were examined in blind fashion in all treatments.
Lipid Peroxidation Measurement
The lipid peroxidation was assessed by measuring the MDA level according to Draper and Hadley. 30 The colonic mucosa was mixed with trichloroacetic acid-hydroxytoluenebutylate (TCA-BHT) solution, 1% BHT (w/v) dissolved in 20% TCA (w/v). The mixtures were centrifuged at 1000 × g and 4°C for 5 minutes, and the supernatant was blended to (.5 N HCl and 120 mM TBA buffered in 26 mM Tris) solution. The mixture was heated at 80°C for 10 minutes. After cooling, the MDA levels were determined using an extinction coefficient for the MDA–TBA complex of 1.56 × 105 M−1 cm−1.
Hydrogen Peroxide Level Assessment
The assessment of the hydrogen peroxide level in colonic mucosa was carried out according to Dingeon et al. 31 Briefly, H2O2 reacts with 4-aminoantipyrine and P-hydroxybenzoic acid in the presence of peroxidase to form a pinkish complex, measured at 505 nm.
Plasma Scavenging Activity Assay
The plasma scavenging activity (PSA) was evaluated using the DPPH radical model.
32
Indeed, 100 μL of plasma was mixed with 2 mL methanolic solution of 2,2-diphenyl-1-picrylhydrazyl at 100 mM and then 1 mL of chloroform was added to the mixture. The obtained product was incubated for 30 minutes at 37°C and centrifuged for 10 minutes at 3000 × g. The supernatant absorbance was measured at 517 nm, and the PSA was determined using the following formula:
Enzymatic Antioxidant Assessment
CAT activity was determined by measuring the initial rate of H2O2 disappearance at 240 nm for 3 minutes, following the Aebi 33 method. Indeed, enzyme extract was mixed with 50 mM of phosphate buffer (pH 7), and the measurements were taken after the addition of 33 mM of H2O2. The catalase activity was determined using the molar extinction coefficient of 40 mM−1cm−1 for H2O2.
The activity of SOD in the colonic mucosa was performed according to Kakkar and Das. 34 Indeed, 1 unit of SOD is defined as the amount of the extract that inhibits the rate of adrenochrome formation by 50%. Briefly, 5 μL of enzyme extract was mixed with 10 μL of bovine catalase (.4 U/mL) and 20 μL of epinephrine (5 mg/mL). The obtained product was combined with a buffered solution of sodium carbonate/bicarbonate (pH 10.2, 62.5 mM). Indeed, the superoxide anion oxides epinephrine to adrenochrome, and the absorbance was measured at 480 nm.
The activity of GPx was measured using the Flohé and Günzler method. 35 In this respect, .2 mL of colonic mucosa was mixed with .2 mL of GSH (.1 Mm), .2 mL of phosphate buffer (.1 M, pH 7.4), and .4 mL of H2O2 (5 Mm). The mixture medium was incubated for 1 min at 37°C, and the reaction was blocked by the addition of 1 mL of TCA (1%). The obtained product was centrifuged at 3000 × g for 10 minutes. The aliquot (.2 mL) from the supernatant was mixed with 500 μL of phosphate buffer (.1 M, pH 7.4) and 500 μL of DTNB (10 mM), and the absorbance was measured at 412 nm. The GPx activity was expressed as nmol of GSH consumed/min/mg protein.
Non-enzymatic Antioxidant Level Determination
The level of thiol groups (-SH) was determined according to the method of Ellman. 36 The homogenates of the colonic mucosa were mixed with 800 μL of phosphate buffer (.25 M, pH 8.2) and 100 mL of EDTA (20 mM). The optical density was measured at 412 nm (A1). Then, 100 μL of DTNB 10 (mM) was added and the product was incubated at 37°C for 15 minutes and a new value was determined (A2). The concentration of thiol groups was evaluated by the difference between A2 and A1 using a molar extinction coefficient of 13.6 × 103 M−1cm−1.
The GSH level was measured colorimetrically by the method of Sedlak and Lindsay. 37 In fact, 5 mL of supernatant was mixed with 4 mL of cold distilled water and 1 mL of TCA (50%). The mixture was stirred for 10 minutes and centrifuged at 1200 × g for 15 minutes. 2 mL of supernatant was mixed with 4 mL of Tris-buffer (.4 M, pH 8.9), and .1 mL of DTNB (.01 M) solution was added to the reaction mixture. The absorbance was measured at 412 nm.
Calcium and Iron Assessment
Calcium content was measured by using Stern and Lewis 38 technique. Ca2+ precipitates as Ca2+ oxalate leading to a complex with o-cresolphthalein, and the mixture was evaluated at 570 nm. The assessment of free iron was established by using the method of Leardi et al. 39 Briefly, the iron dissociated from transferrin-iron complex, reduced by ascorbic acid, reacts with ferrozine to give a chromatophore read at 562 nm.
C-Reactive Protein and Alkaline Phosphatase Activity Determinations
The inflammatory marker assays such as alkaline phosphatase (ALP) activity and C-reactive protein (CRP) levels were evaluated using commercial kits purchased from the company Biomaghreb (ISO 9001 certificate).
Glycemic and Lipid Profile Evaluation
Glycemia, total cholesterol, high-density cholesterol (HDL-cholesterol), and triglyceride levels were measured using commercially available diagnostic kits and whose references are 20127; 20111; 201131; 20132, respectively (Biomaghreb, Ariana, Tunisia).
Statistical Analysis
Data were analyzed using the unpaired Student’s t test or one-way analysis of variance (ANOVA) and are expressed as means ± standard error of the mean (SEM). All statistical tests were two-tailed, and a P-value of .05 or less was considered significant.
Results
Phytochemical Screening of CAB-DE
Phytochemical Screening and IC50 Values of the DPPH Radical-Scavenging Activity of Crataegus azarolus Berries Decoction Extract (CAB-DE) and Gallic Acid (GA).

Data are expressed as mean ± SD; DM: dry matter; GAE: gallic acid equivalent; TAE: tannic acid equivalent; CE: catechin equivalent; QE: quercetin equivalent.
Phenolic Profile of CAB-DE
LC-ESI-MS Analysis of the Crataegus azarolus Berries Decoction Extract.

aThe numbering refers to elution order of compounds from an Aquasil C18 column.
bIdentification was confirmed using 33 authentic commercial standards.

Chromatographic profiles of Crataegus azarolus berries decoction extract (CAB-DE) (assignments of peaks are given in Table 2).
Antioxidant Capacity of CAB-DE
The determination of free radical-scavenging activity using the DPPH test showed that CAB-DE exhibited a significant dose-dependent antioxidant capacity. However, the IC50 of CAB-DE was very important (IC50 = 164.17 ± 4.78 μg/mL) but still higher than that of gallic acid (IC50 = 13.75 ± 2.15 μg/mL), which was used as a reference antioxidant molecule (Table 1).
Qualitative and Quantitative Gross Evaluation of Colon Lesions
Sub-Acute Effects of the Crataegus azarolus Berries Decoction Extract (CAB-DE), 917 Sulfasalazine (SULF) and Gallic Acid (GA) on Acetic Acid-Induced Changes in Colon Mucus Content, the Gross Lesion Score, and Colon Weight to Length Ratio. Animals Were Pretreated for 10 days With Various Doses of CAB-DE (100, 200, and 400 mg/kg, b.w., p.o.), Sulfasalazine (100 mg/kg, b.w., p.o.), Gallic Acid (50 mg/kg, b.w., p.o.), and Distilled Water. Rats Were Challenged With a Single Anal Administration of AA (300 mg kg−1, b.w.) (3%, v/v, 922 5 mL kg− b.w) or NaCl (0.9%) for 24 h.

Data are expressed as means ± SEM. (n = 10), *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.
Sub-acute Effects of the Crataegus azarolus Berries Decoction Extract (CAB-DE), Sulfasalazine (SULF), and Gallic Acid (GA) on Acetic Acid-Induced Changes in Colon Volume Content. Animals Were Pretreated for 10 days With Various Doses of CAB-DE (100, 200, and 400 mg/Kg, b.w., p.o.), Sulfasalazine (100 mg/Kg, b.w., p.o.), Gallic Acid (50 mg/Kg, b.w., p.o.), and Distilled Water. Rats Were Challenged With a Single Anal Administration of AA (300 mg Kg−1, b.w.) (3%, v/v, 5 mL Kg−1 b.w) or NaCl (.9%) for 24 h.

Data are expressed as means ± SEM. (n = 10), *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.
Comparative Histopathology Study
The histopathological study on colonic sections showed that acetic acid induced several complex lesions identified in the mucosa: hematomas, hemorrhagic foci, venous dilation, and neutrophil infiltration. The crypts structure and number were also affected and their upper part has been eroded. The observations showed a disorder in the arrangement of the mucosa affecting their layers in various degrees and acute cell necrosis and apoptosis (Figure 2B). Effect of Crataegus azarolus berries decoction extract (CAB-DE), sulfasalazine, and gallic acid (GA) on acetic acid-induced several disorders in colonic mucosa. Hematoxylin and eosin-stained colonic tissues (magnification, ×100). Animals were pretreated for 10 days with various doses of CAB-DE (100, 200, and 400 mg/kg, b.w., p.o) (C, D, and E), reference molecules: sulfasalazine (100 mg kg, b.w., p.o) (F), gallic acid (50 mg/kg, b.w.,p.o)(G), and NaCl (.9%) (A). Two hours after the last pretreatment, animals received acetic acid (AA, 3%, v/v, 5 mL kg−1, b.w., p.o.) and they were sacrificed after 24 h.
However, pretreatments with varied doses of CAB-DE (100, 200, and 400 mg/kg, b.w., p.o.) exerted remarkable protection against AA-induced deterioration in colonic mucosa structure (Figure 2C–E).
Indeed, this protection was expressed as a regression of hemorrhagic foci, lesions, and congestion. Moreover, CABDE significantly limited neutrophil extravasations and preserved the shape and length of the crypts in a dose-dependent manner. Importantly, pretreatment with sulfasalazine only (100 mg kg, b.w., p.o.) moderately protected the colonic mucosa (Figure 2F), while preventive treatment with gallic acid (50 mg/kg, b.w., p.o.) protected the colonic mucosa against the AA-induced ulcerative effect (Figure 2G).
Effect of AA and CAB-DE on Colonic Lipid Peroxidation and H2O2 Increase
To evaluate the effect of AA and CAB-DE on the oxidative stress conditions, we firstly studied the colonic lipid peroxidation and hydrogen peroxide contents (Figure 3). AA per se significantly increased the colonic MDA (Figure 3A) and H2O2 (Figure 3B) levels when compared to the control group. Pretreatment with CAB-DE (100, 200, and 400 mg/kg, b.w., p.o.) significantly reduced colonic lipid peroxidation (28.56 ± 2.29 ─ 16.66 ± 3.08 nmol/mg protein) and increased H2O2 (38.47 ± 1.89 ─ 24.82 ± 5.71 nmol/mg protein) in a dose-dependent manner. On the other hand, we also showed that GA (50 mg kg, b.w., p.o.) and sulfasalazine (100 mg kg, b.w., p.o.) exerted a significant protective effect against AA-induced MDA and H2O2 increase. Sub-acute effect of Crataegus azarolus berries decoction extract (CAB-DE), sulfasalazine (SULF), and gallic acid (GA) on acetic acid-induced changes in colonic mucosa MDA (A) and H2O2 (B) levels. Animals were pretreated for 10 days with various doses of CAB-DE (100, 200, and 400 mg/kg, b.w., p.o), sulfasalazine (100 mg/kg, b.w., p.o), gallic acid (50 mg/kg, b.w., p.o), and distilled water. Rats were challenged with a single anal administration of AA (300 mg kg−1, b.w.) (3%, v/v, 5 mL kg−1 b.w) or NaCl .9% for 24 h. *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.
Effect of AA and CAB-DE on Plasma Scavenging Activity
As illustrated in Figure 4, acute rectal administration of AA (3%, v/v, 5 mL kg−1, b.w.) induced a significant decline of plasma scavenging activity (28%). However, sub-acute pretreatment with various doses of CAB-DE significantly protected against this imbalance in a dose-dependent manner (35.84─58.06%). Moreover, sulfasalazine (52.78%) and gallic acid (64.57%) treatments significantly corrected this deregulation. Sub-acute effect of Crataegus azarolus berries decoction extract (CAB-DE), sulfasalazine (SULF), and gallic acid (GA) on acetic acid-induced decrease in plasma scavenging-activity levels. Animals were pretreated for 10 days with various doses of CAB-DE (100, 200, and 400 mg/kg, b.w., p.o), sulfasalazine (100 mg kg, b.w., p.o), gallic acid (50 mg/kg, b.w., p.o), and distilled water. Rats were challenged with a single anal administration of AA (300 mg kg−1, b.w.) (3%, v/v, 5 mL kg−1 b.w) or NaCl .9% for 24 h. *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.
Effect of Acetic Acid and CAB-DE on Antioxidant Enzyme Activities
A comparative study between different pretreatment compounds and AA on enzymatic antioxidant activities has been established (Figure 5). As expected, AA (3%, v/v, 5 mL kg−1, b.w.) induced a significant decrease in CAT, SOD, and GPx activities in the colonic mucosa. However, CAB-DE pretreatment abolished the depletion of CAT (Figure 5A), SOD (Figure 5B), and GPx (Figure 5C) activities in a dose-dependent manner. Impressively, the rats who received GA presented the highest antioxidant properties (Figure 5). Sub-acute effect of Crataegus azarolus berries decoction extract (CAB-DE), sulfasalazine (SULF), and gallic acid (GA) on acetic acid-induced decrease in colonic mucosa enzymatic antioxidant levels. Animals were pretreated for 10 days with various doses of CAB-DE (100, 200, and 400 mg/kg, b.w., p.o), sulfasalazine (100 mg kg, b.w., p.o), gallic acid (50 mg/kg, b.w., p.o), and distilled water. Rats were challenged with a single anal administration of AA (300 mg kg−1, b.w.) (3%, v/v, 5 mL kg−1 b.w) or NaCl .9% for 24 h. *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.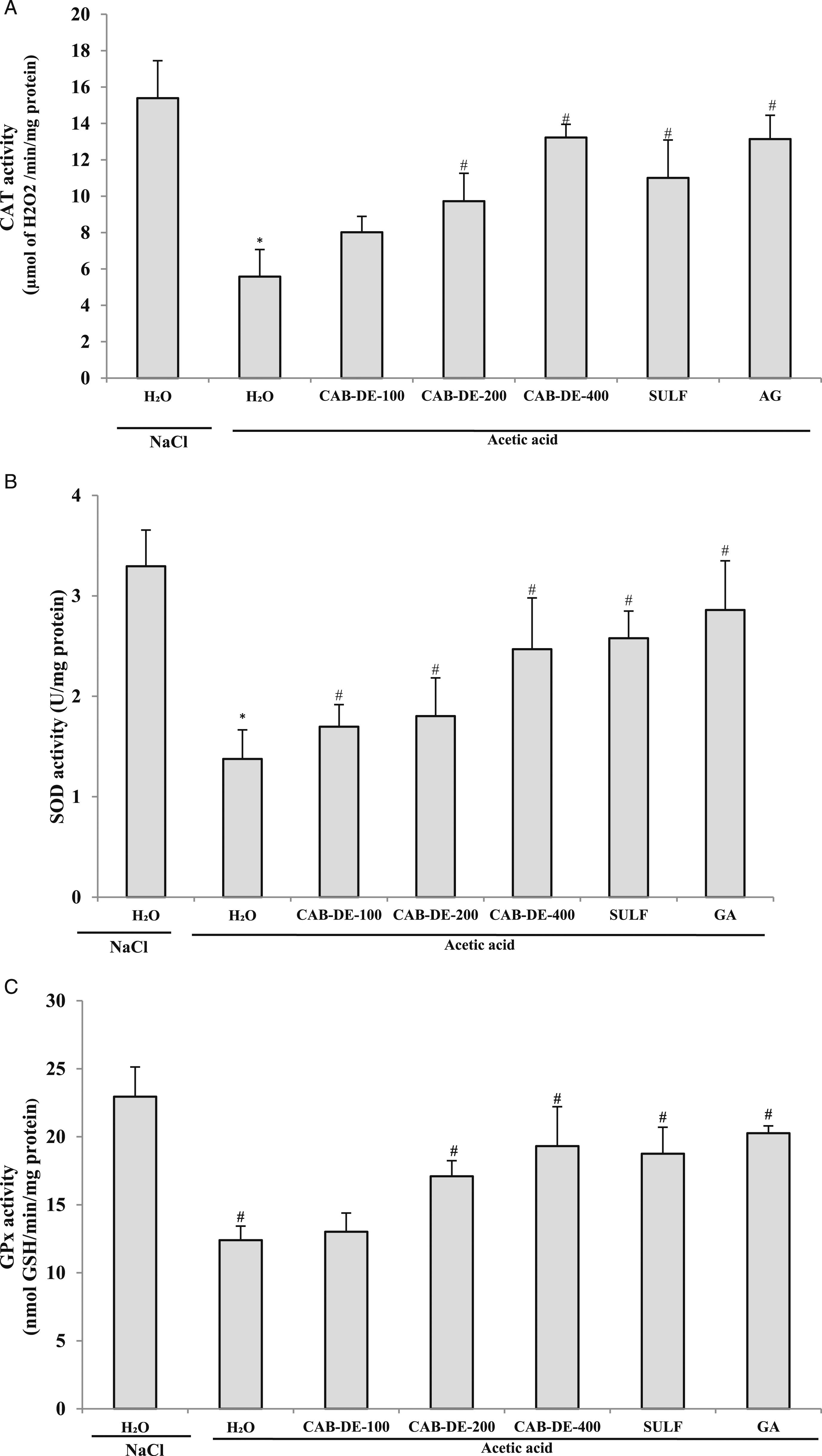
Effect of CAB-DE on Non-enzymatic Antioxidant Contents
The analysis of non-enzymatic antioxidant contents in colonic mucosa demonstrated that AA induced a significant depletion of the thiol group (A) and reduced glutathione (B) when compared to control animals (Figure 6). Pretreatment with CAB-DE preserved the GSH (30.28 ± 2.72 ─ 46.86 ± 4.51 nmol/mg of protein) and -SH group (234.44 ± 8.36 ─ 308.66 ± 9.49 nmol/mg of protein) contents in a dose-dependent manner. Sub-acute effect of Crataegus azarolus berries decoction extract (CAB-DE), sulfasalazine (SULF), and gallic acid (GA) on acetic acid-induced decrease in colonic mucosa non-enzymatic antioxidant levels. Animals were pretreated for 10 days with various doses of CAB-DE (100, 200, and 400 mg/kg, b.w., p.o), sulfasalazine (100 mg/kg, b.w., p.o), gallic acid (50 mg/kg, b.w., p.o), and distilled water. Rats were challenged with a single anal administration of AA (300 mg kg−1, b.w.) (3%, v/v, 5 mL kg−1 b.w) or NaCl .9% for 24 h. *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.
Effect of CAB-DE and AA on Calcium and Iron Levels
Sub-acute Effects of the Crataegus azarolus Berries Decoction (CAB-DE), Sulfasalazine (SULF), and Gallic Acid (GA) on Acetic Acid-Induced Changes on Plasma and Colonic Mucosa Free Iron and Calcium Levels. Animals Were Pretreated for 10 days With Various Doses of CAB-DE (100, 200, and 400 mg/Kg, b.w., p.o.), Sulfasalazine (100 mg Kg, b.w., p.o.), Gallic Acid (50 mg/Kg, b.w., p.o.), and Distilled Water. Rats Were Challenged With a Single Anal Administration of AA (300 mg Kg−1, b.w.) (3%, v/v, 5 mL Kg−1 b.w) or NaCl (.9%) for 24 h.
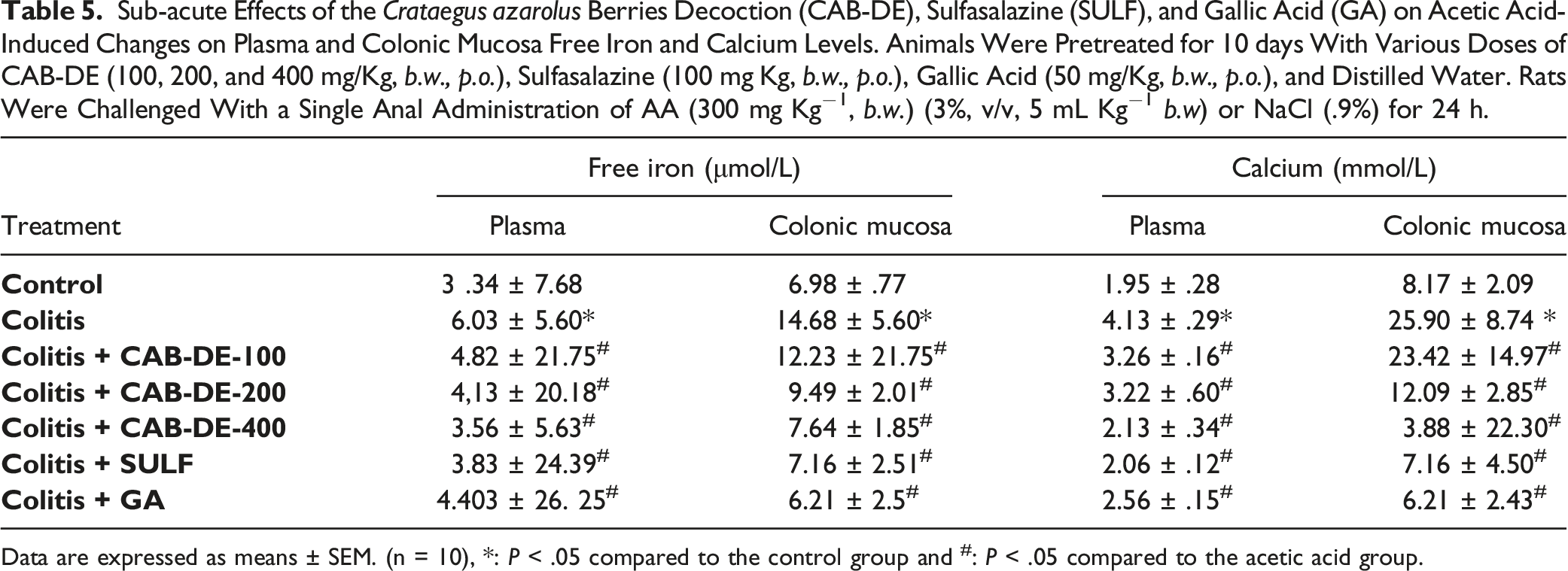
Data are expressed as means ± SEM. (n = 10), *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.
Effect of CAB-DE on Acetic Acid-Induced Inflammation
Sub-acute Effects of the Crataegus azarolus Berries Decoction (CAB-DE), Sulfasalazine (SULF), and Gallic Acid (GA) on Acetic Acid-Induced Changes on Alkaline Phosphatase (ALP) Activity C-Reactive Protein (CRP), Glucose, Total Cholesterol, High-Density Lipoprotein-Cholesterol (HDL-Cholesterol), and Triglycerides Levels. Animals Were Pretreated for 10 days With Various Doses of CAB-DE (100, 200, and 400 mg/Kg, b.w., p.o), Sulfasalazine (100 mg Kg, b.w., p.o), Gallic Acid (50 mg/Kg, b.w.,p.o), and Distilled Water. Rats Were Challenged With a Single Anal Administration of AA (300 mg Kg−1, b.w) (3%, v/v, 5 mL Kg−1 b.w) or NaCl (.9%) for 24 h.

Data are expressed as means ± SEM. (n = 10), *: P < .05 compared to the control group and #: P < .05 compared to the acetic acid group.
Effect of CAB-DE on Glycemia and Lipid Plasma Parameters
We further looked at the implication of the lipid parameters and glycemia metabolism in the protective effect of CAB-DE against acetic acid-induced colitis in rats (Table 6). As expected, we firstly showed that colitis is accompanied by an increase in lipidemia and blood glucose levels. Importantly, CAB-DE pretreatment significantly regulated the level of glucose and all lipid profile parameters in a dose-dependent manner. Additionally, an important protection degree has been showed in GA and SULF groups.
Discussion
In the present investigation, we evaluated the preventive effect of Crataegus azarolus berries decoction extract (CAB-DE) against acetic acid-induced experimentally ulcerative colitis, oxidative stress, inflammation, and metabolic disorders.
In vitro, we firstly showed that CAB-DE contains high levels of secondary metabolites such as total polyphenols and flavonoids as well as total and condensed tannins. However, the extraction technique can affect the level of total phenolic compounds. In fact, we previously showed that the Crataegus azarolus berries aqueous extract obtained by maceration contained a lower level of total phenolic contents than the decoction extract for the same plant part. 40 Our results are in line with the findings of Castro-López et al. 41 In addition, the LC-ESI-MS analysis resulted in the identification of 8 phenolic compounds, among which chlorogenic acid was the major component. In this respect, previous reports demonstrated that this fruit has a very diverse phenolic profile.42,43 However, these discrepancies can be related to edaphic and biotic factors. 44 The extraction technique and solvents have also an important effect on secondary metabolite content. 45
On the other hand, using the DPPH radical-scavenging assay, we found that CAB-DE presents a high scavenging capacity but still lesser than gallic acid which was used as a reference antioxidant molecule. This important antioxidant capacity was due, in part, to the richness in several flavonoid compounds such as procyanidin B2, epicatechin, vitexin, rutin, and isoquercitrin. 46
In vivo, after intra-rectal AA-intoxication (3%, v/v, 5 mL kg−1, b.w.), we noticed a severe and bloody diarrhea. Our results corroborate many previous reports, which explained AA-induced hypersecretion by mucosa degradation and especially apoptosis on the colonic epithelial cells, which are responsible of water and electrolyte absorption. 47
In the present study, we showed that AA induced a decrease in mucus secretion. However, Kühnel 48 explained this deregulation by the destruction of the goblet epithelial cells. We also noticed an increase in the weight of colon and a local congestion of blood vessels. In this respect, Balogun et al. 49 confirmed that this phenomenon is implicated in the amplification of white blood cell production, necessary for the inflammatory reaction.
Importantly, AA (3%, v/v, 5 mL kg−1, b.w., p.a.)-induced ulceration is characterized by the presence of elongated bands of dark red coloring in the distal part of the colon, with edematous areas. These observations are typical to human colitis developing more or less deep lesions and irritations in colonic mucosa. 50 Indeed, acetic acid releases protons making the intracellular space hyper-acid and causing several damages. 51
The microscopic observations demonstrated that AA intoxication caused the alteration of the coating surface, an edema, and epithelium destruction. This damage can gain access to the lamina mucosa, decrease length and number of crypts, and cause acute necrosis of goblet cells and colonocytes. Our results are in line with previous studies showing the same effect of AA. 52 This phenomenon is also characterized by an activation of local inflammatory cells, circulating neutrophil infiltration, and excessive accumulation of arachidonic acid metabolites. 53
However, sub-acute pretreatment with CAB-DE (100, 200, and 400 mg/Kg, b.w, p.o.), GA (50 mg/kg, b.w, p.o.), or sulfasalazine (100 mg kg, b.w, p.o.) protected in a dose-dependent manner against all macroscopic and histological disorders caused by AA. Overall, the degree of protection against AA-induced disorders is ranked in descending order as follows: azerole berries decoction extract, gallic acid, and finally sulfasalazine. This can be explained by the mechanisms of action of each agent or compound. However, the cytoprotection provided by CAB-DE is in part correlated to its richness in phenolic compounds which are also known for their antioxidant and anti-inflammatory properties.54,55 Moreover, tannins have several physiological activities such as blocking the inflammatory mediator synthesis, like prostaglandins and leukotrienes. 56 The study of Pierre et al. 57 has shown that condensed tannins provide the protection and integrity of intestinal physiology; they stimulate mucus secretion by goblet cells, minimize inflammatory reaction, and preserve intestinal profile. Mucus, well-known as a mechanical defense system, limits and inhibits bacterial and viral pathogens from reaching epithelial cells and their adhesion to colonic cells. 58 Mucin 2 (MUC2) plays a very important role in maintaining the mucus layer thickness, the reduction of its secretion, and sulfation being noticed during ulcerative colitis. 59
The important protection showed by gallic acid is due to its ability to reduce the cAMP phosphodiesterase activity, which acts on mast cells by inhibiting their histamine secretions. 60 This latter will have a direct effect on endothelial adhesion molecule expression and minimize neutrophil degranulation and adhesion. 61 In addition, sulfasalazine pretreatment considerably reduced colonic inflammations and eliminated the colonic bacterial flora 57 and its active substance inhibits the mediating axis of the inflammatory response. 62
The present study has also shown that single administration of AA (3%, v/v, 5 mL kg−1, b.w.) induced oxidative stress status in the blood and colonic mucosa. This condition was evaluated by an increase of H2O2 and MDA contents, a depletion of PSA and enzymatic antioxidant activities (SOD, CAT, and GPx), and a decrease of non-enzymatic antioxidant levels (GSH and –SH groups). However, CAB-DE pretreatment significantly protected, in a dose-dependent manner, against the imbalance of oxidative stress parameters induced by AA intoxication. Otherwise, oxidative stress in experimental colitis and regulating effect using plants extract was carried out by previous works.63,64 During inflammatory response, PPNs released reactive oxygen species by membrane and granular NADPH oxidase leading to the superoxide anion liberation and 65 the release of hydroxyl radicals (•OH) or peroxyls (ROO•-) and various highly toxic aldehydes such as malondialdehyde. 66 In the same context, myeloperoxidase (MPO) releases hypochlorous acid, an agent promoting lipid peroxidation, amplifying the inflammatory reaction and releasing ROS. 67 Similarly, during phagocytosis nitric oxide synthase (iNOS) generates an excessive production of nitrogen monoxide (•NO), a very cytotoxic agent, 68 which overcomes the oxidative stress status. Likewise, the highly excessive production of free radicals has been proven to be a potent cytotoxic agent. 69 However, several investigations described the antioxidant properties of phenolic compounds such as phenolic acids, 70 proanthocyanidins, 71 tellimagradin, geraniin, and (-)epigallocatechin gallate. 72 Similarly, flavonoids demonstrate a great inhibitory action on enzymes which generate superoxide radicals. 73 Indeed, the anti-inflammatory and antioxidant activities of alcoholic extracts of berries, flowers, and leaves have been demonstrated in vitro.74,75
In the present investigation, we also found that ulcerative colitis is accompanied by an increase in plasma and colonic mucosa of H2O2, calcium, and free iron levels, which is in accordance with previous studies. 76 This intracellular deregulation is explained by pro-inflammatory cytokine effect, which locally causes an increase in blood supply by congestion and therefore the infiltration of more proteins and leukocytes into the inflamed tissue. 77 Indeed, H2O2 can react with Fe2+ and produce the highly reactive hydroxyl radical (•OH) during the Fenton reaction. 78 The high Ca2 + content in inflamed colons has been explained by the excessive oxidative species production and this phenomenon leads to an increase of electrolytes and water secretion in the intestine. 65 However, other investigations indicated that during inflammation, the endoplasmic reticulum will be stressed and release Ca2+ into the cytosol, which is used in cell signaling. 79 Sub-acute pretreatment with CAB-DE limited and reorganized the H2O2, iron, and calcium homeostasis disorder. The same suggested mechanism has been proved for other plant extracts.64,80
More importantly, we demonstrated that AA increased the marker levels of inflammation in plasma and colonic mucosa such as phosphatase alkaline activity and C-reactive protein content. The pretreatment with CAB-DE, GA, and sulfasalazine reduced the inflammation markers in a dose-dependent manner. In the same context, it has been shown that following a rich diet in fruits and vegetables was inversely correlated with (CRP and IL-6) and other inflammatory parameters such as circulating cytokines (IL-8, IL-13, and IFN-α).81,82 The intestinal alkaline phosphatase function was also described by several other studies to minimize the inflammation level. 83 In this respect, ALP daily treatment of subject with chronic ulcerative colitis decreased the levels of fecal C-reactive protein and calprotectin. 84 In another hand, phenolic compounds were assimilated by the small intestine but most of them were found in the colon. These bioactive substances were therefore in a direct contact with the intestinal epithelium and play an important antioxidant and anti-inflammatory role. 85
In the present work, we also showed that AA-induced colitis was accompanied by a disorder in plasma lipid profile parameters, as assessed by an increase in HDL cholesterol, total cholesterol, and triglyceride levels. However, the sub-acute pretreatment with CAB-DE and GA has significantly minimized these disorders. In this respect, Zapatero 86 demonstrated that Crataegus azarolus berries present a hypolipidemic effect in healthy human. Moreover, condensed tannins and procyanidols of azerole improve lipoprotein metabolism and decrease triglycerides and cholesterol levels. 87 On the other hand, we have also shown that AA intoxication induced hyperglycemia. Our results are in line with those of Maconi et al. 88 Whereas, CAB-DE significantly corrected this metabolic disorder in a dose-dependent manner, which was in accordance with the research of Mostfa et al. 89 In this respect, previous reports also showed that berries and leaves of azerole were used for diabetes treatment. 90
Conclusion
Ours results clearly indicated that CAB-DE significantly protected against AA-induced ulcerative colitis, used as an experimental model for IBD, due in part to its antioxidant and anti-inflammatory properties or also by negatively regulating intracellular mediators and metabolic profile parameters. Overall, our findings support the beneficial effects of Crataegus azarolus berries and their application as a therapeutic/preventive strategy for gastrointestinal diseases.
Footnotes
Acknowledgments
The financial support of the Tunisian Ministry of Higher Education and Scientific Research and the Institution of Agricultural Research and Higher Education are appreciatively acknowledged.
Author Contribution
Houcem Sammari, Saber Jedidi, and Houcine Selmi carried out the experiments, analyzed data, and wrote the article. Mourad Jridi and Ala Ayari participated in the processing and analysis of the data. Hichem Sebai has contributed materials and reagents and assisted in the data treatment and experiment planning. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


