Abstract
Nonlinear dose–response relationships exist extensively in the cellular, biochemical, and physiologic processes that are affected by varying levels of biological, chemical, or radiation stress. Modeling such responses is a crucial component of toxicity testing and chemical screening. Traditional model fitting methods such as nonlinear least squares (NLS) are very sensitive to initial parameter values and often had convergence failure. The use of evolutionary algorithms (EAs) has been proposed to address many of the limitations of traditional approaches, but previous methods have been limited in the types of models they can fit. Therefore, we propose the use of an EA for dose–response modeling for a range of potential response model functional forms. This new method can not only fit the most commonly used nonlinear dose–response models (eg, exponential models and 3-, 4-, and 5-parameter logistic models) but also select the best model if no model assumption is made, which is especially useful in the case of high-throughput curve fitting. Compared with NLS, the new method provides stable and robust solutions without sensitivity to initial values.
Keywords
Introduction
The dose–response relationship is a cornerstone of toxicology and pharmacology. It is commonly observed in many cellular, biochemical, and physiologic processes in the presence of varying levels of biological, chemical, or radiation stress. Toxicologists and pharmacologists routinely conduct dose–response experiments to explore the effects of compounds or substrates at various concentrations on biological systems, such as cells, tissues, and whole organisms. Data generated from experiments are subsequently utilized to model dose–response relationships and fit curves, which allows scientists to interpolate or extrapolate missing information. 1
Dose–response modeling has been extensively studied in the past few decades, as it is of key importance in toxicology and pharmacology. The dose–response relationship usually resembles a sigmoidal curve with both upper and lower limits. Both parametric and nonparametric models have been proposed to fit sigmoidal curves. Considering that nonparametric models are not very reliable with a small sample size, which is often the case in experimental designs of dose–response studies, parametric models are preferred for dose–response curve fitting. The most commonly used approach to fit nonlinear parametric models is NLS. 2 In general, NLS estimates model parameters by making linear approximations and then refines the parameters by successive iterations based on the sum of squares. There are many algorithms available for NLS implementation, such as the Gauss-Newton algorithm, 3 the Levenberg-Marquardt algorithm, 4 and the NL2SOL algorithm. 5 Although NLS is a very powerful method for modeling nonlinear relationships, it has several drawbacks. First, it is highly sensitive to the choice of starting values. 2 The initial parameter values of NLS must be close to the true values, otherwise there may be no convergence. Moreover, poor initial values may lead to the algorithm settling into local minimum solutions instead of globally optimal solutions that define the least squares estimates. 2
The increasing use of high-throughput screening (HTS) magnifies the limitations of NLS in dose–response modeling. High-throughput screening is widely used in the pharmaceutical and biotechnology fields, and in academic, and federal institutes. 6 -8 Specifically, it is being applied in the discovery of toxicity, 9 biopharmaceuticals, 10 cosmetics, catalysts, 11 pesticide research, 12 and plant science. 13 Such screens assay hundreds of thousands of chemical compounds or organic molecules simultaneously against specific targets under given conditions. Dose–response modeling for HTS data is challenging, not only because of the large size and the limitations of NLS but also because the data for each curve are sparse. For example, there are typically only 6 to 10 concentrations for each sample across many of even the most high-profile and high-impact experiments. 14,15 Additionally, many of these experiments also screen a number of individual cell lines. It is reasonable to expect that the response pattern of each response curve will be different across chemicals in a screen, as well as across individuals within a screen for just one chemical. Restricting the type of models that can be fit greatly limits the impact of such experiments. 6 Additionally, considering that thousands of curves must be estimated, it is impractical to fit each one manually. Therefore, it is necessary to develop computational tools that can automatically model high-throughput dose–response data with minimal human supervision.
A number of software and R packages are currently available for curve fitting, such as benchmark dose software (BMDS),
16
In the present study, to further address challenges in high-throughput curve fitting, we expand the functionalities of EADRM by adding a broad range of dose–response models such as exponential models and 3-, 4-, and 5-parameter logistic models. We also compare different model selection criteria (ie, Akaike information criterion [AIC], Bayesian information criterion [BIC], and
Materials and Methods
Evolutionary Algorithm
Evolutionary algorithms as a class are heuristic-based approaches to solve problems that cannot be easily solved in polynomial time and/or in tandem with other methods, acting as a quick way to find a somewhat optimal starting place for another algorithm to work off of. 20 The premise of any EA is to mimic the process of natural selection to evolve models that fit a given application—in this case, finding a dose–response curve model. An EA contains 4 overall steps: initialization, selection, genetic operators, and termination. These steps each correspond, roughly, to a particular facet of natural selection and provide easy ways to modularize implementations of this algorithm category. Simply put, in an EA, fitter members will survive and proliferate, while unfit members will die off and not contribute to the gene pool of further generations, much like in natural selection. In this study, we use a very general EA approach as described in detail below.
Additionally, we compared the curve fitting performance of our EA approach with another popular EA, differential evolution (DE).
21
Differential evolution is a relatively new EA and is a powerful stochastic real-parameter optimization algorithm in current use.
22
Differential evolution operates through similar computational steps as employed by a standard EA and is population-based like genetic algorithms. However, unlike traditional EAs, the DE variants perturb the current-generation population members with the scaled differences of randomly selected and distinct population members. Specifically, the candidate solutions are initialized randomly at first. Then at each iteration and for each candidate solution Generate a trial vector: Randomly swap vector components between Replace
Built-In Dose–Response Models
Log-logistic models, including 3-, 4-, and 5-parameter logistic models, are by far the most commonly used dose–response models. Therefore, their corresponding model functions are built into our tool. In addition, an exponential model is also incorporated into our tool because the dose–response relationship may sometimes not resemble a sigmoid curve.
Equations 1 to 3 show 3 log-logistic models:

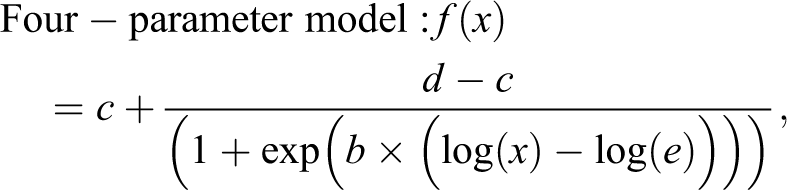

In these log-logistic models,
Equation 4 shows an exponential model:

where β is a scale factor and λ is a growth factor.
Users can choose any model to fit their data. If users do not know which model to use, the tool automatically selects the best model.
Implementation of EA
An EA was implemented to optimize parameters in the selected model. The following describes the optimization process.
A. Initialization
For a faster convergence rate, the initial solution is generated using sensible initialization. That is, the initial parameter values are created based on previous curve fitting experience. For log-logistic models,
B. Evaluation
Each of the random solutions is evaluated by a fitness function to determine how well it fits the data. The fitness function in this study is the BIC, as shown below:

where
C. Selection
Tournament selection is used to select the best solutions from a population.
24
Specifically, the user-defined number of solutions (tournament size N) is randomly selected from the population and then the fitness function is used to select the solution with the best fitness from the N solutions. The process is repeated
D. Mutation
The solutions selected from the current generation are mutated up to ±10% to create the next-generation population of size
E. Termination
The selection and mutation processes are repeated until the user-defined number of generations is reached. Finally, the best solution is selected.
Simulation Data Set and High-Throughput Experimental Data Set
To evaluate the performance of our new tool, real data were used to generate semisynthetic simulations and to demonstrate the performance of the methods on real data. Brown et al 25 performed a high-throughput experiment evaluating the association of genetic variants with differential dose response with anticancer drugs. Lymphoblastoid cell lines (LCLs) were used to perform genome-wide association mapping of the cytotoxic response of 520 European Americans to 29 different anticancer drugs. Details of the experiment and previous results can be found in several previous publications. 25,26 The LCL model has been successfully used to evaluate dose response in a broad range of applications in pharmacology and toxicology. 27
Briefly, Epstein-Barr virus immortalized LCLs were seeded in 384-well plates containing temozolomide (0.10, 0.25, 0.50, 1.0, 2.0, 2.5 mM) obtained from LKT Laboratories. 25 After incubation, the results were quantified on an Infinite F200 microplate reader. 25
A 7-stage quality control (QC) pipeline was used for the cytotoxicity data detailed in a previous study, 28 including the removal of plates containing mostly dead cells, imputation of grossly errant raw fluorescence units (RFUs), QC for the negative control RFUs, and QC for drug vehicle RFUs. The following equation was utilized to compute the percent viability 25 :

where
The final QC procedure includes the following steps. First, ensure that every dose–response curve is monotonic. 25 Second, scale the data so that the mean viability at the lowest drug concentration is 1.0. 25 Third, remove assays containing viabilities above 1.3 or below −0.05. 25 These QC steps result in cleaning 0.5% to 1.5% of data. 25
The data for response to temozolomide from this experiment were used to create simulated data. To generate simulated data sets, small amounts of variation were added to the response values. The variation follows a truncated normal distribution with mean 0 and a standard deviation (SD) of 1, bounded between −1.5 and 1.5.
The real data were also used to demonstrate the methods performance in context of experimental results. Previous results had shown a strong association with differential response to temozolomide with a single-nucleotide polymorphism (SNP) in the
Tool Performance Evaluation
Understanding the impact of parameter choices is important in any EA. 31,32 The simulated data sets were used to investigate the influence of EA parameters on the convergence rate. Evolutionary algorithm parameters include initial population size, equilibrium population size, and initial tournament size. For each parameter being tested, a series of values were evaluated, while other parameters were fixed to the following values: initial population—5000, equilibrium population—200, and tournament size—25. The generated raw data were subsequently averaged.
To tune EA parameters for better performance, 6 different combinations of parameters were evaluated to determine which has the best performance in terms of fitness, variation, and execution time. The simulation experiments were repeated 20 times for each set of parameters, and the mean fitness values, SD, and mean computation time were then calculated.
To compare the initial value sensitivity between EA and NLS, we set up 7 widely ranging starting values for EC50, from 0.0001 to 1000, and then recorded the corresponding results for nonlinear fit with EA and 2 NLS methods: NL2SOL 33 and Gauss-Newton. 34
To investigate this tool’s performance on model selection, 1000 simulation data sets were generated based on a 3-parameter model with added noise magnitude SD 0.01 and another 1000 with added noise magnitude SD 0.1. The tool was then employed to select the appropriate model using different fitness functions (AIC, BIC, and
To demonstrate the utility of using the evolutionary approach, we compared our model fits to curves generated in a random search. To compare this new tool’s performance on high-throughput curve fitting against current computational tools, namely, R package drc and R package DEoptim, our tool was used to fit a high-throughput experimental data set. The mean and SD of
All the code and simulations were written and conducted using the R language. All simulations were conducted in Ubuntu 16.04 LTS using an Intel Xeon Processor E5-2620. An R package implementation is available through CRAN (EADRM.R).
Results
Influence of EA Parameters on Convergence Rate
As evolutionary computation is an iterative optimization procedure, it is of great importance to explore the speed at which an EA converges. Previous studies suggest that the performance of EA is heavily influenced by several parameters such as initial population size, equilibrium population size, and initial tournament size. Therefore, it is necessary to investigate the effect of these parameters on the convergence rate.
Simulation was performed using data generated from 5-parameter logistic models. For each parameter examined, a series of values were evaluated while other parameters were fixed to the following values: initial population—5000, equilibrium population—200, and tournament size—25. The numerical experiments to estimate both convergence rate and running time were performed 50 times, and the raw data were subsequently averaged. As illustrated in Figure 1, all 3 parameters demonstrate similar patterns regarding convergence rate. In general, a small parameter size results in premature convergence on areas of poor fitness, while a relatively large parameter size usually has a good convergence rate. However, the further increase in an already large parameter size does not lead to faster convergence. Figure 2 shows the relationship between a single parameter’s size and execution time. It suggests that there is an exponential trade-off between execution time and tournament size. Additionally, there is a linear trade-off between execution time and initial population size as well as equilibrium population size.
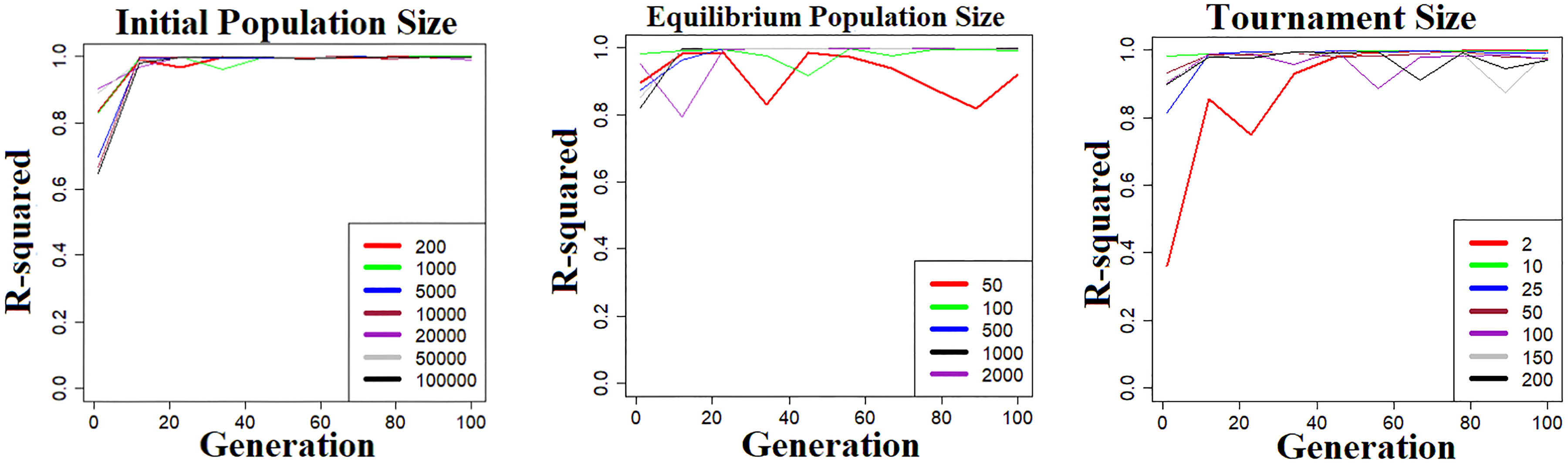
Effect of evolutionary algorithm parameter size on convergence rate. The
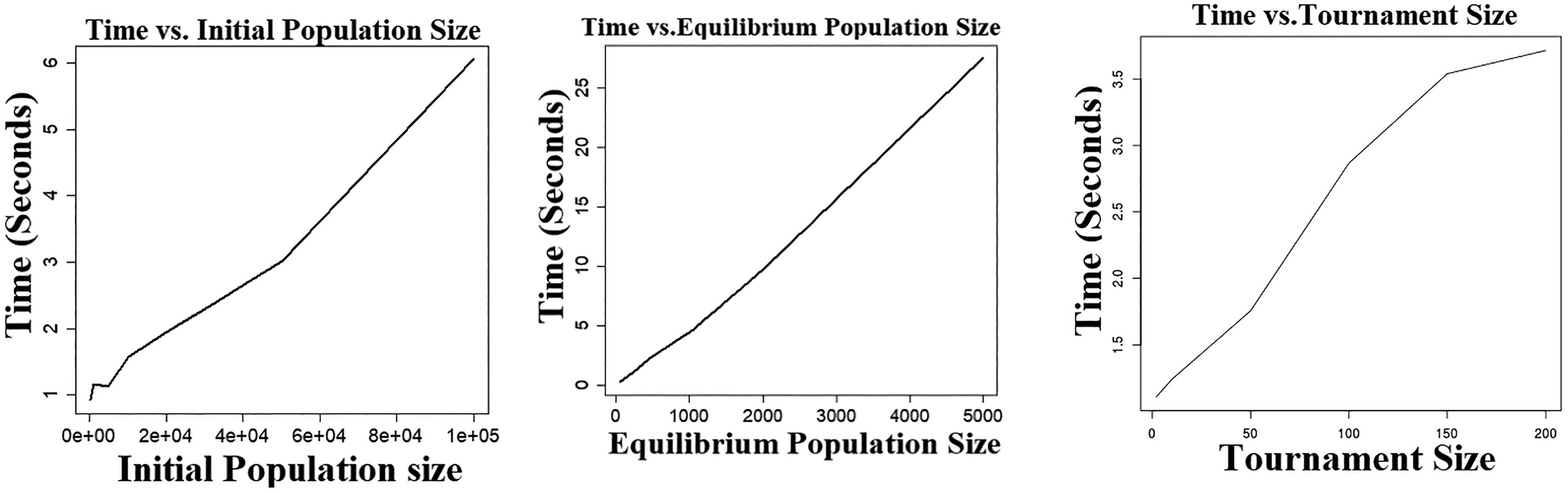
Execution time versus evolutionary algorithm parameter size. The
Parameter Selection in EA
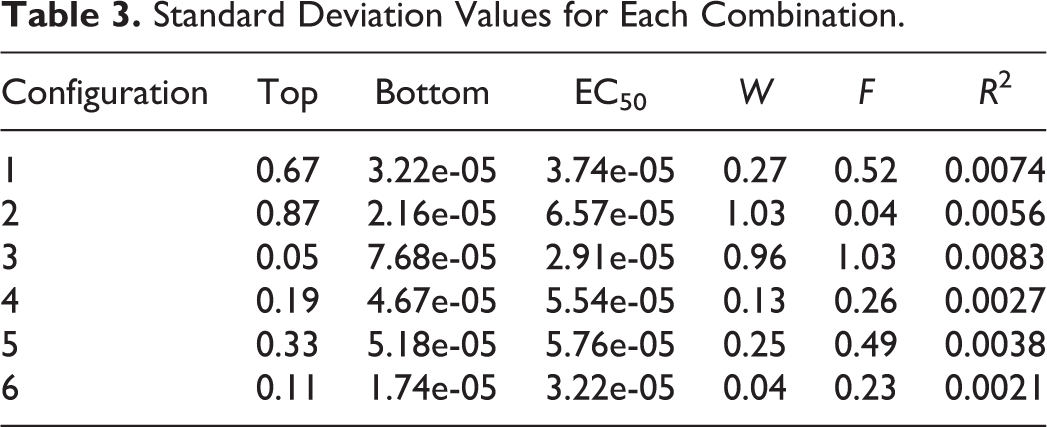
Since EA parameters have a significant impact on the efficiency and ability of EA optimization, it is important to tune these parameters to achieve good performance. In this simulation, 6 parameter combinations (Table 1) were evaluated to determine which has the best performance in terms of fitness, variation, and execution time. The simulation experiments were repeated 20 times for each set of parameters, and the mean fitness values, SD, and mean computation time were then calculated. The results are given in Tables 2 to 4.
Configuration Parameter Values for Each Combination.

Average Parameter Values for Each Combination.

Standard Deviation Values for Each Combination.
Execution Time for Each Configuration.a

a Simulations were conducted in Ubuntu 16.04 LTS using an Intel Xeon Processor E5-2620.
As summarized in Table 2, all 6 sets of parameters returned very good solutions. Set 5 had the best fitness, followed by set 6 and set 3. Table 3 suggests that set 6 generated the most consistent results, followed by set 4 and set 5. In addition, set 1 had the fastest execution time of 23.832 seconds, followed by 58.327 seconds for set 2, presented in Table 4. Although set 5 had slightly better fitness than set 6, it ran about 2.5 times slower than set 5. Considering these factors, set 6 had the best overall performance and was thus selected for the default parameter settings.
Comparison With Random Search
The random search was run so that an equal number of random curve fits to the number of curves evaluated within the EA across all generations. Table 5 presents the results for the best fits generated by the 2 approaches. The EA algorithm very clearly outperforms the random search.
Fitness Summary of EA and Random Search on High-Throughput Simulation Data.
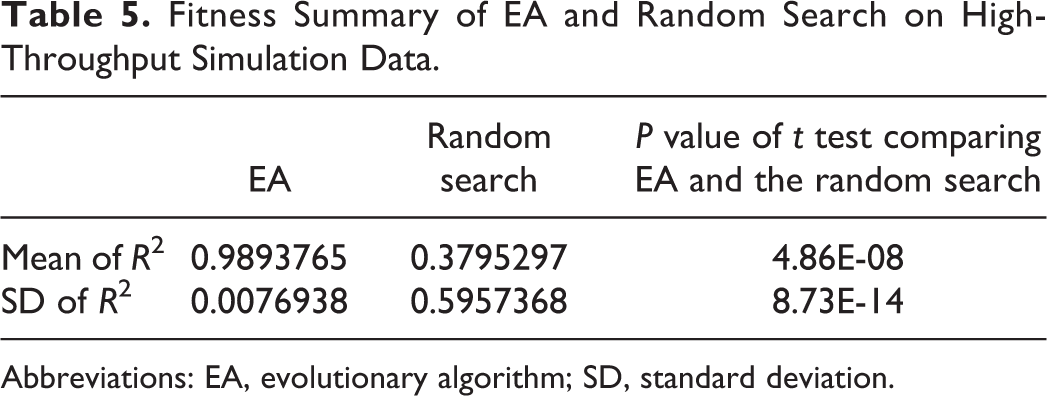
Abbreviations: EA, evolutionary algorithm; SD, standard deviation.
Comparison of Initial Value Sensitivity Between EA and NLS
Nonlinear least squares is a powerful method for modeling nonlinear relationships but has a major disadvantage: sensitivity to the starting values. The initial parameter values of NLS must be close to the true values, otherwise it may not converge or may converge to a local minimum. In this study, the EA is evaluated to assess whether it can overcome this disadvantage of NLS. Considering that EC50 is the most sensitive parameter in the nonlinear-response model, we set up 7 widely ranging starting values for EC50, from 0.00001 to 10 and then recorded the corresponding results for nonlinear fit with EA and 2 NLS methods: NL2SOL and Gauss-Newton. The simulation was conducted using the curve parameters from a 4-parameter logistic model. Tables 6 and 7 summarize the results.
Bias Summary of Initial Value Sensitivity Comparison Between EA and NLS.a

Abbreviations: EA, evolutionary algorithm; NLS, nonlinear least squares.
a NA suggests that an algorithm failed to converge. The true parameter values are as follows:
Fitness Summary of Initial Value Sensitivity Comparison Between EA and NLS.a
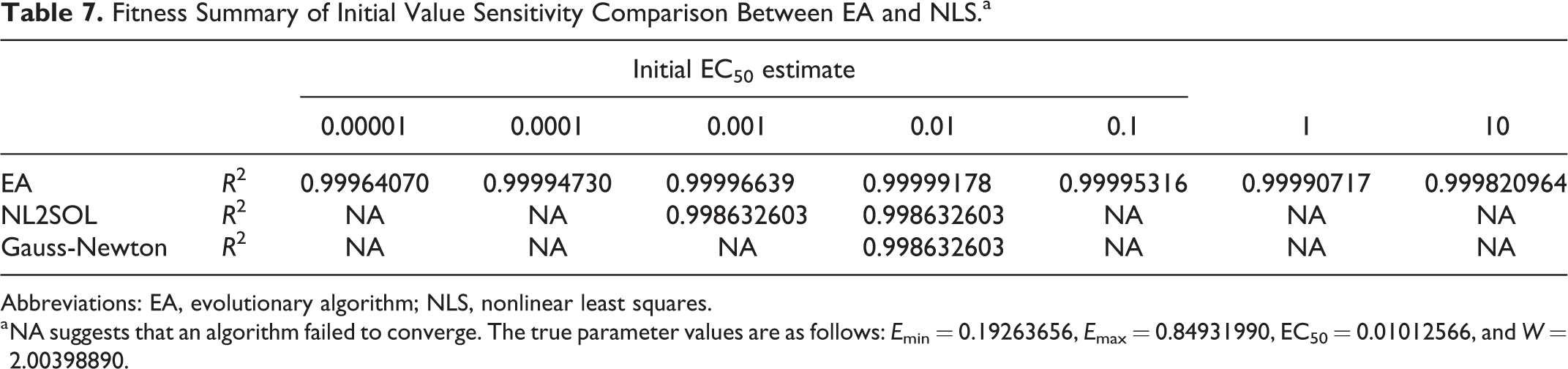
Abbreviations: EA, evolutionary algorithm; NLS, nonlinear least squares.
a NA suggests that an algorithm failed to converge. The true parameter values are as follows:
As expected, both NLS methods produced good solutions with optimum model parameters when the initial EC50 value is close to the true value of 0.01, whereas inappropriate starting values of EC50 led to convergence failure or poor fitness, as presented in Tables 6 and 7. Compared with NLS, EA produced efficient and robust solutions with better fitness for the full range of initial EC50 values, suggesting that EA is relatively insensitive to the choice of initial parameter values.
Model Selection Performance
In addition to modeling nonlinear relationships, our tool can also select the best model from the most commonly used nonlinear dose–response models, including exponential models and 3-, 4-, and 5-parameter logistic models. A major part of the innovation of this method is that it allows analysts to be agnostic to the optimal functional form for the curve fit. To investigate this tool’s performance on model selection, 1000 simulation data sets were generated based on a 3-parameter model with added noise magnitude SD 0.01 and another 1000 with added noise magnitude SD 0.1. The tool was then employed to select the appropriate model using different fitness functions (AIC, BIC, and
Model Selection Frequency for the Different Measures of Goodness of Fit.a

Abbreviations: AIC, Akaike information criterion; BIC, Bayesian information criterion; SD, standard deviation.
a Three-parameter model is the “true” model.
Comparison With Current Computational Tools on High-Throughput Data Analysis
A significant amount of dose–response data is being generated thanks to advances in HTS technology. To evaluate our tool’s performance on high-throughput data analysis, dose–response curve fitting was performed on simulated data sets and our tool was compared with a popular R package DEoptim, which implements the DE algorithm for global optimization. In this comparison, EA automatically selected dose–response models, while Deoptim used 3-, 4-, and 5-parameter dose–response models separately. Table 9 displays the results. It shows that EA has better fitness and higher stability than Deoptim. Furthermore, we performed ANOVA with post hoc test on mean of
Fitness Summary of EA and DEoptim on High-Throughput Simulation Data.
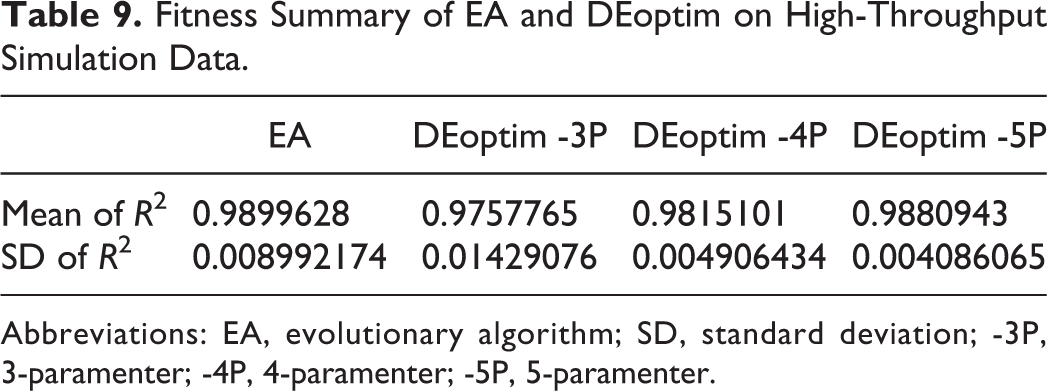
Abbreviations: EA, evolutionary algorithm; SD, standard deviation; -3P, 3-paramenter; -4P, 4-paramenter; -5P, 5-paramenter.
Bias and Variance of Parameter Estimates
Tables 10 displays the results of the bias and evaluations of our method through the simulations, where minimizing both of these measures indicates improved performance. Our results show extremely minimal bias, indicating that the method produces models with both high fitness values and highly accurate parameter estimates.
Bias Summary of EA, DEoptim, and NLS on High-Throughput Simulation Data.

Abbreviations: EA, evolutionary algorithm; NLS, nonlinear least squares; -3P, 3-paramenter; -4P, 4-paramenter; -5P, 5-paramenter.
The bias of the results from DEoptim and NLS was also calculated and compared to our method (given in Table 10). Generally across parameters, our EA approach demonstrated less bias, and the results of ANOVA with post hoc test comparing them demonstrate it is a significant difference (Table 11).

Abbreviations: ANOVA, analysis of variance; EA, evolutionary algorithm; NLS, nonlinear least squares; -3P, 3-paramenter; -4P, 4-paramenter; -5P, 5-paramenter.
Performance on Experimental Data
The real data used were from cytotoxicity assays, where Epstein-Barr virus immortalized LCLs were seeded in plates containing temozolomide. A previous in vitro genome-wide association analysis was conducted with over 2 million QC SNPs and 516 LCLs derived from a Caucasian cohort. It suggests that SNP rs531572 within the
In this study, the EA tool was used to verify if these SNPs were potentially functionally relevant. We examined how the genotype was related to differential dose response in terms of EC50 values. Specifically, the EA tool was used to fit dose–response curves from 516 LCLs and estimate corresponding EC50 values. Subsequently, 1-way ANOVA was employed to test for statistical significance. As shown in Figure 3, rs531572 was found to be highly significantly associated with EC50 values. The

Box plots for the estimated IC50 for temozolomide by genotype for rs531572.
Additionally, the curve fitting performance of our tool was compared with that of R package DEoptim and R package drc on this experimental data set. In this comparison, EA automatically selected dose–response models, while DEoptim and drc used 3-, 4-, and 5-parameter dose–response models separately. Table 12 summarizes the results. It suggests that EA has better curve fitting performance than DEoptim in terms of fitness and stability. It also shows that EA outperforms drc with 3- and 4-parameter models.
Curve Fitting Performance of EA, DEoptim, and drc on High-Throughput Experimental Data.

Abbreviations: EA, evolutionary algorithm; SD, standard deviation; -3P, 3-paramenter; -4P, 4-paramenter; -5P, 5-paramenter.
Software Platform and Availability
This tool was developed using the R statistical language. The code used in this article is available via https://github.com/junmacode/Dose-Response-Modeling-EA. Additionally, an R package EADRM.R is available through CRAN.
Discussion
In the present study, we implemented an EA to model nonlinear dose–response relationships. Our original motivation for this new tool was to address 3 major limitations from current curve fitting tools: the sensitivity of curve fits to initial values with NLS models, the limitation of an analyst having to arbitrarily choose the functional form of a model, and challenges with high-throughput curve fitting due to differences in functional form and other factors across different cell lines, chemicals, and so on.
Our results show that this approach provides stable and robust solutions without sensitivity to initial values. As shown in section “Results,” in spite of dramatically different starting values (from 0.0001 to 1000), EA produced excellent solutions and converged to almost the same parameter values. Unlike NLS, which provides good solutions only when the initial values are very close to the true value, users of our tool do not need to spend time determining an appropriate starting point as they can randomly choose a starting value. One possible and important use case of this approach could be to use the EA to determining reasonable initialization values for downstream use in NLS.
Further, the tool can automatically model high-throughput dose–response data with minimal human supervision. As mentioned, hundreds of thousands of dose–response curves can be generated for a single project due to advances in HTS technology, and it is impossible to fit these curves manually. Our tool provides a good model selection function to automate analysis with minimal analytical intervention. More importantly, since EA uses BIC as the fitness function, it can perform model selection during the optimization process. In contrast, most tools can perform model comparison only after all models are optimized.
Additionally, both simulation and real-data analysis suggest that our tool fits dose–response data better than another popular R package, DEoptim, which implements the DE algorithm for global optimization. There are 2 main reasons for this. First, we have optimized the hyperparameters of evolutionary algorisms specifically for dose–response modeling. Second, our tool is capable of automatically selecting the best dose–response model while only a fixed model can be used for dose–response curve fitting in DEoptim. Our EA produces parameter estimates that are highly accurate, with minimal bias and less bias compared to DEoptim.
Although we are encouraged by these initial results, there are still important caveats that need to be considered, and as possible addressed in future work. Although we implement several functional forms in this toolkit, there are others that should be considered. There is room to extend built-in models, such as the Brain-Cousens models, Cedergreen-Ritz-Streibig models, 35 log-logistic fractional polynomial models, and log-normal models. This will make our tool more powerful and enable it to handle other types of responses. Additionally, the computation time for the current implementation is limiting. As this is an introduction to the approach, ongoing efforts will rewrite the tool in C to significantly reduce run time. For very large HTS efforts, combining this tool with faster approaches may be optimal. In ongoing work, we will implement this approach into the popular BMDS. 16 As mentioned above, the EA could be used to find the best functional form and reasonable initial parameters in a minimal number of generations and then other faster approaches could be used to find final models. Also, using an EA does not address some of the caveats that are inherent to nonlinear curve fitting in general. For example, for data with no asymptote, a unique solution cannot be guaranteed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by intramural funds from the National Institute of Environmental Health Sciences.
