Abstract
Background
Small cell lung cancer is sensitive to chemotherapy and radiotherapy, but local recurrence and distant metastasis occur shortly after treatment. This study aimed to evaluate the real-world value of anlotinib as a maintenance therapy in patients with extensive-stage small cell lung cancer (ES-SCLC) after first-line chemotherapy and consolidative thoracic radiotherapy (CTRT).
Patients and Methods
A total of 150 patients with ES-SCLC treated with first-line chemotherapy and CTRT from April 2017 to December 2021 were retrospectively analyzed. After the completion of chemoradiotherapy, patients received anlotinib according to their desire. The primary endpoints were progression-free survival (PFS) and overall survival (OS) after the first diagnosis, and the secondary endpoints were prognostic factors and safety.
Results
The ORR and DCR of patients with ES-SCLC were 50.0% and 80.3%, respectively, in the anlotinib group and 42.9% and 69.0% in the no-maintenance therapy group. The 3-year OS rates were 27.6% and 12.6% in the anlotinib and observation groups (HR = 2.52, P = 0.003), and the median OS times were 23.8 months and 15.3 months. The 3-year PFS rates were 18.2% and 8.8% in the anlotinib and observation groups (HR = 1.76, P = 0.034) with median PFS times of 11.5 months and 8.8 months. After stratification on the basis of clinical response, patients who achieved CR plus PR after chemoradiotherapy had a longer median OS in the anlotinib and observation groups (34.0 months vs 24.8 months, HR = 2.40, P = 0.009). There were higher incidence rates of hand–foot syndrome (27.3% vs 10.5%, P = 0.001), gingival bleeding/hemoptysis (18.5% vs 4.8%, P = 0.015) and rash (33.3% vs 4.8%, P < 0.001) in the anlotinib group than in the observation group.
Conclusion
Maintenance therapy with anlotinib improved the survival of patients with ES-SCLC after first-line chemotherapy and CTRT. Owing to the small sample size of the real-world trial, the reliability of our study needs to be confirmed in more studies.
Keywords
Introduction
Small cell lung cancer (SCLC) is one of the malignant tumors with the worst prognosis, accounting for approximately 13%-15% of all lung cancer cases.1,2 Most patients with SCLC in China are diagnosed in the metastatic stage. On the basis of the NCCN guidelines, 2 the recommended treatment for SCLC is systemic chemotherapy with or without immunotherapy and palliative radiotherapy. The IMpower133 and CASPIAN trials reported that an improvement in median overall survival (OS) of no more than 3 months with immunotherapy combined with chemotherapy.3,4 SCLC is sensitive to chemotherapy, and local recurrence and distant metastasis occur shortly after treatment. The possible reasons include many residual lesions and rapid proliferation of tumor cells. A literature review indicated that residual disease is one of the most important prognostic factors after chemotherapy and radiotherapy.5,6 The exploration of tolerable therapeutic regimens for ES-SCLC after first-line chemotherapy and CTRT to reduce residual disease and decrease tumor cell proliferation is urgently needed.
Maintenance therapy is a sustained antineoplastic strategy that utilizes highly efficient and low-toxicity treatment regimens to prolong treatment time and further maintain sustained tumor remission. Maintenance therapy has yielded encouraging results in non-small cell lung cancer (NSCLC). 7 Maintenance therapy with bevacizumab or pemetrexed in advanced NSCLC patients improved patient survival and reduced residual disease after chemotherapy.8,9 The Pacific trial also revealed that maintenance therapy with durvalumab can improve the long-term survival of patients with locally advanced non-small cell lung cancer (5-year rates for OS: 42.9% vs 33.4%; 5-year rates for PFS: 33.1% vs 19.0%). 10 However, there are currently no guidelines recommending maintenance therapy for ES-SCLC patients after standard chemotherapy.
Anlotinib, an oral and multitarget tyrosine kinase receptor inhibitor, targets vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR), platelet-derived growth factor receptor (PDGFR), and c-kit proto-oncogene protein (c-Kit). 11 The mechanism of action of anlotinib is correlated with the inhibition of angiogenesis and tumor growth. Various clinical trials have reported beneficial findings in ES-SCLC patients receiving anlotinib. 12 A phase II study by Kong et al indicated that the use of anlotinib as maintenance therapy after first-line chemotherapy for ES-SCLC improved both the PFS and OS of ES-SCLC patients. 13 However, large randomized studies confirming the true value of anlotinib as maintenance therapy after first-line chemotherapy for ES-SCLC are lacking. This study aimed to preliminarily determine the value of anlotinib as a maintenance therapy after first-line chemotherapy combined with consolidation radiation (CTRT) for ES-SCLC in the real world.
Patients and Methods
Patient Eligibility
This study was approved by the Ethics Committee of our institute (NO: 2023-395). Considering this retrospective study, the requirement for informed consent was waived. All the patient details were deidentified. The results of this study complied with the STROBE guidelines. 14 Patients had histological or cytological confirmation of extensive-stage SCLC according to the American Joint Committee on Cancer (AJCC) 2018 TNM staging system. The eligibility criteria were age ≤70 years, WHO performance status 0–1, and weight loss ≤15%. All patients had good lung function (a forced expiratory volume in 1 s [FEV1] ≥ 50% of the predicted value) and underwent the following examinations: brain magnetic resonance scan; ultrasound examination of the neck; neck-chest computed tomography (CT) scan; abdomen CT scan showing the liver, pancreas, kidney, spleen, and retroperitoneal lymph nodes; bone scan; and electrocardiography. In addition, liver and renal function tests, blood cell counts, and markers of lung cancer were measured for all patients.
Study Design
The objectives of this trial were as follows: 1) to observe the feasibility and treatment-related toxicity of maintenance therapy after chemoradiation and 2) to compare the differences in survival between patients with extensive-stage SCLC who received maintenance therapy or no therapy.
Treatments
On the basis of previous multidisciplinary consultations, radiotherapy and chemotherapy were performed once patients were diagnosed. Most patients received palliative radiation after chemotherapy.
Systemic Chemotherapy
Systemic chemotherapy consisted of etoposide 100 mg/m2 d1-3 and cisplatin 75 mg/m2 on Days 1–2 or carboplatin (AUC = 4-5, on Day 1). Antiemetic drugs were administered according to clinical guidelines. All patients received 4‒6 cycles of systemic chemotherapy. Then, consolidation thoracic radiotherapy was performed.
Consolidation Radiotherapy
A CT scan was performed three days before radiation therapy. The images were transferred to a 3D planning system (ADAC Pinnacle 1.5, Philips, USA). Three-dimensional intensity-modulated radiation involving field irradiation technology was delivered to the primary tumor and metastatic mediastinal nodes at a total dose of 50–60 Gy in 2.0 Gy fractions. For residual or new metastases after chemotherapy, stereotactic radiotherapy was used, with a total dose of 45–54 Gy delivered in 5–9 Gy fractions. Treatment was designed with computerized radiation dosimetry and delivered with 6-MV X-rays from a linear accelerator.
Evaluation of Chemoradiation Efficacy
Before maintenance therapy, treatment efficacy was assessed according to the Response Evaluation Criteria in Solid Tumors (RESICT) version 1.1. Radiologic data from computed tomography scans or magnetic resonance images were independently interpreted by two radiologists and one radiotherapy specialist. The effects were categorized as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD).
Maintenance Therapy
All patients were asked whether they would choose to receive maintenance treatment with anlotinib. Two weeks after the end of chemoradiation, 12 mg anlotinib was given orally once a day for 14 consecutive days on a 21-day cycle. When Grade 2 or higher toxicity occurred during treatment, the dose of anlotinib was reduced to 10 mg or 8 mg. Anlotinib was used until tumor progression, the occurrence of intolerable toxicity, patient refusal, or the end of the trial.
Evaluation of Treatment-Related Toxicity
Patients were evaluated at least once every two months by interviews, physical examination, complete blood count, and blood chemistry. A CT scan of the primary tumor and metastatic tumor sites was performed monthly. Proven new metastases or recurrence of primary lesions were defined as treatment failure on the basis of imaging and physical examination. The Radiation Therapy Oncology Group (RTOG) and the Common Terminology Standards for Adverse Events (CTCAE) Version 4.0 standards were used to grade toxicity. During treatment, we monitored patients for signs or symptoms of hematologic, pulmonary, or gastrointestinal toxicity.
Follow-up Evaluation
After completing treatment, patients were evaluated by physical examination. Thoracic CT scans were conducted every 3 months for 2 years and then every 6 months. During maintenance therapy, local progression was defined as the enlargement of primary tumors and/or residual metastases that no longer shrank. The emergence of new metastases was defined as the failure of targeted therapy. The survival time was considered the time from the start of radiotherapy to death or the last follow-up.
Data Analysis
Survival time was considered the time from the start of targeted therapy to treatment failure, death, or the last follow-up. Statistical analyses were conducted with SPSS version 24.0 (SPSS, Chicago, IL). The Kaplan‒Meier method was used to construct curves for local control and overall survival rates. Differences were considered statistically significant if the likelihood ratio was P < 0.05. The log-rank test was used to compare treatments by univariate analyses, and the Cox proportional hazards model was used for multivariate analyses. After chemoradiation, all patients were stratified to compare survival differences according to objective response (CR + PR) or nonobjective response (SD + PD).
Results
Patient Characteristics
From April 2017 to December 2020, 150 patients were retrospectively enrolled in the study at the Radiation Department, Yantai Yuhuangding Hospital (Figure 1). The median follow-up time for the patient was 39 months, with the longest follow-up being 60 months. The clinical characteristics of all patients are shown in Table 1.

The flow chart of patient selection.
Baseline Characteristics of all Patients.
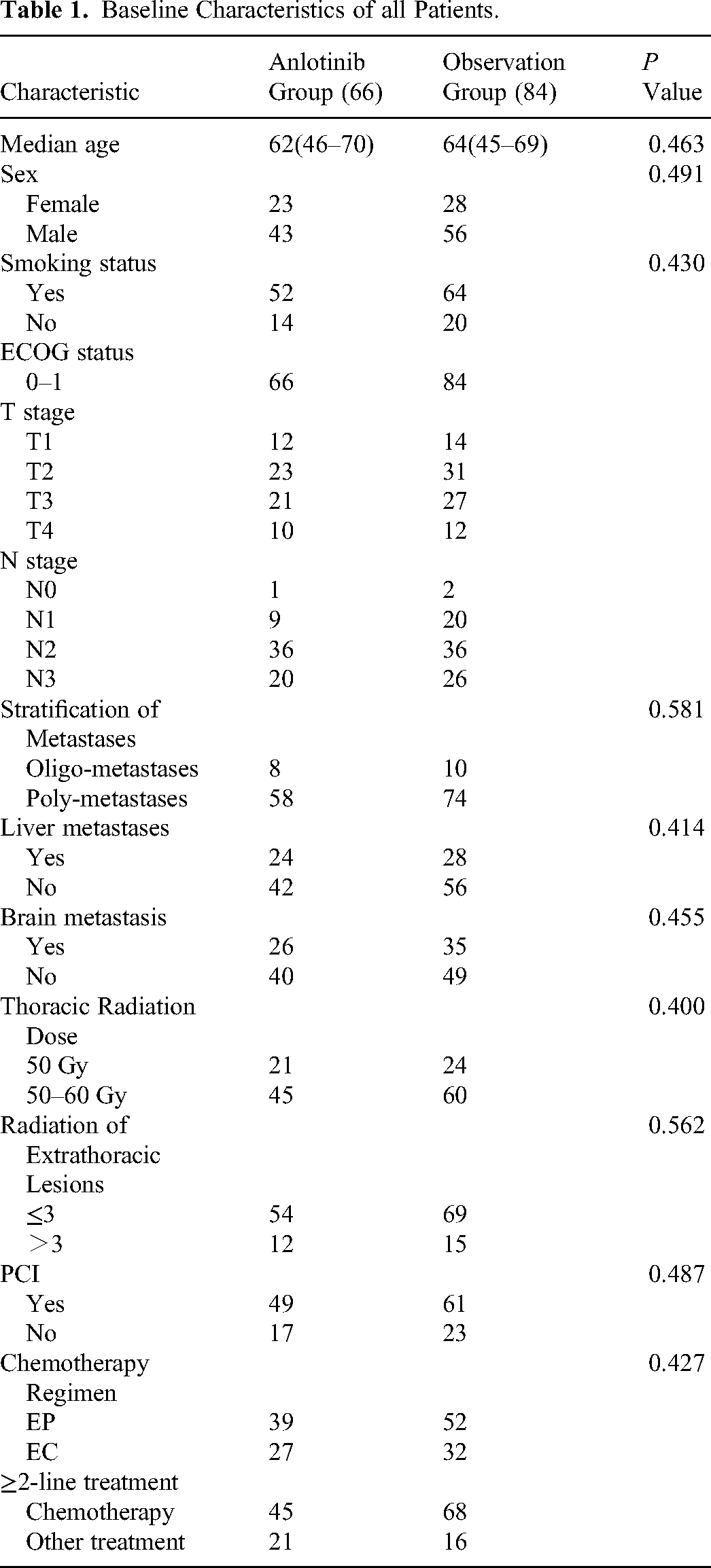
ECOG : Eastern cooperative oncology group. Oligometastases was defined as ≤2 metastatic organs and ≤5 extrathoracic metastatic lesions; otherwise, it was defined as polymetastases. EP regimen was etoposide combined and cisplatin, with EC of etoposide combined and carboplatin. ∗ P < 0.05 was considered significant.
Clinical Response Rate
In the trial, according to the criteria for solid tumor evaluation (CR, PR, SD and PD), there was no significant difference in short-term efficacy between the two groups (P = 0.629). Significant differences in the ORR (42.4% vs 42.9%) and DCR (69.7% vs 69.0%) rates were not observed between the two groups after chemoradiotherapy (Table 2). Nevertheless, the ORR improved from 42.4% to 50.0% after maintenance treatment with anlotinib, and the DCR improved from 69.7% to 80.3%.
Evaluation of Chemotherapy and Consolidation Radiation Efficacy (P = 0.629).

Before maintenance therapy, treatment efficacy was assessed according to RESICT 1.1 by imaging examinations. Complete response: cCR; partial response: cPR; stable disease: cSD; progressive disease: cPD; and sCRT: sequential chemoradiotherapy.
Survival Analysis
The survival curves for all patients are shown in Figure 2. The 1-year, 2-year, and 3-year overall survival rates were 81.7%, 46.1%, and 27.6%, respectively, in the anlotinib group and 70.2%, 26.3%, and 12.6%, respectively, in the observation group (HR = 2.52, P = 0.003). The median overall survival times were 23.8 months (95% confidence interval [CI], 17.60-30.00) and 15.3 months (95% CI, 13.14-17.39) in the anlotinib and observation groups, respectively. The 1-year, 2-year, and 3-year PFS rates were 54.1%, 24.3%, and 18.2%, respectively, in the anlotinib group and 35.3%, 15.5%, and 8.8%, respectively, in the observation group (HR = 2.40, P = 0.034). The median PFS time was 11.5 months (95% confidence interval [CI], 6.50-11.04) in the anlotinib group and 8.7 months (95% CI, 7.52-15.55) in the observation group (HR = 1.76, P = 0.034).
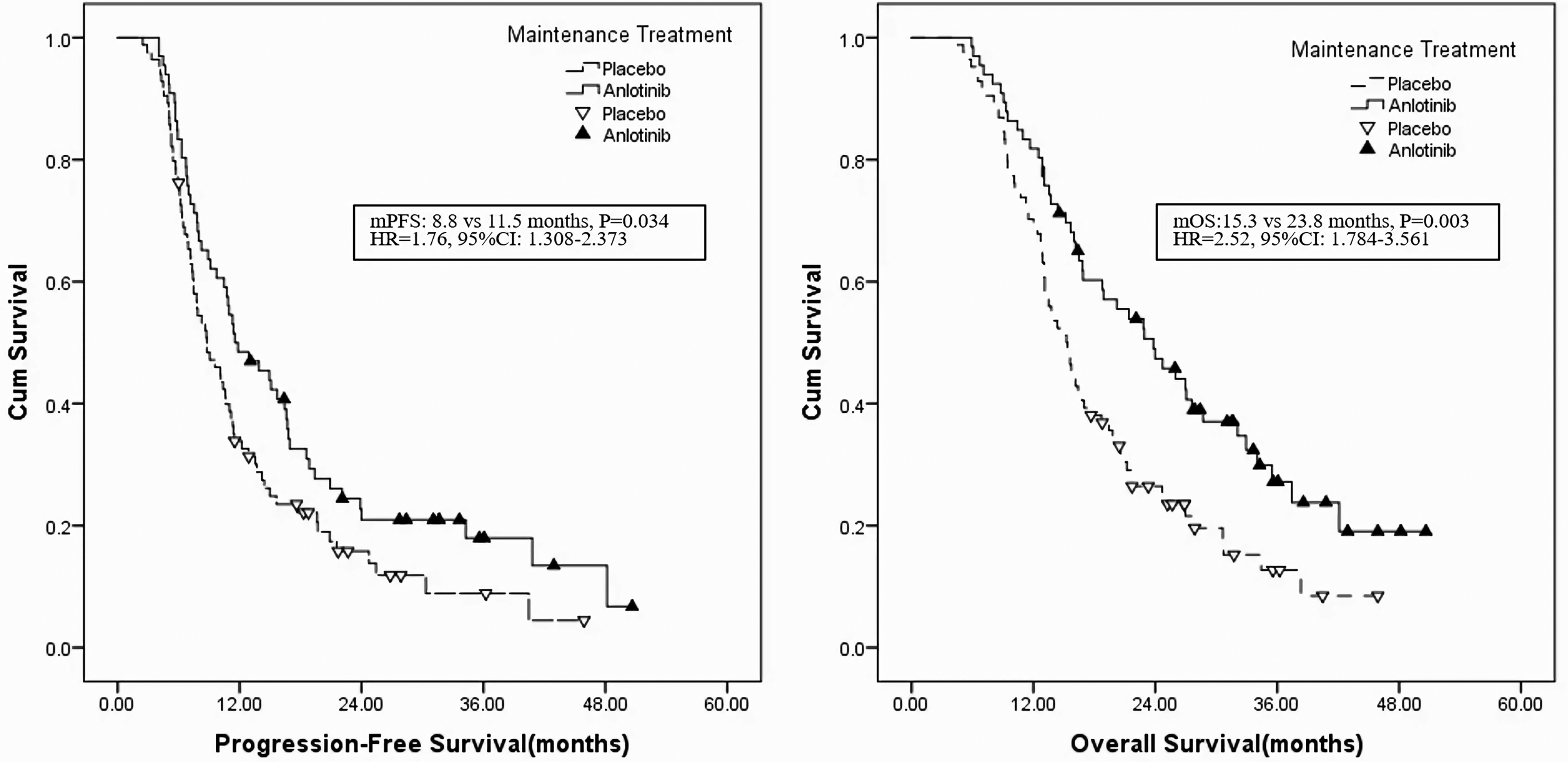
Comparison of PFS and OS between patients treated with anlotinib and observation group.
The clinical response after chemoradiotherapy significantly affected the efficacy of anlotinib according to the stratified analysis (Figure 3). In patients who achieved CR/PR after chemoradiotherapy, OS was longer in the anlotinib group than in the observation group (mOS: 24.8 vs 34.0 months, P = 0.009) (Table 3). No differences in OS were observed between the two groups of patients who underwent SD + PD after chemoradiotherapy (mOS: 12.0 vs 12.8 months, P = 0.150) (Table 3).

After the stratification based on RESICT 1.1 after sequential chemoradiation, overall survival curve between patients treated with anlotinib and observation.
PFS and OS of Patients Stratified Based on RESICT 1.1 After Chemoradiation.
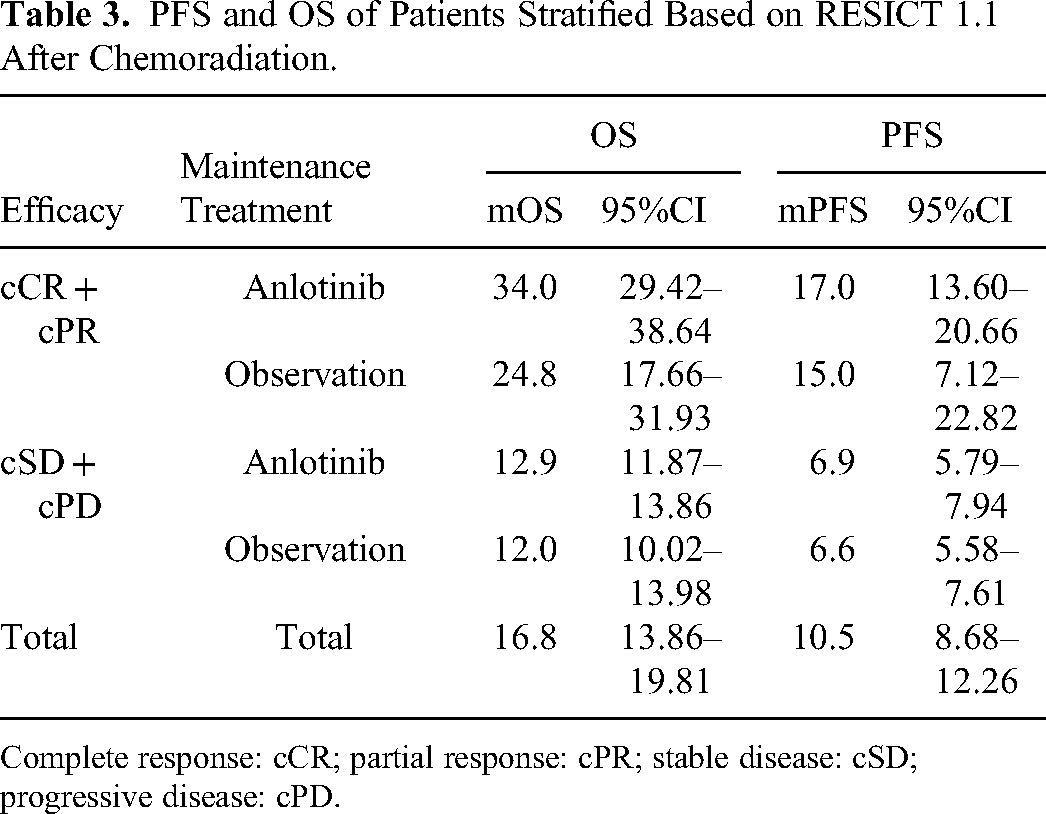
Complete response: cCR; partial response: cPR; stable disease: cSD; progressive disease: cPD.
Patterns of Failure and Prognostic Factors
Multivariate analysis revealed that sex (HR = 5.07, P = 0.021), chemoradiation efficacy (HR = 15.16, P < 0.001), and clinical stage (HR = 4.24, P = 0.043) were prognostic factors for PFS (Table 4). Sex (HR = 5.11, P = 0.024), thoracic radiation (HR = 8.73, P = 0.016), brain metastasis (HR = 14.38, P = 0.004), chemoradiation efficacy (HR = 32.65, P < 0.001) and clinical stage (HR = 3.89, P = 0.0046) were prognostic factors for OS (Table 4). There was a tendency for maintenance treatment with anlotinib to improve OS (P = 0.099).
Cox Regression Analysis of Progression-Free Survival and Overall Survival.

Treatment Toxicity
The incidence of treatment-related toxicities for all patients is shown in Table 5. There was no significant difference between the two groups in terms of treatment-related toxicity (P = 0.123). However, the incidence rates of hand‒foot syndrome (27.3% vs 10.5%, P = 0.001), gingival bleeding/hemoptysis (18.5% vs 4.8%, P = 0.015) and rash (33.3% vs 4.8%, P < 0.001) were significantly greater in the anlotinib group than in the observation group. The Grade 3 or higher toxicities were hypertension (7.6%), hand‒foot syndrome (9.1%), gingival bleeding/hemoptysis (4.5%), and rash (7.6%) in the anlotinib group. Three patients in the anlotinib group died due to sudden cardiac events, acute cerebral thrombosis and chronic kidney disease. Two cases of death from heart disease and cerebral thrombosis were considered to be related to anlotinib, while the other patient had a history of kidney disease, and the cause of death was not related to anlotinib.
Treatment-Related Toxicities in Anlotinib Group (N = 66) and Observation Group (N = 84).

Discussion
Although first-line chemotherapy has a high response rate, most SCLC patients cannot escape disease progression or recurrence. Unfortunately, follow-up treatment for SCLC is limited. The exploration of tolerable therapeutic regimens for ES-SCLC after first-line chemotherapy and CTRT to prevent disease progression is urgently needed. A small-sample phase II clinical trial revealed that maintenance therapy with anlotinib was effective and tolerable after first-line chemotherapy for ES-SCLC, but the real-world value of anlotinib after first-line chemotherapy and CTRT for ES-SCLC patients remains unclear. 13 Consequently, a real-world study was implemented to resolve this issue.
Tumor remission after chemotherapy is an important prognostic factor. Previous studies reported that the response to first-line treatment of SCLC predicts the survival benefit of anlotinib. 15 The survival benefit is greater among patients with cCR after anlotinib, with an mOS of 22.5 months. Moreover, anlotinib has been recommended as a second-line treatment after chemotherapy for patients with PD. The mOS was 4.1 months, and the efficacy of second- or third-line therapy was similar to that reported in the literature.16–18
The increase in treatment lines predicts a lower efficacy of anlotinib in the treatment of SCLC. The mPFS exceeded 8.0 months for first-line treatment with anlotinib but was only 3.6 months for third-line treatment.16,17,19,20 The mPFS of anlotinib, the first-line maintenance treatment for SCLC, was 6.3 months, which was attributed to the emergence of more drug-resistant subclones and a significant increase in tumor burden after multiline treatment failure. 21 Consolidation irradiation of primary and residual metastatic tumors prevented the development and spread of drug-resistant subclones. Zheng et al reported that patients who received higher doses of consolidation radiation had longer PFS (HR = 0.62, P = 0.027) and OS (HR = 0.57, P = 0.019) times. 22
Patients with confined residual disease after treatment have a lower risk of metastasis. 23 These tumors are in an oligometastatic but nonpersistent state. Patients with cCR after chemoradiation have a low risk of metastasis and extended survival time (shown in Figure 3). Patients treated with anlotinib remain in the oligometastatic state for a longer duration.24,25 After the oligometastatic state, treatment rapidly fails, as resistant clones rapidly develop after anlotinib is used for ES-SCLC with a high Ki-67 index.
The prognosis of patients differs between oligometastatic and polymetastatic ES-SCLC. 24 Oligometastatic extensive disease-small cell lung cancer (ED-SCLC) tends to recur locally after chemotherapy, especially in patients with residual disease. New distant metastases are more common in polymetastatic ED-SCLC patients. Consolidation thoracic radiation therapy for oligometastatic ED-SCLC following chemotherapy significantly increased the median OS.26–28 In addition, extrathoracic radiation, but not chemotherapy, may induce a transition from polymetastatic to oligometastatic SCLC in many patients. Extrathoracic radiation improves the median OS of ED-SCLC patients with ≥2 metastases. 29
Previous studies have indicated that drugs that target vascular endothelial growth factor may enhance the efficacy of immunotherapy and that combining anlotinib with immunotherapy may improve treatment efficacy for extensive SCLC, although large randomized clinical trials are still needed. Chen et al reported that the mPFS of patients treated with ICIs plus anlotinib was longer (7.5 months) than that (4.6 months) of patients treated with anlotinib (P = 0.0033), the third-line treatment for extensive-stage SCLC. 30 The mPFS was only 4.6 months for third-line or subsequent treatments. 31 The mOS of SCLC patients was 8.20 months after treatment with anlotinib plus PD-1/PD-L1 inhibitors as second-line treatment. 32
This was a small sample from a real-world trial, and the results indicated that maintenance therapy with anlotinib improved survival after first-line chemotherapy and CTRT in ES-SCLC patients. However, the enrollment population was unbalanced, and the timing and dose of radiotherapy, patient compliance, and dose reduction due to anlotinib toxicity all influence this conclusion. Moreover, the small sample size and bias of the retrospective study may have resulted in a lower incidence of treatment-related toxicities.
Conclusions
In conclusion, our study analyzed the feasibility and efficacy of maintenance therapy with anlotinib after first-line chemotherapy and CTRT for the treatment of ES-SCLC in the real world. Maintenance therapy with anlotinib is a potential and well-tolerated treatment option for the management of ES-SCLC patients after first-line chemotherapy and CTRT. Thus, maintenance therapy with anlotinib warrants further clinical verification.
Footnotes
Author Contributions
JBM, XYM and JS designed the study and drafted the manuscript. WZ, RKZ, SLH and XLW performed the data collection and analysis. All authors read and approved the final manuscrip
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study has been approved by the Ethics Committee of our institute (NO: 2023-395).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
