Abstract
Silver-plated yarn is one of the fundamental materials in smart textiles. However, the plating can be corroded seriously by human sweat during daily wear, degrading the electrical conductivity. To address this issue, this article designed a new set of experimental protocols to faithfully simulate sweat immersion in daily wear. Artificial sweat with pH 5.5 and 8.0 was applied to five types of yarns on a daily basis, and changes in the morphological, electrical and mechanical properties of the yarns before and after treatment were recorded. The results showed increasing resistance of the yarns after exposure to sweat at both pH values. After 10–20 test cycles, almost all samples lost their electrical conductivity, and their mechanical properties also degraded. Yarn service life in acidic sweat was longer than in alkaline sweat. Morphological characterization revealed that the coating on the yarn surface peeled off after treatment. The coating reacted electrochemically with Cl− in sweat, resulting in interruption of the conductive pathway, hence a sudden increase in resistance. Compared to acidic environment, Ag is more prone to undergo redox reaction in alkaline environment. Therefore, silver-plated yarn corrodes faster in alkaline sweat. Finally, the decrease in mechanical properties was due to the swelling behavior of nylon 6 in sweat.
Introduction
In recent years, the emerging smart wearable devices and smart textile products have offered convenient and comfortable user experience, while they also promoted the application of an important new material, that is silver-plated yarn.1–5 Typically, silver-plated yarns are integrated in textiles through knitting, weaving, embroidery, and sewing, 6 enabling functionalities such as sensors,7–9 nanogenerators,10–12 and wires. 13 Currently, these smart textiles have been well applied in many fields including medical, 14 sports, 15 military, 16 etc. in which the performance of silver-plated conductive yarn will directly affect the performance of smart textiles.
Most of previous research on the evaluation of silver-plated yarns focused on the electrical and mechanical properties.17–20 For example, both Ali et al. 17 and Si et al. 18 investigated the coating process of conductive yarns and the impact of stretching and washing on their electrical properties. Won and Yun’s work 19 focused on the daily care of silver-plated conductive yarns, such as the influence of washing methods, drying temperatures, detergents, and other factors on the electromechanical properties of silver-plated conductive yarns. In addition, Liang et al. 20 examined the sensitivity and fatigue of fabrics made from different silver-plated conductive yarns. Research in very recent years also focused on the electromechanical properties of Ag-plated yarns,21,22 as they can be used as sensors and wires for wearable electronics, the former requires large changes in electrical resistance while the latter prefers more stable electrical resistance upon tensile strains. Although the above studies evaluated the performance of silver-plated conductive yarns in both mechanical and electrical perspectives, they neglected the effect of human sweat in practical use.
Human sweat can affect the electrical performance of silver-plated yarns, especially when doing strenuous sports or in hot weather, exposing the yarns to lots of sweat for a long time. Therefore, there have been studies on the sweat resistance of silver-plated yarns. Unfortunately, they simply soaked the yarn directly into artificial sweat,23,24 which is apparently different from the daily wear situations. For instance, Tajin et al. 23 knitted silver-plated conductive yarn into fabric and soaked the fabric in sweat to study its short-term sweat resistance. The experiment had a short testing period and only included electrical performance testing, lacking characterization of the morphology and mechanical property analysis of the fabric after soaking. Park et al. 24 tested the sweat resistance and washability of existing silver-plated fabrics. In the test, they directly immersed the yarn in sweat for a period of time, washed off the surface sweat, and then performed the evaluation. Although this study evaluated the basic parameters of silver-plated yarn in a comprehensive manner, the sweat resistance test still adopted a short-term fashion, which was not consistent with daily use scenarios. In addition, the pH value of the sweat used in the study was insufficient, and they did not take into account the fact that the human sweat pH varies and it can be both acidic and alkaline. Therefore, the previous studies failed to faithfully simulate the interaction between silver-plated yarn and human sweat in daily wear.
In view of the above limitations, this study aims to provide a more realistic and faithful evaluation of sweat resistance of silver-plated yarns. The rationale of our methodology is to simulate a very demanding usage scenario. That is, after doing sports, one’s clothes integrated with silver-plated yarn is soaked in sweat and he or she does not wash them immediately. On the following day, he or she continues to wear this garment to do sports, and in this way he or she wears it day by day. To simulate the above scenario, a testing protocol is established, where artificial sweat is sprayed onto the silver-plated yarns sewn onto a fabric every 24 h until they lose their electrical conductivity. Meanwhile, the surface morphology and electrical conductivity of the silver-plated yarns before and after each treatment with sweat are recorded throughout the experiment and compared with the initial state. According to this testing protocol, silver-plated yarns from multiple manufacturers and with different specifications were explored. In this regard, this study can provide data support and important reference for the optimal design and practical application of silver-plated yarns.
Experiment
Materials
Ag-plated yarns
In order to comprehensively evaluate commercially available silver-coated yarns, we randomly purchased five types of yarns in the Chinese market with a price range of about 280–420 USD/kg. The yarns selected for testing are listed in Table 1, along with their models and parameters. All the yarns were used without further treatment. Note that the yarns only represent the samples we purchased, and thus they are not intended for judging the quality of products or the brands. We selected representative counts of silver-coated yarns, 70D × 3, 70D × 4, and 100D × 3, which are widely used in smart textiles. Additionally, this study helps to evaluate whether the fineness of yarn affects the capability of sweat resistance, providing important insights for future applications of silver-plated conductive yarns in smart textiles.
Specification of silver-plated yarns.

Artificial sweat
Due to the lack of specific standards for testing the sweat resistance of silver-plated conductive yarns, the selection of sweat in this study refers to the most appropriate Chinese national standard, GB/T 3922-2013, “Textile Color Fastness Test for Color Fastness to Perspiration.” 25 Importantly, both the alkaline solution and the acidic solution are exactly the same as those in the International Standard, ISO 105-E04:2013, “Textiles—Tests for color fastness, Part E04: Color fastness to perspiration.” 26 The artificial sweat used in the experiment is shown in Table 2, and the composition of sweat is shown in Tables 3 and 4. The distilled water used, and 0.1 mol/L sodium hydroxide solution was used to adjust the pH of sweat. The pH values were 5.5 and 8.0, respectively, which was according to the composition of artificial sweat specified in GB/T 3922-2013 “Textile Color Fastness Test for Color Fastness to Perspiration.” 25
Basic properties of artificial sweat.

Composition of acidic artificial sweat.

Composition of alkaline artificial sweat.

Fabrics
When a large amount of sweat soaks into clothing, it does not evaporate immediately. Rather, the sweat will slowly corrode the silver-plated yarns over a long period, as a result of the porous structure of fabrics and yarns, and the moisture-absorbing fibers in them. In order to better simulate the real use scenario of silver-plated yarns, this study chose polyester-cotton blended fabric, a common fabric for apparel, as the substrate for research. Cotton fibers have a high moisture regain as a result of the abundant hydroxyl groups and strong moisture absorption capacity. In contrast, polyester has poor moisture absorption but higher mechanical strength. Herein, we chose bleached polyester-cotton blended white fabric (T/C 80/20, Dongguan Huawei Textile Co. Ltd., Guangdong, China) to simulate the apparel fabric. It has a three-up-one-down twill structure. The areal density of the fabric is approximately 190 g/m². In addition, we secured the yarns onto the fabric by sewing to simulate the integration of conductive yarns in smart textiles. Note that the sewing procedure was only applied at the ends of the fabric sample, and no sewing at the middle testing part where sweat was going to be applied. This special arrangement is important, because the sewing process introduces mechanical wear on the coating of the yarn, which may damage the coating and cause potential experimental errors.
Methodology
Sample fabrication
First, randomly take 30 m of yarns from each reel and divide them into 10 sections. Then, cut the middle 0.9 m from each section as the samples for the tests according to the zoning shown in Figure 1. The yellow parts are used for tensile testing, morphological characterization, and composition analysis. Five 60 cm sections are selected from each sample, and the middle 20 cm gray part is cut out as the tensile testing sample. Five 60 cm sections are also selected from each yarn, and 6 cm samples are taken from the head, middle, and tail positions, respectively. Each 6 cm section is cut into two 3 cm samples for morphological characterization and composition testing analysis. The green part is used for sweat resistance testing and linear resistance uniformity testing. From each sample, 5 sections of 30 cm are selected, with the middle 10 cm being the linear resistance uniformity test sample as shown in white. Note that the 10 cm does not need to be cut out. Instead, after the resistance test, the 30 cm will be directly used in the acidic sweat resistance test. Another 5 sections of 30 cm are selected from each yarn in the alkaline sweat test.

Sampling diagram of silver-plated yarns.
Prepair10 pieces of bleached polyester/cotton 80/20 twill fabrics with a size of 26 cm × 36 cm. Each piece of fabric is bound and sewn to prevent the fabric from unraveling during test. After sewing, each piece of fabric measures 25 cm×35 cm. Divide the 10 pieces of fabric into 5 groups, namely 2 pieces in each group. Mark the left upper corner of the fabric with the Xi format, where X represents the type of yarn, see Table 1 for specific numbers, X∈{A, B, C, D, E}; and i represents the type of sweat, see Table 2 for specific numbers, i∈{1, 2}. For example, A1 means the 210D silver-plated yarn from Jinan Yumo Technology and Trade Co., Ltd., and the pH value of the artificial sweat used is 5.5.
Yarn evaluation
Morphological characterization
Firstly, observe the sample appearance using an extended depth-of-field microscope (Keyence VHX-7000, Japan) and record the morphology under 200-fold magnification. Then, gold-spray the surface of the sample and observe its morphology using a scanning electron microscope (ZEISS Sigma 500, Germany). Finally, take energy spectrum photos to observe the distribution of Ag on the yarn surface.
Composition analysis
Perform differential scanning calorimetry (DSC) analysis on each sample using a Differential Scanning Calorimeter (DSC; Diamond DSC, PerkinElmer, USA). Heat each sample (1–2 mg) in a crimped aluminum pan at a scanning rate of 10 °C/min, with a temperature range between 50°C and 480°C. Perform the above analysis under a nitrogen flow rate of 50 mL/min.
Mechanical properties
According to the Chinese standard, GB/T3916-2013, “Textiles - Rolled Yarn - Determination of Breaking Force and Elongation at Break,” 27 five sets of yarn samples (five samples per set, each 20 cm long) are tied at the ends, and the middle 10 cm of the yarn samples are clamped using an electronic universal testing machine (Lan Bo San Si LD23.503, Shenzhen, China) with a pre-tension of 0.02 N and a tensile speed of 30 mm/min.
Linear resistance
Prepare five sections of yarn samples, each of which is 30 cm long and separated by 80 cm. Use a digital multimeter (Hantai HDM3065A, Qingdao, China) to measure the resistance value of the middle 10 cm of each sample and calculate the mean value and standard deviation. Test should be conducted under conditions of (25±2)°C and (65±5)% relative humidity. A pretension of 0.1 N should be applied to the yarn sample in order to make sure the yarn is straight but not stretched. Note that this linear resistance test partially complies with the international standard, EN IEC 63203-201-1. 28 The differences include specimen length and temperature. The former in the standard is 50 cm, while we chose 10 cm. The latter is (23±2)°C or (27±2)°C while we chose (25±2)°C as we situated in the subtropical region. Besides, also note that all the five samples of 30 cm + 80 cm came from the same reel of yarn package and therefore, batch variation was not considered in this test.
Sweat resistance testing
Prepare yarn samples for sweat resistance testing. Fix the prepared yarn samples using stitching onto the corresponding fabric samples and label them, as shown in Figure 2. Place 10 fabric samples under conditions of (25 ± 2)°C and (65 ± 5)% relative humidity for 24 h of conditioning. Use a digital multimeter (Hantai HDM3065A, Qingdao, China) to test the initial (before artificial sweat solution application) resistance of all yarn samples over a distance of 20 cm. Spray 550 mg ± 50 mg of artificial sweat solution at the 10th cm from the middle of each yarn sample, which means the effective distance is 20 cm. After 24 hours conditioning in the constant temperature and humidity laboratory, record the resistance of the samples over a distance of 20 cm. Repeat the above steps until the silver-plated yarn loses its conductivity, assuming that a resistance of 1 MΩ indicates the loss of electrical conductivity. Subsequently, characterize the sweat resistance performance through morphology characterization, electrical performance, and mechanical property testing.

Scheme of the sample layout on fabric. (a) Schematic of sample layout; (b) Picture of one prepared fabric sample.
Characterize the morphology of the yarn samples after sweat treatment. Observe the central part of the samples using an ultra-depth microscope (Keyence VHX-7000, Japan) and take photos with a 200-fold lens. Select the most corroded yarns from each fabric as the samples for SEM characterization. Out of the most corroded yarn sample, cut off the middle 9 cm section, divide it into three equal sections of 3 cm, gold-spray the surface of each sample, and observe the surface morphology using a Scanning Electron Microscope (SEM; ZEISS Sigma 500, Germany). Take energy spectrum photos to observe the distribution of Ag on the yarn surface. Finally, conduct mechanical property testing on the remaining four yarns of each fabric, using the same testing method as before sweat treatment.
Results and discussion
Characterization of raw yarns
Morphology
As shown in Figure 3(a), the silver plating layer of yarn B had good durability, gloss, and uniformity, but there was a slight shedding phenomenon. As seen from the appearance, the yarn was tightly twisted and bound. The gloss of yarn A was second only to yarn B, and yarn D was inferior to yarn A. The silver plating layer of yarn C and yarn E had poor durability and uniformity. Compared to the first three yarns, the two yarns were loose in texture. As shown in Figure 3(b) and (c), the silver content of yarn B was higher, followed by yarns A and D. The silver content of yarns C and E was lower, and the appearance was loose and gloom. Therefore, the quality of yarn in terms of appearance was B > A > D > C > E.

Morphological characterization of five types of Ag-plated yarns. (a) Optical image (×200); (b) SEM image (×2000); (c) EDS image.
Composition analysis
In Figure 4(a), all five sets of yarns showed peaks near 220°C and 450°C, while no peaks were observed at other points. This proves that the compositions of the five yarns are the same. According to previous studies,29,30 the melting temperature of nylon 6 was believed to be around 220°C. Analysis of the characteristic peaks in Figure 4(a) further confirmed that the core material of the five types of yarns was nylon 6. In addition, based on previous research 30 and the energy spectrum diagram in Figure 3(c), it can be confirmed that the characteristic peak at 450°C is silver (Ag).

Properties of Ag-plated yarns (a) DSC curves; (b) stress-strain curves; (c) resistance values of 10 cm yarns.
Mechanical property
The tensile properties of the five types of yarns varied greatly according to Figure 4(b), but the overall fracture strain was all above 40%. Since the fineness of yarns B and E was greater than that of A, C, and D, the former stress was higher than the latter. Under the same yarn count, the strain of B and E was the same, both being 52%. Among them, the breaking strength of E was 1300 MPa, which was higher than that of B, 1100 MPa. Similarly, the breaking strength of A, 750 MPa, and the breaking elongation of 51% were both higher than those of C and D, 400 MPa and 45%, respectively. Therefore, within the same yarn count, the breaking strength of A and E was relatively higher.
Uniformity of Ag plating
As shown in Figure 4(c), A, C, and D yarns are all 210 denier. The resistance values correspond to a yarn gage length of 10 cm as specified in section 2.2.2.4. The resistance and standard deviation of silver-plated yarn A were the smallest, indicating that the silver-plated yarn A had the most uniform electrical resistance, with a linear resistance of 2.2 ± 0.01 Ω/cm. Next is yarn D, with a resistance of 2.3 ± 0.02 Ω/cm. For the samples of B and E, B was more uniform, with a resistance of 1.7 ± 0.02 Ω/cm. Therefore, the order of uniformity of the five yarns’ electrical resistance was A > D > B > E > C. Within the same diameter, the order of electrical conductivity was A > D > C and B > E.
Sweat resistance of Ag-plated yarns
Morphology
Figure 5(a) shows the appearance of the five types of yarns at 200 times magnification after being treated with two types of artificial sweat. The damage to the yarn from sweat No. 2 was higher than that from sweat No. 1, indicating that alkaline artificial sweat with a pH of 8 had a more severe effect on the silver coating. In addition, the surface gloss of all yarns treated with artificial sweat became dull, and the silver coating on the yarn surface was damaged.

Optical image of samples after sweat treatment. (a) Microscopic view; (b) SEM image with energy spectrum.
The SEM and energy spectrum diagrams of the five types of yarns after sweat treatment are shown in Figure 5(b). After the yarns were exposed to sweat, unknown crystals formed on the surface, and some parts were corroded. According to the energy spectrum diagram, it can be confirmed that the silver plating layer on the surface has been damaged. In addition, compared to acidic sweat, alkaline sweat had a stronger destructive power. Under acidic sweat immersion, the sweat-resistant performance of A, B, and D yarns is better than that of C and E yarns.
Electrical resistance
As shown in Figure 6, under the treatment of two types of sweat, the disappearance sequence of conductivity for the five types of yarns is: E, C, D, A, and B. Most of the changes in resistance values were sudden increases, jumping directly from one or two orders of magnitude to mega Ohm level. According to the studies of Shahpar and Payer,31,32 silver-plated yarns mainly react with NaCl under certain conditions, and the reaction equation is as follows:

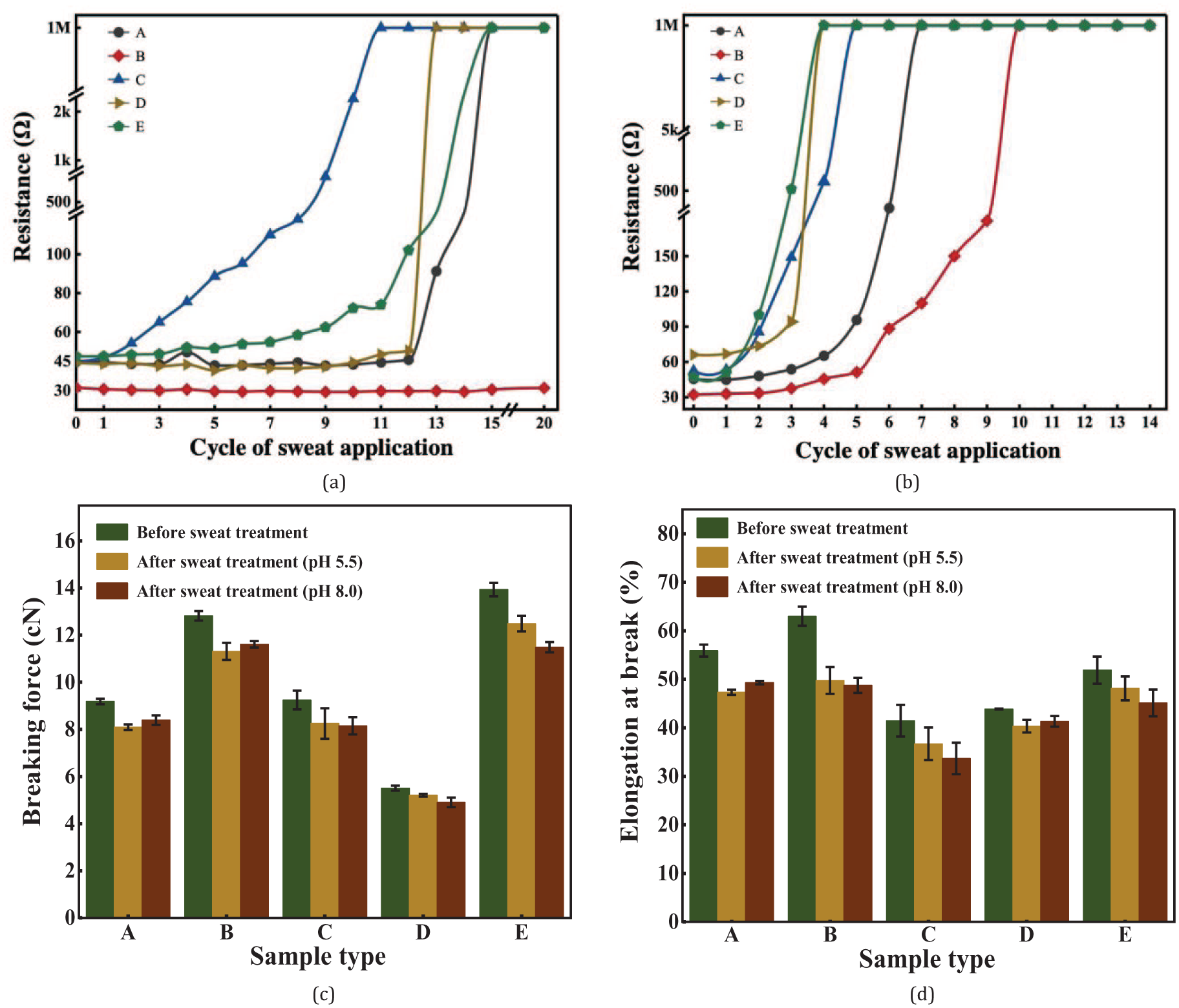
Sweat resistance of samples as shown in electromechanical changes. (a) Resistance changes to acidic sweat; (b) Resistance changes to alkaline sweat; (c) Tensile fracture strength before and after sweat treatment; (d) Tensile elongation at break before and after sweat treatment.
The inorganic salt in sweat underwent electrochemical corrosion with the plated Ag to form AgCl particles. After prolonged exposure to sweat, the surface particles fell off. Once the stable coating on the surface of the silver-plated yarn was destroyed, sweat quickly penetrated into the interior of the yarn, causing the destruction process to accelerate. When a certain part of the coating surface was severely corroded, the resistance increased there, the conductive path of the silver-plated yarn was interrupted, and the resistance therefore increased abruptly. Comparing Figure 6(a) and (b), it can be seen that alkaline sweat was more corrosive to silver-plated yarns. In alkaline sweat, the five types of yarns completely lost their conductivity after a minimum of three test cycles and a maximum of 10 test cycles. In acidic sweat, it took a minimum of 11 test cycles to completely lose conductivity, and even yarn B maintained normal conductivity after 20 cycles. These results are consistent with the work of Goduljan et al., 33 which showed that Ag undergoes redox reactions more easily in alkaline solutions than in acidic solutions. Therefore, the corrosion rate of plated Ag in silver-plated yarns was faster in alkaline sweat, and conductivity dropped more quickly.
Mechanical property
The mechanical properties of five types of silver-plated yarns before and after sweat treatment are shown in Figure 6(c) and (d). After sweat treatment, the yarns lost their electrical conductivity, and the breaking strength and elongation at break both decreased compared to the initial values. According to Figure 6(c), yarn B maintained a constant resistance in acidic sweat, but its mechanical properties deteriorated. Therefore, the silver plating on the surface layer has little effect on the mechanical properties of the yarn, and the mechanical properties of the yarn are mainly determined by the polymer fiber. In this regard, combined with Figure 2, it can be seen that the main reason for the deterioration of the mechanical properties of silver-plated yarns is the swelling behavior of the inorganic salt in sweat with the yarn primary material, which is nylon 6, resulting in a decrease in the mechanical properties of the yarn.
As seen from the above results, samples from these currently available five types of silver-plated yarns cannot meet the actual wearing requirements. To enhance the sweat resistance of silver-plated nylon yarns without compromising electrical conductivity or obstructing the conductive surface, a multifaceted approach is essential. Ultrathin conductive coatings, such as graphene oxide (GO) or polydopamine (PDA), can be applied via layer-by-layer assembly to shield silver from corrosive sweat components while maintaining partial conductivity. Self-assembled monolayers (SAMs) of alkanethiols or fluorinated compounds offer molecular-scale protection, reducing oxidation and sweat interaction without significantly increasing resistance. Co-depositing silver with corrosion-resistant metals like gold or palladium forms stable alloys (e.g. Ag-Au) that resist sulfide/chloride attack while preserving high conductivity. Hydrophobic surface modifications, achieved through fluorinated silanes or plasma-enhanced chemical vapor deposition (PECVD), create nanostructures that repel sweat and minimize ion penetration. For optimal performance, coatings must be uniform and pinhole-free to prevent localized corrosion. Hybrid strategies combining SAMs, alloy plating, and hydrophobic treatments can provide synergistic protection. Rigorous accelerated sweat aging tests are crucial to validate long-term durability.
Conclusion
In order to probe the interaction between silver-plated yarns and human perspiration during daily wear of smart textiles, this paper proposed a simulated wear test scheme for sweat resistance evaluation. According to this scheme, we tested five types of commercially available silver-plated yarns. Type A, B, and D showed good electrical conductivity and uniformity in linear resistance, hence they were more suitable for use in smart textiles. The former can ensure higher performance in electrical signal transmission, while the latter can improve device stability and repeatability. However, there are significant differences in the sweat resistance performance of the five silver-plated yarns, with the alkaline lasting for a minimum of three cycles and the acidic lasting for a minimum of 11 cycles. Among them, type B yarn demonstrated better sweat resistance performance in acidic perspiration, while type E yarn performed poorly. Type B yarn performed best could be due to its higher silver content providing a robust coating and superior mechanical properties. Type E yarn fared worst probably because of its lower silver content, making it vulnerable to sweat. Combining the experimental data from both acidic and alkaline artificial sweat solutions, the ranking of the five yarns’ sweat resistance performance is as follows: B > A > D > C > E. After treatment with artificial sweat, the silver-plated layer on the surface of the yarn peeled off. Meanwhile, the yarns lost its luster and became dull, and there were obvious white particles attached onto the surface of the yarn. The chemical reaction between alkaline artificial perspiration and the silver-plated layer of silver-plated yarn was obviously stronger than that of acidic artificial perspiration, as seen from changes in appearance, resistance, and mechanical property.
In this study, the best-performing yarn withstood only three cycles of sweat treatment before failure, and while Yarn B maintained stable electrical resistance after 20 cycles of acidic perspiration testing, its mechanical properties deteriorated markedly. These findings demonstrate that none of the tested samples meet the practical requirements for long-term wear in smart textile applications. The results revealed significant limitations in their performance. Therefore, we recommend that, enhancing sweat resistance of silver-plated nylon yarns requires a multifaceted approach, integrating ultrathin conductive coatings, SAMs, corrosion-resistant alloys, and hydrophobic modifications, with uniform application and rigorous aging tests to ensure long-term durability and conductivity.
Key limitations of this study include the inability to analyze chemical degradation mechanisms, particularly the effective probing of the formation of AgCl, which may contribute to conductivity loss. Additionally, all samples were sourced from a single yarn reel per type, precluding batch-to-batch variability analysis. The study also did not account for mechanical damage caused by machine washing, a critical factor in real-world usage. Therefore, future research should prioritize the development of advanced fiber materials with enhanced sweat resistance and conductivity stability. Expanding the scope to include more brands, models, and production batches of silver-plated yarns, along with rigorous testing of washing-induced degradation, will be essential to address current gaps and improve the reliability of smart textiles for practical applications.
Footnotes
Acknowledgements
The authors acknowledge the experimental support from Mr. Zhiping Xu and Ms. Huiqiong Zhu.
Authors’ note
Both ZS and YL are postgraduate students. HY is a full professor, and FW is a lecturer.
Author contributions
ZS designed the experiments, performed the experiments, collected the data, interpreted the results, and drafted the manuscript. YL interpreted the results, and drafted the manuscript. HY reviewed the experimental results, and contributed to manuscript preparation. FW conceived the ideas, supervised the experimental design, reviewed the experimental results, and contributed to manuscript preparation.
All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is partly supported by the Guangdong/Hong Kong Joint Foundation of Wuyi University (No. 2019WGALH11), the Foundation of Higher Education of Guangdong, China (No. 2023KCXTD040), and the High-level Talent Start-up Scheme of Wuyi University (No. 2021AL034).
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
