Abstract
Sulforaphane exerts anti-inflammatory activity in inflammatory diseases. The endometriosis (EM) is accompanied by chronic inflammation. The present study aims to explore the therapeutic effects of sulforaphane on EM and its underlying mechanism. An EM rat model was established by transplantation of autologous fragments. The rats were intragastrically administered sulforaphane (5 mg/kg, 15 mg/kg, and 30 mg/kg) for 3 weeks. The volumes of endometriotic foci and adhesion score were calculated at the end of the experiment. Levels of interleukin (IL)-6, IL-10, tumor necrosis factor (TNF)-α, interferon (IFN)-γ, and vascular endothelial growth factor (VEGF) were determined by enzyme-linked immunosorbent assay (ELISA). Expressions of VEGF, B-cell lymphoma/leukemia 2 (Bcl-2), Bax, cleaved caspase-3, PI3K, and Akt in endometrial tissue were determined by Western blotting. Relative expressions of PI3K and Akt were determined by quantitative polymerase chain reaction. Posttreatment of sulforaphane dose-dependently decreased the volumes of endometriotic foci and adhesion score in EM model. Additionally, posttreatment of sulforaphane inhibited levels of IL-6, IL-10, TNF-α, IFN-γ, and VEGF in peritoneal fluid and plasma. Posttreatment of sulforaphane regulated the expressions of VEGF, bcl-2, Bax, and cleaved Caspase-3 in EM model. The underlying mechanism revealed that sulforaphane attenuated EM in the rat model by inhibition of PI3K/Akt signaling pathway.
Introduction
Endometriosis (EM) is a chronic disease characterized by endometrial-like tissue outside the uterine cavity. 1 It has been recognized to be associated with pelvic pain and impaired fecundity in adolescents. 2 There are 6% to 10% of women with reproductive age, 50% to 60% of women with pelvic pain, and as many as 50% of women with infertility affected by EM. 3 Endometriosis is associated with an increased risk of developing into autoimmune diseases, ovarian cancer, and EM-associated adenocarcinoma. 4,5 The first line recommended method for EM is nonsteroidal anti-inflammatory drugs or oral contraceptive pills therapy. 6,7 However, many patients still have pelvic pain despite these medications. Therefore, it is important to discover novel drug candidates for EM therapy.
The underlying mechanisms which induce EM are still not fully understood. However, some studies have demonstrated that the occurrence and development of EM are associated with a disorder of the immune systems. 8,5 Investigations have demonstrated some hallmarks of EM including cell apoptosis and inflammation. 4 For instance, Agic and colleagues have demonstrated that the development of EM is correlated with increased numbers of macrophages and elevations of inflammatory cytokines including monocyte chemoattractant protein-1, interleukin (IL)-6, and tumor necrosis factor (TNF)-α. 9,10 Additionally, some studies have found aberrant expressions of apoptosis markers including B-cell lymphoma/leukemia 2 (Bcl-2) family and caspase-1, 3 in ectopic endometria of EM animal models. 11,12 Therefore, investigation of compounds possessing anti-inflammatory and pro-apoptosis properties might provide a good resource for the discovery of anti-EM drug candidates.
The potential protective effects of consumption of cruciferous vegetables on the occurrence of cancer have been extensively investigated in these years. 13 Sulforaphane is an isothiocyanate isolated from cruciferous vegetables including cabbage, cauliflower, brussels sprouts, and broccoli. Many studies have identified that sulforaphane is a promising compound in chemoprevention and therapy of different types of cancer including prostate cancer, colon cancer, breast cancer, and hepatocellular carcinoma. 14,15 Multiple mechanisms are involved in the chemopreventive and therapeutic effects of sulforaphane on cancer. These include (1) induction of apoptosis, (2) inhibition of tumor angiogenesis, (3) inhibition of inflammation, (4) inhibition of phase I enzymes, (5) induction of phase II detoxification enzymes, and (6) suppression of oxidative stress. 16,14 All these findings encourage us to investigate the effects of sulforaphane on EM. Therefore, the present study aims to explore the effects of sulforaphane in the development of EM and its underlying mechanisms. We identified that sulforaphane ameliorated EM in rat models by multiple effects include (1) inhibition of inflammation-related cytokines including TNF-α, IL-6, IL-10, and interferon (IFN)-γ; (2) inhibition of vascular endothelial growth factor (VEGF); (3) regulation of apoptosis-related biomarkers including Bcl-2, Bax, and cleaved caspase-3; and (4) inhibition of PI3K/Akt signaling pathway.
Materials and Methods
Reagents
Sulforaphane was purchased from Sigma Aldrich (St. Louis, Missouri). Sterile phosphate–buffered saline solution and dimethyl sulfoxide (DMSO) were purchased from Beyotime Biotechnology (Haimen, Jiangsu, China). Sterile 0.9% sodium chloride (NaCl) solution was prepared by our laboratory.
Animals
Female Sprague Dawley rats (weighing 200-250 g, 6 weeks) were purchased from Nanjing Model Animal Institute (Nanjing, China). The rats were housed in a 12-hour light–dark cycle and fed under experimental conditions with a temperature of 22°C to 24°C and humidity of 50% ± 5%. The animal experiments performed in this study were approved by Animal Care and Use Committee in Quanzhou First Hospital Affiliated to Fujian Medical University.
Establishment of the EM Model
A rat model of EM was established according to the previously reported methods with minor modifications. 17,18 All the surgical operations were performed under sterile conditions. The rats underwent anesthesia with 3% pentobarbital sodium (intraperitoneal injection). The abdominal hair was shaved and the adnominal cavity was exposed. After that, endometrial tissue was harvested from the myometrium and trimmed into 5 mm × 5 mm pieces. The autologous fragments were transplanted into the inner surface of the abdominal wall. Next, the abdominal cavity was closed with sutures and the rats could recover from anesthesia.
Drug Administration
Sulforaphane was dissolved in DMSO (final concentration < 0.1%) as a stock solution. The stock solution was diluted using 0.9% NaCl solution into 3 doses including 5 mg/kg, 15 mg/kg, and 30 mg/kg. The animals were divided into 6 groups (n = 8). In group 1, the rats underwent sham surgery and were intragastrically administered the same volume of 0.9% NaCl solution. In group 2, the rats underwent sham surgery and were intragastrically administered sulforaphane at a dose of 30 mg/kg. In group 3, the rats underwent surgery and intragastrically administered the same volume of 0.9% NaCl solution. In the group 4 to 6, the rats underwent surgery and were intragastrically administered sulforaphane at doses of 30 mg/kg, 15 mg/kg, and 5 mg/kg. Two treatment methods including pretreatment and posttreatment were applied in this study.
All rats were sacrificed and the volumes of endometriotic foci and adhesion score were calculated. The volumes of endometriotic foci were calculated according to formula, which is π/6 × length × width × height. Adhesion score of the endometriotic lesion was calculated according to the Blauer scoring system. To evaluate the effects of sulforaphane pretreatment on EM, the volumes of endometriotic foci and adhesion score were calculated 3 weeks after surgery. To evaluate the effects of sulforaphane posttreatment on EM, the volumes of endometriotic foci and adhesion score were calculated 6 weeks after surgery.
Enzyme-Linked Immunosorbent Assay
Plasma and peritoneal fluid were collected. Vascular endothelial growth factor, IL-6, IL-10, IFN-γ, and TNF-α were determined using an ELISA kit (BioLegend, San Diego, California), according to the manufacturer’s instructions.
Isolation of Total RNA and Quantitative Polymerase Chain Reaction (qPCR)
Trizol reagent was used to extract total RNA from ectopic endometria, according to the manufacturer’s instructions (Takara, Japan). To remove DNA contamination, RNase-free DNase I was used. Primers for PI3K, Akt, and β-actin were used for amplification of these genes. The sequences are as follows. PI3K forward: 5′-AAC ACA GAA GAC CAA TAC TC-3′, and reverse: 5′-TTC GCC ATC TAC CAC TAC-3′; Akt forward: 5′-TCA CCT CTG AGA CCG ACA CC-3′, and reverse: 5′-ACT GGC TGA GTA GGA GAA CTG G-3′; and β-actin forward: 5′-CCT GGC ACC CAG CAC AAT-3′, and reverse: 5′-GGG CCG GAC TCG TCA TAC-3′. To analyze the accuracy of the PCR reaction, the Melt curves were used. To evaluate the expressions of genes, 2−△△ Ct values were calculated. The mRNA expression value of PI3K and Akt was normalized to that of β-actin.
Western Blotting
The rats were harvested and ectopic endometria were collected. The protein was extracted according to the previously reported methods. 19,20 Briefly, tissues were cut into small pieces and then homogenized in radioimmunoprecipitation assay buffer supplied with protease inhibitors. Next, the lysate was centrifuged at 12000g to remove insoluble material. The supernatant was collected and protein concentrations were qualified using a Bicinchoninic acid protein assay kit (Thermo Fisher, Waltham, Massachusetts)
Equal amounts of protein were separated by 10% sodium dodecyl sulfate (SDS) gel. Next, the SDS gel was then transferred to a polyvinylidene fluoride membrane and 5% nonfat milk was used to block the membrane. After the membrane was probed with antibodies against VEGF, Bcl-2, Bax, cleaved caspase-3, PI3K, Akt, p-Akt, and β-actin overnight at 4°C, the membrane was then incubated with appropriated secondary antibodies. ChemiDoc MP imaging system (Bio-Rad, Hercules, California) was used to examine chemiluminescence. The expressions of proteins of interest were normalized to the internal control β-actin.
Statistical Analysis
SPSS (SPSS, Chicago, Illinois) was used to statistical analysis. All data were expressed as mean value (SD). To evaluate the significance, one-way analysis of variance with multiple comparisons and Student-Newman-Keuls test were performed. A P value less than.05 was considered as a statistical significance.
Results
Effects of Sulforaphane on the Volumes of Endometriotic Foci and Adhesion Score in the EM Rat Model
To explore an ideal treatment method of sulforaphane on the EM rat model, rats were pretreated or post-treated with sulforaphane. After that, the volumes of endometriotic foci and adhesion score were evaluated. As shown in Figure 1A, we observed endometriotic foci with massive volumes in the EM group. Similarly, the highest adhesion score was observed in the EM group (Figure 1B).

Effects of sulforaphane on the volume (A) and adhesion score (B) of endometriotic lesion in a rat model of EM (n = 8). Data are presented as mean (SD). *P < .05, **P < .01 compared to pretreatment group. # P < .05, ## P < .01 compared to posttreatment of EM group. EM indicates endometriosis; SD, standard deviation.
We found that sulforaphane post-treated significantly decreased the volumes of endometriotic foci and adhesion score when compared with those in the EM group. Interestingly, we found that pretreatment of sulforaphane (5 mg/kg) showed less endometriotic foci volumes and lower adhesion score than those in sulforaphane (5 mg/kg) post-treated group. However, posttreatment of sulforaphane at doses of 30 mg/kg and 15 mg/kg significantly decreased the volumes of endometriotic foci and adhesion score when compared with those in the sulforaphane pre-treated group (30 mg/kg and 15 mg/kg). Therefore, the posttreatment of sulforaphane was selected in the following experiments in this study.
Effects of sulforaphane on the Levels of TNF-α, IL-6, IL-10, and IFN-γ in Plasma and Peritoneal Fluid
Endometriosis is an inflammatory process and inflammatory cytokines were elevated in the development of EM. 4 We next evaluated the anti-inflammatory effects of sulforaphane in the EM rat model. Several inflammation-related cytokines including IL-6, IL-10, TNF-α, and IFN-γ in plasma and peritoneal fluid were evaluated. As shown in Figure 2A and B, levels of IL-6, IL-10, TNF-α, and IFN-γ in plasma were significantly increased in the EM group when compared with those in the sham group (P < .01). Posttreatment of sulforaphane significantly decreased levels of IL-6, IL-10, TNF-α, and IFN-γ in plasma when compared to those in in EM group.

Effects of sulforaphane on the levels of cytokines in plasma and peritoneal fluid (n = 8). Levels of TNF-α (A and E), IL-6 (B and F), IL-10 (C and G), and IFN-γ (D and H) in plasma and peritoneal fluid were determined by ELISA. Data are presented as mean (SD). *P < .05, **P < .01, ***P < .001 compared to sham group. # P < .05, ## P < .01 compared to EM group. ELISA indicates enzyme-linked immunosorbent assay; EM, endometriosis; IFN-γ, interferon; IL, interleukin; SD, standard deviation; TNF-α, tumor necrosis factor-α.
Similarly, levels of IL-6, IL-10, TNF-α, and IFN-γ in peritoneal fluid were significantly increased in the EM group when compared to those in the sham group (Figure 2C and D, P < .001). In addition, posttreatment of sulforaphane significantly decreased levels of IL-6, IL-10, TNF-α, and IFN-γ in the peritoneal fluid when compared with those in the EM group. Notably, sulforaphane significantly decreased levels of IL-6, IL-10, TNF-α, and IFN-γ in peritoneal fluid and plasma in a dose-dependent manner.
Effect of Sulforaphane on the Protein Levels of VEGF in the Plasma, Peritoneal Fluid, and Ectopic Endometria
We investigated the effects of sulforaphane on the protein expressions of VEGF in the plasma, peritoneal fluid, and ectopic endometria. As shown in the Figure 3A and B, levels of VEGF in the plasma and peritoneal fluid were significantly increased in the EM group when compared to those in the sham group. Interestingly, sulforaphane (30 mg/kg and 15 mg/kg) post-treated significantly decreased levels of VEGF in peritoneal fluid and plasma in a dose-dependent manner.

Effect of sulforaphane on the levels of VEGF in the EM models (n = 8). Levels of VEGF in peritoneal fluid (A) and plasma (B) were determined by ELISA. Expressions of VEGF in ectopic endometria were determined by Western blotting. Data are presented as mean (SD). *P < .05, **P < .01, ***P < .001 compared to sham group. # P < .05, ## P < .01 compared to EM group. ELISA indicates enzyme-linked immunosorbent assay; EM, endometriosis; SD, standard deviation; VEGF, vascular endothelial growth factor.
Next, we evaluated the expressions of VEGF in the ectopic endometria. As shown in the Figure 3C and D, the expressions of VEGF in the ectopic endometria were significantly increased in the EM group when compared to those in the sham group. However, sulforaphane (30 mg/kg and 15 mg/kg) post-treated significantly decreased the expressions of VEGF in the ectopic endometria when compared with that in the EM group. These results supported the effects of sulforaphane on angiogenesis in part by inhibition of expressions of VEGF.
Effects of Sulforaphane on the Expressions of Apoptosis-Related Factors in the EM Rat Model
Aberrant expressions of apoptosis markers including Bcl-2 family and caspases enzymes have been reported in the development of EM. 12 Therefore, we further investigated the effects of sulforaphane on the expressions of apoptosis-related factors in the EM rat model. As shown in the Figure 4A-C, we found that a decrease in expressions of Bcl-2 while an increase in expressions of Bax in the sulforaphane (30 mg/kg and 15 mg/kg) treated groups when compared to those in the EM group. Additionally, we also observed that expressions of cleaved caspase-3 were significantly increased in the sulforaphane (15 mg/kg and 30 mg/kg) treated groups when compared to those in the EM group (Figure 4D, P < .05, P < .01). These results supported that sulforaphane attenuated EM in part by the regulation of apoptosis-related proteins including Bcl-2, Bax, and caspase-3.
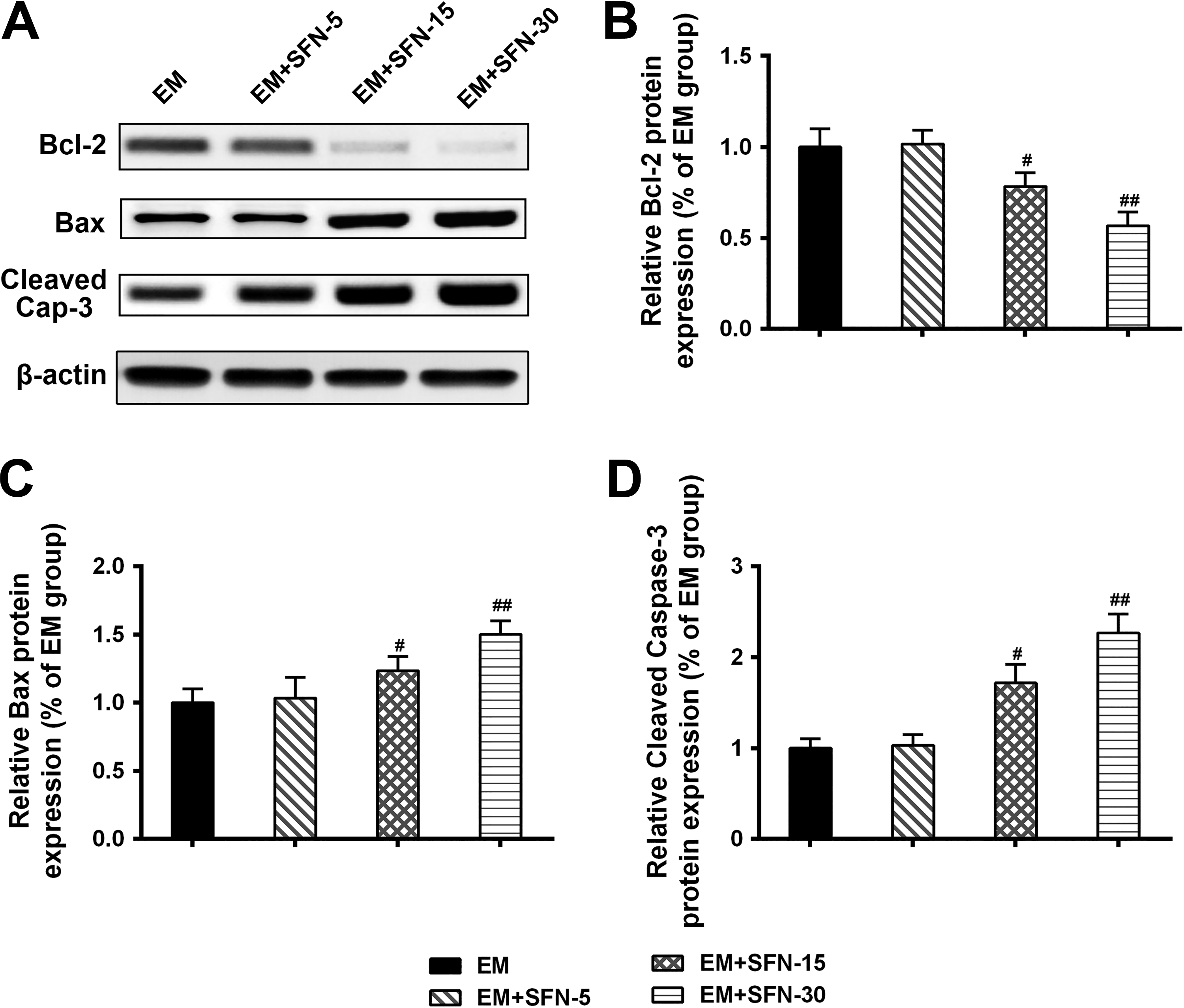
Effect of sulforaphane on the expressions of apoptosis-related genes in the EM models (n = 8). A-D, Expressions of Bcl-2 (B), Bax (C), and cleaved caspase-3 (D) in ectopic endometria were determined by Western blot. Data are presented as mean (SD). # P< .05, ## P < .01 compared to EM group. EM indicates endometriosis.
Effects of Sulforaphane on Expressions of PI3K/Akt Signaling Pathway in the EM Rat Model
We explored the underlying mechanisms of sulforaphane on the development of EM. PI3K/Akt signaling pathway has been reported to play an important role in a series of cellular events including cell proliferation and apoptosis. Activation of PI3K/Akt signaling pathway accelerates expressions of inflammatory cytokines and mediators. Using Western blotting and qPCR, we examined the expressions of PI3K, p-Akt, and Akt in ectopic endometria tissue. We found that protein expressions of PI3K and p-Akt were significantly decreased in sulforaphane (15 mg/kg and 30 mg/kg)-treated groups when compared with those in the EM group (Figure 5A-C, P < .05, P < .01). Interestingly, when we examined the mRNA expressions of PI3K and p-Akt, we observed that only PI3K was significantly increased in sulforaphane (30 mg/kg)-treated group when compared to that in the EM group (Figure 5D and E, P < .05).

Effects of sulforaphane on the expressions of PI3K/Akt signaling pathway in the EM models (n = 8). A-D, Protein expressions of PI3K (B), p-Akt (C), and Akt (D) in ectopic endometria were determined by Western blotting. (E and F) mRNA expressions of PI3K (E) and Akt (F) in ectopic endometria were detected by qPCR. Data are presented as mean (SD). # P < .05, ## P < .01 compared to EM group. EM indicates endometriosis; qPCR, quantitative polymerase chain reaction; SD, standard deviation.
Discussion
Sulforaphane is an isothiocyanate isolated from cruciferous vegetables including cabbage, cauliflower, brussels sprouts, and broccoli. 21 It has garnered significant interest due to its potential therapeutic effects in many types of cancer. 14,15 More importantly, sulforaphane exists anticancer properties by multiple mechanisms including the regulation of inflammation, inhibition of angiogenesis, and induction of apoptosis. 16,14 It is interesting to explore the effects of sulforaphane on other diseases.
Endometriosis has been identified to be associated with pelvic pain and impaired fecundity in adolescents. 1,2 There are 6% to 10% of women with reproductive age, 50% to 60% of women with pelvic pain, and as many as 50% of women with infertility affected by EM. 3 Although the underlying mechanisms which induce EM are still not fully understood, many studies have demonstrated its occurrence and development associated with a disorder of the immune systems. Additionally, increasing population of apoptotic cells, elevation of inflammatory cytokines, and angiogenesis biomarkers, have been identified as the hallmarks in the development of EM. 11,12 It is worthwhile exploring compounds with properties including inhibition of inflammation, induction of apoptosis, and inhibition of angiogenesis. Therefore, in this study, for the first time, we aim to explore the therapeutic effects of sulforaphane for EM.
A rat model of EM was established and volumes of endometriotic foci and adhesion score were evaluated. Blauer scoring system is well-known to grade intraabdominal adhesion and widely used to evaluate in the EM model. 17,18 In this study, endometriotic foci with massive volumes and the highest adhesion score were observed in the EM group, indicating a rat model of EM was successfully established. Next, we evaluated the protective effects of sulforaphane on rat EM. The results demonstrated that treatment of sulforaphane (15 mg/kg and 30 mg/kg) significantly decreased the volumes of endometriotic foci and adhesion score when compared with those in the EM model, suggesting protective effects of sulforaphane on rat EM. We further explored the underlying mechanisms of sulforaphane on the EM.
Inflammation has been reported to play an important role in the development of EM. 9,5 For instance, in patients with EM, levels of pro-inflammatory cytokines such as IL-6 and TNF-α, growth factors, and other inflammatory mediators in the peritoneal fluid and plasma are dramatically increased. 22 -24 In addition, an increase of activated macrophages and natural killer cell is also observed in the patients with EM. 23 In this study, we evaluated the effects of sulforaphane on the expressions of inflammation-related cytokines in rat EM. The results demonstrated that posttreatment of sulforaphane dose-dependently decreased levels of IL-6, IL-10, TNF-α, and IFN-γ in peritoneal fluid and plasma, indicating its protective effects on EM are in part by inhibition of inflammatory cytokines including IL-6, TNF-α, and IFN-γ. Interestingly, we observed that the levels of IL-10 in the EM and sulforaphane post-treated groups were much higher than those in the Sham group. IL-10 is known as an anti-inflammatory cytokine. We infer that there may be a compensatory mechanism which acts to regulate the levels of inflammatory cytokine in rat.
Vascular endothelial growth factor is an important biomarker of angiogenesis and has been identified as a key factor for the regulation of EM by maintaining endometriotic cells growth and survival. 5 Inhibition of VEGF has been identified as an effective method to reduce the volume of ectopic endometrial explants. 25 In the present study, we found that the expressions of VEGF in the peritoneal fluid, plasma, and ectopic endometria were significantly increased in the EM group. Sulforaphane (30 mg/kg and 15 mg/kg) post-treated dose-dependently decreased the expressions of VEGF in the peritoneal fluid, plasma, and ectopic endometria when compared to that in the EM group. These results supported that inhibition of VEGF by sulforaphane suppressed the growth of endometriotic foci. Interestingly, in a previous study, sulforaphane has been reported to inhibit VEGF expression in human colon cancer cells. 26 Our data, along with this study, supported sulforaphane is able to suppress VEGF expression in colon cancer and EM.
Apoptosis is crucial to maintaining tissue homeostasis. Some studies have been reported that abnormal expressions of apoptosis markers include Bcl-2 family and caspase-1, 3 of ectopic endometriotic tissues in EM. 11,10 We then evaluated the effects of sulforaphane on the apoptosis. Interestingly, we observed that a decrease of expressions of Bcl-2 while an increase in expressions of Bax and cleaved caspase-3, in the sulforaphane (30 mg/kg and 15 mg/kg) treated groups when compared to those in the EM group. These results are in part in agreement with the pro-apoptosis effects of sulforaphane on brain malignant glioma, in which sulforaphane regulates mitochondrial and MEK/ERK-mediated apoptosis pathway. 27 Our results demonstrated that effects of sulforaphane on EM are in part by the regulation of apoptosis-related proteins including Bcl-2/Bax and cleaved caspase-3.
To our knowledge, sulforaphane exists chemopreventive and therapeutic effects by multiple mechanisms including induction of apoptosis, inhibition of tumor angiogenesis, inhibition of inflammation, and suppression of oxidative stress. 14,21 Interestingly, we found that sulforaphane attenuated EM in rat models by the regulation of inflammation, induction of apoptosis and inhibition of angiogenesis. These results demonstrated that the regulation of inflammation, apoptosis, and angiogenesis might be a strategy, which can be used to treat EM.
Furthermore, we further explored the potential target of sulforaphane on EM. PI3K/Akt signaling pathway has been identified to play an important role in the regulation of cellular functions including cell growth, proliferation, and apoptosis, and activation of immune cells including T cells and macrophages. 28 In addition, PI3K/Akt signaling pathway also regulates angiogenesis in cancers and normal tissues. 29,28 In this study, for the first time, we found that PI3K and p-Akt were significantly decreased in sulforaphane (15 mg/kg and 30 mg/kg) treated groups. These results suggested that sulforaphane ameliorated EM in rat models in part by inhibition of PI3K/Akt signaling pathway. However, the effects of sulforaphane on other signaling pathways associated with the development of EM should be investigated in further studies.
Conclusion
The present study, for the first time, identified that sulforaphane ameliorated EM in rat models by multiple effects. First, treatment of sulforaphane decreased levels of inflammatory cytokines including TNF-α and IL-6 in peritoneal fluid and plasma. Second, treatment of sulforaphane decreased the expressions of VEGF in the peritoneal fluid, plasma, and ectopic endometria. Third, treatment of sulforaphane regulated the expressions of apoptosis-related biomarkers including Bcl-2, Bax, and cleaved Caspase-3. Moreover, we revealed that sulforaphane-ameliorated EM in rat models is by inhibition of PI3K/Akt signaling pathway.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
