Abstract
This article, discuss the effect of finishing polyester/cotton blended fabric (PET/C) with alkali and Titanium dioxide nanoparticles (TiO2 NPs) simultaneously. The treatment conditions such as NaOH and TiO2 NPs concentrations, reaction temperature and duration will be investigated. The effect of addition NPs on alkaline treatment conditions will prove through weight loss and carboxylic content. The ability of PET/C fabrics for loading with NPs during alkaline treatment was investigated by using SEM, EDX, and FTIR measurements. The effect of finishing of PET/C blended fabric with the suggested method on antimicrobial activity and ultraviolet protection was investigated. The simultaneous finishing of PET/C blended fabrics with alkali and TiO2 NPs showed excellent ultraviolet protection and high antimicrobial activity against Gram-positive (Bacillus mycoides), Gram-negative (Escherichia coli), and nonfilamentous fungus (Candida albicans). The functional performance imparted to PET/C fabrics by the suggested approach are durable in repeated laundering processes, even after five Launder-Ometer washes.
Introduction
Nowadays, polyester fibers are one of the most important and prevalent fibers in the textile industry. Aside from the superior characteristics of PET fabrics are tensile strength, dimensional stability and abrasion resistance. But these fibers have certain drawbacks such as low water absorbency, pilling tendency, and accumulation of electrostatic charges also been reported. To overcome these drawbacks, blending with cotton fiber is recommended, so better water absorbency, handle, and comfort can be achieved.1,2
Recently, functional finishing of PET/C blended fabrics with NPs in order to impart them a new functional performance for the convenience of consumer and to expand the areas of their use.3–6 Today, with the spread of COVID-19 pandemic, there has become an urgent necessity for imparting fabrics antimicrobial property to maintain health and reduce infection to suit the public use and medical sector. As well as protection from ultraviolet rays for men of the armed forces, police and workers in desert sites, whose nature of work requires them to be exposed to sunlight for long periods.
Scouring of PET/C blended fabrics with moderate concentration of NaOH aqueous solution is one of the vital industrial stages in dyeing and finishing factories aiming to verify the removal of starch. With the use of higher concentrations, partial hydrolysis of PET filaments and mercerization process of cotton fibers occur.7–10 High concentration of NaOH has a dual effect on PET/C blended fabrics. (a) The ester bond in PET fibers hydrolyzed, unique properties such as silk-like handle and antistatic are acquired to PET fibers. (b) Mercerization of cotton fibers which results in the enhancement some of physical properties of cotton fabrics.11,12
The possibility of utilizing the conventional alkaline hydrolysis of PET fibers for enhancement of the binding efficiency of TiO2 NPs has been studied by Montazer and Sadighi. 13 Many researchers established that alkaline hydrolysis of PET fibers leads to a considerable chemical and topographical change of fibers where scission of polymer chains takes place.14–16 Namely, the carboxylic and hydroxyl groups are anticipated to be introduced and this can be utilized for the improvement of binding of TiO2 NPs.
However, no one has worked on the effect of doping PET/C fabrics in a bath containing nano-metal oxides during alkali treatment and shown their effect on the functional performance of finished fabrics. Based on the abovementioned, this article provides a new study and facile finishing approach by simultaneously treatment of PET/C blended fabric with sodium hydroxide and TiO2 NPs. This technique is paving the way for NPs to be widely implemented on a wet processing line in industry, making them more cost-effective for textile finishers.
Experimental work
Materials
Desized and Bleached polyester/cotton blended fabric (PET/C) 100% Trevira (165 g m−2) woven fabric was used throughout this work provided by local textile industries. To clean the fabric from the impurities, the fabric was scoured in a solution containing 2 g L−1 nonionic detergent for 30 min at 60°C with liquor to goods ratio (M:L) of 1:40, then rinsed with distilled water and dried at ambient temperature.
All chemical used in this work (Titanium dioxide nanoparticles (TiO2 NPs) emulsion (aqueous, 30%)) – Sodium Hydroxide (NaOH), hydrochloric acid (were purchased from Fluka and have been used as received).
Bacillus mycoides (
Methods
Alkaline treatment of PET/C fabric
The alkaline treatment of PET/C blended fabric was carried out by using a high temperature, high pressure laboratory dyeing machine. Required volume from the prepared and calibrated of NaOH aqueous solution was placed in stainless-steel bowl. The fabrics samples were immersed in the solutions and the sealed bowls were rotated in a closed bath containing ethylene glycol at selected temperature. The liquor-to-fabric ratio (M:L) was 1:50. The bath temperature increased at rate of 2°C/min. After the predetermined durations, the samples were removed from the bath, rinsed repeatedly with distilled water, neutralized with a solution of 1% hydrochloric acid and rinsed. The samples were then dried at 100°C, cooled in a desiccator, and weighed. The weight loss is expressed as relative WL was calculated according to the equation:
where: W1 and W2 are the weights of the samples before and after alkaline treatments, respectively.
PET/C fabric loaded by TiO2 NPs
The treatment of PET/C blended fabric with TiO2 NPs was carried out according to the above mentioned method in presence TiO2 NPs by using suitable concentrations (0.05–1.0 g L−1).
Durability testing method
In order to evaluate the Ti2O NPs adhesion to the PET/C blended fabrics, the treated fabric was washed five cycles according to AATCC Standard Test Method (61-1989). One cycle takes about 45 min in presence 2.0 g L−1 nonionic detergent, every one washing cycle equal to five typical home launderings. In our approach, washing of textiles was performed at washing temperature settings of 71°C. After this process, the textiles were dried at 105°C.
Analysis
Carboxylic content
Carboxylic content was determined according to the method described in Daul et al. 17
Antimicrobial activity
Antimicrobial activity of PET/C blended fabrics loaded with TiO2NPs was quantified using the following methods:
Shake flask method
In this method the antimicrobial activity of immobilized antimicrobial agents is determined under dynamic contact conditions according to ASTM standard test method 2149 (2001).
Surface morphology analyzed by SEM and EDX
The structures and surface morphology of the alkali treated PET/C blended fabrics and loaded with TiO2 NPs were characterized by scanning electron microscopy (SEM, JEOL JSM T2O). Electron dispersive Emission X-ray. EDX mode was applied for the elemental composition analysis. Gold layer was coated on the fabrics surfaces before the examination.
FTIR analysis
The surface chemistry of the PET/C blended fabrics, partially hydrolyzed and loaded by TiO2 NPs were investigated using the Fourier Transformation Infrared (FT-IR) spectrometer, model NFXUS 670, NICLET USA. The measurements were carried in the spectral range from 4000 to 500 cm−1. Reflection percentage measurement technique (R%) was applied to all investigated samples.
Ultraviolet protection factor (UPF)
UPF was determined using UV-Shimadzu 3101 PC Spectrophotometer. It is a double beam direct ratio measuring system. It consists of photometer unit and a PC computer. The UPF values were automatically calculated on the basis of the recorded data in accordance with Australia/New Zealand standard AS/NZS 4395:1995. 18
UV protection and classification according to AS/NZS 4395:1995

Results and discussion
The present finishing method was based on doping PET/C blended fabric with an aqueous solution containing both NaOH and TiO2 NPs; taking in our mind that the weight loss % (WL%) during this finishing approach depends upon the treatment conditions; aiming to show the effect of TiO2 NPs on the reaction medium. It would be of great importance to study the effect of different finishing factors to determine the suitable conditions for imparting new functional performance to PET/C blended fabrics. The WL% resulting from this finishing method was measured under different conditions including NaOH and TiO2 NPs concentrations, treatment temperature, and duration.
Given below are the factors affecting on the alkali treatment of PET/C blended fabric in presence TiO2 NPs.
Factors affecting alkali treatment
Effect of NaOH
Alkali treatment of PET/C blended fabrics leads to a partial hydrolysis (PETC → H) of fabric surface and produces new properties. 19 The dissociation of ester bonds takes place with alkaline treatment of PET molecules by NaOH in two ways: unimolecular and bimolecular reactions, in which one and two hydroxide ions react with ester bond, respectively. The polymer chains by progressive reaction with alkali are hydrolyzed to its monomer unit’s terephthalate anions and ethylene glycol (Scheme 1).

Effect of alkali on polyester fiber.
Alkali treatment associated with a significant increase of OH and COOH groups on the surface of polyester fibers. 19 These data were experimentally proved by the estimation of functional groups created on the surfaces of fabrics before and after the alkaline treatment through carboxylic content measurement.
Table 1 showed that, the surface of PET/C → H in general, had an increase in its carboxylic content rather than parent. Alkali treatment causes outstanding increase in carboxylic content from 6.2 to 22.4 meq/100 g fabric. Therefore, the interaction between the partially hydrolyzed textile surface by alkali and TiO2 NPs was electrostatic in its nature, since the textile surface was negatively charged as a result of the activation and formation of COOH and OH groups on the surface. This could be enhancement the loading of TiO2 NPs onto the fabrics surfaces, similar observations were reported.20–22
Carboxylic content for PET/C blended fabrics before and after treatment with aqueous NaOH solutions.

Alkali treatment conditions: [NaOH], 1.5 mol L−1; duration, 60 min; temperature, 100°C; M:L, 1:20.
Effect of NaOH on WL%
It was observed that (Figures 1–4), alkaline treatment (at constant concentration from TiO2 NPs equal 0.2 g L−1) of PET/C → H fabric accompanied by a decrease in its weight. Hydrolysis was found to proceed, but with a low WL (1.6%, after 10 min at 80°C) when polyester fabric was subjected to aqueous NaOH solution having low concentration (0.5 mol L−1). The WL was directly proportional with both NaOH concentration and treatment temperature. For example, WL of 13.0% was gained at NaOH equal to 2.0 mol L−1. The results appear on Figures 1–4. Also, revealed that alkaline treatment of PET fabrics proceeds nonlinearly with the concentration of NaOH.

Effect of [NaOH] on the WL% of alkali treated PET/C blend fabric. Treatment conditions: [TiO2 NPs], 0.2 g L−1; temperature, 80°C; M:L, 1:50.

Effect of [NaOH] on the WL% of alkali treated PET/C blend fabric. Treatment conditions: [TiO2 NPs], 0.2 g L−1; temperature, 90°C; M:L, 1:50.

Effect of [NaOH] on the WL% of alkali treated PET/C blend fabric. Treatment conditions: [TiO2 NPs], 0.2 g L−1; temperature, 95°C; M:L, 1:50.

Effect of [NaOH] on the WL% of alkali treated PET/C blend fabric. Treatment conditions: [TiO2 NPs], 0.2 g L−1; temperature, 100°C; M:L, 1:50.
The results showed that, the extent of nonlinearity was directly proportional with reaction duration. This finding may be due to two possibilities. First, resulting from the concentration of hydroxylate anion groups (OH−) decreases with the degree of reaction. Second, when a large regain of the sample has been hydrolyzed, the surface area decreases accordingly, thus decreasing the reaction rate (a second order relationship).
Validity of equation (1) is verified in Figure 5 where WL/C values have been plotted against C. All plots were straight lines as required by equation (1). Values of constants (a) and (d), obtained from the intercept and slope, respectively of the straight lines. It was clear from Table 2 that values of constant (d), which was a measure of the extent of nonlinearity, is for PET/C → H fabrics as compared to that of polyester fabrics treated with alkali in presence TiO2NPs nearly the same.


Test of validity relationship for the description of data indicating the dependence of WL of alkali treated PET/C blended fabric on concentration of NaOH solutions for different intervals of treatment time (min) : 10; (◊), 20; (□), 30; (∆), 45; (×), 60 (*). (a) PET/C → H and (b) PET/C → (H + 0.2 TiO2 NPs).
Values of constants (a) and (d) of alkali treated PET/C blended fabric at 100°C.
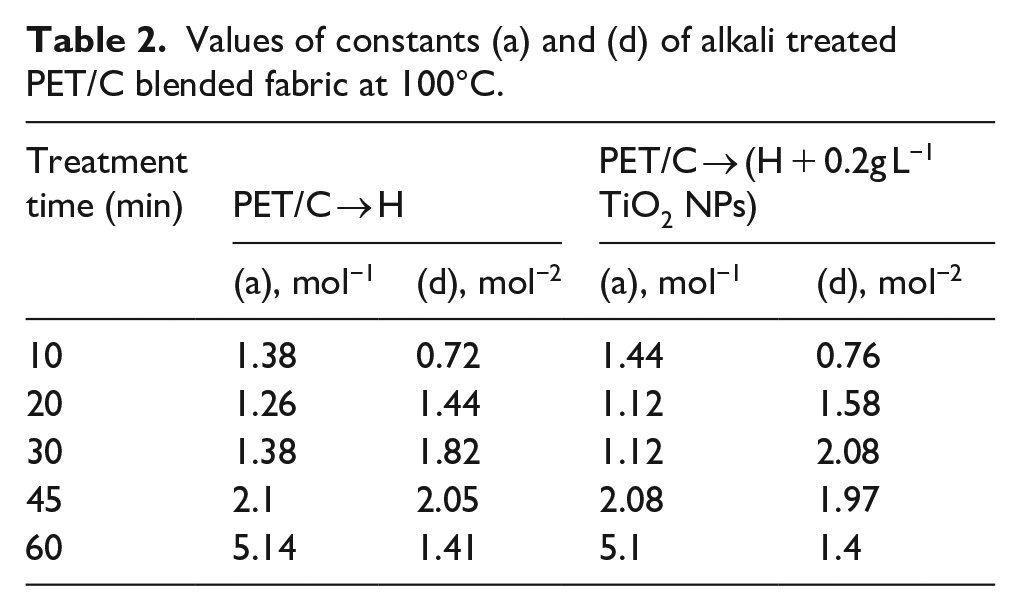
Where, (a) and (d) are constants dependent upon treatment time and temperature, satisfactorily describes the experimental data.
Finishing time
Figures 1–4 and 6 illustrates the percentage WL of PET/C → H fabrics respectively on treatment with 0.5–2.0 mol L−1 sodium hydroxide aqueous solutions at 80–100°C with constant concentration from TiO2 NPs equal 0.2 g L−1 for various time periods. According to this data, alkaline hydrolysis proceeds linearly with time at constant temperature and depends on the initial concentration of the alkali. The slope of the straight lines obtained between weight loss and time increases with increasing concentration of the alkali. This was valid irrespective of the type of treated PET/C fabrics. A linear dependence of weight loss on treatment time has also been observed by other researchers.21,22 The relationship between weight loss, WL, and treatment time, t, can be expressed as equation (2):


Dependence of WL% of alkali treated PET/C blended fabric on treatment time. Reaction conditions: [NaOH], mol L−1: (◊), 0.5; (□), 1; (∆), 1.5; (×), 2.0; [TiO2 NPs], 0.2 g L−1 treatment temperature, 100°C; material:liquor ratio (M:L), 1:50.
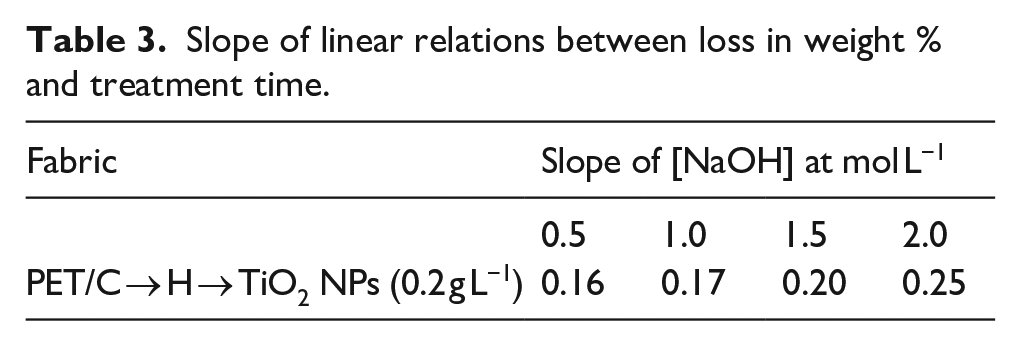
Where, b is a constant dependent on temperature and alkali concentration. However, a high level of weight reduction was observed in the case of PET/C blended fabric. The slopes of these linear curves (Table 3) indicated equal values of weight reduction in the case of PET/C blended fabrics, when the reaction was carried out in presence or absence TiO2 NPs.
Slope of linear relations between loss in weight % and treatment time.
Finishing temperature
The dependence of WL of PET/C → H fabrics on the finishing temperature was shown in Figures 7–10. It can be seen that, the extent of WL increases with increasing the finishing temperature. When the temperature was lower than the Tg of PET (~80°C), 23 the slope was steeper. The observed nonlinear dependence of WL on the concentration of NaOH and finishing temperature was resulting from the enhanced hydrolysis of polymer chains (ester bonds) with increasing NaOH concentration. 21 The steep rise in WL at treatment temperature higher than the Tg may be attributed to above this value the polymer bonds have high degree of freedom to move and thus more easily interact with NaOH. The results obtained showed the same behavior after adding TiO2 NPs to the reaction medium.
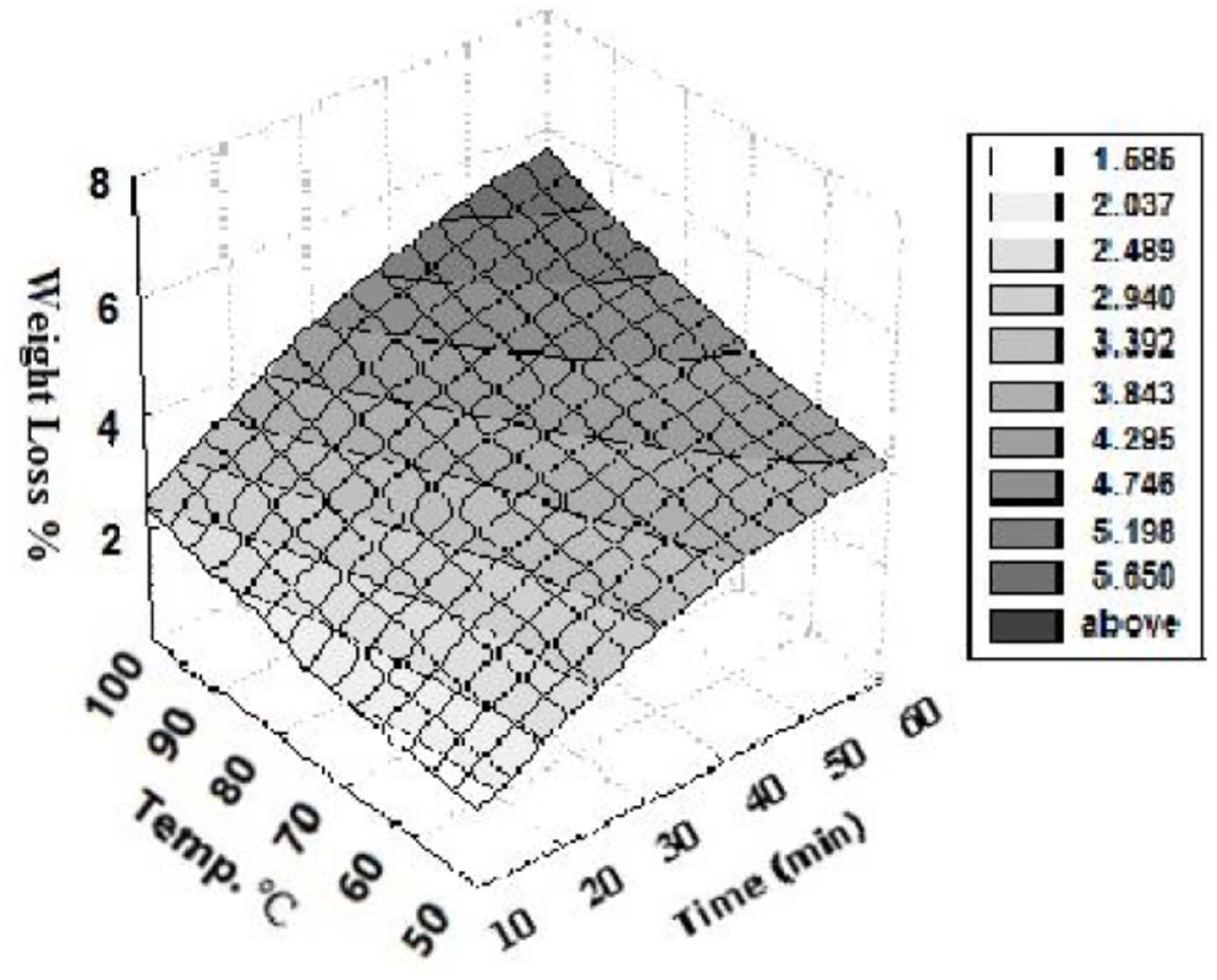
Effect of treatment temperature on WL% of alkali treated PET/C blended fabrics. Treatment conditions: [NaOH], 0.5 mol L−1; [TiO2 NPs], 0.2 g L−1; M:L, 1:50.

Effect of treatment temperature on WL% of alkali treated PET/C blended fabrics. Treatment conditions: [NaOH], 1.0 mol L−1; [TiO2 NPs], 0.2 g L−1; M:L, 1:50.

Effect of treatment temperature on WL% of alkali treated PET/C blended fabrics. Treatment conditions: [NaOH], 1.5 mol L−1; [TiO2 NPs], 0.2 g L−1; M:L, 1:50.

Effect of treatment temperature on WL% of alkali treated PET/C blended fabrics. Treatment conditions: [NaOH], 2.0 mol L−1; [TiO2 NPs], 0.2 g L−1; M:L, 1:50.
TiO2 NPs concentrations
The effect of incorporation of various concentrations of TiO2 NPs (0.05–1.0 g L−1) in the finishing bath containing 1.5 mol L−1 NaOH, on the WL for all treated samples are shown in Table 4. It is clear that, the presence of TiO2 NPs, even with any concentration, in the finishing bath not plays a significant role in alkaline hydrolysis of all treated fabrics. For example, the presence of 0.05 g L−1 of TiO2 NPs during the treatment at 60 min gives 9.8 WL% for PET/C fabrics, when the reaction precedes in absence of TiO2 NPs nearly the same value obtained. It is also seen that the weight loss % is not dependent on the TiO2 NPs concentration.
Effect of TiO2 NPs concentration on the weight loss % of alkali treated PET/C blended fabrics.

Treatment conditions: [NaOH], 1.5 mol L−1; temperature, 100°C; M:L, 1:20.
The increase of TiO2 NPs concentration from 0.05 up to 1.0 g L−1 is not accompanied by any change in the extent of weight loss. However, both the fast rate of initial weight loss and the maximum weight loss were found to be dependent on the concentration of NaOH in the finishing bath only.
It is well known that Ti atoms can be bound to carboxyl groups through different modes which are shown in Scheme 2. Carboxyl groups can be bound in a mono-dentate mode to form an ester-like linkage or they can bind with each of their two oxygen atoms either to one Ti atom (bi-dentate chelating) or to two of them (bi-dentate bridging).24,25 In addition, they can interact with the TiO2 surface through hydrogen bonding either with a surface bound hydroxyl group and/or a lattice oxygen atom. 25

Possible binding modes of a COOH group to TiO2 NPs.
Based on the abovementioned results, it could be state that under any alkaline treatment conditions, presence or absence TiO2 NPs in the alkaline medium hadn’t effect on the weight loss % (neither facilitate nor inhibit the alkali hydrolysis). The point which paves the way for applying TiO2 NPs in the scouring step on the wet processing line to achieve better fabric performance (smooth surface and enhancement in dye-ability) as a result of weight reduction and moreover, the produced PET/C blended fabrics could acquire new and durable functional finishing performance as antimicrobial and Ultra Violet protection due to loading the PET/C fabrics with TiO2 NPs.
Characterization of PET/C fabrics loaded with TiO2 NPs
Scan electron microscope (SEM)
Pristine and finished PET/C blended fabrics were examined by using SEM to investigate the presence of deposited TiO2 NPs and/or incorporate NPs on the surfaces of fabric (Figure 11). It was shown that the surface topography of parent fabric was retained stable and did not observe shrinkage. At higher magnification, the surfaces of the fabric were more distinguished. Parent fabric (Figure 11(a)) had smooth, clean, and without any etches can be seen on its surface.

SEM micrographs of alkali treated PET/C blended fabric* and loaded with TiO2 NPs (X3000): (a) PET/C (Blank), (b) PET/C → H, (c) PET/C → (H + 0.2 g L−1 TiO2 NPs), (d) PET/C → (H + 0.4 g L−1 TiO2 NPs), (e) PET/C → (H + 0.6 g L−1 TiO2 NPs), (f) PET/C → (H + 0.8 g L−1 TiO2 NPs), and (g) PET/C → (H + 1.0 g L−1 TiO2 NPs).
The effect of alkaline treatment on the surface of PET/C blended fabric was clarified in Figure 11(b), where the surfaces of PET/C → H fabric became pitted with vacuoles distributed randomly on the fabric surfaces. The surface of alkali treated fibers were rough that can be due to etching of NaOH to the fabric surfaces which resulting from hydrolysis of short chains and amorphous regain in the polymer matrix. A few numbers of pits on the fiber surface can be observed on the fabrics by adding TiO2 NPs to the reaction medium. Also, some fibers started to link to each other and a thin film were covering the structure of fabrics surfaces. Figure 11(c) appeared that, some distributed aggregates of TiO2 NPs on the surface of the finished fabrics.
Figure 11(g) showed that, at higher magnification appeared a random distribution of TiO2 NPs on the surface of the alkali treated fabric compared to the hydrolyzed fabric in absence NPs, and had some cracks and vacuoles produced on its surface under the effect of alkaline hydrolysis, these cracks enhanced a more TiO2 NPs entrapped inside them. The particles had large size could be easily removed from the fiber surface, whereas the small one could penetrate the polymer matrix and adhere more strongly to the fabric.26,27
The shape and size of the PET/C → (H + TiO2) fibers covered with deposited layers; and any further increase in the TiO2 NPs concentration is accompanied by increase in the deposited materials on the fiber surfaces (Figure 11(d)–(f)).
Emission dispersive X-ray (EDX)
The presence of TiO2 NPs on the PET/C fabrics surfaces was proven by using SEM and EDX. The spectra of PET/C fabrics in EDX mode loaded with TiO2 NPs after one washing cycle were shown in Figure 12. This figure showed that, the atomic structure of loaded fabrics was Ti and O2. This finding was proved even after one washing cycle (five home washings), TiO2 was still bind to the PET/C fabrics surface (Table 5). EDX measurements also revealed higher atomic weight % of Ti on partially hydrolyzed PET/C fabrics and loaded with Ti NPs, this atomic content increases by increasing concentration of Ti in the reaction medium (Ti atomic weight % was 0.33 increased to 3.31). This was more conformation that TiO2 NPs had a good adhesion to the alkali treated fabrics.

EDX Micrographs of alkali treated PET/C blended fabric* and loaded with TiO2 NPs (X3000): (a) PET/C → (H + 0.2 g L−1 TiO2 NPs), (b) PET/C → (H + 0.4 g L−1 TiO2 NPs), (c) PET/C → (H + 0.6 g L−1 TiO2 NPs), (d) PET/C → (H + 0.8 g L−1 TiO2 NPs), and (e) PET/C → (H + 1.0 g L−1 TiO2 NPs).
Ti content on the surfaces of alkali treated PET/C blended fabric and loaded with TiO2 NPs.

Treatment conditions: [NaOH], 1.5 mol L−1; duration, 60 min; temperature, 100°C; M:L, 1:20.
After one washing cycle; AATCC Test Method (61-1989).
Fourier transform infrared spectroscopy (FTIR)
The infrared spectrum of parent PET/C fabric (Table 6) shows the characteristic peaks attributed to >C=O (carbonyl) and OH (hydroxyl) groups respectively. After alkali treatment, the intensity of characteristic peaks ascribed to OH at 3549.9 cm−1 and the peak attributed to >C=O (carboxylic acid) at 1713.2 cm−1 was increased, but no change was observed at other peaks. This proves the creation of hydroxyl OH and carboxyl COOH functional groups on the PET/C surface as a result of alkali hydrolysis.
FT-IR absorption bands of alkali treated PET/C blended fabrics* and loaded with TiO2 NPS.

Treatment conditions: [NaOH], 1.5 mol L−1; duration, 60 min; temperature, 100°C; M:L, 1:20.
After one washing cycle; AATCC Test Method (61-1989).
The intensities of the peaks ascribed to OH and COOH increased for PET/C fabrics treated with NaOH compared with parent PET/C showing a higher formation of hydroxyl and carboxyl functional groups. Also, comparing partially hydrolyzed PET/C, and alkali treated in presence TiO2 NPs with parent fabrics indicated a decrease in the intensities of the peaks ascribed to C=O (carboxylic acid) at 1713.2 cm−1. This is proving the possibility of reaction between TiO2 NPs and polymer chains.
The FTIR charts of alkali treated PET/C fabrics showed appearing new peaks at the position 848 cm−1 with PET/C, which proves the ionic interaction between carboxylate groups existed on the fabric surfaces and Ti present in the reaction medium, similar finding was reported. 21
The functional performance of PET/C fabrics loaded with TiO2 NPs
The antimicrobial
The antimicrobial activity of alkali treated PET/C fabrics and loaded with TiO2 NPs was investigated. Table 7 indicates that, all PET/C fabrics showed, after one washing cycle, higher antimicrobial activity against the previously mentioned three microorganisms. In fact, the % reduction colony forming unit (% CFU) for all tested PET/C fabrics samples was significant, whereas it was null for all the blank samples. The role of activation of PET/C fabrics with alkali hydrolysis in presence TiO2 NPs on the antimicrobial activity seems to be more significant as the samples were laundered repeatedly in Launder-Ometer.
Antimicrobial activity of alkali treated PET/C blended fabrics loaded with TiO2, determined by shake flask method.

Treatment conditions: [NaOH], 1.5 mol L−1; duration, 60 min; temperature, 100°C; M:L, 1:20.
After one washing cycle; AATCC Test Method (61-1989).
It was found that, the bioactivity of the PET/C treated by NaOH in presence TiO2 NPs became significantly different (Table 8). The reduction in bioactivity of PET/C fabrics activated occurred progressively as the number of washing cycles increased. Under these treatment conditions fabrics had lost about 11%, its antimicrobial activity against Gram-positive bacteria B. mycoides after five Launder-Ometer washes.
Ultraviolet protection factor (UPF) of alkali treated PET/C blended fabrics and loaded with TiO2 NPs.

Treatment conditions: [NaOH], 1.5 mol L−1; duration, 60 min; temperature, 100°C; M:L, 1:20.
After one washing cycle; AATCC Test Method (61-1989).
CFU percentage of TiO2 loaded PET/C blended fabrics revealed that increases in TiO2 NPs from 0.2 to 1.0 g L−1 for all of the modified fabrics caused a significant enhancement in the % CFU values; this meant that the TiO2 NPs particles had a biocide effect to microbes, and it proved the antimicrobial property of the TiO2 NPs treated fabrics.
Several mechanisms were proposed for the antimicrobial activity of metal ions. It was reported that the toxic effects of metals on microorganisms can be due to antioxidant depletion, DNA damage, impaired membrane function and/or interference with nutrient assimilation. Some metal ions such as Cu2+, Co2+, Zn2+, and Ti2+ increase intracellular reactive oxygen species, leads to DNA damage, membrane activity disturbance, and inhibition of particular enzyme activities that are vital for cell growth. Metal ions bind to the proteins, enzymes and other electronegative chemical groups of the cell wall and cell membrane of the microorganism causing structural changes that disrupt the permeability leading to lyses of the cells.
Metal ions can access inside the bacterial cell, complexing with apoenzyme or displacing any metal ion that essential for the enzymatic action preventing protein synthesis and/or inhibiting nucleic acid synthesis. Metal catalyzed oxidation of several amino acids leading to loss of catalytic activity of the enzymes. Also, some metals activate cell autolytic enzymes leading to bacterial death.28,29
These results proved that, the feasibility of applying simultaneous treatment of PET/C blended fabrics with sodium hydroxide and TiO2 NPs as finishing method to produce PET/C fabrics imparted antimicrobial property.
Ultraviolet protection properties
The effect of activation of simultaneous treatment of PET/C fabric with NaOH and TiO2 NPs, on UV protection efficiency was investigated. The rate of UV protection was quantified and expressed via UPF values that are given in Table 8. It was found that, the UPF factor for pristine PET/C blended fabric was equal to 13.3 (Poor). Activation with alkali treatment in presence TiO2 NPs leads to deposition onto the above mentioned PET/C fabric surface NPs leading to a significant increase in UPF factor to the level corresponding to UPF rating of 45–50+ which assigns the excellent UV protection. After 10 washing cycles the UPF values for PET/C fabric was decreased to 40–50+, this assigns excellent UV protection. These results showed good laundering durability of PET/C fabrics activated with alkali and loaded with TiO2 NPs.
The UPF values of the nano-TiO2 loaded PET/C → H fabric revealed that, increases in TiO2 NPs for all of the alkali modified fabrics caused a significant enhancement in the UPF values; this meant that the TiO2 NPs particles blocked the UV rays, and it proved the UV protection property of the TiO2 NPs treated fabrics.
Conclusion
The present work confirmed the possibility of applying the simultaneous treatment of PET/C blended fabric with alkali in presence TiO2 NPs as a finishing method to bind NPs to the fabrics. Partial hydrolysis of PET/C fibers was accompanied by erosion of some parts of their surfaces. This leads to creation of COOH and OH groups as a result of dissociation some of ester bonds. At the same time, mercerization occurs for the cotton fibers. So that, alkali treatment had the ability to enhancement the binding efficiency of some TiO2 NPs to the surface of PET/C blended fabrics. On the basis of the obtained results, EDX measurements appeared a higher atomic weight % of Ti on partially hydrolyzed PET/C fabrics and loaded with Ti NPs. The FTIR charts of alkali treated PET fabrics showed appearing new peaks at the position 848 cm−1 with PET/C, which proves the ionic interaction between carboxylate and hydroxyl groups existed on the fabric surfaces and Ti present in the reaction medium. The factors affecting on the alkali treatment of PET/C fabric in presence TiO2 NPs have been studied. The effect of surface activation method on antimicrobial activity and UV protection efficiency of polyester fabrics was evaluated. It was found that the TiO2 NPs-loaded PET/C fabrics showed antimicrobial and UV protection properties. They exhibited a strong bioactivity and UPF values even after 10 washing cycles, indicating their excellent laundering durability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

