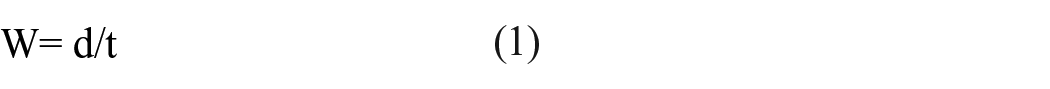Abstract
Antibacterial woven polyester fabrics are drawing much attention in the medical and textile industries because they can provide many health-promoting properties that prevent infection while providing a barrier to the external environment. In this work, we developed and investigated two different polyester fabrics to demonstrate the antibacterial, non-adhesive, and moisture wicking properties of a multicomponent wound dressing. Vertical wicking of a braided layer composed of Poly-l-lactide (PLLA) yarn of 4-deep-grooved (4DG) fibers was tested using porcine blood serum to mimic wound exudate viscosity. Bacterial inhibition of Pseudomonas putida was measured after exposure to gentamicin sulfate (GS)-incorporated PLLA skin-contact layers of two different shaped yarns. Poly-ethylene glycol (PEG) was added to the GS PLLA contact layer to further prevent non-specific cellular attachment. Results indicated vertical wicking of moisture at 0.88 mm/s. While a specific wicking rate for adequate wound moisture regulation has not been indicated in the literature due to wound and patient variability, we assert that rapid removal of excess fluid from the wound bed, as demonstrated by our fabric is an essential component for a comprehensive wound dressing. Antibacterial experiments showed that GS exhibited both bactericidal and bacteriostatic trends by killing bacteria and further inhibiting growth, respectively. The surface treatment of PEG on PLLA contact layers showed less cellular attachment, promoting non-adhesion during dressing changes. Ultimately, using woven polyester medical textiles in wound care allows for optimizing parameters, such as antimicrobial uptake/release and moisture wicking for personalized healing. The construct studied here may serve as a tunable platform for development of wound dressings with specific antibacterial, moisture, and tissue adhesion requirements.
Introduction
Antibacterial woven polyester fibers with demonstrated biocompatibility have shown significant advancements in the medical and textile industries because of the critical need to manage personal hygiene, disease prevention, and spread of infection. 1 Unlike natural materials, many synthetic polyesters create versatility in both fabrication and usage. 2 Polyester fibers that can be used to fabricate medical textiles degrade relatively slower, ultimately allowing them to withstand longer periods of use. 3 Many forms of biocompatible polyester fibers and fabrics provide the opportunity to incorporate antibiotics, antiseptics, and growth factors for drug delivery.4–6 Fabrics may be composed of fibers of different size and cross-sectional shape for modulation of drug release. 7 Additionally, textile fabrication methods such as weaving or knitting have the advantage of controlling the porosity, dimension, and strength of the fabric.
Traditionally, weaving is a fabrication method where linear materials are interlaced in perpendicular directions in multiple configurations to form an integrated structure. Previously, materials were only incorporated into the weft and warp directions which resulted in 2D fabrics that were strong, warmth-keeping, flexible, and drape-able. Weaving is also capable of creating 3D fabrics, by arranging warp yarns in a 3D form and inserting weft yarns at different levels in multiple directions. The advantages of these 3D fabrics are being able to create forms/shapes with end-use in mind, reducing waste when making these forms/shapes, and increased structural integrity. 8 These fabrics offer the strength, pore structure, and defined geometry necessary for medical applications such as scaffolds for tissue engineering, artificial skin grafts, and wound dressings. 9
Weaving usually results in more dimensionally stable and robust structures compared to knitting. Knitting is formed by creating loop stitches in rows that intermesh with the following and previous row to form tubular or flat fabrics. The knitted loops are capable of being extended in the X and Y direction, meaning knit fabrics are much more elastic than woven fabrics. More fibers are involved in the creation of knitted materials compared to any other textile technology, which enables knitting to create complex 2D and 3D structures. 9 The yarn material, knitting process, and types of stitches determine the mechanical and physical properties of the knitted fabric. Weft-knitted structures offer superior control over pore size, fiber alignment, and porosity of the construct compared to warp, while warp-knitted structures offer more flexibility compared to weft. 10 The extensibility of knitted fabrics makes them a great candidate for load-bearing applications, however, creating fabrics with adjustable properties in multiple directions is difficult with knitting. 10
Recent technology has allowed for the development of extruding fibers with unique cross- sections for the purpose of drug delivery performance, moisture wicking, comfort, bulkiness, and tactility.11–13 Some common shapes of fibers are round, scalloped oval, cruciform, lobular, trilobal, square with voids, star-shaped, and hexachannel. 14 One shape in particular, 4-deep-grooved (4DG) is unique because of its eight microchannels created by the protrusions from the main fiber body. 15 This shape was designed with deep grooves or channels along the longitudinal axis of the fiber. 16 The grooves provide channels that can serve as ducts to move fluid spontaneously and provide large surface areas (up to 300% that of round fibers) for a given denier per filament.
Therefore, the increase in surface area with respect to round fibers allows for an increase in drug loading, ultimately serving as a benefit for infection management. Additionally, 4DG fibers also contribute to moisture wicking due to microcapillary spaces that can displace liquid. 17 Excess moisture travels through the channels of the fiber and moves around the fabric by running along the weave. Ultimately, moisture reaches the outside of the fabric and evaporates into the air, enabling faster drying times for wicking fabrics due to limited absorption. 18 There are many factors that can alter the rate of wicking, such as fiber material, geometry, fiber count, twisting level, and tension. Wicking also occurs in-plane where there is a spreading of the liquid in the textile layer and through-plane where liquid is transported from the surface to the other side of the fabric. 17 Because of the 4 deep channels that the 4DG shape provides, a medical textile fabric made out of a multifilament yarn of 4DG cross-section may allow for ideal movement of viscous moisture for applications such as performance wear, transdermal patches, and more prominently wound dressings.
Wearable biomedical textiles
There are many examples of wearable textiles engineered for biomedical applications. These products need to meet the constraints of both a wearable fabric and a biomedical tool. For example, Cheung et al. created fabric using chitosan/cotton blends at varying concentrations (0/100, 5/95, 10/90) that can be used in apparel or accessories to maximize convenience and comfort while also promoting wound repair and inhibiting microbe growth. The yarns were spun via an an-static spinning approach and then incorporated into the fabric. The size of the wound treated with the highest concentration of chitosan was smallest after 10 days compared to the other groups. However, the strength of chitosan fibers may be insufficient in certain therapeutic applications. 19
Mihailovic et al. 20 created a weft-knitted polyamide/elastane compression material treated with gentamicin sulfate and Piceas abies to determine the effect of antimicrobial treatment on the strength-related properties of the fabric. Treatment with antimicrobial agents resulted in an increase in the stiffness, tensile strength, and bending modulus compared to untreated fabric samples, but the tensile modulus values were not affected. The authors found that the fabric was effective against many microorganisms, and when applied on human legs, resulted in a decrease in leg circumference. This study found that a compression material that can produce the desired pressure can be created by altering the production technique, changing structural parameters of the knitted fabric, and selecting appropriate antimicrobial treatment parameters.
Biomedical textile wound dressings – current state of art
Various papers have used blended yarns to create dressings that can be used in wound care. Ozel et al. created a core-shell composite fiber using braided poly(glycolide/
Yang and Hu created a superabsorbent fabric consisting of three layers: two hydrophobic layers that were made of polyester/spandex, and the superabsorbent layer in the middle made of superabsorbent yarns (superabsorbent fiber (SAF) or superabsorbent fiber thicker (SAFT)). The superabsorbent yarn was knitted into the spacer layer of the fabric. The superabsorbent fabrics had better absorbency, higher air permeability, heat insulation, and faster wetting speed. This means that superabsorbent fabrics can be used on exuding wound dressings to facilitate the wound healing process. 23 Wu et al. created warp-knitted spacer fabrics (WKSFs) that were designed and manufactured for pressure ulcer prevention and care. The air permeability, water transport, compression performance were studies. WKSF is more compliant to deformation, meaning it can provide better support to the human body. The fiber materials used were 0.2 and 0.16 mm polyester monofilaments for the spacer yarns and polyester multi-filaments of 300D/96F for the surface layer yarns. 24
Mihai et al. investigated the physical and mechanical properties of Lempur, acetate, cotton, and cotton/rayon blends. Their results were then used to design four conceptual woven structures that can be integrated into wound dressings. 25 Such work facilitates the development of personalized wound dressings.
Therefore, the recent technology made possible by antibacterial polyester composites of different macro- and micro- structure has allowed for the development and evaluation of two unique woven layers of a multicomponent wound dressing. The first is a woven, wicking and evaporative layer composed of a 4DG multifilament yarn necessary for moisture wicking to ensure evaporation while preventing maceration. In this study, the wicking capability of a woven, braided, poly-
Materials and methods
Extrusion process
PLLA (Jamplast, Inc. Ellisville, MO USA) was melt-spun into 36 filaments of round or 4DG cross-sections using a large-scale continuous twin screw melt-extruder and spinneret system (Hills Incorporated, Melbourne, FL). Differential scanning calorimetry (DSC) was used to obtain the glass transition (Tg) and melting temperatures (Tm) needed to derive set points for the extruder’s four heating zones. Final heating zones for PLLA were 185°C, 205°C, 220°C, and 210°C. The fiber diameter was controlled by the pump speed at the extruder outlet and the drawing rate of the sequential rollers and spool used to collect the fiber post-extrusion. The resultant fibers were coalesced into a 300 μm (outer diameter) yarn with a 438 denier.
Braiding the moisture wicking layer
The PLLA yarn was braided in the full load regular pattern (1 over 2 under 2) using a 25.4 mm diameter mandrel. The multifilament was braided at 15 pics per inch using a KL80d carrier with tension and ratchet springs of 0.45 and 0.3 mm, respectively. Four rollers were used at a speed of 35 rpm.
Porcine serum extraction
Tuck et al. developed guidelines that SOPs for the collection, handling, processing, and storage of serum should include. They mentioned that the SOPs should include factors like allowing time for clot formation, temperature, the number of freeze-thaw cycles, and hemolysis. These factors influenced the creation of our porcine serum extraction protocol. 26 Two (2) liters of blood were collected from three porcine donors in 10 mL blood collection tubes (BD, Franklin Lakes, NJ, USA). Upon collection, the whole blood was set to clot and left undisturbed at room temperature for 30 min. Each tube was centrifuged at 1500 rpm for 10 min. Immediately after centrifugation, serum was aliquoted and stored at −20°C until use.
Vertical wicking analysis
Wicking analysis was evaluated using AATTC 197. PLLA braids (7 cm) were cut and dried in a vacuum chamber for 30 min. Porcine serum was thawed to room temperature. A solution of 3 mL of serum and 1 mL of trypan blue dye (Gibco™, Thermo Fisher Scientific, Eugene, OR, USA) was used as the 5 mm tall wicking absorbate and added to a 100 mL beaker. The PLLA braid was added to the beaker and the stopwatch started immediately after. Time was stopped once the dyed serum solution reached 7 cm. Vertical wicking rate can be calculated using equation (1).
W, vertical wicking rate (mm/s) where d is the wicking distance in millimeters and t is the wicking time in seconds.
Pre-treatment and knitting the skin-contact layer
Due to mechanical weakness, the PLLA yarn with filaments of 4DG cross-section were pretreated to give strength to the yarn by adding 150 twists per meter using a twisting machine (Model C10, Agteks, Istanbul, Turkey). PLLA yarn was knitted using a jersey knit construction with a 220-needle circular knitting machine with a 54-gage needle (Model FAK, Lawson-Hemphill, Swansea, MA). The machine can create fabrics containing 20 needles per inch, operate at a maximum speed of 160 m/min, and produce yarns between 400 and 100 denier. Contact layers were cut into round disks of 2 cm or 1.25 cm diameters for antibacterial analysis and biocompatibility and LIVE/DEAD™ experiments, respectively.
Cleaning process
Before GS incorporation, each contact layer was cleaned and prepared for bacteria or cell culture via the protocol outlined in Figure 1. Contact layers were washed in 70% ethanol followed by a 5-min drying period and rinsed in sterile deionized water followed by a 30-min drying period. Those contact layers used for biocompatibility and LIVE/DEAD™ experiments also received a 15-min soaking period in Dulbecco’s Modified Eagle Medium (DMEM) (R&D Systems, Inc. Minneapolis, MN, USA) with ultraviolet (UV) light exposure on each side of the contact layer. Lastly, contact layers were dried for 1 h.

Illustration of the pre-treatment of the skin contact layer protocol.
Antibiotic incorporation
Gentamicin Sulfate (GS) was the antibiotic chosen and incorporated into the skin-contact layer. GS is an aminoglycoside, used to treat infections that contain strains of Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Bacillus, and Subtilis. 27 The antibiotic has concentration-dependent bactericidal and bacteriostatic activity that inhibits bacterial protein synthesis. Contact layers of round or 4DG yarn were submerged in solutions of 0, 10, 100, 1000, and 10,000 μg/mL of GS (Thermo Scientific™, Waltham, MA) and sterile DI H2O, and soaked overnight.
PEG surface treatment
The treatment of PEG was added as a thin surface layer to the PLLA/GS contact layers to introduce non-adhesive properties. About 400 mg of 1500 Da or 4600 Da PEG (Sigma-Aldrich, St. Louis, MO USA) was dissolved in 10 mL of sterile DI H2O. About 1 mg of GS was added to each solution. Contact layers were submerged in 2 mL of the PEG/GS solution and left to sit for 48 h. The resulting contact layers were left to dry overnight.
X-ray photoelectron spectroscopy
To confirm the surface treatment of PEG, 1 cm × 1 cm × 0.32 mm PEG/PLLA contact layers were loaded, separately, into a sterile load-lock chamber. Once the pressure registered 10−5 Pa, the samples were transferred to the analysis chamber. After the pressure dropped to 10−7 Pa, the spectra of each sample were collected.
Antibacterial efficacy testing
The contact layers of various GS concentrations were tested to determine the antibiotic efficacy against Pseudomonas putida. P. putida is a Gram-negative bacteria commonly found in soil and many other environmental niches that can cause serious infection and outbreaks in humans, particularly in unclean open wounds. 28
Liquid Broth analysis
Using culture tubes, P. putida was inoculated in 4 mL of sterile Liquid Broth (LB), Lennox (Fisher Scientific, Fair Lawn, NJ, USA) and incubated at 37°C and 5% CO2 overnight. The starting optical density (OD) was measured from each culture tube using a NanoDrop 2000 (Thermo Fisher Scientific, Waltham, MA, USA). Diluting the P. putida in LB was performed to ensure consistent starting OD between experiments. About 1.25 mL of P. putida in LB were added to each well using 12-well non-treated plates. Contact layers were submerged in each well. The OD was measured from each well at hours 1, 3, 6, 9, 24, 48, 72, 96, and 120. This protocol follows the recommendation by Gulati et al. 29 on how to carry out the most efficient, conventional, and reliable broth microdilution.
Disk diffusion method
A “fabric disk diffusion assay” was used to determine the antimicrobial properties of the contact layer. 29 P. putida was cultured in sterile LB and incubated at 37°C and 5% CO2 and agitated on a shaker plate overnight. After resuspension, the starting OD was measured. The bacterial solution was diluted 1000-fold with sterile deionized water. About 100 μL of the new diluted bacterial solution was added to an agar plate. Using plating beads (Zymo Research, Irvine, CA, USA), the solution containing P. putida was homogeneously spread over the entire agar. 1.25 cm diameter contact layers of each GS treatment were placed flat in the center of the agar plate. Each plate was incubated at 37°C and 5% CO2 overnight. The zone of inhibition (ZOI) was determined by measuring the diameter of the region where there was no apparent growth of P. putida around the contact layer.
Biocompatibility and cell adhesion analyses
Biocompatibility tests were performed simultaneously using AlamarBlue™ (Invitrogen, Thermo Fisher, Eugene, OG, USA) and Quant-iT™ PicoGreen™ (Thermo Fisher Scientific, Eugene, OG) assays to determine whether the material and GS elution of the contact layers promoted healthy cell growth. AlamarBlue™ and Quant-iT™ PicoGreen™ assays measure cell growth through metabolic response and DNA quantification, respectively.
The mesenchymal stem cells used in this study were murine bone marrow stromal D1 cells. Cells were first cultured in T-75 polystyrene culture flasks (Corning; Fisher Scientific, Waltham, MA, USA) to confluency in a growth medium cocktail consisting of DMEM, fetal bovine serum (FBS) (Life Technologies; Carlsbad, CA, USA), and antibiotic-antimycotic (AA) (Life Technologies, Carlsbad; CA, USA) at 37°C and 5% CO2. Growth medium was changed every 2–3 days. Cells were removed from their flask via trypsinization and split into a 24-well non- treated, low attachment polystyrene culture plates (Corning; Fisher Scientific, Waltham, MA, USA) with the starting cell density of 17,000 cells/well. Each contact layer was submerged over the cells immediately after cell splitting. Cell proliferation was analyzed at hours 24, 72, and 120.
AlamarBlue™ assay
At each time point, 65 μL of AlamarBlue™ dye was added to each well and incubated at 37°C and 5% CO2 for 3 h. About 200 μL was taken from each well and added to a 96-well plate (Corning; Fisher Scientific, Waltham, MA, USA). The plate was read using a Synergy MX Multi- Mode Reader at an excitation wavelength of 560 nm and an emission wavelength of 590 nm.
Quant-it™ picogreen™ assay
The remaining cell solution with AlamarBlue dye was removed, and each well was rinsed with 600 μL of 1× Phosphate Buffered Saline (PBS) (Life Technologies Limited; Thermo Fisher, Grand Island, NY, USA). To obtain double-stranded DNA (dsDNA) in solution, the PBS was removed, and cells were ruptured via trypsinization for a 2-min incubation period and sonication for 20 s. The trypsin solution of each well was centrifuged and removed. The dsDNA was resuspended using TE buffer (Invitrogen; Thermo Fisher Scientific, Eugene, OR, USA). Samples were frozen at −20°C until analysis. After each time point, samples were thawed. About 1 mL of Quanti-iT PicoGreen dsDNA reagent (Invitrogen; Thermo Fisher Scientific, Eugene, OR, USA) was added to each sample. A standard curve was created (Table 1) to determine the relation between fluorescence and dsDNA concentration.
Standard curve concentrations for dsDNA detection.

Each standard and unknown sample was added to a 96-well plate. The plate was read using a Synergy MX Multi-Mode Reader at an excitation wavelength of 480 nm and an emission wavelength of 520 nm.
Live/dead™ analysis
Fibroblasts used in this study were from a murine 3T3 cell line. Cells were first cultured in T-75 polystyrene culture flasks to confluency in a growth medium cocktail consisting of DMEM, FBS, and AA at 37°C and 5% CO2. Growth medium was changed every 2–3 days. Cells were removed from their flask via trypsinization and split into a 24-well non-treated, low attachment polystyrene culture plates with the starting density at 17,000 cells/well. Each PEG treated contact layer was submerged over the cells immediately after cell splitting. Cell proliferation and attachment were analyzed at 24, 72, and 120 h. Cell media from each treatment was removed and cells were rinsed with 100 µL of 1× PBS. About 200 µL of staining solution containing Calcein and ethidium homodimer (Invitrogen Ltd.; Paisley, UK) diluted in PBS was added to each well and incubated at 37°C and 5% CO2 for 30 min. Cells were imaged under transparent, Texas red, and green fluorescent filters.
Statistical analysis
Antibacterial effects considered all combinations of contact layers to determine differences in shape and concentration impact. Given the factorial nature of this study, all combinations were not tested for biocompatibility and LIVE/DEAD™ testing. All experiments included three replications (n = 3) and analyzed using a T. Test function using Microsoft Excel and a level of significance α = 0.05.
Results and discussion
This study includes a factorial set of treatments based on PLLA yarn geometry and GS concentration to determine moisture wicking and antibacterial efficacy for a polyester medical textile wound dressing. PLLA was chosen as the material for the moisture wicking and skin-contact layers due to its versatility in extruding shaped yarns, mechanical properties conducive to knitting and braiding, and antibiotic uptake and release. The GS incorporation method via diffusion was determined as the most effective method in releasing GS to create a bactericidal response. GS has a large 3-dimensional structure which makes dying the molecule into the PLLA fibers difficult as discovered in previous work. The 4DG shape has a larger surface area relative to round yarn, and thus was chosen as an additional condition when testing its effect on uptake and release.
Cross Sectional Area of the 4DG Mono and Macro-structure and Knitted Contact Layer Images of the cross-sectional area of the 4DG monofilament and yarn, as well as the knitted contact layer are shown in Figure 2. Figure 2(b) shows each of the 36 4DG filaments that make up the shaped yarn, used in the woven layer. The contact layer in Figure 2(c) specifically depicts the geometric shape during the knitting process as well as the breathability of the layer. The jersey knit construction provided a light, relatively smooth skin-contacting surface with low retention of heat and moisture given the need for this layer to pass through moisture to the subsequent absorptive and wicking layers of the multi-layer dressing.

(a) Scanning electron microscope (SEM) image of a wicking fiber. Adopted with permission from John Wiley and Sons, 30 (b) cross sectional area of 4DG yarn, where red and blue outlines represent macro- and microcapillaries, and (c) knitted contact layer of round yarn.
Wicking analysis
A solution of dyed porcine serum with viscosity 1.6 cP, similar to that of wound exudate, was used as the absorbate of the vertical wicking test. The sample fabric was tested three times. After being vertically exposed to the serum solution, the 4DG braided layer was able to wick the serum upwards at an average wicking rate of 0.88 ± 0.265 mm/s when calculated with equation (1). After an average of 1 min and 25 s, the dyed serum reached the boundary of the fabric. The vertical wicking rate seems to wick the serum faster after initial exposure of priming of the microchannel and has a logarithmic trend, indicating that there were higher vertical rates at initial exposure. The vertical wicking rate can be seen in Figure 3.

Representation of vertical wicking of dyed porcine serum through the 4DG channels of the braided PLLA yarn at different time periods (min:secs).
The braided medical textile layer was designed for the purpose of moisture wicking and the prevention of autolytic debridement via maceration. The macrostructure of the layer was designed to be braided, which is predicted to manage large forces and mechanical strain from weight bearing activity, since this layer will be adjacent to shoes, orthotics, or flooring. The wicking capabilities of the layer are made possible by the unique 4DG shape of the individual fibers making up the multifilament yarn that is eventually braided into the overall layer. The eight different legs form four uniquely shaped and sized ducts that move fluid spontaneously, and for this purpose transport excess exudate that can overwhelm the healing process.
The results found in this experiment are similar to those found by Liu et al. while investigating the wicking of twisted yarns. They studied polyester fibers with an average radius of 8.4 µm and length of 400 mm that were made into yarns composed of 37 fibers. Their results also show that the initial velocity of liquid is much higher in early stages. As time progresses, the liquid becomes slower until equilibrium is reached. At around 200 s, equilibrium was reached, and using equation (1), we can determine that their average wicking rate was 2 mm/s. 31
XPS analysis
GS contact layers were treated with PEG to prevent non-specific binding of proteins leading to some cell attachment, as seen in the PicoGreen™ cell viability study. The method for treating PEG on the surface of the contact layer was confirmed using XPS analysis. The survey spectra of the PLLA/PEG contact layers shown on Figure 4(a) indicates that there are only two elements within the sample, carbon and oxygen at binding energies of approximately 285 and 540 eV. Figure 4(b)–(d) depict the normalized peaks of the PLLA contact layer, PLLA/1500 PEG contact layer, and PLLA/4600 PEG contact layer, respectively. The contact layer with PEG shows an additional peak at approximately 286 eV, where there is an additional C-O bond. As the density of the PEG increases, the peak area increases. Therefore, the total peak of area of O-C=O, C-C-C=O, and C-C present in PLLA, decreases in contact layers treated with PEG by 15% and 34% for PEG 1500 and PEG 4600, respectively.

(a) XPS survey spectra. Peak fitting of (b) PLLA contact layer, (c) PLLA/1500 PEG contact layer, and (d) PLLA/4600 PEG contact layer.
Antibiotic efficacy of GS incorporated contact layers via liquid broth and disk diffusion analysis
Antibacterial skin contact layers of all shape and concentration combinations were submerged in liquid cultured P. putida for testing. Previous work showed that the 4DG fiber had the ability to uptake more GS, as seen in its elution profile due to the significantly higher surface area.
The OD was measured at both short and long term intervals. The percent growth was then determined using the ratio of the current OD reading to that of the initial OD reading (t = 0). When treating contact layers of round and 4DG shaped yarn, all concentrations showed a bactericidal effect within the first 24 h, where there was an exponential decay in bacterial growth (Figure 4). It appears that although the 4DG shape of the yarn contributes to a higher release profile of GS compared to round yarn as indicated by its higher surface area, the initial release above the indicated minimum inhibitory concentration (~1 µg/mL) 32 of GS from each shaped yarn seemed to have a similar impact on the inhibition of P. putida. Because all concentrations reached the MIC, the antibacterial profile was independent of concentration.
The amount of GS that the contact layer initially contained could have played a role in the antibacterial behavior. During the short-term intervals, all treatments contributed to a bactericidal effect, where the bacteria growth rate decayed over time. For concentrations below 1000 µg/mL, the bacteriostatic effect occurred after 24 h where bacteria growth remained inhibited. The two highest diffusion concentrations, being 1000 and 10,000 µg/mL, caused a subtle increase in bacteria growth after 24 h, indicating some minor antibacterial resistance. Therefore, the ideal effect from concentrations below 1000 µg/mL determined the chosen concentrations at 1, 10, and 100 µg/mL for the cell viability and adhesion studies.
The effects of antibiotic release as a function of shape can be seen in Figures 5 and 6.

Percent growth of P. putida from contact layers of 4DG yarn.

Percent growth of P. putida from contact layers of round yarn.
Unlike the liquid broth antibacterial analysis, the disk diffusion study showed that the zone of inhibition was dependent on concentration treatments for all concentrations. Contact layers of all treatments were placed on agar cultured P. putida. At 24 h, the zone of inhibition was determined by measuring the diameter across the area where there were no bacteria colony forming units (CFU) present, as shown in Figure 7. The combinational effect of yarn shape and GS diffusion concentration is shown in Figure 8. As concentration increases by a factor of 10, the ZOI increases linearly as indicated in Figure 8. There is no significant impact on the ZOI from the shape of the yarn similarly to the efficacy profile in liquid cultured P. putida.

(a–d) P. putida exposed to contact layers with round yarn treated with concentrations between 10 and 10,000 µg/mL respectively. (e–h) P. putida exposed to contact layers with 4DG yarn treated with concentrations between 10 and 10,000 µg/mL, respectively.

ZOI of contact layers with round or 4DG GS treated yarn.
Bajpai et al. created cotton cellulose/polyacrylic acid fibers and loaded 88 mg or 95 mg of GS on 1 g of fiber. They found that as GS content increased, the amount of GS bound to fibers increased, which caused the antibacterial action of fibers to be more effective and the zone of inhibition to increase. 33 These results are consistent with the results found in this experiment, as an increase in GS concentration also resulted in the diameter of the ZOI increasing.
Cell viability and adhesion
After performing antibacterial analyses for all conditions of contact layers, the metabolic activity and DNA content of D1 mouse bone marrow stromal cell was studied over a duration of 5 days to determine any cytotoxic effects from the release of GS as well as cell attachment preference.
Due to the factorial experimental design, cells were only exposed to contact layers of round yarn treated with concentrations of 1, 10, and 100 µg/mL. Non-treated 24-well plates were used to prevent cell attachment for a better indication of cell adherence preference. The cell metabolic activity was measured via AlamarBlue™ fluorescent agent. Normalized metabolic activity is described as the percent viability. Each experimental condition’s percent viability was calculated using the fluorescent readings of the experimental condition and the negative control condition, which included the cultured D1 cells without any presence of a contact layer. Each treatment had a percent viability greater than 95% relative to the negative control condition, indicating that both the PLLA contact layer and eluted GS did not harm metabolic activity. Additionally, there appears no difference in concentration treatment indicating that our tested elution concentrations effective in inhibiting bacterial growth, 1–100 µg/mL, do not create any cytotoxic response.
In line with similar demonstration of the non-cytotoxicity of GS, Wang et al. created poly(dimethyl siloxane) (PDMS) incorporated with either GS or triclosan (TCA) to determine cytotoxicity. They found that the density of human lens epithelial cells (HLEC) on PDMS and PDMS-GS films was less than the density on tissue culture plates, but the films were biocompatible as the cells maintained normal morphology and spreading. However, the PDMS-TCA films were cytotoxic as the number of HLEC decreased and they did not maintain normal morphology and spreading. Cell viability was then evaluated by a Cell Counting Kit-8 (CCK-8) assay which showed that activity on the PDMS and PDMS-GS was more or less the same (more than 91% of that on TCPS), while activity on PDMS-TCA was 66.5% of that on TCPS. 34
In addition to metabolic activity, DNA concentration was measured using Quant-iT™ PicoGreen™ Assay. A standard curve of known DNA concentrations was used to calculate the DNA content from D1 cells adhered to either the contact layer or the well plate in order to determine cell attachment preferential behavior. Each experiment contained a well (blank condition) that had actively growing cells without the presence of a contact layer. It appears that for each concentration condition, cells preferred to attach to the contact layer as a result of high surface area for adhesion and lack of pretreatment on the polystyrene 24-well plates. This particular study is important because it shows that PLLA alone does not allow for non-adhesion properties required for dressing changes. Therefore, a surface treatment is needed to prevent cell adhesion, while creating a biocompatible environment that does not alter GS release and antibacterial efficacy. PEG was chosen as an additional surface treatment due to its relative surface resistance to protein and non-specific cellular binding for the prevention of unnecessary debridement during dressing changes. 35 When the contact layer is treated with either PEG 1500 and 4600, there is less cell attachment, and the fibroblasts prefer to adhere to the untreated well plate.
Yu and Kohn created tyrosine-PEG-derived poly(ether carbonates) and performed experiments to determine cytotoxicity and attachment on rat lung fibroblasts and murine osteoblast-like MC3T3 E1 cell. They discovered that when the copolymer contained a larger percent of PEG, the rat lung fibroblasts and osteoblasts were less capable of attaching and growing on the copolymer surface. Calcein AM and Trypan blue were used for viability testing in the osteoblasts and showed that they remained viable during the entire culture period. These results also demonstrate that the elimination of growth and cell attachment was not a result of PEG-containing copolymer cytotoxicity, but a reflection of the altered surface properties. 36
Live/dead™ adhesion analysis
3T3 fibroblasts were exposed to GS incorporated PLLA and PLLA/PEG contact layers. After exposure for 24, 72, and 120 h, the fibroblasts were stained with diluted ethidium homodimer and calcein. Green fluorescently labeled cells indicate healthy, viable fibroblasts and red fluorescently labeled cells indicate dead fibroblasts. At each time point for all conditions seen in Figure 9, there remained nearly 100% viability indicating that the GS elution, as well as the PEG surface treatment does not negatively impact cell viability. Additionally, there is an increase in cell growth for all treatments as time increases. The control PLLA contact layer depicts some cell attachment to the contact layer over time as shown in Figure 10. When PEG 1500 and 4600 are introduced to the surface of the contact layer, there is less cell attachment seen under fluorescent microscopy. This decreased cell attachment is an important pre-cursor to prevention of tissue adhesion during the wound healing process. Tissue adhesions to wound dressings have the potential to create reinjury upon dressing changes, which may further delay healing.

LIVE/DEAD™ images of 3T3 fibroblasts exposed to PEG treated GS incorporated PLLA contact layers (PEG MW (Da):GS concentration (µg/mL)) showing cell proliferation.

LIVE/DEAD™ images of 3T3 fibroblasts exposed to PEG treated GS incorporated PLLA contact layers (PEG MW (Da):GS) showing cell attachment.
The biocompatibility of GS and PEG was also explored in a separate study when Same et al. created a nanocomposite hydrogel based on poly-e-caprolactone (PCL)–PEG—PCL and natural gelatin (PCEC/GEL) reinforced with halloysite nanotube (HNT). They investigated the viability of human dental pulp-derived stem cells (h-DPSC) using the microculture tetrazolium (MTT) assay in PCEC/GEL/HNT-GS, PCEC/GEL/HNT, and PCEC/GEL and found that the proliferation and survival of cells increased for all samples. These results also demonstrate that the combination of GS and PEG is biocompatible and does not negatively impact cell viability. 37
Conclusion
This work explored the combination of two polyester medical textiles for the advanced treatment of chronic wounds in a multicomponent wound dressing. The vertical wicking properties of a braided medical textile layer composed of 4DG shaped fibers were measured. Not only did the unique channels of the braid promote wicking, but it readily moved moisture of higher viscosities needed for a typical wound model. Antibiotic efficacy of two different shaped polyester yarns was measured and it was discovered that both shapes eluted antibiotic above the MIC against P. putida, causing both a bactericidal and bacteriostatic effects. Additionally, the elution for optimal GS concentrations did not negatively impact healthy cell metabolic activity. However, cells gravitated toward the skin contact layer indicating the need for a non-adherent surface treatment. Finally, the influence of a PEG-incorporated skin-contact layer resisted non-specific cellular binding and therefore, can prevent autolytic debridement during dressing changes.
Outcomes of this work provide insight into advancements of antibacterial, woven polyester composites for the medical and textile industries and more specifically optimize wound care management through manipulating fabric parameters for personalized healing.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) acknowledge funding for this research from the Greenville Health System (Prisma Health) Health Sciences Diabetes Reseach Seed Grant.