Abstract
This study suggests solutions to improve the adhesive of textile electrodes and the body signal occasion such as surface electromyography (sEMG). For this, a textile electrode embedded in the forearm band was produced using a knitting technique with two designs. The effects of skintight conditions on the forearm band during sEMG measurement were evaluated according to the pattern reduction rate (PRR) of the forearm band, ranging from 0% to 30%. Finally, an adhesive intermediate electrode, conductive gel, between the skin and the textile electrode was introduced to improve the unstable skin contact. The sEMG measurements confirmed that the sEMG signal was detected stably without excessive skintight conditions, such as a more than 20% PRR by applying the adhesive intermediate electrode. Skin-electrode impedance evaluation results also showed a similar tendency. Therefore, the knitted electrode developed in this study uses an adhesive intermediate electrode to improve the electrode performance and provide comfortable wearing conditions. This electrode is a practical electrode substrate for bio signal monitoring that allows the detection of muscle activity in a comfortable wearing condition for a long time.
Introduction
The main study areas of smart textiles encompass the electronics industry, communication systems, military applications, the entertainment sector, and the health industry. 1 Within healthcare systems, wearable smart textiles offer ease of use while ensuring help, diagnosis, rehabilitation, and therapy. 2 Furthermore, they present an appealing method for monitoring physiological characteristics and vital parameters of the human body, spanning from sports to healthcare, rehabilitation, and self-diagnosis. 3 With significant advancements in bio textile systems, real-time monitoring of bio signals has become feasible. In signal monitoring, electrodes are used to measure the body functions, such as electrocardiogram (ECG) and electromyography (EMG) or breathing rate, as well as bio impedance or body temperature. 1 Electrodes fabricated on flexible textile substrates represent a revolutionary breakthrough in wearable health monitoring due to their lightweight nature, breathability, comfort, and ability to conform to the body’s contours. Many studies have focused on flexible textile electrodes to seamlessly integrate bio-potential monitoring into everyday garments, ensuring non-intrusive monitoring with the comfort, and flexibility to conform to the curvilinear body shape.4,5 Textile electrodes can measure the myoelectric potentials of the skeletal muscles using a recording technique known as electromyography (EMG) when muscles are activated. EMG is typically measured using invasive and noninvasive methods. When EMG is measured using a noninvasive method, measurements were conducted on the skin surface using a disposable wet silver/silver chloride (Ag/AgCl) electrode because it is almost non-polarizable, particularly for low frequencies. Generally, wet electrodes comprise a gel layer to reduce the skin-electrode contact impedance and adhesive padding to improve contact with the skin. Although wet Ag/AgCl electrodes can provide a high quality signal, they face several challenges when considering their practical applications, especially during long term monitoring. The signal quality decreases over time, and the cable connecting the pad to the amplifier is a recurrent source of movement artifacts.6,7 In addition, because these disposable electrodes cannot be reused, the inconvenience and environment of having to be replaced every time after use is a disadvantage. In addition, the adhesive layer causes added discomfort while wearing the electrode and at the time of removal because of the need for mechanical peeling for electrode detachment. The requirement of skin preparation can also involve abrasion of the outer skin layer. Textile electrodes embedded in garments, which do not require the careful placement of sensors or wires and appear to allow unrestricted movement, might be a solution to the limitations of traditional wet electrodes. Textile electrodes cover a larger area of the muscle-to-surface EMG (sEMG).8–10 However, they have a few drawbacks, such as an intermittent adhesion between the electrode surface and skin that can cause fluctuations in electrical resistivity.11,12 Textile electrodes are difficult to adhere to the skin because of their unique characteristics. This becomes a factor causing issues such as weak signal pick up, low signal-to-noise ratio, high noise, or motion artifacts during sEMG measurements.5,13 Thus, when measuring sEMG, it is inevitable to wear the textile electrodes tightly to ensure adherence to the skin. This restricts the wearer’s free movement and reduces the wearer comfort during prolonged measurements. Therefore, it is necessary to develop a textile electrode that can measure sEMG while wearing it with a proper skintight condition and comfort similar to that of general clothing. Various techniques exist to incorporate conductive materials into a garment, including weaving, embroidering, printing of conductive inks, and attachment of conductive sheets to the textile and knitting of conductive yarns. 11 Of these well-established techniques, knitting has desirable characteristics critical for wearable textile electrodes, such as their stretchability, softness, permeability, and flexibility arising from the interlocking loops. Hence, an area of connected conductive yarns is realized, and electrical current can flow within a yarn and from yarn to yarn. Moreover, the loops allow the fabric to stretch horizontally and vertically, providing easy movement and a comfortable fit.13,14 This stretchiness is advantageous for garments needing to accommodate body movement. Hence, textile electrodes produced by knitting (here in after called knitted electrode) can adjust to the curvature of the human body while being comfortable with the skin, and intimate skin-electrode contact is enabled because the highly stretchable and good stretch recovery of the knitted structure meets the requirements of bio signal monitoring. 12 Moreover, when using knitting as a manufacturing technique, there is the opportunity to seamlessly integrate electrodes into a garment to produce wearables, meaning that only one production step is required, and almost no waste is produced. 1 These advantages make knitted electrodes a suitable substrate for fabricating wearable textile electrodes integrated in clothing for bio signal monitoring, providing advantages for user comfort in terms of freedom of movement. 1
Therefore, this study aims to develop the knitted electrodes were fabricated on a knit substrate by knitting for sEMG measurement. In addition, this work introduces an adhesive intermediate electrode (here in after, AI electrode) such as commercial conductive hydrogel between the skin and the knitted electrode to improve the efficiency of the textile electrode. It is most important to find a method of maintaining stable contact between the electrodes and the skin surface by minimizing intermittent contact. The conductive yarns are then filled with AI electrode, acting as an interface between the skin and the exposed knitted electrodes. The significance of this study is that it has the advantages of a wet electrode and the advantages of the existing textile electrode by introducing the AI electrode into the textile electrode. As a result, the freedom of movement and a comfortable fit can be given to the wearer while measuring bio signals. The performance of textile electrodes can be improved by applying adhesion to the electrode to enhance skin-electrode contact.
For this, first, the knitted electrodes were fabricated by intarsia technique using an automated flat knitting machine to separate the exposed surfaces of the electrode and transmission line part. Intarsia knitting is the insertion of other yarn segments into the fabric to form “patterns,” such as polka dots and stripes. Hence, knit capacitive materials can be knitted within the textile during manufacture, as with any other conductive yarns. 14 The design of knitted electrodes was optimized by designing two versions: single-faced electrodes and double-faced electrodes embedded in the forearm band. Depending on the design, the difference in the electrode performance was evaluated by sEMG measurements and skin-electrode impedance. Second, the effect of skintight conditions according to the pattern reduction rate (PRR) when wearing the forearm band on electrode performance was evaluated by measuring the sEMG signal and skin-electrode impedance. Finally, the AI electrode such as commercial conductive gel was applied onto the surface of the knitted electrode to improve adhesion with skin, and the resulting sEMG signal measurement performance was evaluated. In addition, the reusability of knitted electrodes was evaluated by measuring the electrode performance after washing five times. These results can serve as a database to expand the utility of knitted electrodes for wearable and reusable smart clothing and provide valuable guidelines for practical sEMG monitoring applications.
Experimental details
Fabrication of knitted electrode
A forearm band was fabricated with an embedded array of two knitted electrodes with the transmission line. The knitted electrodes were used to record sEMG signals continuously from the biceps brachii of the forearm. The forearm band with knitted electrodes embedded was produced using a 14-gauge flatbed knitting machine (CMS 330 HPW TT SPORT, STOLL, Germany) with STOLL M1 Plus 7.0.038 software. The part of electrodes and transmission line was knitted by four-ply conductive yarn from the AMANN group (Silver-tech, AMANN group, Bönnigheim, Germany), which has a 22 tex and resistivity of 530 Ω/m. The surrounding fabric of two electrodes had a single jersey structure made from nylon (150 denier) and spandex (70 denier) yarns to provide and maintain consistent conformal contact with the skin. A single jersey called a plain stitch is recognized by its flat, uniform appearance, having interlocking “V” shapes on the front and crescent on the back. 15 Both band edges were knitted by a double jersey structure with a two-ply of non-conductive yarn (cotton, CM 40s). A double jersey, also known as double knit, is a type of knitting technique that produces a fabric with two layers of interlocking stitches. Unlike a single jersey, with a single layer of stitches, a double jersey is more stable and maintains its shape. In this study, two types of forearm bands were fabricated by changing the design of electrodes as single-faced (Figure 1(a) and (c)) and double-faced (Figure 1(b) and (d)). Among the knitting techniques, the intarsia method was chosen for knitting the electrode and transmission line. Intarsia knitting can produce separate designs using different yarn feeders for each section within a row or round. The transmission line area of both samples was knitted in the same cross-section to prevent contact with the skin. The band of sample 1 was knitted to be a single-faced electrode, where the external (or outer) side was non-conductive to insulate the electrodes in the internal (or inner) side from the ambient environment. For sample 2, which has a double-faced electrode by conductive yarn, the electrode areas with the electric path were knitted using the intarsia method; the conductive yarns of the electrodes were exposed on both sides, as shown in Figure 1(b). Other surfaces of the conductive part were all covered with a thermoplastic polyurethane (TPU) film to prevent sEMG contamination by unintended signals. 16 The snap connectors were sewn with the conductive silver-plated yarn in the internal (or inner) side of the transmission line so that it could transmit the sEMG signals. Figure 1 presents a prototype of two knitted electrodes.

The pictures of sample 1 (a) and 2 (b), the front, back side of sample 1; single-faced electrode (c), and the front, back side of sample 2; double-faced electrode (d).
Design of forearm band
The forearm band was designed with a width and length of 32 cm and 9 cm, respectively, but the width was changed according to the pattern reduction rate (PRR), as shown in Figure 2. The band was fastened by velcro on the edges so it could be fitted securely to the forearm of each wearer. The electrode part was positioned over the anterior forearm. The inter-electrode spacing is defined as the center-to-center distance between the conductive area of the two electrodes. The internal (or inner) area has two knitted electrodes equally spaced according to the circumference of the band. The width and height of the conductive area were approximately 25 mm × 30 mm (rectangular) (usual shrinkage occurred after knitting and steaming; therefore, the areas varied slightly) in the mediolateral and longitudinal directions, respectively, and the inter-electrode distance (IED) was 40 mm according to the SENIAM (surface EMG for the noninvasive assessment of muscles) and previous studies.15,16 The transmission line area of each electrode was set to 30 mm × 10 mm. The line was knitted in connection with the electrode area but was designed to avoid contact with the skin by being exposed to the front of the forearm band.
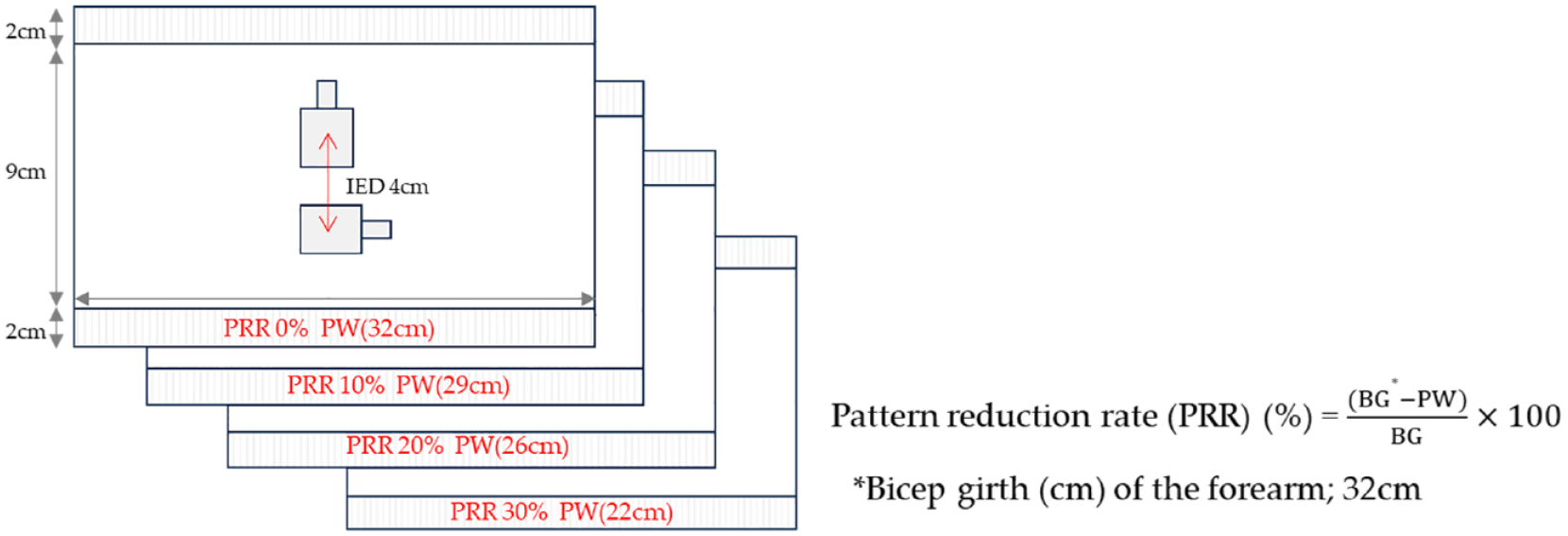
Design of the knitted electrode integrated in forearm band of different PRR (%).
Characterization
Surface resistance
The surface resistance of the knitted electrodes according to the different designs was examined using a four-point probe conductivity meter (CMT-100 Series, AIT Co., Ltd. Korea). The front and back sides of each sample were measured separately before and after introducing an AI electrode. Due to the inherent characteristics of textiles, the surface of the electrode is not smooth, and yarn protrusions are formed by knit stitches so that all samples were measured 10 times, and the average value was used.
sEMG measurement methods
The knitted electrodes were validated by comparing the sEMG signals detected simultaneously from the same muscle using disposable Ag/AgCl electrodes (Kendall LTP, Covidien, MA, USA) and knitted electrodes. The performance of the knitted electrodes in detecting sEMG signals was validated by analyzing the sEMG signal characteristics with three variables: the electrode knitted designs, PRR, and AI electrode. The sEMG signal was collected from the dominant forearm of one participant (male, 24 years old) to reduce the variations arising from individual differences in body impedance during forearm movements. Two knitted electrodes of the same size (25 mm × 30 mm) were placed 20 mm apart (center-to-center) and positioned on the biceps brachii long head according to the SENIAM recommendations.17,18 A ground electrode, Ag/AgCl electrodes (Kendall LTP, Covidien, MA, USA), was positioned at the dorsal surface of the elbow joint, the electrically neutral bony prominence. In this study, skin preparation before testing (i.e., hair removal, abrasion of the skin, and cleaning with alcohol wipes was not conducted, considering that people would not implement skin preparation when wearing sEMG suits in practical use. 19 The electrode system was connected to a physiological signal monitoring wireless device (iWireR-ECG:H21027, iXorx Systems Inc., USA). The obtained signals were delivered to the amplifier (IX-TA-ROAM Data Recorder, iXorx Systems Inc., USA) using LabScribe 23 version software and filtered in the range of 30 to 500 Hz for the EMG measurements. A notch filter of 60 Hz was also applied to minimize the recording noise. The data were full-wave rectified and averaged with a 100 ms time constant to draw the amplitude of the signals.16,20 The knitted electrode performance was evaluated using sEMG measurements in three test series. In series 1, the influence of skintight conditions on the arm by tightly wearing the forearm band, referred to as the different sizes of the forearm band, and then sEMG was measured depending on the different PRR values. In series 2, the effect of AI electrode applied to the knitted electrode surface was evaluated by measuring sEMG signals. The AI electrode used in this study is a commercial conductive hydrogel purchased from Parker (Signagel®, U.S.A.). In series 3, after using the knitted electrode with AI electrode, it was washed and dried five times to show the reusability of the knitted electrode by measuring electrode performance.
Series 1: Influence of pattern reduction rate (PRR)
The effects of PRR on sEMG measurement were tested by preparing the forearm band in different PRRs from 0% to 30%, as shown in Figure 2. The PRR was applied only in the width reduction of the forearm band, not in the length reduction. Each proximal width (PW) of the forearm band samples was calculated using equation (1):
where BG is the bicep girth (cm) of the forearm (Figure 3). According to equation (1), the forearm band of PRR 0% covered the arm without any tightness on the arm. In all samples for testing the influence of PRR, the other electric configurations were identical when testing the electrode sizes.

Illustrate of the measuring process and the measuring posture for the sEMG signal of biceps brachii.
Series 2: Influence of adhesive intermediate electrode (AI electrode)
The AI electrode was applied in this study to improve the adhesiveness between the electrodes and skin without high tightness against the body during wearing. The effects of the AI electrode between the electrode and skin were evaluated by applying 0.8 g of AI electrode onto the two knitted electrodes surface in the forearm. After applying AI electrode to the electrode surface, it was maintained for 5 min to allow even penetration of AI electrode into the surface. Afterward, it was worn on the participant’s forearm to measure sEMG.
Series 3: Reusability of knitted electrode
Given that the objective of this work was to present a scalable and reusable solution for sEMG-based control of the knitted electrode, the reusability of the knitted electrode was investigated after washing and drying five times to test the performance consistency. The washing procedure was performed according to the standard ISO 6330:2012. The samples were washed for 15 ± 0.5 min in 40 ± 2°C temperature washing solution with 3 g/l washing powder concentration as followed previous study. 21 After the knitted electrode was washed five times, it was dried for 60 min at room temperature. The AI electrode was applied to the dried knitted electrode, and the electrode performance was then evaluated and compared using the SNR of the sEMG signal.
Data processing and experimental protocol
The root-mean-square (RMS) operations were performed for sEMG signal data processing. The value was calculated to validate the utility of the sEMG signals. Before testing muscle flexion, the baseline sEMG signals were collected in the muscle relaxation for 10 s to measure the baseline electrode noise. During muscle flexion, the sEMG signals were calculated by averaging 2 s during 5 s of the activated phase. The signal-to-noise ratio (SNR) of the sEMG signal is defined as the RMS of the signal during EMG activity (during muscle flexion) divided by the RMS of the EMG activity (during muscle extension), as expressed in equation (2):
Figure 3 shows the testing posture for measuring the sEMG signal by knitted electrodes embedded in the forearm band. First, the baseline sEMG signals were collected in the arm relaxation position for 10 s to measure the baseline electrode noise. The bicep curls movements were conducted using a dumbbell (2 kg) to induce the sEMG signal by muscle flexion. The elbow was bent approximately 90° to lift the dumbbell (muscle extension) and return to the natural straight arm position (muscle flexion). These movements were conducted continuously by five trials for forearm extension and flexion. At that time, each phase lasted 5 s. This study also tested one subject; the individual differences in muscle activities were not considered. The experiment was conducted at room temperature under 60% relative humidity (RH). The test movement was conducted five times to record the sEMG signal from one subject during intermittent muscle flexion measurements. Except for the first and last trials, three flexions from five were used to calculate the average activated sEMG for comparison.
Skin-electrode impedance
The skin-electrode impedance was measured using AI electrode and the degree of PRR for measuring the biceps brachii muscle sEMG. A disposable Ag/AgCl electrode was used as the reference electrode, and the forearm band was worn so that two knitted electrodes could be located on the biceps brachii muscle. The knitted electrode was measured by attaching a disposable electrode to the electrode surface to facilitate a connection with a skin electrode-impedance analyzer (ZIVE P2 ELECTROCHEMICAL WORKSTATION, Won-A tech. Co. Ltd., Seoul, Korea).19,22 The skin-electrode contact impedance was measured using an AC sinusoidal signal at the 1–100 kHz frequency range. 23 Two designs of knitted electrodes were used for the test: sample 1) a single-faced electrode sample knitted with electrodes only on the surface in contact with the skin, and sample 2) a double-faced electrode sample knitted with electrodes on both the surface and surface in contact with the skin. The degree of PRR of the band was changed to 0%, 10%, 20%, and 30%, and the use of AI electrode on the knitted electrode area was varied and measured three times. The skin-electrode impedance at 100 Hz, targeting the peak frequency component for EMG, 24 was analyzed to compare the electrode fabricated by varying the measuring conditions.
Results and discussion
Sheet resistance
The thickness of the two samples was 1.8 ± 0.2 mm (sample 1) and 1.12 ± 0.05 mm (sample 2), respectively. Sample 1 was thicker than sample 2 because sample 1 was knitted in two layers with different yarns, such as conductive yarn and nylon, using an intarsia technique. This knitting technique also affects the sheet resistance. In the case of sample 1, the sheet resistance was 0.14 ± 0.05 Ω/sq as the conductive yarns were exposed on the back side of the electrode. In contrast, the sheet resistance value was not measured on the front side of the electrode area because the conductive yarns were inlayed inside the non-conductive yarns and were not exposed. In the case of sample 1, conductive and non-conductive yarns are supplied simultaneously and knitted using the intarsia technique, so the contact point between the conductive yarns was less than that of sample 2. Hence, sample 1, which was designed as single-faced electrodes, prevents unstable signal collection because of the external contact that may occur when worn and can enable stable signal collection even when worn for a long time during the sEMG measurement. On the other hand, in the case of sample 2, which was designed as a double-faced electrode, the sheet resistance was 0.42 ± 0.12 Ω/sq (front side) and 0.35 ± 0.04 Ω/sq (back side) which were measured to be higher than that of sample 1. The conductive yarns were exposed on both sides, increasing the number of conductive yarn stitches between loops in both the course and wale directions, resulting in increased sheet resistance. After applying AI electrode to the textile electrode surface of sample 1, the sheet resistance changed to 0.09 ± 0.03 Ω/sq, and the front side of the electrode was not also measured, even though AI electrode was introduced. In the case of sample 2, the sheet resistance of the front side was slightly changed to 0.28 ± 0.17 Ω/sq, whereas the back side was retained same value. The stitch density of the two samples was similar. The stitch density is the number of stitches per unit area, obtained by multiplying the number of courses per inch by the number of wales per inch. The stitch densities were 600 (sample 1) and 540 (sample 2) (Table 1).
Thickness, stitch density, and sheet resistance of knitted electrodes with two samples.

sEMG measurement
Figures 4 and 5 show the raw sEMG signals of commercial Ag/AgCI electrode and sample 1 with or without AI electrode according to the change in PRR, respectively. The raw sEMG signal was intended to confirm the noise of each sample and the magnitude of the signal obtained during muscle activation. Three flexions from five, except for the first and last trials, were used to calculate the average activated sEMG for comparison. As shown in Figure 4, the baseline noise and active signal of the reference and commercial Ag/AgCI electrodes were 0.056 ± 0.01 mV and 2.83 ± 0.55 mV, respectively.
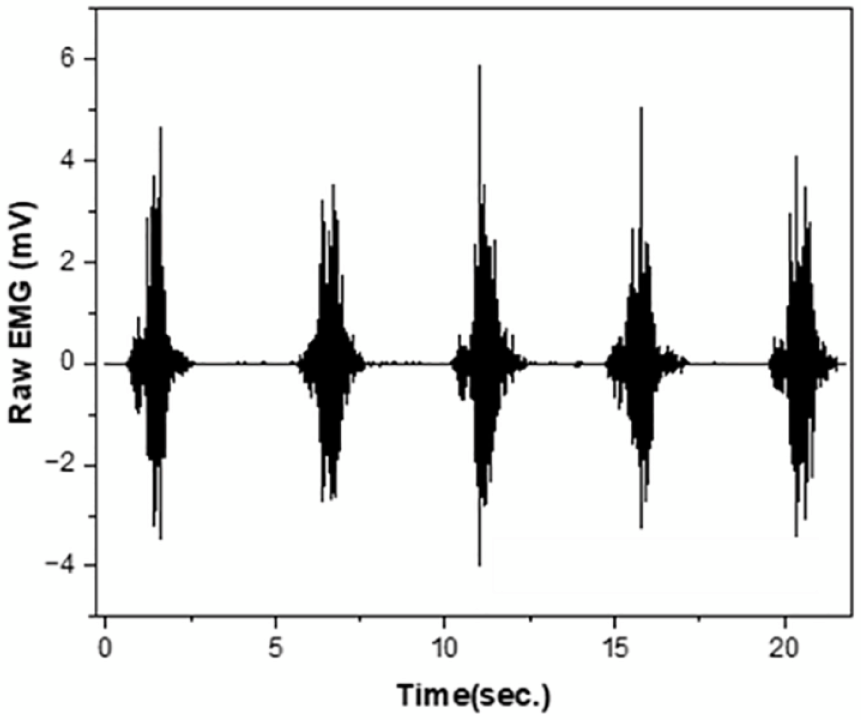
Raw sEMG signals retrieved from commercial Ag/AgCI electrode as reference electrode.

Raw sEMG signals retrieved from sample 1 without AI electrode (a–d) and with AI electrode (e–h) according to variation of PRR 0% to 30% of forearm band.
First, the sEMG signal of sample 1 showed significant differences according to the increase in PRR and the application of AI electrode. In the absence of AI electrode, the baseline noise was 0.37 ± 0.06 mV, 0.17 ± 0.04 mV, 0.37 ± 0.26 mV, and 0.14 ± 0.11 mV, and the active signal was 3.32 ± 0.23 mV, 1.3 ± 0.1 mV, 1.05 ± 0.22 mV, and 1.43 ± 0.11 mV along the PRR of 0%, 10%, 20%, and 30%, respectively (Figure 5 (a)–(d)). After AI electrode was applied to the electrode surface of sample 1, the baseline noise was 0.18 ± 0.05 mV, 0.014 ± 0.04 mV, 0.017 ± 0.05 mV, and 0.01 ± 0.004 mV and the active signal was 3.15 ± 0.24 mV, 2.2 ± 0.56 mV, 1.96 ± 0.2 mV, and 1.15 ± 0.1 mV, at 0%, 10%, 20%, and 30%, respectively, as shown in Figure 5(e) to (h). The sEMG signals during muscle flexion increased as the PRR was increased. After applying AI electrode, the highest active signal (2.2 ± 0.56 mV) and low baseline noise (0.014 ± 0.04 mV close to 0) was obtained at a PRR of 10%, which is similar to the commercial Ag/AgCI electrode. Regardless of whether AI electrode was used, the highest active signal and the largest noise signal were indicated in sample 1 with 0% PRR. This was determined by measuring the unstable and irregular signals, as shown in Figure 5(a). In the case of knitted electrodes, the signal collection is unstable when worn loosely because of the unstable contact between the electrode and the skin surface. This result shows that sEMG signal collection is possible only when the electrode is in close contact with the skin at proper skintight conditions.
Figure 6 shows the sEMG signal analysis results for sample 1 after filtering raw data and converting it to the root mean square (RMS), the calculated mean and standard deviation of the three acquired signals. The SNR operations were performed using the following equation: SNR = RMSbaseine/RMSactivated, where RMSbaseline and RMSactivated represent the average RMS values of the sEMG signals measured at muscle extension and muscle flexion, respectively. The SNR of the reference electrode, the commercial Ag/AgCI electrode, was 26.86 ± 1.6. First, the SNRs of the sEMG signals measured from sample 1 were suggested in Figure 6(c). The SNR of sample 1 increased consistently and significantly as the PRR was increased during muscle extension. The results suggested that the SNR value for the sEMG signal was lower under the condition where the PRR was loose (fit loosely on the body). The knitted electrode performance was improved along the increase of PRR. The highest SNR value (12.24 ± 1.4) was obtained at PRR 30%. Thus, a decreasing tendency was observed in the baseline sEMG signals, and an increasing tendency was noted in the activated sEMG signals as the PRR was increased. Hence, the skintight condition is required when measuring the sEMG signal by the knitted electrode to maintain electrode surface contact with the skin. On the other hand, in the presence of AI electrode, the highest SNR value (25.2 ± 1.24) was obtained at a PRR of approximately 20%, but a higher degree of those caused slight decreases in the SNR value. Hence, AI electrode affects the increase in SNR value. These results revealed a higher signal than the SNR value of approximately 3.61 to 7.03 from a previous study that developed the knitted electrode. 25
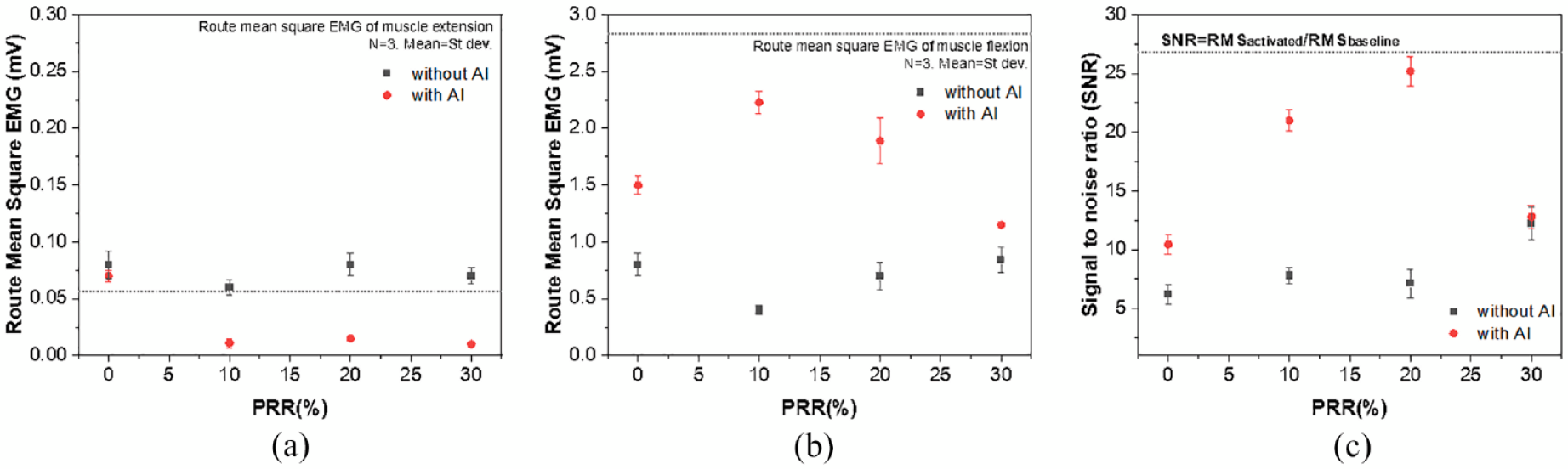
Average rectified sEMG of the baseline electrode noise during muscle extension (a); sEMG amplitude during muscle flexion (b) retrieved from sample1; (c) signal-to-noise ratio (SNR). The dashed lines indicate the values obtained from the commercial Ag/AgCI electrode. The values were calculated by averaging three consecutive measurements from three samples of sample 1.
Figure 7 shows the raw sEMG signals of sample 2 with or without AI electrode according to the change in the PRR. In the absence of AI electrode, the baseline noise of sample 2 was 0.11 ± 0.01 mV, 0.1 ± 0.03 mV, 0.05 ± 0.04 mV, and 0.02 ± 0.004 mV, and the active signal was 1.61 ± 0.15 mV, 1.45 ± 0.12 mV, 1.68 ± 0.04 mV, and 1.29 ± 0.15 mV along the PRR 0%, 10%, 20%, and 30%, respectively (Figure 7(a)–(d)), which is a similar result to sample 1. After AI electrode was applied to the electrode surface of sample 2, the sEMG signal changed as the baseline noise decreased. The baseline noise was 0.04 ± 0.01 mV, 0.02 ± 0.006 mV, 0.018 ± 0.035 mV, and 0.043 ± 0.027 mV at 0%, 10%, 20%, and 30%, respectively, and the active signal was 0.76 ± 0.09 mV, 1.71 ± 0.301 mV, 1.65 ± 0.035 mV, and 1.09 ± 0.096 mV at 0%, 10%, 20% and 30%, respectively.

Raw sEMG signals retrieved from sample 2 without AI electrode (a–d) and with AI electrode (e–h) according to variation of PRR 0% to 30% of the forearm band.
Figure 8(c) shows that the SNR value increased as the PRR increased. The highest value of SNR (16.58 ± 0.97) was evaluated at a PRR of 30% in the absence of AI electrode. Sample 2 showed better performance with PRR > 20% because the knitted electrode of the PRR 30% is in close contact with the skin and has sEMG signal acquisition performance. After AI electrode was applied, the highest SNR value of 19.36 ± 0.2 was observed at PRR = 10%, while a slight decrease was shown at PRR = 30%, showing a similar tendency to sample 1. Accordingly, (1) the electrode design had no significant effect on the electrode performance; (2) both knitted electrodes required moderate skintight conditions when wearing a forearm band that is in close contact with the skin and has sEMG signal acquisition performance. Hence, the application of AI electrode support is similar to that of the commercial Ag/AgCI electrode without heavy tightening fitting conditions on the body.

Average rectified sEMG of baseline electrode noise during muscle extension (a); sEMG amplitude during muscle flexion (b) retrieved from sample 2; (c) signal-to-noise ratio (SNR). The dashed lines indicate the values obtained from the commercial Ag/AgCI electrode. The values were calculated by averaging three consecutive measurements from three samples of sample 2.
Skin-Electrode Impedance
When acquiring bio signals using electrodes, accurate sEMG signal acquisition performance can be confirmed according to the degree of contact between the skin and the electrodes. Accordingly, the skin-electrode impedance of the knitted electrodes embedded in the forearm band for sEMG measurements was measured according to the degree of PRR, with/without AI electrode and by the different designs of the knitted electrodes, as listed in Table 2.
Skin-electrode impedance value of knitted electrodes according to use AI electrode and degree of PRR of forearm band.
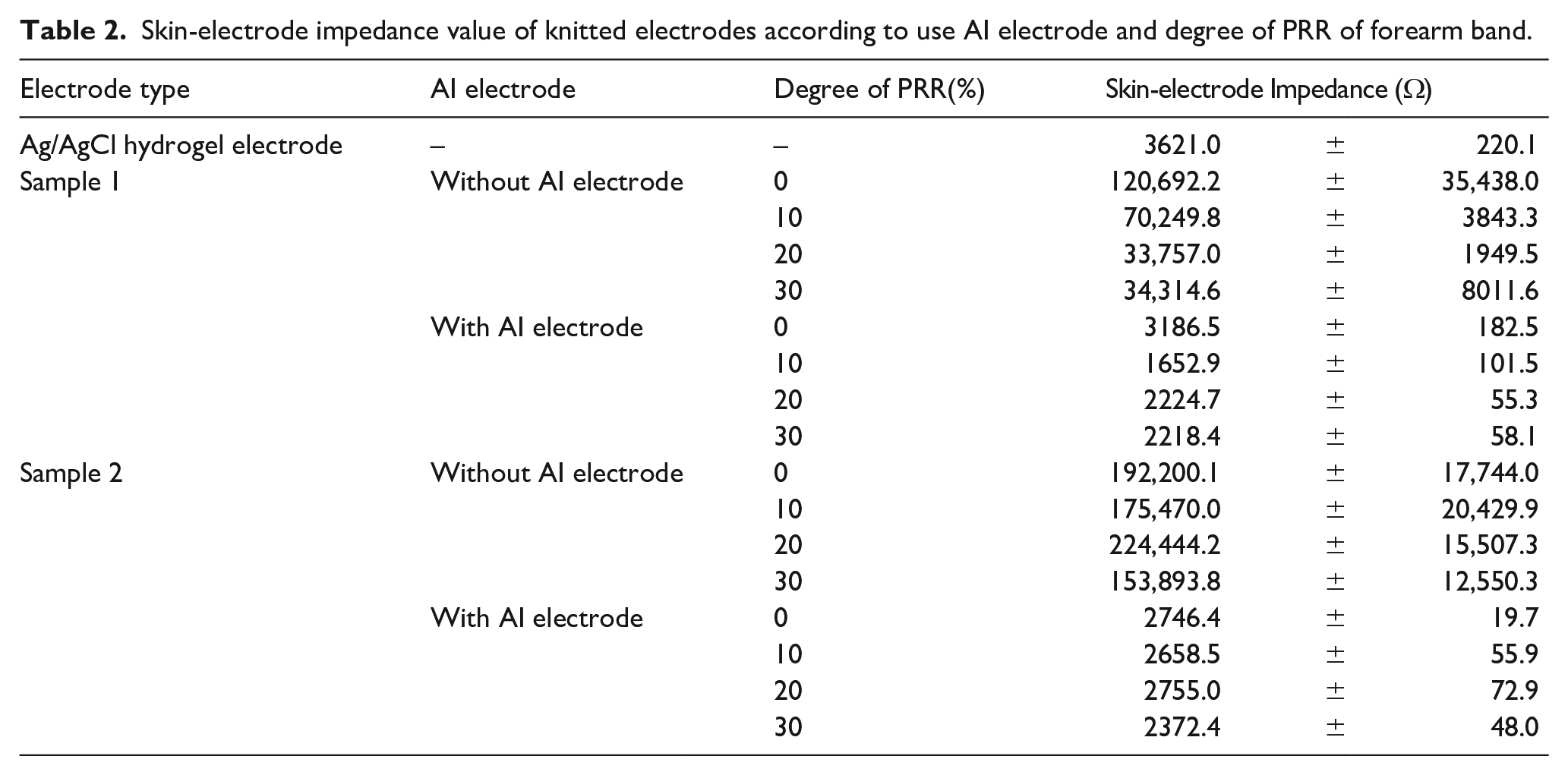
The skin-electrode impedance by the designs of knitted electrodes at PRR = 0% increased in the order of sample 2 (with AI electrode) < sample 1 (with AI electrode) < Ag/AgCl hydrogel electrode < sample 1 (without AI electrode) < sample 2 (without AI electrode). For each type of electrode, in the case of samples 1 and 2, to which no AI electrode was applied, the impedance value was more than 40 times higher than that of the Ag/AgCl hydrogel electrode, which is a wet electrode. Hence, the adhesion to the skin of the dry electrode is not excellent compared to the Ag/AgCl hydrogel electrode. On the other hand, the two samples of knitted electrodes to which the AI electrode was applied were at least 10–80 times different from the sample without it when in contact with the skin. In addition, compared with the conventionally used Ag/AgCl hydrogel electrode, the impedance of sample 2 with AI electrode was approximately 2700 Ω, which was lower than that of the Ag/AgCl hydrogel electrode. The skin-electrode impedance was decreased by improving contact with the skin while making the dry electrode and skin surface wet by introducing AI electrode.22,26
As a result of skin-electrode impedance analysis of knitted electrodes for sEMG measurement according to the degree of PRR, the two samples of knitted electrodes without AI electrode showed a decrease in skin-electrode impedance as the degree of compression increased. In general, the contact area of an electrode is one of the most important factors in measuring the skin-electrode impedance. As the contact area increases, the electrode contact area with the skin also increases, so the skin-electrode impedance decreases.26,27 In the case of the knit, it has excellent stretchability because of its structural characteristics. Hence, the area can expand easily according to tension. Therefore, as the degree of PRR increases, and the knit stretches, the band and the gap between the loops forming the knitted electrodes located in the band also increase, confirming that the impedance value decreased. On the other hand, both knitted electrodes using AI electrode showed different results. After using AI electrode, both electrodes showed an overall skin-electrode impedance value of less than 3000 Ω regardless of the degree of compression, which was superior to the Ag/AgCl hydrogel electrode. Hence, when the AI electrode is applied to the knitted electrode, it improves the contact force between the textile and the skin. In addition, the conductivity is increased as it penetrates between the loops in the knitted electrode because it contains moisture. Therefore, by introducing AI electrode to the knitted electrode, the dry electrode and the electrical performance were superior to the existing disposable electrode.
Reusability of knitted electrode
The reusability of the knitted electrodes after several wash cycles was assessed using the forearm band. The knitted electrode of two samples was washed five times, and the sEMG measurement was conducted. AI electrode was applied to the knitted electrode surface after five washing cycles. As shown in Figure 9, two knitted electrodes were resistant to the repetitive wash cycle process. In the case of sample 1 wearing at PRR = 20%, which showed the highest SNR value, the SNR value was changed from 19.7 ± 1.6 to 16.3 ± 1.5 after five wash cycles. Sample 2 at PRR = 10% showed the highest SNR value, which changed from 18.7 ± 0.2 to 15.3 ± 2.3 after five wash cycles. Both samples showed an approximate 17% decrease. Hence, the sEMG signal SNR did not deteriorate after five wash cycles.
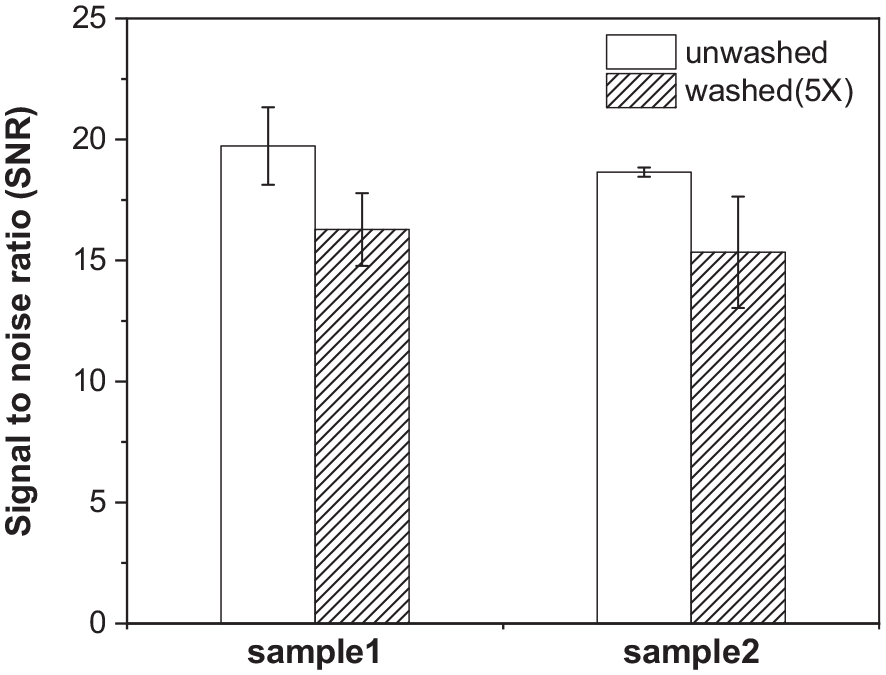
SNR value of unwashed and washed (5X) knitted electrode with AI electrode.
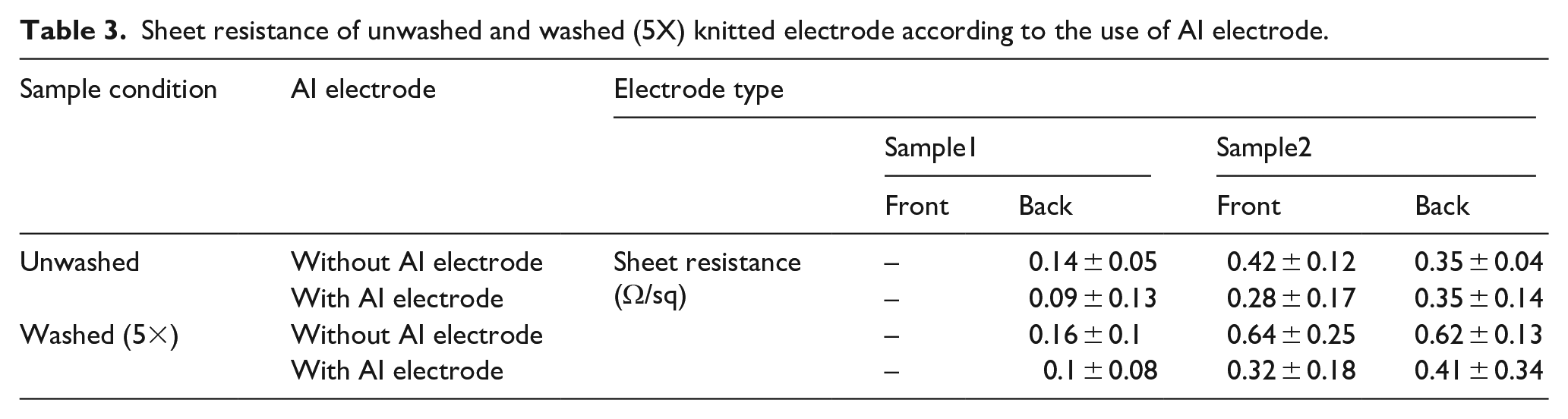
Finally, the sheet resistance was also measured after five wash cycles to characterize the durability of knitted electrodes with AI electrode to repetitive washing. The wash cycle led to a negligible change in sheet resistance for the two types of samples. After five wash cycles, the sheet resistance of sample 1 (back side) was approximately 0.2 ± 0.04 Ω/sq, as listed in Table 3. After applying AI electrode, the sheet resistance was 0.1 ± 0.02 Ω/sq, which remained after washing. The measured sheet resistance of sample 2 was 0.6 ± 0.07 Ω/sq (front side) and 0.6 ± 0.05 Ω/sq (back side). After applying AI electrode, it changed to 0.3 ± 0.1 Ω/sq (front side) and 0.4 ± 0.1Ω/sq (back side). The electrical properties of the knitted electrode were not adversely affected by repeated washing, and the application of AI electrode helped promote the durability of the knitted electrode. Hence, it has reusability, which is one of the critical advantages of textile electrodes. Therefore, the knitted electrode can be reusable with stable electrode performance in the presence of AI electrode owing to its better consistency, stability, and durability.
Sheet resistance of unwashed and washed (5X) knitted electrode according to the use of AI electrode.
Conclusions
In bio signal monitoring, dry textile electrodes generally introduce higher, unstable skin-electrode impedances, which might lead to noise and reduced signal quality. This study evaluated a method to improve sEMG signal quality by enhancing intermittent adhesion between textile electrodes and skin. For this, the AI electrode as electrode-skin interface was applied to the knitted electrode, and the sEMG measurement was conducted according to the designs of the knitted electrode and the degree of PRR, respectively. The following results were obtained. (1) The knitted electrode designs did not significantly affect electrode performance. In sample 2, however, which was knitted as double-faced by conductive yarns, the conductive yarns were exposed at both sides to contact the body occasionally during forearm movement. (2) Without AI electrode, the highest sEMG signal was obtained under the heavy skintight condition, such as PRR = 30%. On the other hand, after AI electrode was applied, the sEMG signal was collected under the loosely skintight condition, such as PRR = 10–20%. The recorded signals showed good quality and skin-electrode impedance after using AI electrode. 3) The reusability of the knitted electrode was demonstrated by a repetitive wash cycle. The knitted electrode with AI electrode retained its electrode performance after five wash cycles. Therefore, the electrical property of the knitted electrode was not loosened significantly by repeated wash cycles. Furthermore, the application of the AI electrode support promoted the durability of the knitted electrode; thus, it has reusability, which is one of the critical advantages of textile electrodes. Overall, knitted electrodes can be reusable with stable electrode performance owing to their better consistency, stability, and durability. This study showed that the knitted electrodes embedded in the forearm band provide stronger sEMG signals than the commercial Ag/AgCI electrode. The application of AI electrode as a textile electrode-skin interface improved its adhesion to the skin. As a result, the freedom of movement and a comfortable fit can be given to the wearer while measuring bio signals. In conclusion, the performance of textile electrodes can be improved by applying adhesion to the electrode to enhance the skin-electrode contact. Therefore, the proposed textile electrode with AI electrode is an attractive solution for long-term EMG monitoring and will be a subject of future studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) Funded by the Ministry of Education (No. NRF-2021R1G1A1012189) and the Korea Institute of Industrial Technology as “Development of microfactory-based technology for future smartwear manufacturing (kitech EH- 24-0006).” It was also supported by the Ministry of Education of the Republic of Korea and the National Research Foundation of Korea (No. NRF-2021R1I1A3057957).


