Abstract
Background and Aim
This study investigated the dose-dependent cardioprotective effects of Pterocarpus indicus extract (P.indicus; LD: 27, MD: 54, HD: 108 mg/kg) against isoproterenol (ISO)-induced myocardial ischemia in rats, focusing on oxidative stress, apoptosis, inflammation, and fibrosis.
Experimental procedure
Rats were pretreated for 14 days and divided into control, ISO, propranolol, and P.indicus (LD, MD, HD) groups. Myocardial injury was assessed via ECG, histopathology, serum biomarkers (CK-MB, LDH, cTnT, IL-6, IL-1β, TNF-α), oxidative markers (SOD, MDA, GSH-Px), and apoptosis-related proteins (Bax, Bcl-2, Caspase-3). Nrf2/HO-1 expression was analyzed by western blot. Phytochemical profiling was performed via UPLC-MS/MS and HPLC. Pharmacokinetics, safety, and therapeutic-regimen efficacy were also evaluated.
Results
P.indicus at MD and HD significantly attenuated ISO-induced ECG abnormalities, histopathological damage, serum injury markers, inflammatory cytokines, and fibrosis (all P < 0.01). Treatment suppressed oxidative stress (restored SOD/GSH-Px, reduced MDA) via Nrf2/HO-1 activation and inhibited apoptosis via Bax/Bcl-2/Caspase-3 modulation. The efficacy of MD (EC50 ∼50 mg/kg) was confirmed by dose-response analysis. Pharmacological inhibition established that both Nrf2 activation and caspase-3 inhibition are necessary for P.indicus’s effects. PK studies showed systemic exposure to key compounds (quercetin-3-O-glucoside, ferulic acid, linoleic acid), and a 14-day safety assessment revealed no hepatorenal toxicity. Post-injury therapeutic administration also conferred significant protection.
Conclusion
P. indicus extract confers dose-dependent cardioprotection against ISO-induced myocardial injury through dual modulation of the Nrf2/HO-1 and Bax/Bcl-2/Caspase-3 pathways, with the medium dose (54 mg/kg) identified as optimally effective and well-tolerated.

Keywords
Introduction
According to the WHF (2024), cardiovascular diseases (CVDs) have remained the leading cause of mortality, accounting for over 33% of global deaths, with ischemic heart disease responsible for 9.1 million deaths in 2019 alone. 1
Current primary treatments for myocardial ischemia include thrombolysis, coronary stent implantation, and coronary artery bypass grafting.2,3 However, while these interventions restore perfusion, they also induce significant reactive oxygen species (ROS) generation, exacerbating myocardial damage and increasing mortality risk.4,5 Thus, identifying novel diagnostic targets and intervention strategies is imperative.
A key pathophysiological mechanism is oxidative stress, which drives myocardial injury through multiple interconnected processes. 6 Excessive ROS during ischemia activates inflammasomes, promoting inflammatory cytokine release and recruitment of immune cells, thereby amplifying post-ischemic inflammation and contributing to cardiomyocyte death. Moreover, ischemia-hypoxia increases mitochondrial membrane permeability, facilitating the release of apoptogenic factors; in combination with ROS, this accelerates apoptosis.7-9 Therefore, targeting oxidative stress and its downstream effects represents a promising therapeutic approach. 10 Central regulators of these processes include the Nrf2 pathway, which governs antioxidant responses, NF-κB-mediated inflammation, and the Bcl-2/Bax axis regulating mitochondrial apoptosis.11,12 Simultaneous modulation of these pathways may yield synergistic benefits in myocardial ischemia.
Chinese herbal medicines are characterized by their synergistic multi-component and multi-target effects and have demonstrated significant efficacy in the prevention and treatment of ischemic heart disease.13,14 Pterocarpus indicus Willd (P. indicus), a traditional remedy with documented cardioprotective potential, contains bioactive phytochemicals such as flavonoids, lignans, and phenolic acids, which exhibit antioxidant, anti-inflammatory, and vasodilatory properties.15,16 Preclinical studies in non-cardiac models have shown that P. indicus extracts reduce oxidative stress by scavenging free radicals and modulating inflammatory pathways such as NF-κB and NLRP3.17,18 In vitro experiments further suggest that its flavonoid-rich fractions inhibit platelet aggregation and improve endothelial function, 19 aligning with its traditional use for symptoms resembling cardiovascular inflammation (“heart-heat syndrome” characterized by pain and heat sensations in TCM).
While conventional therapies (such as beta-blockers, statins, ACE inhibitors) effectively manage risk factors like hypertension and dyslipidemia, they often inadequately address reperfusion injury and persistent oxidative stress following revascularization.20,21 Traditional applications of P. indicus in “nourishing the heart” (cardiotonic effects) and “clearing heat” (anti-inflammatory actions) may complement these approaches by targeting residual pathophysiology, including oxidative stress, inflammation, and apoptosis.22,23 For instance, lignans from P. indicus have attenuated neuronal apoptosis via Wnt/β-catenin pathway in cerebral ischemia models, 24 suggesting analogous cardioprotective mechanisms.
Based on this evidence, we hypothesized that P. indicus extract provides dose-dependent cardioprotection in isoproterenol (ISO)-induced myocardial ischemia by alleviating oxidative stress, inflammation, and apoptosis. This study aimed to validate this hypothesis through integrated physiological, biochemical, and molecular assessments.
Materials and Methods
Drug Extraction
The heartwood of P. indicus was purchased from the Tibetan medicinal materials market in Lhasa, Xizang., China. The botanical identity of the material was authenticated by Professor Sun Fangyun from Xizang Minzu University (Xianyang, China). The raw material (2.0 kg) was weighed and recorded prior to extraction. The P. indicus extract was prepared as follows: the heartwood of P. indicus was crushed using an oscillating Chinese herbal medicine grinding machine (DFY-200, Dade Pharmaceutical Machinery, Taizhou, China) and the contents were then soaked in 75% ethanol for 24 h. A heated reflux extraction was performed twice to obtain an extract containing the ethanol-soluble components of P. indicus, with a material-to-liquid ratio of 1:10 (g/mL). The filtrates were then combined and concentrated under reduced pressure using a circulating water vacuum pump (SHZ-III, Shanghai Xiande Experimental Instrument Co., Ltd., Shanghai, China) until the ethanol odor was absent. The concentrate was suspended in 500 mL of water prior to fractionation and equal amounts of ethyl acetate and collected supernatant was combined. The ethyl acetate fraction obtained from the extraction was further concentrated under reduced pressure and freeze-dried into powder for storage. The freeze-dried powder (182.3 g) was weighed to calculate extraction yield (9.12% ± 0.34% w/w). The obtained extract was formulated into a solution containing 1 g of crude drug per mL and stored at 4°C for future use.
Phytochemical characterization of the extract was performed as follows:
Total polyphenolic content was determined via the Folin-Ciocalteu method: 100 µL of extract solution was mixed with 4.9 mL distilled water and 0.5 mL Folin-Ciocalteu reagent. After 3 min, 1.5 mL of 20% Na2CO3 was added, and the mixture was incubated at 40°C for 20 min. Absorbance was measured at 765 nm using a UV-visible spectrophotometer, with gallic acid as the standard. The total polyphenolic content was 32.6 ± 1.8 mg gallic acid equivalents per gram of extract (mg GAE/g).
Total flavonoid content was quantified using the aluminum chloride colorimetric assay: 1 mL of extract solution was mixed with 10% AlCl3 and 1 M CH3COOK, incubated at room temperature for 30 min, and absorbance was measured at 415 nm with rutin as the standard. The total flavonoid content was 18.3 ± 1.2 mg rutin equivalents per gram of extract (mg RE/g).
Reagents
Isoprenaline hydrochloride (abbreviated to ISO hereafter, batch number hy-b0468) was purchased from the MedChemExpress Company (Shanghai, China) and propranolol (batch number p913470) was purchased from Macklin Reagent Company (Shanghai, China). ELISA kits were purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd. (Shanghai, China): lactate dehydrogenase (LDH, 2408007), superoxide dismutase (SOD, 2407008), malondialdehyde (MDA, 2408005), and glutathione peroxidase (GSH-Px, 2408006) ELISA kits were purchased from Beijing Solarbio Science & Technology Co., Ltd (Beijing, China). Creatine kinase (CK-MB, 202411), cardiac troponin T (cTnT, 202211), tumor necrosis factor-alpha (TNF-α, 202406), interleukin-6 (IL-6, 202406), and interleukin-1β (IL-1β, 202406) ELISA kits were purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd. (Shanghai, China). Bcl-2 Associated X protein (Bax, 50599-2-1 g) and B-cell lymphoma-2 (Bcl-2, 60178-1-1 g) antibodies were purchased from Wuhan Sanying Biotechnology Co., Ltd. (Wuhan, China) and Caspase-3 (Cleaved caspase-3, ab85309), heme oxygenase 1 (HO-1, ab13248), HRP goat anti-mouse IgG (H+L, ab6789) antibodies were purchased from Abcam Trading (Shanghai) Co., Ltd. (Shanghai, China). Nuclear factor erythroid 2-related factor 2 (Nrf2, A1244) antibody was purchased from ABclonal Biotechnology Co., Ltd. (Wuhan, China). HRP goat anti-rabbit IgG (H+L, S0001) antibody was purchased from Affinity Biosciences Co., Ltd. (Shanghai, China). A BCA assay kit (cat. no. P0009) was purchased from Shanghai Beyotime Biotechnology Co., Ltd (Shanghai, China). β-actin (Cat. No. GB15001-100, Servicebio, China) and β-tubulin (Cat. No. ZB15140-HRP-100, Servicebio, China) antibodies were diluted 1:5000 and incubated overnight at 4°C. Quercetin-3-O-glucoside (≥98%, Sigma-Aldrich, USA), Ferulic Acid HPLC Assay Kit and linoleic acid (≥99%, Solarbio, China)) were used as reference standards. Methanol (HPLC-grade) and formic acid (LC-MS-grade) were purchased from Merck (Germany).
Animal Model
Animals
Adult male Sprague-Dawley (SD) rats (SPF grade; weight: 200 ± 20 g) were obtained from Chengdu Dashuo Experimental Animal Co., Ltd. (Sichuan, China; license No. SCXK 2020-030). Rats were housed in individually ventilated cages under standard SPF conditions with environmental enrichment (nesting material). Following a 1-week acclimation period with ad libitum access to food and water, animals underwent electrocardiogram (ECG) screening. Only rats meeting the following criteria were included: normal ECG waveform, body weight of 180 – 220 g, and absence of visible health abnormalities (e.g., alopecia, lethargy). Exclusion criteria included abnormal ECG findings, body weight outside the specified range, or clinical signs of illness. All procedures were approved by the Institutional Animal Ethics Committee of Xizang Minzu University (Approval No. 2024088) and conducted between March and May 2024. Humane endpoints (>20% body weight loss or severe immobility) were predefined, though no animals met these criteria.
Experimental Design
Sixty eligible rats were randomized via computer-generated sequence to 6 groups (n = 10):
Control (saline oral + s.c.)
ISO model (saline oral + ISO 85 mg/kg s.c.) 25
Propranolol (30 mg/kg oral + ISO s.c.)
P. indicus LD (27 mg/kg), MD (54 mg/kg), HD (108 mg/kg) oral + ISO s.c.
All groups received daily oral gavage for 14 days. On days 7-14, ISO groups received daily ISO injections (85 mg/kg s.c.) 30 min post-gavage.
To evaluate the therapeutic potential of P. indicus, a seventh group was included (n = 8):
P. indicus therapeutic (P.indicus-THE): 54 mg/kg (oral) + ISO (s.c.)
Therapeutic regimen: P. indicus treatment was initiated concurrently with and continued after ISO induction (days 1-14 oral, days 1-7 ISO), simulating clinical intervention upon disease onset.
Sample size was determined via a priori power analysis (G*Power v3.1) using a moderate effect size (Cohen’s f = 0.35), α = 0.05, and β = 0.20, confirming n = 10/group provided >80% power to detect significant differences in primary biochemical and histological endpoints. The therapeutic group size (n = 8) was set based on the expected large effect size in key endpoints from the established efficacy of the MD dose. The doses of P. indicus extract (27, 54, and 108 mg/kg) were selected through an integrated strategy. This was initially guided by the human equivalent dose (1:6.15 conversion; Chinese Materia Medica 26 ) and subsequently refined by an internal pilot study (n = 5/group), which identified 50 mg/kg as the approximate EC50 for reducing ISO-induced CK-MB elevation—a finding consistent with the final dose-response analysis (As shown in the Dose-Response Analysis section). The low (LD), medium (MD), and high (HD) doses were set at 0.5x, 1x, and 2x the pilot EC50. To minimize bias: 1) Group allocation was performed by an independent researcher not involved in experiments; 2) All treatments were prepared by a separate technician to be visually indistinguishable and administered using blinded protocols; 3) Endpoint assessors (ECG/histology/biochemistry) were fully blinded to group allocation through coded samples until analysis completion. Daily evaluations included survival, body weight, fur condition, and activity levels.
ECG Monitoring
Twenty-four hours after the last ISO injection, the rats in each group were anesthetized by intraperitoneal injection with 2% pentobarbital sodium (30 mg/kg) and fixed in a supine position on a mouse board. The BL-420A biological function experimental system (Chengdu Taimeng Software Co., Ltd, Chengdu, China) was used to record the ECG of lead II and ST segment changes observed within 60 min. Successful myocardial ischemic modeling was indicated by a T-wave inversion or abnormal increase, and ST-segment elevation or reduction relative to the control group. Additionally, QRS complex duration and the incidence of ventricular premature beats (VPBs) over the recording period were analyzed.
Biochemical Indices Detection in Rat Serum and Myocardial Tissues
After ECG recording, a laparotomy was performed and 3 mL of blood was collected from the abdominal aorta. The blood was centrifuged at 3500 rpm for 10 min to separate the serum, aliquoted and stored at −80°C for future use. Serum CK-MB, LDH, cTn-T, and inflammatory cytokine (IL-6, IL-1β, and TNF-α) levels were detected by ELISA according to the manufacturer’s protocols and quantified using a full-wavelength microplate reader (FSP120-AAAN2, Thermo Fisher Scientific, China). After blood collection, rats were euthanized by overdose intraperitoneal injection of 2% sodium pentobarbital (150 mg/kg) (in compliance with the AVMA Guidelines for the Euthanasia of Animals), ensuring painless death before rapid thoracotomy to isolate cardiac tissues.
To obtain myocardial tissue, rats were thoracotomized and the hearts were excised. The tissue was cut into small pieces using ophthalmic scissors and homogenized (KZ-III-F; Servicebio, Wuhan, China) in ice-cold water. After centrifugation at 3500 rpm for 15 min, the supernatant was collected, and myocardial SOD, GSH-Px, and MDA levels were determined via ELISA according to the manufacturer’s protocols.
To evaluate P. indicus extract-induced systemic toxicity, serum liver function markers (ALT, AST) and kidney function markers (BUN, Cr) were also measured via commercial kits (Nanjing Jiancheng Bioengineering Institute) following manufacturers’ protocols; detection was performed using the same microplate reader to ensure consistency.
Cardiac Index and Histopathological Examination of Rat Myocardial Tissue
To determine the cardiac index, rats were weighed, anesthetized, and the hearts were excised. The hearts were rinsed in pre-cooled physiological saline to remove any blood, blotted dry with filter paper, and weighed. The cardiac index was calculated as the ratio of heart weight to body weight (cardiac index = heart weight/body weight, expressed as mg/g), which reflects myocardial hypertrophy or edema associated with ischemic injury (elevated values indicate pathological remodeling). For liver and kidney index determination: Livers and kidneys were dissected immediately after heart excision, rinsed with ice-cold saline, blotted dry, and weighed using an analytical balance (0.1 mg precision, Mettler Toledo). Liver index (mg/g) = liver weight (mg)/body weight (g); kidney index (mg/g) = total kidney weight (mg)/body weight (g).
For histopathological examination, rat hearts were fixed in 4% paraformaldehyde solution for 24 h, embedded in paraffin, and sectioned at 4 μm using a microtome (Leica, Germany). Sections were then stained with hematoxylin and eosin (H&E), with myocardial damage observed under a digital light microscope (BA210Digital, Motic Group Co., Xiamen, China). Myocardial damage was scored semi-quantitatively on a scale of 0 to 4 points, 27 based on the assessment of 3 key pathological features: the degree of myofiber disarray, the extent of inflammatory cell infiltration, and the area of necrosis. The scoring criteria were as follows:0 points: No pathological changes observed. 1 point: Pathological changes involving less than 25% of the observed myocardium. 2 points: Pathological changes involving 25% to 50% of the observed myocardium. 3 points: Pathological changes involving 50% to 75% of the observed myocardium. 4 points: Pathological changes involving more than 75% of the observed myocardium. The total score was the sum of individual criteria (range: 0-12), normalized to a 0-4 scale (total score/3) for simplicity. Histopathological scoring was performed independently by 2 blinded pathologists. Inter-observer reliability was confirmed with excellent agreement (intraclass correlation coefficient = 0.89). Final scores represent consensus evaluations.
Masson’s Trichrome Staining for Myocardial Fibrosis and Infarct Assessment
To evaluate myocardial fibrosis and collagen deposition—key histological hallmarks of ischemic injury and remodeling—heart sections were subjected to Masson’s trichrome staining. Paraffin-embedded heart sections (4 μm) were deparaffinized, rehydrated through a graded alcohol series, and stained using a Masson’s Trichrome Stain Kit according to the manufacturer’s instructions. Briefly, sections were stained in Weigert’s iron hematoxylin working solution to visualize nuclei, followed by staining in Biebrich scarlet-acid fuchsin solution to differentiate muscle fibers and cytoplasm. Subsequent treatment with phosphomolybdic-phosphotungstic acid solution allowed for differential binding of aniline blue, which specifically stains collagen fibers. After dehydration and clearing, sections were mounted with neutral balsam. Stained sections were imaged under a digital light microscope. The fibrotic (blue-stained) area was quantified as a percentage of the total left ventricular area using ImageJ software by an observer blinded to the group assignments.
Detection of Bcl-2, Bax, and Casp-3 mRNA Expression via Quantitative Real-Time PCR (qPCR)
Rat myocardial tissue was transferred to RNase-free EP tubes containing 1 ml of TRIzol (BM1144; Hefei Bomei Biotechnology Co. Ltd, Hefei, China). Samples were then homogenized, total RNA was extracted, and the obtained RNA was quantified and qualified using a NanoDrop microvolume spectrophotometer (NanoDrop One, Thermo Fisher Scientific, Shanghai, China). Primers were designed and screened using Primer Premier software and were based on full-length gene sequences obtained from the NCBI database. All primers were synthesized by Shanghai Sangon Biotechnology Co., Ltd. (Shanghai, China), and purified using ULTRAPAGE. The primer sequences (5′→3′) were as follows:
β-actin (internal control): F = 5’-GGGAATCGTCGTGACATT-3’, R = 5’-GCGGCAGTGGCCATCTC-3’. Bax: F = 5’-AGACACCTGAGCTGACCTTGGAG-3’, R = 5’-TTCATCGCCAATTCGCCTGAGAC-3’. Bcl-2: F = 5’-GGGCTACGAGTGGGATACTGGAG-3’, R = 5’-CTCAGGCTGGAAGGAGAAGATGC-3’. Caspase-3: F = 5’-CTGGCACACGGGACTTGGAAAG-3’, R = 5’-GCGATGACTCAGCACCTCCATG-3’. Samples were then evaluated on a CFX Connect real-time fluorescent qPCR instrument (BioRad, Shanghai, China) under the following conditions: initial denaturation at 95°C for 10 min, followed by 45 cycles, each consisting of denaturation at 95°C for 5 s, annealing at 55°C for 30 s, and extension at 72°C for 30 s. β-actin was utilized as an internal reference gene and relative gene expression levels were calculated using the 2−ΔΔCT method.
Western Blot Analysis of Bax/Bcl-2/Casp-3 and Nrf2/HO-1 Myocardial Protein Expression
Myocardial tissue was collected from each group and lysed with RIPA lysis buffer (G2002-100 ML; Servicebio, Wuhan, China) to extract total proteins, with total protein concentrations determined using a BCA kit per the manufacturer’s protocols. Proteins (30 µg/lane) were separated by SDS-PAGE using 12% gels for Bax (21 kDa), Bcl-2 (26 kDa), and cleaved caspase-3 (17 kDa), and 15% gels for Nrf2 (68 kDa) and HO-1 (32 kDa). Tris-Glycine buffer (BL2352 A, Biosharp, Hefei, China) was used for all gel runs to ensure optimal resolution of proteins across the molecular weight range. The PVDF membrane was blocked in 5% skim milk diluted in TBST buffer (BL346 A, Biosharp, Hefei, China) and incubated on a shaker for 2 h. After blocking, membranes were incubated with Bax (1:5000), Bcl-2 (1:2000), cleaved caspase-3 (1:1000), Nrf2 (1:2000) or HO-1 (1:1000) primary antibodies at 4°C overnight. Samples were then washed with TBST and secondary antibodies (1:5000) were added and incubated at 37°C for 2 h. Following 3 washes, samples were incubated with ECL luminescent reagents (G2161-200 ML, Servicebio, Wuhan, China), visualized using a chemiluminescence detection system (Tanon 5200 Multi, China), and analyzed using Image J software, with β-tubulin used as an internal control.
Analysis of P. indicus Components
For liquid chromatography–mass spectrometry (LC-MS), an Orbitrap Q-Exactive mass spectrometer was coupled with an UltiMate 3000 RS UPLC system (Thermo Fisher Scientific). P. indicus extract (100 µL) was combined with 300 µL of methanol, vortex-mixed for 10 min, centrifuged at 13000 rpm for 10 min, and the supernatant was collected for analysis. Chromatographic separation was performed using an AQ-C18 column (2.1 × 150 mm, 1.8 µm, Welch) at a flow rate of 0.30 mL/min and a temperature of 35°C, with a 0.1% formic acid in water aqueous phase and a methanol organic phase. Separation was carried out based on a chromatographic gradient. From 1 to 30 min, the proportion of the aqueous phase changes as 98% - 80% - 50% - 20% - 5% - 98% (corresponding to 1, 5, 10, 15, 20 and 28 min), and the proportion of the organic phase is correspondingly 2% - 20% - 50% - 80% - 95% - 2%.
Mass Spectrometry was performed under electrospray ionization (ESI) in positive and negative ionization modes with a range of 100-150 m/z and spray voltages of 3.2 kV for each. Full-MS/dd-MS2 was employed with resolutions of 70000 and 17500, respectively. Data acquisition was completed for 30 min under the following conditions: capillary temperature of 300°C, high-purity argon collision gas, and high-purity nitrogen sheath and auxiliary gas. All raw MS data was analyzed using CD3.3 (Compound Discoverer 3.3, Thermo Fisher) and searched against the mzCloud database.
HPLC Quantitative Analysis of Major Bioactive Compounds
(1) Chromatographic conditions:
Quantitative analysis was performed using an Agilent 1260 Infinity II HPLC system (Agilent Technologies, USA) equipped with a diode array detector. The chromatographic conditions were aligned with the LC-MS profiling method to ensure consistency: Column: AQ-C18 column (4.6 × 150 mm, 5 µm, Welch); Column temperature: 35°C; Flow rate: 1.0 mL/min; Mobile phase: A (0.1% formic acid in water) and B (methanol). The gradient program was set as follows: 0-1 min, 2% B; 1-5 min, 2-20% B; 5-10 min, 20-50% B; 10-15 min, 50-80% B; 15-20 min, 80-95% B; 20-28 min, 95% B; 28-30 min, 95-2% B; followed by 5 min re-equilibration at 2% B. The detection wavelengths were optimized for each analyte: 0-12 min at 320 nm (for ferulic acid); 12-18 min at 370 nm (for quercetin-3-O-glucoside); 18-30 min at 205 nm (for linoleic acid). The injection volume was 10 μL. (2) Standard preparation:
Authentic reference standards of quercetin-3-O-glucoside (≥90% purity), ferulic acid (≥99% purity), and linoleic acid (≥99% purity) were used. Primary stock solutions (1 mg/mL) were prepared by dissolving accurately weighed standards in methanol. Working standard solutions for calibration curves were prepared by serial dilution to 7 concentration levels: quercetin-3-O-glucoside (1-100 µg/mL), ferulic acid (0.5-50 µg/mL), and linoleic acid (5-200 µg/mL). (3) Sample preparation:
The P. indicus extract was accurately weighed (10.0 mg) into a 10 mL volumetric flask, dissolved in methanol via 15 min ultrasonic extraction, cooled to room temperature, made up to volume with methanol, and mixed thoroughly. An aliquot of this solution was centrifuged at 12000 rpm for 10 min, and the supernatant was filtered through a 0.22 µm PTFE membrane prior to HPLC analysis. (4) Method validation:
The method was validated according to ICH guideline Q2(R1) for the following parameters:
Linearity: Calibration curves were constructed by plotting peak area against concentration (7 points each). Correlation coefficients (R2) were calculated.
Precision: Intra-day (n = 6 replicates in one day) and inter-day precision (n = 3 days, duplicates each day) were evaluated and expressed as the relative standard deviation (RSD) of retention time and peak area.
Accuracy: Determined by standard addition recovery tests at 3 levels (80%, 100%, 120% of the estimated content in the sample), each in triplicate. Results are expressed as mean recovery (%) and RSD.
Limits of Detection (LOD) and Quantification (LOQ): Determined by serial dilution of standards to signal-to-noise ratios (S/N) of 3 and 10, respectively.
System suitability: Parameters including resolution (Rs ≥ 1.5), theoretical plate count (N ≥5000), and tailing factor (T ≤1.2) were evaluated for each analyte to ensure optimal system performance prior to sample analysis. (5) Quantification:
The concentrations of the target compounds in the sample solutions were calculated using the respective external standard calibration curves. The final content in the dry extract was expressed as mg per gram (mg/g). All samples were analyzed in triplicate, and results are presented as mean ± standard deviation.
Pharmacokinetic Study
A preliminary pharmacokinetic study was conducted in healthy Sprague-Dawley rats (n = 6). After a single oral administration of P. indicus extract at the medium dose (54 mg/kg), blood samples (0.3 mL) were collected from the tail vein at pre-dose (0 h), 0.25, 0.5, 1, 2, 4, 8, 12, and 24 h post-dosing. Plasma was separated by centrifugation and stored at −80°C until analysis. The concentrations of quercetin-3-O-glucoside and ferulic acid in plasma were determined using the aforementioned validated HPLC-DAD method. Pharmacokinetic parameters, including the maximum plasma concentration (Cmax), time to reach Cmax (Tmax), area under the plasma concentration-time curve from 0 to 24 h (AUC0-24h), and elimination half-life (t1/2), were calculated using non-compartmental analysis (Phoenix WinNonlin, Version 8.3).
In Vivo Mechanistic Validation Using Pathway Inhibitors
To establish the causal role of the Nrf2/HO-1 and Casp-3 pathways in P. indicus extract’s cardioprotection, specific pharmacological inhibitors were administered in vivo. Forty rats were randomized into 5 groups (n = 8):
MD: Received P. indicus extract (54 mg/kg, p.o., days 1-14) and ISO (85 mg/kg, s.c., days 7-14).
MD + ML385: Received P. indicus extract and ISO, with the Nrf2 inhibitor ML385 (30 mg/kg, i.p., 30 min before each P. indicus extract gavage).
MD + ZF: Received P. indicus extract and ISO, with the Casp-3 inhibitor Z-DEVD-FMK (1 mg/kg, i.p., 30 min before each P. indicus extract gavage).
ISO + Vehicle: Received inhibitor vehicles and ISO.
Inhibitor efficacy was confirmed by western blot analysis of myocardial HO-1 and Cleaved casp-3. Cardiac injury (serum cTn-T), oxidative stress (myocardial SOD, MDA) were assessed. A significant reversal of P. indicus extract’s protective effects in inhibitor co-treatment groups would demonstrate pathway necessity.
Statistical Analysis
All data were processed and analyzed using GraphPad Prism 9.0 (GraphPad Software, USA), with results expressed as mean ± standard deviation (SD). Prior to statistical comparisons, data normality and variance homogeneity were verified via Shapiro-Wilk and Levene’s tests, respectively. One-way analysis of variance (ANOVA) was performed for group comparisons, followed by Tukey’s honestly significant difference (HSD) test for pairwise post-hoc analysis. Statistical significance was defined at P < 0.05.
Results
P. indicus Ameliorates ISO-Induced Cardiac Dysfunction and Structural Damage
Effect of P. indicus in ISO-Induced Myocardial Ischemic Model Examined via ECG
Continuous ECG was monitored for 60 min. The control group showed no significant abnormalities (ST-segment elevation: 0.005 ± 0.002 mV). In contrast, the ISO group exhibited a marked ST-segment elevation (0.386 ± 0.025 mV, P < 0.01 vs. control). Pretreatment with propranolol (PRO group) significantly attenuated this elevation (0.024 ± 0.033 mV, P < 0.01 vs. ISO). Both medium-dose (MD: 0.285 ± 0.089 mV) and high-dose (HD: 0.036 ± 0.015 mV) P. indicus also significantly reduced ST-segment elevation compared to the ISO group (P < 0.01 for both), whereas the low-dose (P. indicus-LD: 0.329 ± 0.067 mV) showed no significant difference (P > 0.05; Figure 1). No other notable ECG abnormalities were observed. Effects of P. indicus extract on ECG indicators in ISO-induced myocardial ischemic rats. (A) ST segment elevation (mV). (B) Width of the QRS complex (s). (C) Ventricular premature beat (VPB) count (times/60 min). (D) Representative ECG tracings for each group. Data in panels (A-C) are presented as mean ± SD. **P < 0.01 vs. control group; ##P < 0.01 vs. ISO group. Abbreviations: LD, low-dose; MD, medium-dose; HD, high-dose.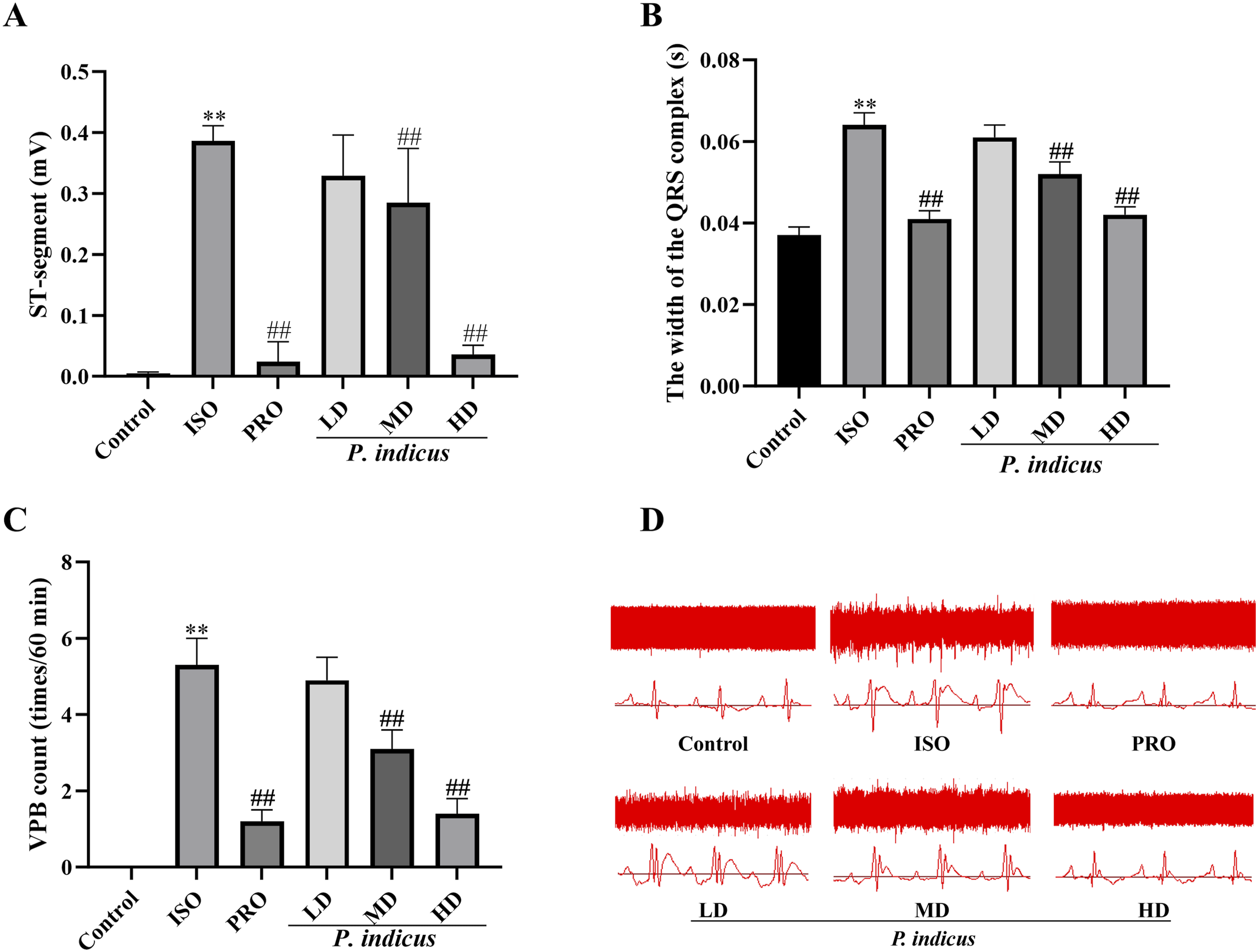
QRS duration, an indicator of ventricular conduction, was normal in the control group (0.037 ± 0.002 s). ISO administration induced significant QRS widening (0.064 ± 0.003 s, P < 0.01 vs. control). Both PRO and P. indicus-HD normalized QRS duration (0.041 ± 0.002 s and 0.042 ± 0.002 s, respectively; P < 0.01 vs. ISO). The MD group showed a significant but intermediate reduction in QRS duration (0.052 ± 0.003 s, P < 0.01 vs. ISO), while the LD group had no significant effect (0.061 ± 0.003 s, P > 0.05 vs. ISO).
No ventricular premature beats (VPBs) were detected in the control group (0 ± 0 beats/60 min). ISO-induced rats exhibited frequent VPBs (5.3 ± 0.7 beats/60 min, P < 0.01 vs control). Propranolol pretreatment significantly suppressed VPBs (1.2 ± 0.3 beats/60 min, P < 0.01 vs. ISO). A similar suppressive effect was observed in the P. indicus-HD group (1.4 ± 0.4 beats/60 min, P < 0.01 vs. ISO). The MD group showed a moderate reduction in VPBs (3.1 ± 0.5 beats/60 min, P < 0.01 vs. ISO), whereas the LD group had no significant effect (4.9 ± 0.6 beats/60 min, P > 0.05 vs. ISO).
Histopathological Changes in Myocardial Tissue (H&E Staining and Cardiac Index)
In the control group, the myocardial architecture was well-preserved, with myofibers exhibiting regular alignment, intact cross-striations, and centralized nuclei. No inflammatory infiltration or necrosis was observed (Figure 2). Compared to the control group, the ISO group displayed severe myocardial damage, including myofibers with varying degrees of necrosis, increased fibrous tissue formation, and significant inflammatory cell infiltration. A pathological score of 3.41 ± 0.23 was assigned (P < 0.01 vs. control). Pretreatment with propranolol and P. indicus extract at medium (MD) and high doses (HD) significantly alleviated these pathological changes, showing reduced necrosis, diminished inflammatory cell infiltration, and restored myofiber alignment. The pathological scores were significantly lower (all P < 0.01 vs. ISO group). The low-dose (LD) group showed no significant improvement. P. indicus Treatment Mitigates Histopathological Damage in Myocardial Tissue. (A) Representative H&E-stained sections. To clearly display the pathological changes, images include both ×200 overview and ×400 high-magnification fields (insets) of the key observed areas. Specific features are indicated: myocardial fiber rupture (black arrow), lymphocyte infiltration (blue arrow), widened intercellular spacing (orange arrow), fibroblasts (green arrow), and myocardial cell atrophy with reduced volume (yellow arrow; clearly visible in ×400 magnification as shrunken cell size and diminished cytoplasm). (B) Representative images of heart measurements (gross heart size/morphology) for each group, reflecting morphological changes. (C) Pathological scores (evaluated based on the severity of myocardial fiber rupture, inflammatory infiltration, and structural disorganization) showing that P. indicus treatment significantly reduced scores compared to the ISO group (##P < 0.01), indicating alleviated tissue damage; scores in all treatment groups remained higher than the Control group (**P < 0.01). (D) Cardiac index (Heart weight/Body weight ratio) demonstrating that P. indicus treatment reversed the ISO-induced abnormal index, with high-dose (HD) showing the most significant recovery (##P < 0.01 vs. ISO group), consistent with mitigated myocardial injury. Values are presented as mean ± SD. Low-Dose (LD), Medium-Dose (MD), High-Dose (HD).
Consistent with the histopathological damage, the cardiac index (heart weight/body weight ratio) was significantly elevated in the ISO group relative to the control group (P < 0.01), indicating myocardial hypertrophy or edema. This increase was significantly attenuated in the PRO, MD, and HD groups (all P < 0.01 vs. ISO), but not in the LD group (P > 0.05).
Assessment of Myocardial Fibrosis via Masson’s Trichrome Staining
Myocardial collagen deposition, indicative of fibrosis and ischemic scarring, was assessed separately using Masson’s trichrome staining (Figure 3). Myocardial tissue from the control group exhibited minimal blue staining, indicating normal collagen distribution and the absence of significant fibrosis (fibrotic area: 0.05 ± 0.03%). In stark contrast, hearts from the ISO model group displayed extensive blue-stained collagen deposition, particularly in the perivascular and interstitial spaces, confirming severe ischemia-induced fibrosis (fibrotic area: 34.6 ± 2.5%, P < 0.01 vs. control). Effects of P. indicus on Myocardial Fibrosis in ISO-Induced Myocardial Ischemia Rats as Assessed by Masson’s Trichrome Staining (200x). Figure depicts masson’s trichrome staining results and quantitative analysis of myocardial fibrosis across different experimental groups.(A): Representative masson’s trichrome - stained myocardial sections (Left) and corresponding pseudo - colored images (Right, with red indicating collagen - positive areas for enhanced visualization). Groups include: Control (Normal control), ISO (Isoproterenol - induced myocardial ischemia model), PRO (Propranolol - treated positive control), and P. indicus at Low Dose (LD), Medium Dose (MD), and High Dose (HD). In masson’s staining, blue staining represents collagen fibers, while red staining represents myocardial fibers.(B): Quantitative analysis of the masson - positive area ratio (Percentage of collagen - positive area relative to the total myocardial area). Data are presented as mean ± SD. **P < 0.01 vs. Control group; ##P < 0.01 vs. ISO group.
Treatment with propranolol and P. indicus extract at medium (MD) and high doses (HD) significantly attenuated this pathological collagen deposition. The fibrotic area was reduced to 11.2 ± 2.1% (PRO, P < 0.01), 19.5 ± 2.8% (MD, P < 0.01), and 12.8 ± 2.4% (HD, P < 0.01) compared to the ISO group. The low-dose (LD) group showed a trend towards reduction, but the change was not statistically significant (28.95 ± 2.1%, P > 0.05). These findings demonstrate that P. indicus extract effectively mitigates ISO-induced myocardial fibrosis in a dose-dependent manner.
P. indicus Attenuates Myocardial Injury and Systemic Inflammation
ISO administration induced significant cardiac injury, markedly elevating serum levels of CK-MB (+51.5%), LDH (+95.4%), and cTn-T (+130.2%) compared to the control group (all P < 0.01, Figure 4). These increases were substantially attenuated by high- and medium-dose P. indicus treatment, which reduced CK-MB by 22.8-31.5%, LDH by 37.3-52.0%, and cTn-T by 31.3-50.0% vs the ISO group (all P < 0.01), showing efficacy comparable to propranolol. The low dose did not elicit significant reductions in these injury markers (all P > 0.05). Effects of P. indicus Extract on Serum Cardiac Injury Markers and Pro-inflammatory Cytokines in Isoproterenol (ISO)-Induced Myocardial Ischemia Rats. (A) Creatine Kinase - MB (CK - MB); (B) Lactate Dehydrogenase (LDH); (C) Cardiac Troponin T (cTn - T); (D) Interleukin - 6 (IL - 6); (E) Interleukin - 1β (IL - 1β); (F) Tumor necrosis factor - α (TNF - α). Groups include control (Normal Control), ISO (Isoproterenol - induced myocardial ischemia model), PRO (Propranolol - treated positive control), and P. indicus at Low Dose (LD), Medium Dose (MD), and High Dose (HD). Data are presented as mean ± SD. **P < 0.01 vs. Control group; ##P < 0.01 vs. ISO group.
A similar pattern was observed for inflammatory cytokines (IL-6, IL-1β, TNF-α), which were elevated by 160-265% in the ISO group (P < 0.01). Medium and high doses of the extract produced broad anti-inflammatory effects, significantly suppressing all 3 cytokines by 27.9-63.0% (P < 0.01). In contrast, the low dose demonstrated a selective and modest effect, reducing only TNF-α by 17.1% (P < 0.05) without significant effects on IL-6 or IL-1β (P > 0.05). The low inter-individual variability in TNF-α reduction (RSD = 8.2%) further supports a consistent yet concentration-limited (threshold) effect, where higher doses are required for comprehensive anti-inflammatory activity.
P. indicus Activates the Nrf2/HO-1 Pathway to Counteract Oxidative Stress
To evaluate oxidative stress, SOD, GSH-Px and MDA were examined via ELISA in myocardial tissues. ISO challenge induced severe oxidative stress, reducing SOD by 60% and GSH-Px by 53% while elevating MDA by 122% vs. control (all P < 0.01, Figure 5). Medium/high-dose P. indicus significantly attenuated these alterations, restoring SOD to 70-150% of the levels in the ISO group, increasing GSH-Px by 35-97%, and reducing MDA by 26-48% (all P < 0.01 vs. ISO). Propranolol showed comparable efficacy. Low-dose treatment selectively elevated GSH-Px by 35% (P < 0.05) without significantly affecting SOD (+19%, P > 0.05) or MDA (−10%, P > 0.05), suggesting a nonlinear dose-response where glutathione-dependent pathways are preferentially activated at lower concentrations. P. indicus Counteracts Oxidative Stress by Activating the Nrf2/HO-1 Pathway A: Detection of SOD activity in myocardial tissues; B: Detection of GSH-Px activity in myocardial tissues; C: Detection of MDA content in myocardial tissues; D: Protein expression level of Nrf2 in myocardial tissues; E: Protein expression level of HO-1 in myocardial tissues; F: Western blot bands of Nrf2, HO-1 and β-tubulin (Internal reference). Values are presented as a mean ± SD. *P < 0.05, **P < 0.01, Compared with the Control group; #P < 0.05, ##P < 0.01, Compared with the ISO group. Low-Dose (LD), Medium-Dose (MD) and High-Dose (HD).
Concurrently, Western blot analysis revealed downregulation of the key antioxidant transcription factor Nrf2 and its downstream target HO-1.
Expression of Related Proteins in the Hearts of Rats in Each Experimental Group (
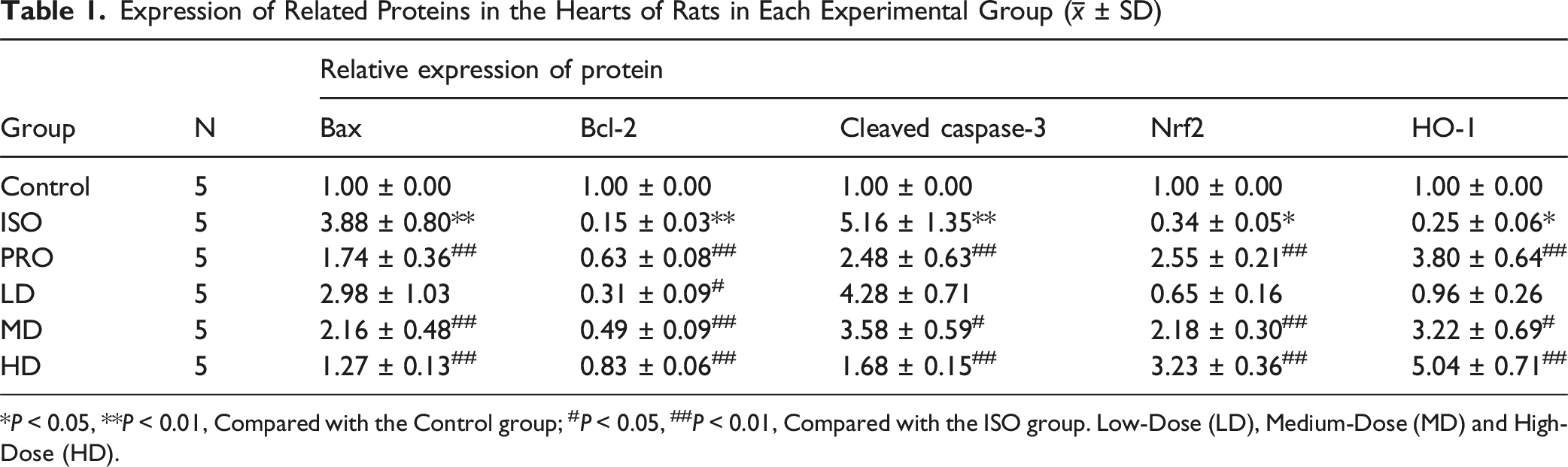
*P < 0.05, **P < 0.01, Compared with the Control group; #P < 0.05, ##P < 0.01, Compared with the ISO group. Low-Dose (LD), Medium-Dose (MD) and High-Dose (HD).
P. indicus Modulates the Bcl-2/Bax/Caspase-3 Axis to Suppress Apoptosis
The cardioprotection offered by P. indicus was further linked to the inhibition of cardiomyocyte apoptosis. qPCR analysis revealed ISO-induced dysregulation of apoptosis markers vs control: Bax increased 3.8-fold, Casp-3 increased 1.9-fold, and Bcl-2 decreased 79% (all P < 0.01, Figure 6). Medium/high-dose P. indicus significantly reversed these changes, reducing Bax by 45-63% and Casp-3 by 62-76% while elevating Bcl-2 2.4-3.4-fold vs ISO (all P < 0.01). Propranolol showed comparable effects. Low-dose treatment did not significantly alter Bax (+31% vs. ISO, P > 0.05), Casp-3 (−18%, P > 0.05), or Bcl-2 (+64%, P > 0.05). This transcriptional resistance contrasts with low-dose efficacy in glutathione regulation, suggesting apoptotic pathway modulation requires higher therapeutic thresholds. P. indicus Modulates the Bcl-2/Bax/Caspase-3 Axis to Suppress Apoptosis. A: mRNA expression of Bax in myocardial tissues; B: mRNA expression of Bcl-2 in myocardial tissues; C: mRNA expression of Caspase-3 in myocardial tissues; D: Protein expression of Bax in myocardial tissues (Normalized to β-tubulin); E: Protein expression of Bcl-2 in myocardial tissues (Normalized to β-tubulin); F: Protein expression of cleaved Casp-3 in myocardial tissues (Normalized to β-tubulin); G: Western blot bands of cleaved Casp-3, Bcl-2, Bax and β-tubulin (Internal reference). Values are presented as a mean ± SD. **P < 0.01, Compared with the Control group; ##P < 0.01, Compared with the ISO group. Low-Dose (LD), Medium-Dose (MD) and High-Dose (HD).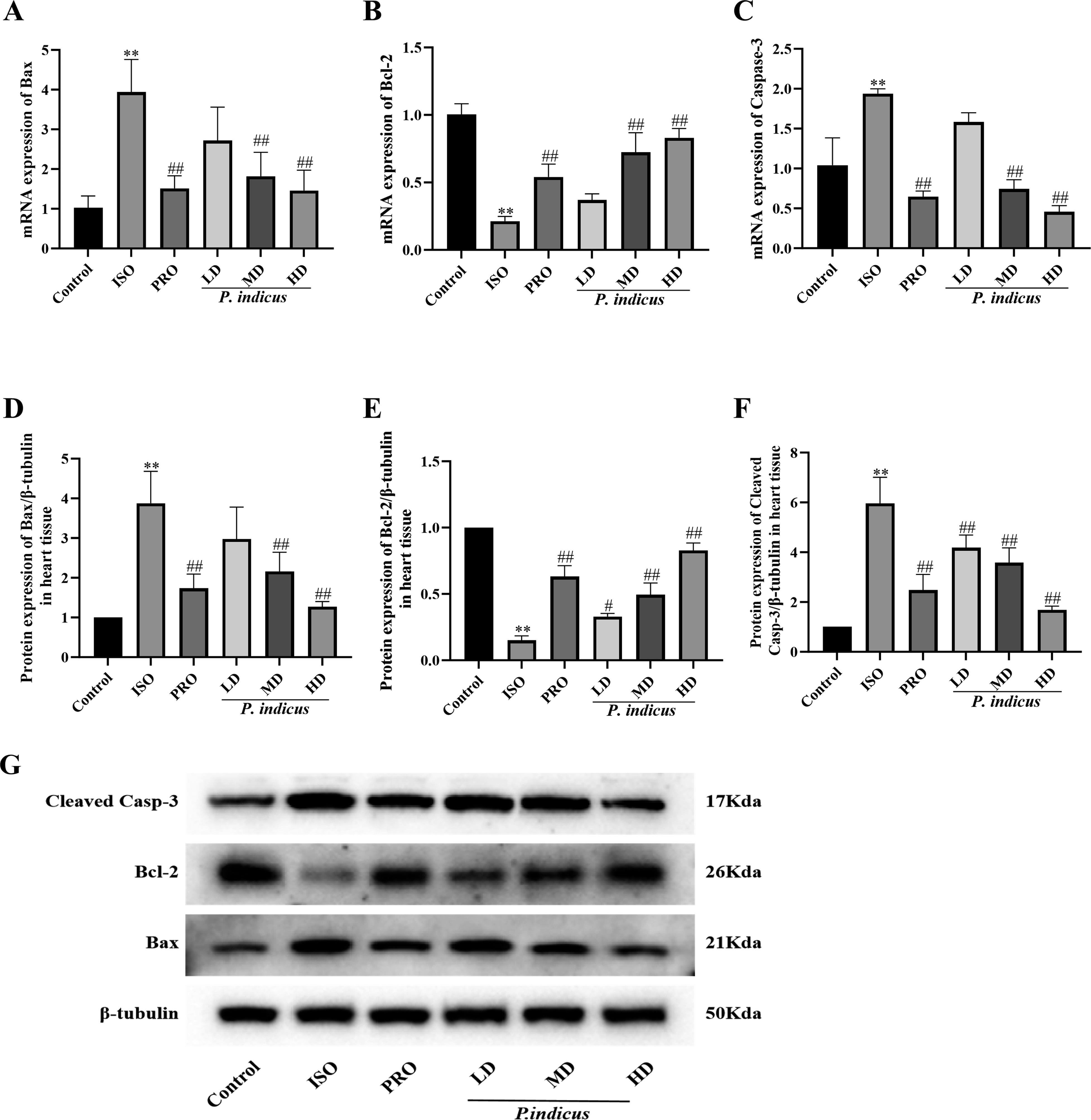
To further evaluate Bax, Cleaved Casp-3 and Bcl-2, western blot analysis was performed and showed the same trends as the mRNA expression in the ISO group, with Bax and Cleaved Casp-3 up-regulated, while Bcl-2 was down-regulated relative to the control (P < 0.01, Figure 6). Furthermore, consistent with the qPCR findings, the PRO and medium- and high-dose groups showed decreased Bax and Cleaved Casp-3 and increased Bcl-2 expression relative to the ISO group (P < 0.01). However, in the low-dose group, Cleaved Casp-3 expression was significantly decreased, while Bcl-2 was increased (P < 0.01) relative to the ISO group, but there was no significant change in Bax expression (P > 0.05).
Phytochemical Profiling and Dose-Response Relationship of P. indicus
Chemical Composition Analysis of P. indicus
The chemical composition of P. indicus was analyzed via Q-Exactive LC-MS, with a base peak ion current chromatogram generated (Figure 7). By matching molecular weights, fragment ions, and retention times to literature, 14 compounds were identified and categorized with specific examples: amino acids (L-tryptophan), alkaloids (harmane), organic acids (ferulic acid), polyphenols (gallic acid), flavonoids (quercetin-3-O-glucoside), and fatty acids (linoleic acid) (Table 2). Key bioactive compounds with reported cardioprotective effects were identified, which may contribute to the observed reductions in apoptosis and inflammation: Quercetin-3-O-glucoside (flavonoid): Known to inhibit apoptotic pathways by upregulating Bcl-2 and downregulating Bax/Casp-3, and to activate Nrf2/HO-1 for antioxidant defense—consistent with our findings in treated groups. Linoleic acid: A polyunsaturated fatty acid that suppresses pro-inflammatory mediators, potentially contributing to the reduced ischemic damage observed. Ferulic acid: Exhibits antioxidant activity by scavenging free radicals, which may enhance the protective effects of the Nrf2/HO-1 system. These compounds, either individually or synergistically, likely underpin the cardioprotective effects of P. indicus in our model. Phytochemical Profiling of P. indicus. A: Base peak ion current chromatograms of P. indicus extract analyzed via Q-Exactive LC-MS; B: HPLC chromatogram of mixed standard solution containing Quercetin-3-O-Glucoside, Ferulic Acid, and Linoleic acid; C: HPLC chromatograms of individual standard compounds (Quercetin-3-O-Glucoside, Ferulic Acid, Linoleic acid); D: Content determination of Linoleic Acid, Quercetin-3-O-Glucoside, and Ferulic Acid in P. Indicus Extract. Peaks: 1, Quercetin-3-O-Glucoside; 2, Ferulic Acid; 3, Linoleic acid. Chemical Composition Identification of P. indicus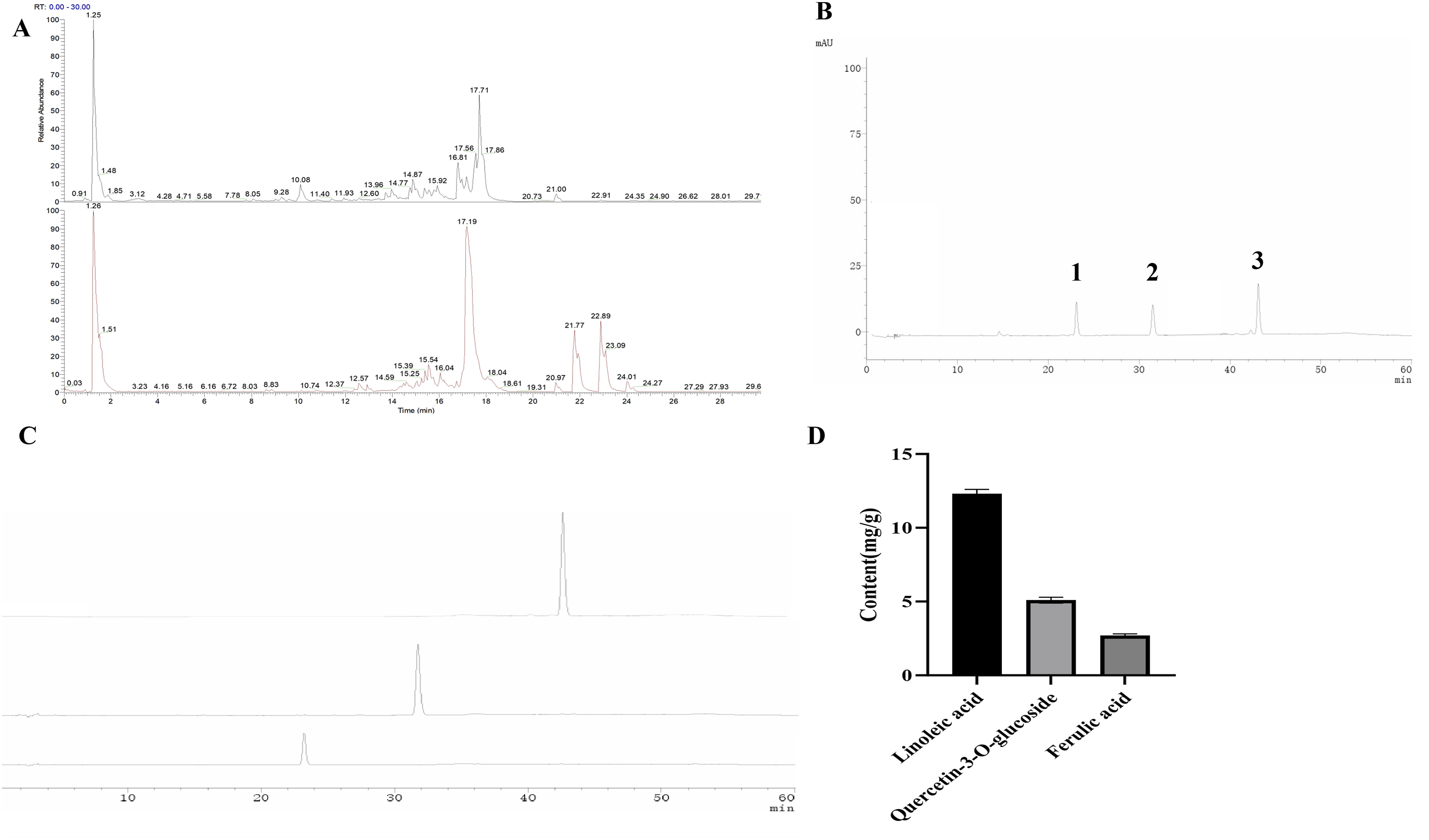

Quantitative Analysis of Major Bioactive Compounds in P. indicus Extract via HPLC
High-performance liquid chromatography (HPLC) coupled with a diode array detector (DAD) was used to quantify three key bioactive compounds—quercetin-3-O-glucoside, ferulic acid, and linoleic acid—in P. indicus extract. Method validation and quantitative results are detailed below. (1) Method Validation Results for HPLC Quantitative Analysis
In accordance with ICH guidelines Q2(R1), method validation was conducted to confirm the reliability of the HPLC method for quantifying the target compounds in P. indicus extract. All three compounds exhibited excellent linearity over their respective concentration ranges (quercetin-3-O-glucoside: 1-100 µg/mL, y = 1254.7x + 85.3, R2 = 0.9996; ferulic acid: 0.5-50 µg/mL, y = 985.2x + 42.1, R2 = 0.9994; linoleic acid: 5-200 µg/mL, y = 542.8x + 128.6, R2 = 0.9993). Intra-day and inter-day precision RSD values were all <3.0% (quercetin-3-O-glucoside: 1.95%/2.48%; ferulic acid: 1.78%/2.33%; linoleic acid: 2.15%/2.67%). Mean recovery rates ranged from 96.5% to 103.2% (RSD <3.2%). The method demonstrated adequate sensitivity for trace analysis with LOD values of 0.3-2.0 µg/mL and LOQ values of 1.0-6.0 µg/mL. System suitability parameters confirmed reliable performance (resolution Rs > 1.5; theoretical plate count N >5000; tailing factor T <1.15). These results collectively confirm the method’s suitability for quantitative analysis. Key validation parameters are summarized in Table 3. (2) Quantification of Target Bioactive Compounds in P. indicus Extract Summary of Method Validation Parameters for the HPLC Quantitative Analysis of Target Bioactive Compounds Rs, resolution; N, theoretical plate count; T, tailing factor.
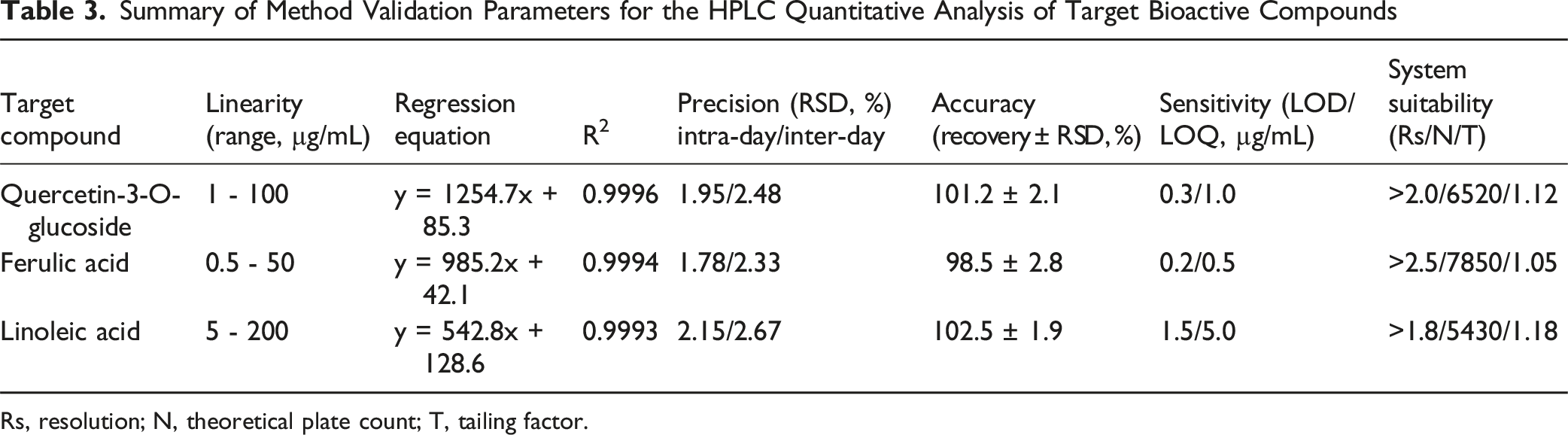
Contents of Target Bioactive Compounds in Three Batches of P. indicus Extract (mean ± SD, n = 3)

The quantified compounds showed consistent levels across 3 production batches: linoleic acid (12.3 ± 0.3 mg/g), quercetin-3-O-glucoside (5.1 ± 0.2 mg/g), and ferulic acid (2.7 ± 0.1 mg/g). The RSD values for all compounds across batches were <5%, indicating excellent batch-to-batch consistency of the P. indicus extract (Figure 7). Furthermore, LC-MS profiling revealed a broader phytochemical composition, suggesting that the overall cardioprotective activity may involve synergistic contributions from multiple constituents, consistent with the multi-component nature of herbal extracts.
Dose-Response Analysis
EC50 Values of P. indicus Extract for Key Cardioprotective Endpoints

The convergence of EC50 values around 50 mg/kg indicates that the medium dose (54 mg/kg) is situated precisely at the half-maximal effective concentration for these key cardioprotective endpoints, representing an optimal balance between efficacy and dose exposure. The designation of MD as the “optimal dose” is based on this efficacy profile; a comprehensive assessment of the therapeutic window would require formal toxicological evaluation at the high dose. Furthermore, the high dose (108 mg/kg) produced effects near the maximal response plateau, with no significant added benefit over the MD for most endpoints, which confirms the optimality of the MD and suggests a saturation of the therapeutic effect.
Pharmacokinetic Profile, Safety, and Therapeutic Potential
Systemic Safety Assessment
The systemic safety of P. indicus extract was evaluated through hepatorenal function markers and organ indices. As summarized in Figure 8, no significant differences were observed in serum ALT, AST, BUN, or Cr levels among groups, with all values remaining within normal physiological ranges and showing no dose-dependent trends. Liver and kidney indices were likewise comparable across groups, indicating an absence of organ hypertrophy or atrophy. Effects of P. indicus Extract on Serum Liver and Kidney Function Markers and Organ Indices in Isoproterenol (ISO)-Induced Myocardial Ischemia Rats. (A) Alanine Aminotransferase (ALT); (B) Aspartate Aminotransferase (AST); (C) Blood Urea Nitrogen (BUN); (D) Creatinine (Cr); (E) Liver index; (F) Kidney Index. Groups include control (Normal control), ISO (Isoproterenol - induced myocardial ischemia model), PRO (Propranolol - treated positive control), and P. indicus at Low Dose (LD), Medium Dose (MD), and HighDose (HD). Data are presented as mean ± SD
P. indicus extract treatment did not exacerbate the mild, non-significant parameter increases observed in the ISO model group. These results collectively demonstrate that 14-day P. indicus extract administration induces no detectable hepatorenal toxicity or organ damage at the tested doses, supporting its favorable safety profile and the selection of the medium dose (54 mg/kg) as a well-tolerated therapeutic regimen.
Pharmacokinetic Profile of P. indicus Extract Marker Compounds
The systemic exposure to key bioactive compounds in P. indicus extract was evaluated following a single oral dose of 54 mg/kg. The mean plasma concentration-time profiles of quercetin-3-O-glucoside, ferulic acid, and linoleic acid are presented in Figure 9, and the corresponding pharmacokinetic parameters are summarized in Table 6. Plasma Concentration-Time Profiles of Marker Compounds in P. indicus Extract. A: Plasma concentration-time profile of quercetin; B: Plasma concentration-time profile of ferulic acid; C: Plasma concentration-time profile of linoleic acid Pharmacokinetic Parameters of Marker Compounds in Rat Plasma After a Single Oral Administration of P. indicus (54 mg/kg) (mean ± SD, n = 6)
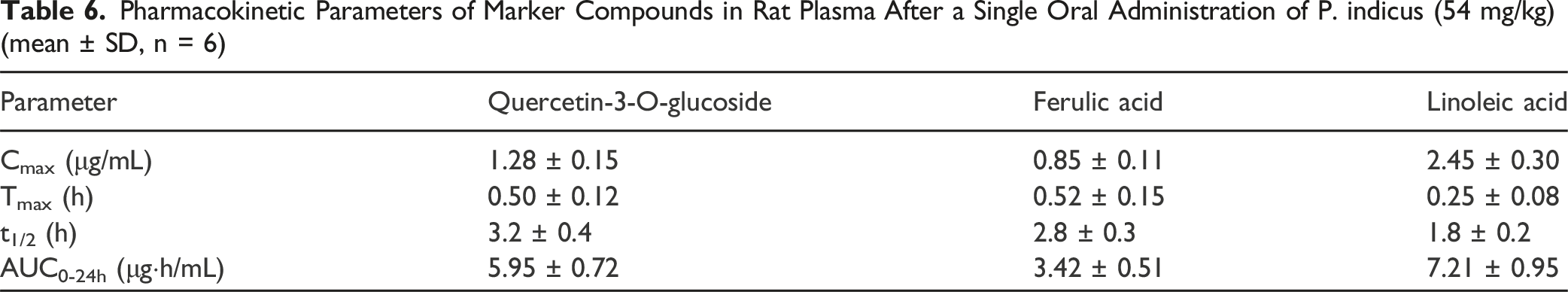
All three marker compounds were rapidly absorbed into the systemic circulation. Linoleic acid exhibited the most rapid absorption, reaching its maximum plasma concentration (Tmax) at 0.25 h post-administration, earlier than quercetin-3-O-glucoside and ferulic acid (both at 0.5 h). The peak concentration (Cmax) of linoleic acid was 2.45 ± 0.30 μg/mL, which was notably higher than that of the phenolic compounds.
Thereafter, plasma concentrations of all three compounds declined. The elimination half-life (t1/2) of linoleic acid was 1.8 ± 0.2 h, shorter than that of quercetin-3-O-glucoside (3.2 ± 0.4 h) and ferulic acid (2.8 ± 0.3 h), indicating a faster clearance rate.
The total systemic exposure, expressed as the area under the plasma concentration-time curve from 0 to 24 h (AUC0-24h), was 5.95 ± 0.72 μg·h/mL for quercetin-3-O-glucoside, 3.42 ± 0.51 μg·h/mL for ferulic acid, and 7.21 ± 0.95 μg·h/mL for linoleic acid. Despite its shorter half-life, linoleic acid achieved the highest overall exposure due to its high initial concentration.
These data provide the first direct evidence of systemic exposure to the key marker compounds of P. indicus extract, establishing a preliminary pharmacokinetic basis for its observed in vivo efficacy. The distinct pharmacokinetic profiles of the phenolic antioxidants and the fatty acid suggest potential complementary roles in the cardioprotective effects.
Therapeutic Efficacy of P. indicus
To evaluate the clinical relevance of P.indicus, we investigated its efficacy when administered concurrently with and after ISO-induced injury (therapeutic regimen). The results demonstrated that P. indicus (54 mg/kg) initiated after the onset of ischemic injury still conferred significant cardioprotection across multiple key endpoints, although the effect magnitude was generally slightly attenuated compared to the prophylactic regimen (Figure 10). Effects of P. indicus in Therapeutic and Prophylactic Regimens on Various Cardiac and Oxidative Stress - Related Indicators. Panels A - C show the levels of Creatine Kinase - MB (CK - MB), Lactate Dehydrogenase (LDH), and Cardiac Troponin T (cTn - T), respectively. Panels D - F display the levels of Superoxide Dismutase (SOD), Glutathione Peroxidase (GSH - Px), and Malondialdehyde (MDA), respectively. Values are presented as a mean ± SD. **P < 0.01, relative to the Control group; ##P < 0.01, relative to the ISO model group. Medium-Dose (MD) and P. indicus therapeutic (THE)
In Vivo Mechanistic Validation of Cardioprotective Pathways
Pharmacological inhibition confirmed that Nrf2/HO-1 and caspase-3 pathways are necessary for P.indicus-mediated cardioprotection (Figure 11). The Nrf2 inhibitor ML385 abrogated P.indicus-induced HO-1 upregulation, while the caspase-3 inhibitor Z-DEVD-FMK reversed the suppression of cleaved caspase-3. Consequently, both inhibitors attenuated the P.indicus-induced reduction in serum cTn-T. ML385 specifically nullified P.indicus’s antioxidant effect by suppressing SOD activity and elevating MDA content, whereas Z-DEVD-FMK selectively blocked its anti-apoptotic action. Control experiments ruled out intrinsic cardiotoxicity for either inhibitor. These findings establish that Nrf2 activation and caspase-3 inhibition are both essential for P.indicus’s cardioprotective efficacy in vivo. In Vivo Mechanistic Validation of P. Indicus-Mediated Cardioprotective Pathways. A: Serum cTn-T level detection; B: Myocardial SOD activity detection; C: Myocardial MDA content detection; D: Western blot bands of HO-1, Cleaved Caspase-3 and β-Tubulin (Internal reference) in myocardial tissues; E: Protein expression level of HO-1 in myocardial tissues; F: Protein expression level of Cleaved Caspase-3 in myocardial tissues. Values are presented as a mean ± SD. **P < 0.01 vs. ISO; ##P < 0.01 vs. MD; ISO: Isoproterenol; MD: Medium-Dose P. indicus; ML385: Nrf2 Inhibitor; ZF: Caspase-3 Inhibitor Z-DEVD-FMK.
Discussion
The aim of this study was to explore the protective effect and mechanisms of P. indicus in an ISO-induced myocardial ischemic rat model. To construct the rat model, ISO, as a β-adrenergic receptor agonist, was chosen due to its ability to trigger a series of pathological processes similar to myocardial ischemia, including an accelerated heart rate and increasing myocardial oxygen consumption, thereby leading to mitochondrial dysfunction, oxidative stress response and myocardial cell apoptosis. 28 The successful establishment of the ISO-induced myocardial ischemia model was confirmed by significant ECG abnormalities (ST-segment elevation), elevated serum cardiac enzymes (CK-MB, LDH, cTn-T), and histopathological damage. P. indicus intervention, particularly at medium and high doses, markedly ameliorated these ischemic injury markers. The absence of significant changes in hepatorenal function parameters and organ indices across all P. indicus extract-treated groups suggests a favorable preliminary safety profile for the extract at the administered doses.
Oxidative stress plays an important role in myocardial ischemic injury and as such, SOD, MDA and GSH-Px were selected as indicators of oxidative stress. SOD is an important antioxidant metalloenzyme that can catalyze the disproportionation of superoxide anion free radicals and maintains the balance between oxidation and antioxidation. 29 As an end product of lipid peroxidation, MDA levels reflect the rate of lipid peroxidation and the degree of tissue damage. 30 GSH-Px exerts an antioxidant effect through catalytic reactions. 31 As expected, the ISO group showed significantly reduced SOD and GSH-Px, and increased MDA relative to the control.32,33 Following P. indicus intervention, the opposite trends were observed. Notably, the identified flavonoid hispidulin, rich in phenolic hydroxyl groups, may scavenge reactive oxygen species (ROS) directly, as reported in previous studies, thereby inhibiting MDA accumulation and enhancing SOD/GSH-Px activity. This is consistent with the cardioprotective effects of other flavonoid-rich herbal extracts, but P. indicus shows a unique dose-dependent response, with medium/high doses specifically targeting the Bcl-2/Bax/Casp-3 pathway.
Inflammatory cytokines are important factors that exacerbate myocardial ischemic injury. Herein, serum inflammatory cytokines IL-6, IL-1β and TNF-α were increased in the ISO group relative to the control, with levels significantly reduced following P. indicus treatment. This finding suggests that P. indicus may play a myocardial protective role by reducing the release of inflammatory factors, thereby reducing inflammatory damage in myocardial tissue. It is worth noting that propranolol, a non-selective β-adrenergic receptor antagonist, exerted comparable protective effects by blocking ISO-induced β-receptor overactivation, reducing myocardial oxygen demand, and inhibiting downstream pro-inflammatory and pro-apoptotic signaling—serving as a valid positive control to validate the cardioprotective efficacy of P. indicus.
Although the low-dose P. indicus extract (27 mg/kg) did not significantly ameliorate most ischemic parameters, it selectively reduced TNF-α levels and modestly enhanced GSH-Px activity. This suggests a threshold effect for broader anti-inflammatory and antioxidant responses, possibly due to differential sensitivity of signaling pathways. Future studies should investigate inter-individual variability and potential subpopulations of “responders” even at low doses, which may inform personalized dosing strategies.
Moreover, further examination identified the Nrf2/HO-1 pathway that plays a key role in antioxidative stress and suppresses inflammatory cytokines, 34 including Il-6 and Il-1β. During cellular ischemia or hypoxia, Nrf2 is activated and translocated into the nucleus to activate the expression of antioxidant enzymes such as HO-1. The results of this study showed that P. indicus can significantly up-regulate Nrf2/HO-1 expression and thereby reduce the inflammatory and oxidative stress associated with myocardial ischemic injury.
Apoptosis plays a significant part in myocardial ischemia, with the Bcl-2/Bax/Casp-3 signaling pathway being a key regulator of apoptosis. 35 Herein, both Bax and Casp-3 mRNA and protein expression levels were up-regulated, while Bcl-2 was down-regulated, in the ISO group relative to the control. However, P. indicus treatment promoted the expression of anti-apoptotic Bcl-2 and down-regulated pro-apoptotic protein Bax and its downstream target Casp-3. The protective effects were strictly dose-dependent, with MD/HD (but not LD) significantly activating the Nrf2/HO-1 antioxidant pathway (Figure 6D-E). While LD showed no change in Nrf2 protein expression (P > 0.05 vs. ISO), MD/HD increased Nrf2 by 40%/60% (P < 0.01 vs. ISO), respectively, leading to enhanced HO-1 expression and reduced MDA levels. This dose threshold likely reflects the minimal concentration required to trigger Nrf2 nuclear translocation and subsequent anti-apoptotic effects, as Bcl-2/Bax ratio was only significantly increased in MD/HD (P < 0.01 vs. LD).
To further characterize P. indicus, its chemical constituents were identified using LC-MS and divided into several categories including amino acids and their derivatives, alkaloids, organic acids and their esters, polyphenols and flavonoids, lipids and fatty acids. Amino acids and their derivatives contain amino, carboxyl and hydroxyl groups. Amino and carboxyl groups participate in metabolic reactions in the body and hydroxyl groups regulate the redox state in cells and play a role in myocardial cell energy metabolism and antioxidant defense system,36,37 reducing myocardial ischemic injury.
Upon component identification, it was found that natural flavonoids are present in P. indicus. Flavonoids are polyphenol derivatives of plant origin and have been implicated in numerous activities such as antioxidant, anti-inflammatory, anti-apoptotic, and anti-thrombotic activity, leading to their role in cardioprotection. 38 Of the identified alkaloids, trigonelline contains a pyridine ring structure and plays a role by binding to intracellular receptors or enzymes to regulate inflammatory signaling pathways, reduce the release of inflammatory factors and reduce inflammation. 39
Among organic acids and their esters, nicotinic acid and propyl gallate were identified. Nicotinic acid, also known as vitamin B3, contain a carboxyl group that participates in redox reactions. The carboxyl group reduces oxidative stress damage by participating in electron transfer or synergistic action. 40 In propyl gallate, the phenolic hydroxyl group scavenges free radicals, and the ester group affects absorption and distribution to enhance the antioxidant effect. 41 Additionally, the polyphenol hispidulin has multiple phenolic hydroxyl groups that can scavenge free radicals, interrupt the chain reaction of lipid peroxidation, and regulate antioxidant enzyme systems, such as SOD/GSH-Px/MDA, to enhance the antioxidant capacity of the heart. 42
Among the lipids and fatty acids, linolenic acid is a polyunsaturated fatty acid with carbon-carbon double bonds. As a cell membrane component, it affects membrane stability and has anti-inflammatory and antioxidant effects. It protects myocardial ischemic injury by reacting with free radicals and regulating the synthesis and release of inflammatory mediators.43,44 Compared to other well-studied botanicals such as resveratrol or quercetin, which also exhibit antioxidant and anti-inflammatory properties, P. indicus extract contains a unique blend of bioactive compounds including hispidulin, trigonelline, and linoleic acid. This multi-component profile may confer synergistic cardioprotective effects targeting both oxidative stress and apoptosis simultaneously. Moreover, the dose-dependent response observed here—with medium dose (54 mg/kg) achieving efficacy comparable to high dose—suggests a favorable safety profile relative to some high-dose flavonoid interventions reported elsewhere.
In conclusion, this study demonstrates that P. indicus extract confers significant cardioprotection in ISO-induced myocardial ischemic rats, primarily through reducing oxidative stress, inflammation, and apoptosis, correlated with modulation of the Nrf2/HO-1 and Bcl-2/Bax/Casp-3 pathways. However, these protein-level associations do not establish causality, and future studies should employ targeted interventions—such as Nrf2 or Casp-3 inhibition and Bcl-2/Bax knockdown—to verify mechanistic specificity. The medium dose (54 mg/kg) is recommended as optimally effective and safe for further development. These findings provide a foundation for advancing preclinical and clinical applications of P. indicus in cardiovascular therapeutics.
In addition to the pharmacodynamic effects, we characterized the preliminary pharmacokinetic behavior of P. indicus extract’s key marker compounds. The rapid absorption and measurable systemic exposure of quercetin-3-O-glucoside and ferulic acid provide a pharmacokinetic basis for the observed in vivo efficacy. The fact that significant cardioprotection was achieved at doses yielding these plasma concentrations supports the biological relevance of the quantified markers. Future studies could explore formulation strategies to enhance the bioavailability and prolong the systemic exposure of these active constituents.
This study has several limitations. First, although 3 bioactive compounds were quantified and linked to relevant mechanisms, the individual contribution of each constituent, as well as potential synergies among them, remains to be clarified through bioactivity-guided fractionation and testing of pure compounds. Second, the specific role of signaling pathways such as Nrf2/HO-1 and the functional relevance of other identified compounds require further validation using in vitro or genetic approaches. Moreover, the prophylactic treatment regimen employed here does not fully reflect the clinical scenario of post-ischemic intervention; thus, studies using a therapeutic dosing protocol are needed to enhance translational relevance. Finally, while the ISO-induced model recapitulates key features of myocardial injury such as oxidative stress and apoptosis, it models catecholamine toxicity rather than coronary occlusion-reperfusion. Therefore, caution is warranted in extrapolating these results directly to human myocardial infarction, and further validation in ischemia-reperfusion models is recommended.
Conclusion
This study demonstrates that P. indicus extract exerts dose-dependent cardioprotective effects in ISO-induced myocardial ischemic rats, primarily associated with the attenuation of oxidative stress, inflammation, and apoptosis. The medium dose (54 mg/kg) is recommended as the optimal efficacious dose for future preclinical studies. These findings provide a preliminary foundation for developing P. indicus as a therapeutic agent, pending further investigation into its mechanisms of action, pharmacokinetics, and therapeutic efficacy in post-infarction models.
Footnotes
Ethical Considerations
Xizang Minzu University, No. 2024088.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Natural Science Foundation of Tibet Autonomous Region (Grant No. XZ202001ZR0015G), the Natural Science Research Project of the Department of Education of Shaanxi Province (No. 23JK0697), the 2023 Project of the Shaanxi Provincial Education Science ‘14th Five Year Plan’ (No. SGH23Y2487), and the Internal Research Project of Xizang Minzu University (No. 22MDY03 and 22MDZ02).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
