Abstract
This study aimed to evaluate the efficacy and safety of “highly exposed Chinese herbal medicine” combined with apatinib as maintenance treatment following first-line or second-line chemotherapy in patients with ES-SCLC. A total of 23 patients with ES-SCLC were included in this single-arm prospective study (ChiCTR2100045255). “Highly exposed Chinese herbal medicine” combined with apatinib was administered each day after the chemotherapy for maintenance treatment. The primary endpoint of the study was median PFS, while the secondary endpoints included median OS, DCR, ORR, AE, and the association of “highly exposed Chinese herbal medicine” with PFS and OS. Three and 16 patients achieved partial response (PR) and stable disease (SD), respectively, and four patients were with disease progression (PD). The ORR of the patients was 13.0%, DCR was 83.0%, median PFS was 5.0 months, and median OS was 18.0 months. The major AE included secondary hypertension and hand-foot syndrome. Oral intake of Chinese herbal medicine for ≥ 6 months was associated with longer PFS. Hand-foot syndrome was an independent predictive factor for PFS. The statistical analysis suggested no independent influencing factors for OS. “Highly exposed Chinese herbal medicine” combined with apatinib is effective and relatively safe as the maintenance treatment for ES-SCLC patients who undergo first-line or second-line chemotherapy.
Keywords
Introduction
Small-cell lung cancer (SCLC) is a type of high malignant tumor with high invasiveness and rapid growth rate, which could metastasize in relatively early stages. About 80% of SCLC patients are diagnosed when the tumor is already in the advanced stage; the 5 year survival rate is only 1%–2%.1,2 Extensive-stage small-cell lung cancer (ES-SCLC) is highly sensitive to first-line chemotherapy and radiotherapy. Although the objective response rate (ORR) for ES-SCLC is between 60% and 70%, the median survival rate is still low (approx. 8–10 months). ES-SCLC patients generally present with disease progression and recurrence within 1 year after chemotherapy, while the response to second-line therapy is unsatisfactory.3-5 The regimen of therapy with PD-1 inhibitor combined with EP(etoposide plus platinum-containing chemotherapy) for 4 cycles, followed by sequential PD-1 inhibitor (atezolizumab or durvalumab) for maintenance therapy, has shown to be relatively effective against ES-SCLC; yet, the median OS and median PFS after therapy remain low. In addition, this treatment regimen is not economically convenient.6,7 Therefore, investigating new maintenance therapy regimens following first-line/second-line chemotherapy to increase patients' survival has become an urgent requirement in clinical practice.
In recent years, integrative Chinese and Western medicine has been highlighted in the medical field, which is also a trend in comprehensive cancer treatment. 8 While Traditional Chinese Medicine (TCM) highlights the macroscopic and external factors, such as clinical manifestations and regulations of the internal environment of patients, Western medicine highlights microcosmic and internal mechanisms, such as tumor regression rate and underlying mechanisms. 9 It is believed that integrative Chinese and Western medicine could become a new important method for the maintenance therapy for ES-SCLC.
Apatinib, an oral small-molecular tyrosine kinase inhibitor (TKI), is a targeted anti-angiogenesis drug developed in China. Apatinib can specifically and competitively bind to the AF binding site of VEGFR-2, consequently blocking the transduction of downstream signals and thus inhibiting angiogenesis of tumor tissues. 10 Apatinib has been approved by the Chinese National Food and Drug Administration (CFDA) and recommended for third-line therapy of metastatic gastric cancer.11,12 Recent evidence has also suggested that apatinib has substantial effects in treating non-small-cell lung cancer.13-15 In this study, we investigated the efficacy and safety of apatinib as the maintenance treatment following first-line or second-line chemotherapy in patients with ES-SCLC.
The molecular mechanism of Yi-Qi-Tong-Luo-Jie-Du formula is based on the theory of “Tumor Toxicity” and “Disease Collaterals” in TCM. “The Tumor Angiogenesis” described in modern medicine is consistent with the conspectus of “Disease Collaterals” described in TCM. “Disease Collaterals” are the channels for tumors that acquire normal Qi and blood, the channels for tumor toxin to spread to other viscera and throughout the body. “Disease Collaterals” participate in various pathological events of tumors, including the development, progression, and metastasis, which play an important role in tumor suppression. 16
In the Yi-Qi-Tong-Luo-Jie-Du formula, Astragalus (Huang Qi) is expert in tonifying Qi and raising Yang, benefiting the body and strengthening the exterior. The pharmacological effects are to enhance immune function and significantly increase the activity of natural killer cells (NK cells).The main effects of Atractylodes macrocephala (Bai Zhu) are to benefit Qi and strengthen the spleen, dry dampness, and promote water circulation. The pharmacological effects contain (1) modulation of gastrointestinal motility; (2) anti-tumor; (3) antibacterial and detoxification; and (4) enhancement of immune function. The anti-tumor mechanisms of Poria (Fu Ling) are to inhibit nucleoside transport and DNA synthesis in tumor cells; enhance the ability of macrophages to kill tumor cells; and activate the body’s tumor immune surveillance system. The main effect of roasted chicken’s gizzard-membrane (Ji Nei Jin) is to strengthen the stomach and promote digestion. After oral administration, gastric juice secretion, acidity, and digestive power are increased, and gastric motility is enhanced. The main effects of liquorice (Gan Cao) are to tonify the spleen, remove phlegm and cough, clear heat, and detoxify of the body. The main effects of Hedyotis diffusa (Bai Hua She She Cao) are dredging disease collaterals, clearing heat and dampness, and detoxification. Cichorium (Shan Ci Gu) is mainly used to clear heat and detoxify the body, disperse nodules, and dredge disease collaterals. Coastal glehnia root (Bei Sha Shen) is mainly used to nourish yin, clear the lungs, benefit the stomach, and promote the production of body fluid. Chinese wolfberry (Gou Qi Zi) is mainly used to improve the immunity of the body and to prevent and fight cancer. Tangerine peel (Chen Pi) is used to regulate Qi and strengthen the spleen, dry dampness, and dissolve phlegm. Thunberg fritillary (Zhe Bei Mu) is mainly used to clear heat and reduce phlegm, disperse nodules, and detoxify.
According to the Chinese Medicine Formular theory, Yi-Qi-Tong-Luo-Jie-Du formula has the function of benefiting Qi and dredging disease collaterals, dispersing nodules, and detoxifying. A large dose of Astragalus (Huang Qi) is used in this formula to reinforce the healthy qi. Atractylodes macrocephala (Bai Zhu), Poria (Fu Ling), chicken’s gizzard-membrane (Ji Nei Jin), and tangerine peel (Chen Pi) are used to strengthen the spleen and benefit qi, thus, enriching and nourishing the healthy qi and drive away evil. Cichorium (Shan Ci Gu), Hedyotis diffusa (Bai Hua She She Cao), and Thunberg fritillary (Zhe Bei Mu) enhances the function of disease collaterals dredging, heat clearing, nodules dispersing, and detoxifying. Coastal glehnia root (Bei Sha Shen) and Chinese wolfberry (Gou Qi Zi) enhance the effect of yin nourishing. Finally, liquorice (Gan Cao) is used to harmonize the effects of all the herbs in the formula.
In previous studies, we have already found that the combination of Yi-Qi-Tong-Luo-Jie-Du formula with chemotherapy for advanced lung cancer can significantly improve the efficacy and reduce clinical symptoms. Related studies have demonstrated that “highly exposed Chinese herbal medicine,” for example, continuous intake of Chinese herbal medicine for ≥ 6 months, may improve progression-free survival (PFS) and overall survival (OS) of patients.17,18 The aim of this study was to evaluate the efficacy and safety of the Yi-Qi-Tong-Luo-Jie-Du formula combined with apatinib as maintenance treatment following first-line or second-line chemotherapy in patients with extensive-stage small-cell lung cancer (ES-SCLC). Our findings will provide new theoretical evidence for the clinical practices for the treatment of tumors with integrative Chinese and Western medicine.
Methods
Patient Selection
Patients with ES-SCLC treated in the Oncology Department of Integrated Traditional Chinese and Western Medicine, the First Affiliated Hospital of Anhui Medical University from October 2017 to September 2020, were included in this study. The inclusion criteria were as follows: (1) with small-cell lung cancer (extensive stage); (2) age between 18 and 70 years; (3) with at least one measurable lesion (according to the CT/MRI evaluation, the maximum diameter of the tumor was ≥ 10 mm), according to the RECIST 1.1 criteria; (4) had already undergone 4–6 cycles of first-line or second-line chemotherapy, and were in the status of SD; and (5) with the ECOG score of 0–2 points. The exclusion criteria were as follows: (1) those treated with other anti-angiogenesis drugs; (2) allergic to apatinib; (3) could not tolerate long-term Chinese herbal medicine treatment; (4) accompanied with cardiovascular diseases (such as heart diseases or uncontrolled hypertension) or hematological diseases; (5) with liver or renal insufficiency; (6) with severe psychological disorders and could not adhere to the treatment; (7) with poor compliance and high risk of loss to follow-up; (8) with hemorrhagic tendency; (9) underwent major surgeries within 28 d before participating in this study; and (10) with perforation or abdominal fistula within 28 d before participating in this study.19,20
The initial screening resulted in 27 patients, of which 23 completely met the inclusion criteria. Four patients were additionally excluded due to the following reasons: one due to limited pathological stage, one due to short life expectancy ( ≤ 1 month), one had mixed tumor type containing small cells, and one due to incomplete baseline assessment. Finally, four patients were with disease progression, 13 patients died, one patient dropped off due to adverse events, and the treatment is still ongoing for five patients at the data cutoff (Figure 1). The profile of patients enrolled in the trial.
This trial (No. ChiCTR2100045255) was approved by the Ethics Committee of the Cancer Center of the First Affiliated Hospital of Anhui Medical University and was performed according to the Declaration of Helsinki. Written informed consent was obtained from all the patients.
Treatment
According to the study protocol, apatinib with the initial dose of 500 mg, once per day was provided for the patients; 28 days’ treatment was considered as one cycle. The Chinese herbal medicine Yi-Qi-Tong-Luo-Jie-Du formula (containing Huang Qi 40 g, Bai Zhu 10 g, Fu Ling 10 g, Bei Sha Shen 10 g, Gou Qi Zi 10 g, Shan Ci Gu 10 g, Chen Pi 10 g, Ji Nei Jin 20 g, Gan Cao 5 g, Bai Hua She She Cao 20 g, and Zhe Bei Mu10 g) was prepared as a decoction for oral administration, twice per day; administration during 28 continuous days was considered as one cycle. The continuous intake of Chinese herbal medicine for ≥ 6 months was considered as “highly exposed Chinese herbal medicine.” The Chinese herbal medicines were prepared by the First Affiliated Hospital of Anhui Medical University. The maintenance treatment was continued until disease progression or the occurrence of intolerable adverse events.
The dose of apatinib was allowed to be adjusted as 500 mg and 250 mg doses every other day, or 250 mg once per day, due to the concerns of adverse responses. When grade 3 or 4 hematological or non-hematological AE occurred, the dosing of apatinib was temporally stopped. In addition, the dose was reduced after the AE was alleviated to ≤ grade 1. However, if the AE did not alleviate after 2 weeks, the treatment was discontinued, and the patient’s participation in this study was ended. 20 If moderate gastrointestinal responses occurred during the oral intake of Chinese herbal medicine, the Chinese herbal medicine was temporally stopped for ≤ 14 d and then was continued at the original dose after the symptoms disappeared.
Response and Toxicity Evaluation
The treatment responses were evaluated according to the RECIST 1.1 criteria. The first evaluation was performed on the 28 d after treatment, after which the patients were evaluated every two cycles until disease progression. Imaging examinations, including CT and MRI, were performed for all the patients to evaluate the tumors every two cycles, and blood routine examination, liver and renal function, tumor markers, coagulation functions, and electrocardiogram (ECG) were evaluated every cycle. The AE were evaluated according to the Common Terminology Criteria Adverse Events Version 4.0 issued by the National Cancer Institute (NCI CTCAE4.0).
Outcomes and Assessment
The primary endpoint of this study was median progression-free survival (PFS). The secondary endpoints included medial overall survival (OS), disease control rate (DCR), objective response rate (ORR), association of “highly exposed Chinese herbal medicine” with PFS and OS, and adverse events (AE). PFS was defined as the time from the start of follow-up after randomization to disease progression, deaths due to any causes, or end of the follow-up. The medial OS referred to the time that exact 50% of patients survived. DCR included the patients achieved complete response (CR), partial response (PR), and stable disease (SD). ORR included the patients who achieved CR and PR.
Statistical Analyses
Statistical analysis was performed using SPSS, version 25 (IBM, USA), and statistical graphs were plotted by R software (survfit and ggplot2 package). Continuous data are presented as medians (inter-quartile ranges [IQR]), and categorical variables are expressed as numbers and percentages. Survival analyses were estimated using the Kaplan–Meier method with log-rank statistics. PFS and OS were used as the study effect index. Cox proportional hazards models were used to identify potential independent predictors of PFS and OS. In a first step univariate, Cox regression analyses were performed. We included explanatory variables with P < 0.20 in the multivariable COX models. Variables in the final model were selected with an Enter method. P < 0.05 was considered statistically significant in the final model.
To estimate the sample size, the lowest limit of median PFS of the patients receiving maintenance treatment was set at 4.1 months. 21 According to the calculation, when the statistical power (1−β) was 0.8 and one-sided α was 0.025, the sample size of 21 could result in a probability of 80% for PFS > 4.1 months. Therefore, the sample size of 23 in this study was applicable.
Results
Baseline Patient Characteristics
Baseline Patient Characteristics.

Treatment Responses
Treatment Responses.

The data presented as n (%) and n (%; 95% CI).
Waterfall Plot of Measurable Lesion Response
According to the results of the Waterfall plot, the maximum diameters of tumors ranged between −0.60% and 54.30% in patients who received the treatment of Yi-Qi-Tong-Luo-Jie-Du formula combined with apatinib. Among the 16 patients with SD, the tumor regression rate in three patients reached −14.1%, −15.1%, and −16.5%, respectively (Figure 2). The Waterfall plot shows the percentage of the tumor regression rate in the 23 patients who received treatment of Yi-Qi-Tong-Luo-Jie-Du formula combined with apatinib, comparing with the baseline level. The tumor regression rates were arranged in increasing order. The colors of the bars indicated different responses during the treatment. According to the RECIST 1.1 criteria, the upper dash line indicates that the tumor diameter increased by 20%. The lower dash line indicates that the tumor diameter decreased by 30%, compared with the baseline levels.
Survival Outcome
Survival analysis was performed in 23 ES-SCLC patients who received the Yi-Qi-Tong-Luo-Jie-Du formula combined with apatinib. Based on the Kaplan–Meier and log-rank tests, the median PFS of the cohort was 5.0 months (95%, 4.0-18.0), and the median OS was 18.0 months (95%, 15.0-NA) (Figure 3). Using Kaplan–Meier to estimate the survival of ES-SCLC patients receiving the maintenance treatment by Yi-Qi-Tong-Luo-Jie-Du formula combined with apatinib. A, PFS; and B, OS. The gray areas in the plots show the 95% confidential intervals. The dash line shows the median survival. The lower tables show the number of patients included in the analysis at each time point. Abbreviation: NA, not applicable; OS, overall survival; PFS, progression-free survival.
Median PFS and Median OS of Previous Lines of Treatment.

P values < .05 are statistically significant.
The treatment response evaluation and survival analysis findings suggested that patients with ES-SCLC could benefit from the regimen of maintenance treatment consisting of Yi-Qi-Tong-Luo-Jie-Du formula combined with apatinib.
Adverse Events
Summary of Adverse Events.

Cox Regression Analysis
Factors That Influenced the PFS of Patients With ES-SCLC by Cox Regression Analysis.

*P < .2.
Factors That Influenced the OS of Patients With ES-SCLC by Cox Regression Analysis.
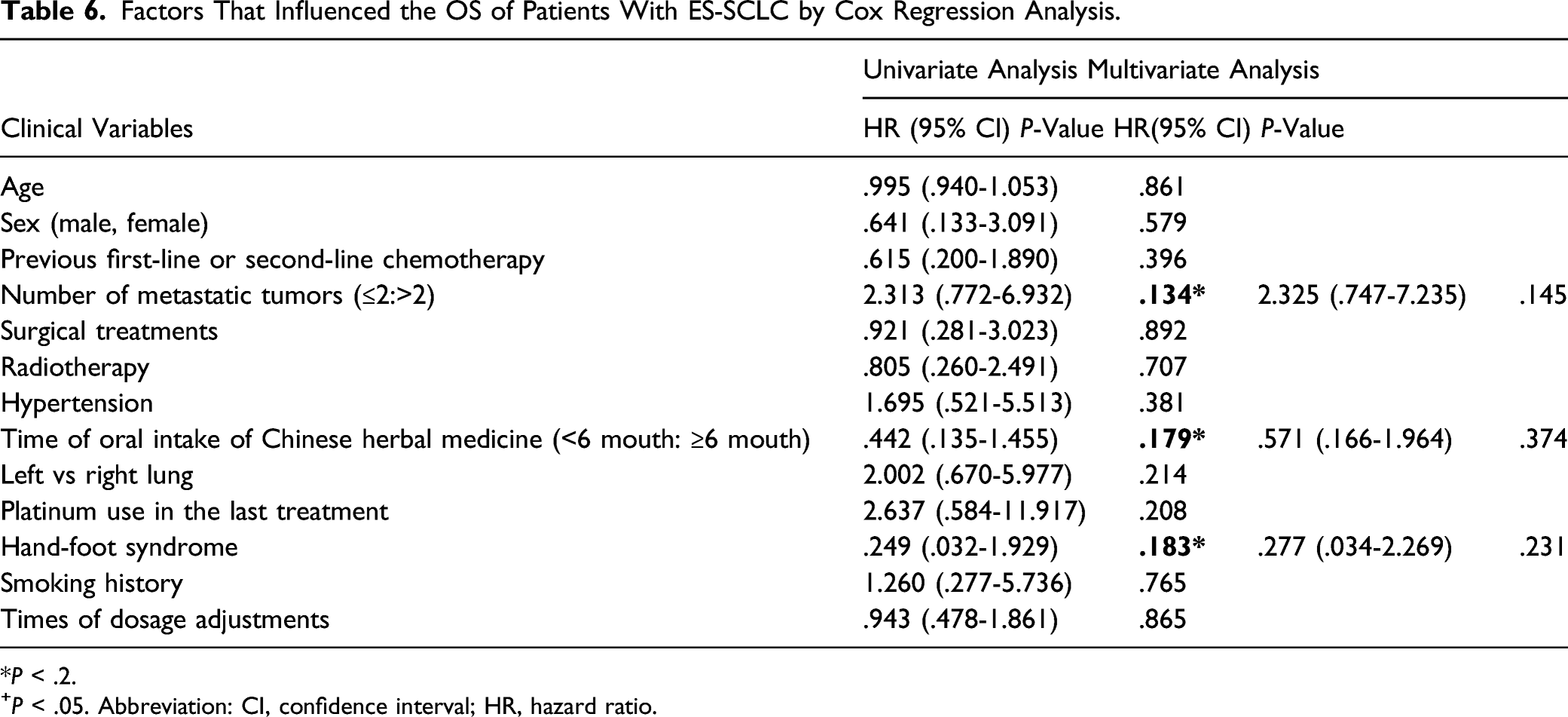
*P < .2.
+P < .05. Abbreviation: CI, confidence interval; HR, hazard ratio.
Dosage Adjustments
Of the 23 patients who received the treatment regimens, the dosage of Yi-Qi-Tong-Luo-Jie-Du formula combined with apatinib (500 mg, once per day) was continued in 7 patients. The dose of apatinib was reduced two times in 10 patients (from 500 mg to 250 mg, once per day in the first adjustment; 250 mg every other day in the second adjustment; the dose of Apatinib was adjusted back to 250 mg, once per day in two patients after the AE alleviated to grade 2). The dose of apatinib was reduced once in 6 patients. During the oral intake of Chinese herbal medicine, moderate gastrointestinal responses occurred in five patients; the treatment was stopped temporally for ≤ 14 d, and then resumed at the original dose after the symptoms disappeared. No significant association of the times of dosage adjustments with PFS (P = 0.557) of OS (P = 0.865) was found.
Discussion
Although several treatment methods have been proposed, the overall prognoses and OS in patients with ES-SCLC still remain unsatisfactory. The major reason is the drug resistance in the patients with disease progression after the first-line chemotherapy, which consequently leads to extremely poor responses to the second-line chemotherapy. 22 Integrative Chinese and Western medicine has been suggested as an alternative treatment (maintenance treatment) for ES-SCLC. To the best of our knowledge, this is the first study that investigated the efficacy and safety of “highly exposed Chinese herbal medicine” combined with apatinib as the maintenance treatment for patients with ES-SCLC.
The findings of this study showed that the ORR was 13.0% (95% CI; 0.0-26.7), DCR was 83.0% (95% CI; 67.6-98.4), median PFS was 5.0 months (95% CI, 4.0-18.0), and median OS was 18.0 months (95% CI, 15.0-NA). The major AE included secondary hypertension and hand-foot syndrome. According to the multivariate analysis results, oral intake of Chinese herbal medicine for ≥ 6 months was associated with longer PFS (P = 0.033). Hand-foot syndrome was an independent prognostic factor for PFS (P = 0.039). Yet, no independent influencing factors for OS were discovered.
The findings of the IMpower133 study23,24 showed that therapy with the regimen of atezolizumab combined with EP (etoposide plus platinum-containing chemotherapy) for four cycles, followed by sequential atezolizumab could improve the median PFS (5.2 vs 4.3 months) and OS (12.3 vs 10.3 months) in patients with extensive-stage small-cell lung cancer compared to placebo combined chemotherapy group. Moreover, the CASPIAN study showed that first-line therapy by the regimen of durvalumab combined with the etoposide/carboplatin or cis-platinum for four cycles, followed by sequential maintenance treatment by durvalumab could improve the median OS (13.0 vs 10.3 months) in extensive-stage small-cell lung cancer patients compared with the single chemotherapy group.25,26 Both studies reported that the median OS was only increased by over 2 months. The findings of this study showed that the improvement in median PFS was not substantial, but the median OS was considerably increased. However, the bias from the single-arm study could not be ruled out.
Maintenance treatment by anti-angiogenesis drugs for ES-SCLC has already shown preliminary effects. Yet, previous studies mainly investigated the effects of monotherapy. 27 A recent study used sunitinib for the maintenance treatment following the chemotherapy by irinotecan/carboplatin in ES-SCLC patients and showed that the 1 year OS was 54% and median PFS was 7.6 months (95% CI 5.7-9.0). 28 A single-center retrospective clinical study reported that the median PFS was 4.1 months and median OS was 12.5 months in ES-SCLC patients who received apatinib for maintenance treatment following chemotherapy. 21 Compared with monotherapy by apatinib, maintenance treatment by “highly exposed Chinese herbal medicine” combined with apatinib showed advantages in both median PFS and OS in ES-SCLC patients, suggesting that the Yi-Qi-Tong-Luo-Jie-Du formula has an important role in the maintenance treatment of ES-SCLC.
A prospective, small sample-sized study performed by the Oncology Department, Guang’anmen Hospital (China Academy of Chinese Medical Sciences) showed that ES-SCLC patients who received oral intake Chinese herbal medicine for ≥ 7 months following chemotherapy had longer PFS. 10 Similar findings were demonstrated in this study. In detail, multivariate analysis showed that oral intake of Chinese herbal medicine for ≥ 6 months was associated with longer PFS (P = 0.033). The findings also demonstrated that even patients with disease progression had longer survival time, which could be associated with Chinese herbal medicine’s long-term use. Chinese herbal medicine could regulate the tumor microenvironment and consequently reduce the invasive capacity of tumors, thus increasing the PFS.
It has been reported that the toxic side effects of VEGFR TKI could be used as a biomarker for the evaluation of treatment responses.29,30 A recent study evaluated the apatinib monotherapy (500 mg, once per day) in patients with gastric cancer and found that hand-foot syndrome, among the common AE, was an independent prognostic factor for both PFS and OS. 31 In this study, the dose of apatinib was 500 mg, once per day, which was safe and controllable; grade 3 AE only occurred in three (13%) patients (including hypertension in one patient [33%] and hand-foot syndrome in two patients [67%]). Other patients presented with grade 1-2 AE, and no grade 4 AE was observed. In addition, no hemorrhagic or perforation events or AE-induced death occurred in the study. The further multivariate Cox regression analysis showed that hand-foot syndrome was an independent prognostic factor for PFS (P = 0.039). Hand-foot syndrome is a common treatment-related AE that could be managed by topical application of drugs. Previous studies have demonstrated that hand-foot syndrome is one of the most common side-effects of anti-VEGF treatment, which has also been suggested as a biomarker predicting the responses and prognoses of anti-VEGF treatment. 32
The dose of apatinib in different previous clinical trials on solid tumors ranged from 500 to 750 mg once per day.33,34 Therefore, the initial dose of apatinib in this study was 500 mg once per day. However, the clinical findings showed that most Chinese SCLC patients could not tolerate this dose. Appropriate intervention could generally control the AE, and almost all the patients could continue the treatment after dosage adjustment. Therefore, we suggested that the initial dose of Apatinib should be 500 mg and 250 mg, once every other day.
However, there were also several limitations in this study. For instance, this study was a single-arm study, without “other Chinese herbal medicine therapy” or “other antiangiogenic therapy” as the control groups may lead to bias; some patients are still in the treatment and have not reached the end point of PFS evaluation. In addition, the sample size was relatively small, and the doses were frequently adjusted. More multi-center clinical studies with larger sizes are needed to further verify our findings.
Conclusion
In summary, our data suggest that “highly exposed Chinese herbal medicine” combined with apatinib is effective and relatively safe as the maintenance treatment for ES-SCLC patients who undergo first-line or second-line chemotherapy. Three and 16 patients achieved partial response (PR) and stable disease (SD), respectively, and four patients were with disease progression (PD). The ORR of the patients was 13.0%, DCR was 83.0%, median PFS was 5.0 months, and median OS was 18.0 months. The major AE included secondary hypertension and hand-foot syndrome. Oral intake of Chinese herbal medicine for ≥ 6 months was associated with longer PFS. Hand-foot syndrome was an independent predictive factor for PFS. The statistical analysis suggested no independent influencing factors for OS.
Footnotes
Acknowledgments
The authors would like to thank Professor Song Wu from the Anhui University of Traditional Chinese Medicine for his help with statistical analyses.
Author’s Contributions
MQW and HLJ conceived and coordinated the study, designed, performed, and analyzed the experiments, and wrote the paper. MZ, YC, TW, and PL carried out the data collection, data analysis, and revised the paper. All authors reviewed the results and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation for Youth Development Fund of the First Affiliated Hospital of Anhui Medical University (2019-2896) and the National Natural Science Foundation of China (81673908). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
