Abstract
The in-situ embedding of nano-palladium (PdNPs) within the viscose textile was hydrothermally carried out in different media (acid & alkaline) in order to gain multi-finishing properties. Cationization of viscose (Q-viscose) was performed using diallyl dimethyl ammonium chloride, to improve the fabric’s functionality. The size of PdNPs in the supernatants was quite small (6.9 & 10.3 nm), while aggregated particles of Pd was seen over the fabrics. Alkaline condition was shown to enhance the accessibility of viscose textile and consequently denser Pd masses were detected. Due to the in-situ embedding of PdNPs, color of viscose turned to yellow-brownish, while deeper color obtained for Q-viscose. UV protection factor (UPF) declare that; Q-viscose had very good to excellent UV-blocking (UPF = 38.9 – 49.7). UV-blocking was stand until 10 washing cycles and good to very good blocking (UPF = 20.3 – 33.6) was recorded. The modified Q-viscose exhibited very good microbial protection (against bacteria & fungi) and 85.4–87.8% from the microbes was dead. Partial durability for microbial reduction was obtained, while the microbial mortality reached 58.9–61.2% after 10 repetitive washings. Hence, long-lasting UV-blocking viscose textiles with antimicrobial character were successfully prepared via in-situ embedded of PdNPs.
Introduction
Our life depends on sunlight because it creates vitamin D, that supports the body’s physiological processes. 1 Due to its durability, vitamin D can be produced by the skin through a sequence of chemical reactions brought on by exposure to ultraviolet B (UVB) radiation from sunlight. 2 The proper timing of sun exposure stimulates metabolism, improves blood circulation, and boosts resistance to various infections. Sunburns, dermatitis, wrinkles, and skin cancer are all brought on by the sun’s ultraviolet radiation (UVR) penetrating human skin.3,4 Despite this, many people like to soak up the sun because it is good for their health and raises their risk of developing skin diseases over time because of UVR action. Sun protection, by avoiding the bad impact of sunlight with wearing protective clothes and accessories, is a great trend that helps to protect the human body from harmful sun radiation. 5 There are several ways to combat the harmful effects of UVR such as by wearing protective clothing, applying sunscreen, and reducing the amount of time you spend in the sun. 6 Regardless to sunscreen lotions, textiles and accessories such as hats, shoes, umbrellas, awnings, and baby carrier covers are the primary source of UV protection. 7 Previously, when it was realized that textiles could shield wearers from the spread of bacteria and diseases, the textile industry were considered with creating antimicrobial fibers. Consequently, textiles were developed for widespread use in a range of antimicrobial treatments and recently became essential for use in personal care, hotels, hospitals, and sports. Three additional essential requirements must be met by textile antimicrobial finishing in addition to getting rid of undesirable microorganisms and stopping the spread of illnesses, such as; (i) safety: the product must not be allergic or irritate skin when applied to textile materials; (ii) compatibility: the antimicrobial finishing agent used must not negatively impact textile industrialization; and (iii) durability: the reagent must be resistant to leaching, washing, and drying. Healthcare textiles are made for personal hygiene, health, and surgical applications. As a kind of medical clothing, antimicrobial textiles protect patients from sanitary problems caused by coming into contact with bacteria that cause infections or odors.8,9 The microbial growth can cause undesirable aesthetic changes or rotting damage, which reflects in bad effects on the textile performance. Consequently, the marketing of different antimicrobial textile finishing agents has been significantly increased. The structural composition, cost-effectiveness, processing technique, and environmental impact of these antimicrobial textile finishing agents is known to be varied. 10
Nanotechnology is numerously reported for production of functional textiles as a relatively new advancement in clothing technology. Textile-based nano-products combine all the advantages of nanomaterials to produce and modify high-values without sacrificing the comfort and flexibility of the substrates.11,12 Various textile materials, including fibers, fabrics, natural and synthetic substitutes, or conventional fabrics were acquired enhanced properties via chemical modification with nanoparticles and clothing via composing of nanofibers. 13 Because of their distinct physicochemical and biological properties, metal nanoparticles (MNPs) were identified as a subject of extensive research on textile functionalization. Unlike large, conventional additives, MNPs have remarkable surface properties that allow for greater effects, making them essential to this technological advancement. 14
The properties of nanostructures include antimicrobial, insect, oil/water, odor-control, fire retardant, heat-resistant, weather resistant, sensor-based, thermo-regulating, antistatic, electrically conductive, anti-wrinkle, self-cleaning, and conductive properties. Functionalization of textiles with nanostructures were extensively investigated via using metal oxide and metal nanoparticles for imparting various required functions such as UV-resistance, antimicrobial activity, photoactive, conductive and self-cleaning. 4 MnO2, MnFe2O3, ZnO, Cu2O, TiO2 and Al2O3 were applied onto natural, synthetic and blended textiles for imparting UV-protective properties, photocatalytic active, self-cleaning and antimicrobial activity.8,15–21 Furtherly, metallic nanoparticles including silver, palladium and gold were largely used for obtaining colored, antimicrobial, conductive and self-cleaned/photocatalytic active and UV protective textiles.22–25
MNPs are useful in many different fields of research because of their outstanding qualities. Although there are many different approaches that can be used for synthesis of NPs, they can be broadly categorized into two groups: (I) top-down approach and (II) bottom-up approach. 26 Nanoparticles are produced from bacterial, fungal, and plant sources using the bottom-up approach. Such type of NPs exhibits a wide range of technological and scientific uses. Micro – organisms and plants contain amines, phenolic compounds, carbohydrates, proteins, amino acids, aldehydes, and carboxylic acids that reduce metal (M+2) to metal (M0) nanoparticles.27,28 Palladium is one of the most active metals as a catalyst because it efficiently de-halogenates wastewater and reduces hydrogen. A unique catalytic activity is attributed to its electron structure and rapid absorption of hydrogen into bulk palladium. As a member of the platinum group of metals, palladium is considered a costly noble metal. Palladium is found in group 10 (Pd4d105s0) of the periodic table. The fifth O-shell in palladium’s electron configuration is entirely empty, which is a unique phenomenon that distinguishes its characteristics from those of elements in the 10 group. It is the least density and has the lowest melting point of all the metals in the platinum group. 29 Palladium dissolves slowly in hot or concentrated sulfuric acid and concentrated nitric acid, and it splits into hydrochloric acid.
An experimental technique for creating antimicrobial and excellent UV-protective viscose textile through in-situ immobilized PdNPs was presented in the current work. The in-situ immobilization of PdNPs was investigated under acidic and alkaline condition, while the effect of cationization was also studied. Estimation of the particle size in the supernatant solution was carried out by the transmission microscope. Pd-modified textile was investigated by scanning microscope, infrared, X-ray diffraction and X-ray photoelectron. The functions of the modified viscose were measured via evaluation of the colorimetric properties, UV-blocking character and antimicrobial protection. Additionally, the durability was tested via measurement the acquired functions after the repetitive washing.
Materials and experiments
Materials and chemicals
Laboratory-grade materials included palladium chloride (PdCl2, 99%, Sigma-Aldrich), sodium hydroxide, (NaOH, 99%, Merck), sodium carbonate (Na2CO3, ≥99.5%, Sigma-Aldrich), acetic acid (glacial, CH3COOH, ≥99.7%, Sigma-Aldrich), diallyl dimethyl ammonium chloride (C8H16NCl, DADMAC, ≥97%, Sigma-Aldrich) and non-ionic surfactant (commercial) were used as supplied. El-Nasr Company for spinning, weaving, and dying (El-Mahallah El-Kubra, Egypt) provided bleached plain weave viscose fabric.
Procedure
Viscose fabric cationization
According to published research, cationization was used for chemical modification of viscose textile. The experimental procedure was used to mix diallyl dimethyl ammonium chloride (DADMAC) with sodium hydroxide solution at a molar ratio of 2 (NaOH):1 (DADMAC). The viscose samples were padded with two dips and two nips of this mixture followed by squeezing to a wet pick-up of approximately 100%. 7 Ten minutes of drying at 80°C and 3 min of curing at 130°C were applied to the viscose fabric. Consequently, samples of cationized viscose (Q.V) were washed with tap water and 1% acetic acid, for neutralization. Fabrics were then subjected to several cycles of washing before being dried at 80°C. 30
In situ, implantation of PdNPs within viscose matrix
Details for preparation step of Pd-modified viscose fabrics.
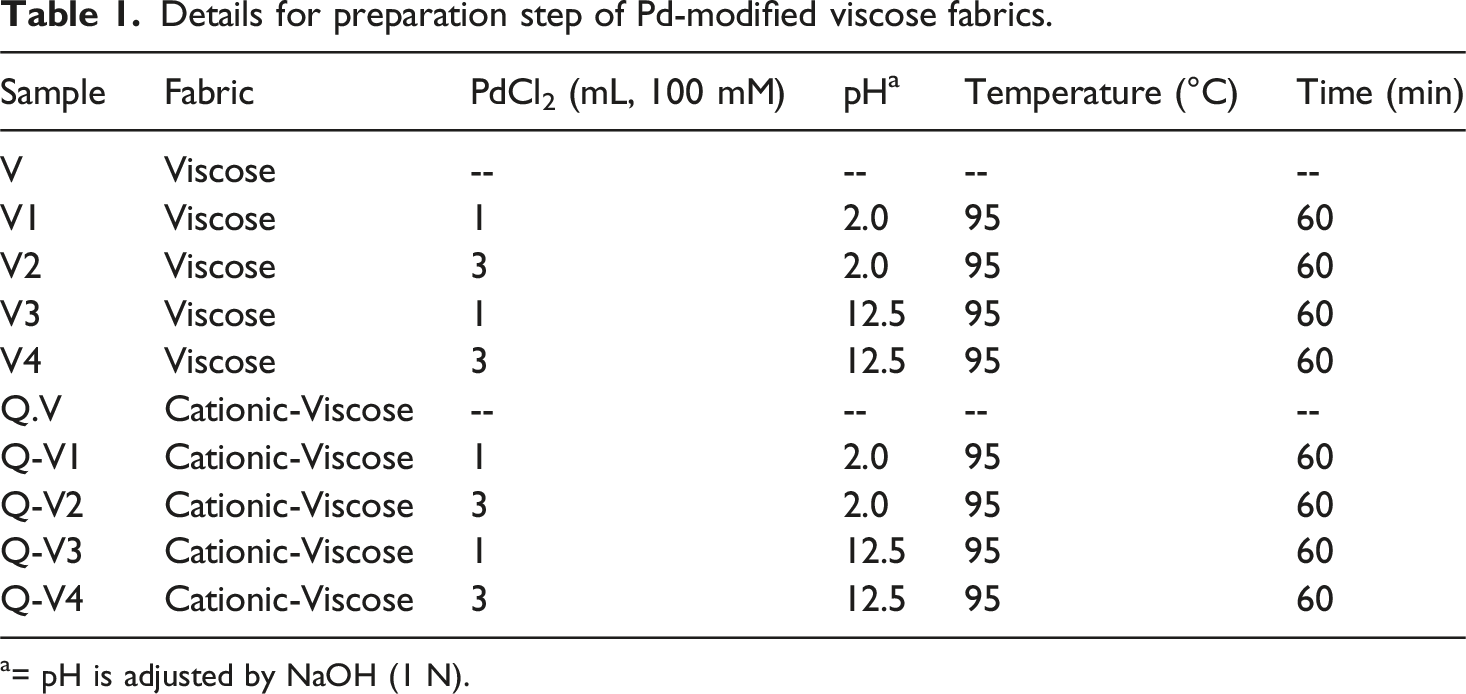
a= pH is adjusted by NaOH (1 N).
Characterization and instrumental analysis
PdNPs in the residual solutions were firstly confirmed by detecting UV-visible absorption spectra in the wavelength range of 200–800 nm via using the spectrophotometer (Agilent 5000 UV–Vis-NIR Systems, version 1.12). While, high-resolution transmission electron microscope (JEOL-JEM-1200; Japan) was used for evaluating the geometric form and size distribution of the PdNPs in supernatant. Size distribution of PdNPs was assessed using 4 pi analysis software (USA) for a minimum of 50 particles.
High-resolution scanning electron microscope (HRSEM Quanta FEG 250 with a field emission gun, FEI Company, Netherlands) was used to characterize the handled fabrics. Energy dispersive X-ray analyzer (EDAX AME-TEK analyzer) was utilized to measure the elemental analysis. Jasco FT/IR 6100 spectrometer was applied to detect the infrared spectra for the fabric. The applied scanning time and scanning rate was 64-time and 2 mm/sec, while 2 cm−1 resolution was used. Furthermore, fabrics were characterized by powder X-ray diffraction at room temperature using a Philips X'Pert MPD diffractometer system. While, the diffraction patterns were estimated monochromatically (Cu Kα X-radiation at 40 kV, 50 mA, and λ = 1.5406 Å). By using X-ray photoelectron spectroscopy (XPS, Kratos Ultra system), the valence state of palladium on the surface of viscose textiles was assessed. X-ray beam (Al Ka; 1486.6 eV) was used to irradiate the sample surface under extremely high vacuum. The remaining content and thermal characterization were evaluated using a TA Instrument TGA Q5000 with a heating rate of 10 K/min and an air flow of 25 mL/min. The sample’s mass was roughly 5 mg, and high-temperature platinum pans was used.
Pulsed xenon lamp (UltraScan Pro, Hunter Lab, USA) spectrophotometer was used to measure the colorimetric parameters (absorbance, color strength, L, a*, b* and whiteness) of the fabrics. While, L*, a*, and b* were the color coordinate parameters that represented lightness (black/white, 0/100), reddish/greenish ratio (+/ −), and the yellowish/blueish ratio (+/ −), respectively. 23 Two measurements were made in order to estimate the average values.
Before and after the successive preparation of PdNPs, the transmission spectral results (T%) for ultraviolet irradiation (UVR) over fabrics were estimated using a JASCO V-750 Spectrophotometer (Japan) with two nm intervals. AATCC test method 183-2010.44 was used to predict UV protection factor (UPF) and resistance in the UV-A (315–400) and UV-B (280–315) regions. In order to determine the average and standard deviation, estimation was done twice for every sample.
All modified viscose fabrics before and after washing underwent an antimicrobial test against three distinct pathogens using the “standard quantitative method”. 31 Three pathogenic strains were tested for the fabrics’ antimicrobial activity presented in Escherichia coli ATCC-25,922 (G-ve bacteria, E. Coli), Staphylococcus aureus ATCC-6538 (G+ve bacteria, St. Aureus) and Candida albicans ATCC-10,231 (fungi, C. Albicans). The microbial strains, meanwhile, were encouraged to grow in a suspensible form in the agar, while the plate filled with 100 μL of the microbial colloids. Following that, pieces of fabric were suspended in the plate and incubated for a day at 37°C under shaking. The absorbance of the samples was detected at 550 nm using “JASCO UV 630 spectrophotometer”, while the slipped calipers were used to estimate the reduction percentage in the microbial growth. For each sample, two repetitive experiments are performed, while the average and standard deviation are both considered.
Results and discussion
Self- immobilization of PdNPs
According to our knowledge, no previous studies were interested in in-situ immobilization of PdNPs within viscose for functionalization, whereas, embedding of PdNPs within silk and cotton was limitedly carried out using the hydrothermal process.11,32 The in-situ demonstration of PdNPs for long-lasting functional viscose finishing was studied in the presented work. The effect of in-situ-immobilization for PdNPs within viscose matrix in order to improve UV-resistance, antibacterial activity, and long-lasting coloring was investigated. On the other hand, fabrics were previously cationized by diallyl dimethyl ammonium chloride (DADMAC) to activate the functional groups of cellulosic materials, which increased the fabrics’ availability for contact with Pd ions and consequently stabilized and improved the self-embedding of PdNPs via steric or electrostatic stabilizations.
10
Since the cellulosic building blocks of viscose with the terminal OH- groups may help in reduction of Pd ions, while the produced PdNPs were directly embedded within the intermolecular spaces of fabrics.
33
In case of Q-viscose, cationizing group could share in the reduction and stabilization process and subsequently the embedded amounts of PdNPs may be higher. An illustration for the interaction mechanism of PdNPs with viscose textiles is presented in Figure 1. The schematic preparation for Pd-modified viscose fabrics.
UV–visible spectral mapping data
The initial confirmation for serial clustering of PdNPs was obtained visually by monitoring the color changes in the reaction liquors. The production of PdNPs was responsible for this color adjustment.34,35 Figure 2(a) represents the spectral data of the residual solution after removing the fabrics, as a function of pH, temperature, reaction time, and concentration, before and after cationization. The color of the reaction solution changed from light yellow to brown. The spectra declare that the specific Pd+2 peak at 284 nm is disappeared in case of using the alkaline medium (V3, V4, Q.V3, Q.V4) rather than the acidic medium (V1, V2, Q. V1, Q.V2), while new peak at 220 – 248 nm is detected. This is due to charge transfer from the cellulosic material reducible functional groups to Pd+2 during the redox reaction forming metallic Pd0 in the form of PdNPs. Characterization of PdNPs in the supernatants; 
TEM micrographs and size distribution
The morphology and geometry of the suspended PdNPs in the medium containing fabrics could be ascertained using TEM micrographs (Figure 2(b)), where the size distribution was estimated.36,37 Based on the spectra data (Figure 2(a)), two samples in the basic medium (V3, Q.V3) were selected considering viscose and cationized viscose. Data reveal that in the supernatant solution, PdNPs were effectively clustered with spherical shaped particles. The estimated size distribution and particle size were 3.0 – 12.0 nm (6.0 – 16 nm) and 6.9 nm (10.3 nm) in case of viscose (cationized viscose). The particle size of PdNPs was not considerably changed by using viscose of Q-viscose. This data is furtherly confirmed the successful clustering of Pd in nano-dimension by action of viscose fabrics as cellulosic material. While, viscose’s constituents demonstrated a strong propensity for redox interactions with palladium ions in order to nucleate the required NPs. Additionally, the obtained PdNPs were shown with polycrystalline structure including dots with structure of nan-shell. The selected area in electron diffraction with the shown arcs approved the high orientation crystal structure of PdNPs .
SEM pictures and EDX information
Following the in-situ immobilization of nanopalladium, SEM images were utilized to analyze the surface and geometrical properties of the textiles, as illustrated in Figure 3 (viscose) and Figure 4 (Q.vicose). SEM micro-images, energy dispersive x-ray (EDX) signals and elemental analysis were all utilized to verify the effect of conditions on the reliable embedding of PdNPs in the textile.7,11,38 The micro-images declared that the aggregated palladium particles were obviously seen over the textile fibrils, while much denser masses of palladium were observed by using alkaline medium compared with that of acidic medium. The amount of embedded PdNPs increased by increasing concentration of palladium and it observed higher in case of Q.viscose. From EDX analysis, the characteristic peak of palladium was detected beside those of carbon, oxygen and nitrogen approving that the observed particles are Pd. The reported observation supported the suggested mechanism and confirm the in-situ immobilizing of PdNPs in the viscose textile matrix. SEM micro-images and EDX analysis for the synthesized Pd-modified viscose fabrics; SEM micro-images and EDX analysis for the synthesized Pd-modified viscose fabrics; 

FTIR
The infrared spectroscopy (FTIR) is used to inform the chemical interaction of the applied materials, and to follow up how the treatment processing affected the structure. As seen from Figure 5, the native viscose textile showed five characteristic bands for O-H stretching vibration (3296 cm-1), aliphatic C-H symmetric stretching (2872 cm-1), carbonyl vibration (1608 cm-1), C-O-C asymmetric stretching (1359 cm-1) and C-C symmetric stretching (1018 cm-1), which referring to the cellulose structure.32,39 The all mentioned bands were retained after the successive in-situ growth of PdNPs which reflected that the fabrics’ structure is not obviously affected by PdNPs immobilization. In addition, two new bands are recorded at 877 and 1417 cm-1 assigning for O-Pd and consequently confirmed the role of fabrics’ functional groups in reduction of Pd ions inside the textile matrix. There is no obvious difference in the infrared spectral results between viscose and Q-viscose. FTIR spectral analysis for the Pd-modified viscose fabrics.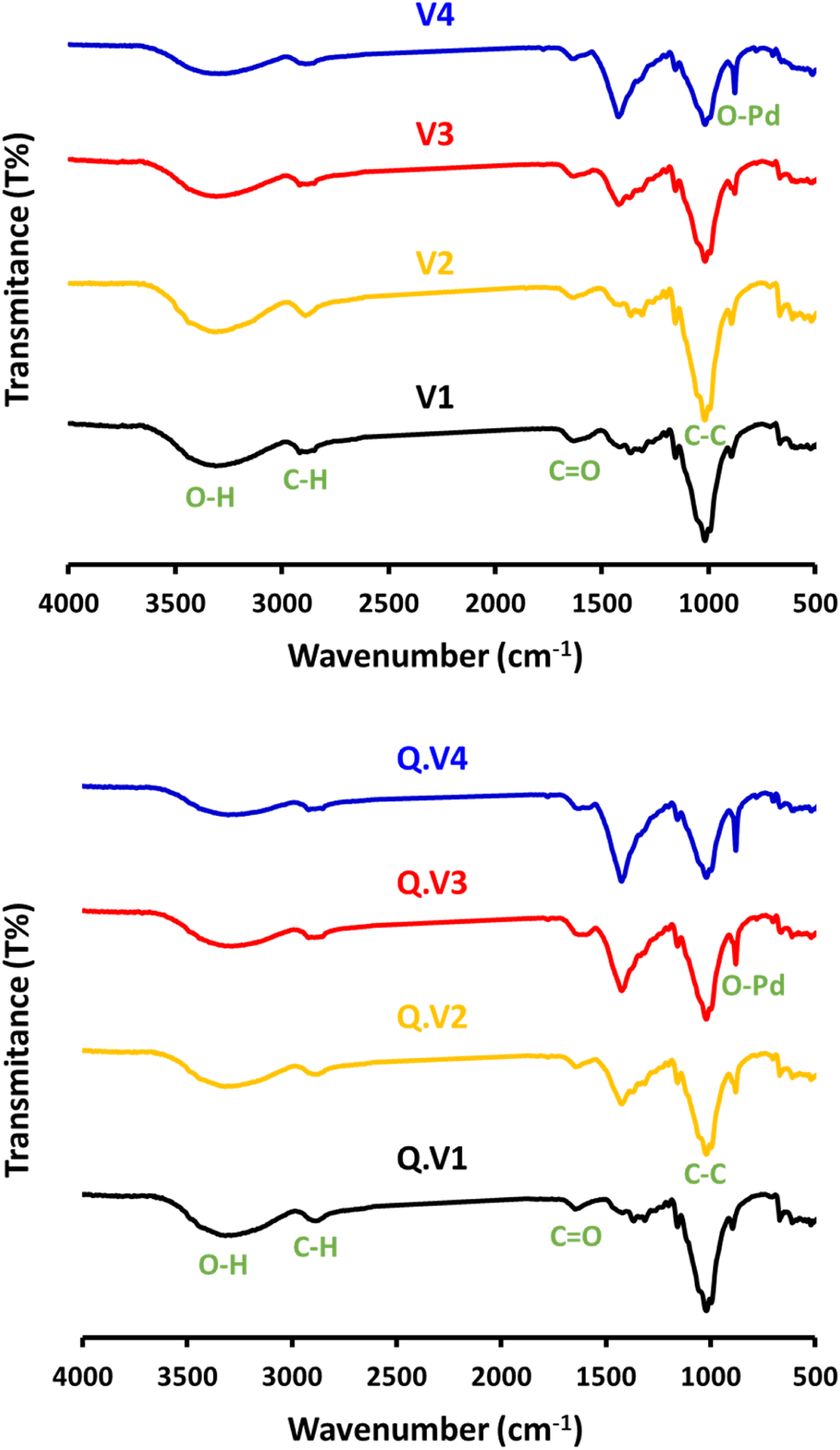
XRD patterns
Figure 6 depicts the XRD data for the textile fabrics before and after the in-situ embedded of PdNPs. Viscose textile exhibited two main diffraction bands at 2θ = 20.7°, and 22.6°, which are attributed to the crystal structure of cellulose II of viscose. While, new diffraction band at 2θ = 14.6° was detected for Q-viscose which reflects the changing in crystal structure of viscose after cationization process. By in-situ embedding of PdNPs, a new diffraction band at 2θ = 45.4° was appeared which a signing for (200) of the “face centered crystalline (Pd) (JCPDS data number 89–4897 card).11,22 The intensity of the characteristic band for PdNPs (45.4°) was increased by increasing the concentration of palladium and further increment was observed in case of Q-viscose. This confirms the influence of cationization in improving the fabric’s accessible for regulative implantation for more PdNPs. XRD analysis for the Pd-modified viscose fabrics.
XPS analysis
XPS spectral mapping for the Pd-modified viscose fabric (Q.V4) was analyzed (Figure 7) in order to give more additional confirmation about the chemical interaction between viscose and Pd as well as the ionic state of Pd.22,40,41 The study of the spectrum revealed that the binding energies of C1s, O1s, N1s were localized around 278-290, 526-536 and 393-405 eV, respectively. C1s bands were determined with binding energies of 282.1, 284.3 and 286.7 eV, which corresponded to C–H, C–C, and C–O, respectively. The two binding energy peaks recorded for O1s at 530.1 and 532.5 eV are referred to C-O and C-O-O, respectively. One binding energy band for N1s was detected at 399.5 eV related to N-H. Two characterized energy bands were seen for Pd3d at 336.2 and 341.5 eV assigning for 3d3/2 and 3d5/2 of metallic palladium (Pd0) which approved the successive in-situ immobilization of PdNPs within the viscose matrix. While, the energy band for Pd3d at 347.8 eV is corresponded to the 3d5/2 of Pd-O,
40
confirming the coordinative interaction between the embedded PdNPs and the alcoholic groups of the cellulosic material. XPS spectral analysis for the Pd-modified viscose fabric (Q.V4).
TGA data
Thermal stability is a significant property to explain the rearrangement and association of the cellulose chains in viscose, and how immobilization of PdNPs affected the thermal stability and physiochemical properties of textile. Both of the thermo-gravimetric (TG) percentage (%) and the thermal derivatives (dTG) percentage/rate (%/min) were analyzed for four samples (V, V4, Q.V and Q.V4) and were presented in Figure 8. The decomposition process of the all tested textile samples was observed mainly over two stages related to the dehydration and degradation of textiles. As seen for viscose, water removal was observed around 100-130°C with weight loss of 4.6 % and cellulose chains degraded at maximum temperature up to 345°C with weight loss of 49.9%, while the thermal derivative was −11.0 %/min at 345°C. The decomposition of viscose after immobilization of Pd was slightly improved, while the weight loss was 45.3% °C at 345°C with degradation rate of 13.6 %/min., the cationized viscose (Q-viscose) showed much higher weight loss (77.9%) at 345°C comparing with viscose due to the decomposition of cationic groups, while the weight loss was significantly reduced to be 48.6% after in-situ embedding of PdNPs. The obtained data showed the improvement in the thermal stability of the viscose textile after the immobilization of PdNPs and subsequently stabilize the viscose textile at the elevated temperature. Thermal analysis for the Pd-modified viscose fabric; 
Colorimetric analysis
Color measurements result for the Pd – modified viscose fabrics.


Color properties for the Pd-modified viscose fabrics;
UV-protection
Viscose is semi-synthetic textile that is broadly suitable for a variety of uses, due to their strong characters as breathability, smoothness, and biodegradation. However, there are some negative characteristics, such as high flammability, weak UV protection, and susceptibility to the microbial attack. According to recent reports, different types of nanoparticles, oxides of metals and metal organic complexes, along with certain polymer-based materials, were successfully used to insert distinctive functional qualities into textiles.39,42–46 Herein, the protection of ultraviolet radiation (UVR) was investigated for the viscose textiles after the in-situ immobilized PdNPs. UVR transmission (T%) was firstly evaluated as seen in Figure 10 and the UVR protection parameters (UPF, UVA and UVB) were all estimated and summarized in Table 3. Ultraviolet radiation (UVR) transmission through the Pd-modified viscose fabrics. Ultraviolet protection results for the Pd –modified viscose fabrics before and after washing.

The transmission was considerably lowered from 51.8–53.6% for viscose textile to 19.5–24.7% and 2.4–5.5% PdNPs modified viscose in acid and alkaline medium, respectively. For Q-viscose, further reduction in the transmission was observed by PdNPs immobilization, to be 4.5–15.1% and 1.9–3.2% for textile modified in acidic and alkaline medium, respectively. The estimated UPF for viscose/Q-viscose was gradually enlarged after PdNPs immobilization from 1.9/4.0 (in sufficient) to 4.3 – 5.0/14.7 – 16.5 (in sufficient) under acidic treatment and to 22.0 – 40.9/38.9 – 49.7 (good – excellent) under alkaline treatment. The evaluated data declare that the in-situ embedding of PdNPs enhanced the UV blocking character of viscose textiles, while higher UV protection was detected for the modified textiles in alkaline medium and at higher Pd concentration. The high accessibility and swelling of cellulose in alkaline medium are resulted in high Pd uptake which may explain the higher UV protection data for samples modified in alkaline medium.
As analogous with the published studies, the UV-blocking data for viscose textile by in-situ immobilization of PdNPs were obviously greater than those for other textiles impregnated with metallic/nano-metallic such as silver, copper, titanium,22,23,39,42–47 carbon nanostructures43,48 and Ln/Ca based metal organic framework.49,50 Similarly excellent UVR blocking was recorded for textiles modified with graphene oxide@AgNPs 51 and ZIF/MIL metal organic complexes.46,52 The UV protective agents are divided two classifications presented in organic compounds and inorganic compounds. The organic compounds characterized by the high absorption coefficient 53 and consequently they acted in absorption of the UVR. While, the inorganic compounds (including metal) improve the UV protection through reflection and distribution of the UVR by the metallic particles.46,54 Therefore, the embedded PdNPs is reported to act in reflecting the incident UVR and prevent its passing through the textiles, achieving excellent UV protection.
Antimicrobial activity
Biological activities for the Pd –modified viscose fabrics before and after washing.

Microbial protective textiles were recently obtained through immobilization of metallic particles including (Ag, Au),56–59 Ag@natural dye & Pd@natural dye, 22 oxides (TiO2, ZnO, Cu2O)60–67 and metal organic complexes.10,46,68 Considering the reported results of the mentioned materials, similar microbial protection data were observed in the presented study, regardless to the measurement used technique. The mechanism for the microbial protection of the Pd-modified viscose textiles could be explained due the effect of reactive oxygen species (ROS).69–76 The embedded PdNPs can generate ROS such as singlet O and OH radicals, which consequently suggested to act in the mortality of microbes via cell death. The generated ROS were supposed to interact and penetrate the cell wall of microbes followed by the oxidative stress motivation and leaded to inhibit the replication of gene. Furtherly, the ROS may cause mitochondria dysfunction, lipids oxidation, cell wall destroying (by action of necrosis or apoptosis) and finally the microbial cell is damaged.
Durability
The impact of repetitive washing on the UV-protection and antimicrobial performances for the viscose textile modified with PdNPs was investigated, while the results were shown in Table 3 and Table 4. UV-blocking was measured for viscose after 5 and 10 washing cycles. The UV blocking results were reduced after washing, and viscose without cationization (V3, V4) showed insufficient UV-blocking (UPF = 11.6 – 16.3) (Table 3). While, Q-viscose exhibited good – excellent protection (UPF = 29.2 – 48.7) and good – very good protection (UPF = 20.3 – 33.6) after 5 and 10 washings. These promising data reflect the stability/durability of the PdNPs within the textile matrix against the repetitive washing and very good UV-protection achieved for the modified textiles after 10 washing cycles.
The microbial activity was tested for the Pd-modified Q-viscose after 5 and 10 consecutive washes (Table 4). The microbial mortality was lowered after washing to be 43.9 – 52.1 % for Q.V2 and 69.4–72.7% for Q.V4 after 5 washes, while the mortality was further decreased to 41.9–50.9% and 58.9–61.2% for Q.V2 and Q.V4, respectively. The obtained results notified that, the in-situ embedded viscose textile was exhibited with good microbial protection after 5 washing and substantial microbial reduction after 10 repetitive washings, which reflects the reasonable antimicrobial durability. Cationization of viscose is significantly enhanced the durability of the gained functions (UV blocking and antimicrobial characters) through stability of and crosslinking with the in-situ immobilized PdNPs and prevent its easily leaching to the washing liquor.
Conclusion
Herein, multi-finished viscose textile (colored, UV blocking and antimicrobial) was successfully designed by the hydrothermally in-situ embedded PdNPs. The immobilization of PdNPs within textiles was studied in different media (acidic and alkaline medium) and the effect of cationization was also investigated. The in-situ embedded PdNPs was confirmed by the micrographs, XRD and XPS. The cationization process acts in improving the insertion of PdNPs and denser masses of particles were seen over the Q-viscose fibrils. The modification in alkaline medium showed much better results due to the increment of the accessibility of viscose which consequently improved Pd uptake. The color of viscose textiles was changed to yellow-brownish color and darker color observed for Q-viscose. After the in-situ embedding in alkaline medium, the Q-viscose textiles exhibited very – excellent UV blocking (UPF = 38.9–49.7). Q-visocse textile showed quite durable UV protection and good – very good blocking (UPF = 20.3 – 33.6) was determined after 10 repetitive washings. The microbial mortality against the tested bacteria and fungi, reached 85.4–87.8% for Q-viscose after embedding of PdNPs in alkaline medium. After 5 and 10 washings, the microbial mortality became 69.4–72.7% and 58.9–61.2%, respectively. The durability of the gained functions (UV-blocking and antimicrobial) is related to the stability of the embedded PdNPs within the viscose textile matrix against the consecutive washing, which was achieved by cationization to prevent its easy release during washing. It may eventually be stated that the hydrothermal embedding of PdNPs can be intriguingly applied to design the long-lasting excellent UV-blocking and antimicrobial viscose textiles. The in-situ process could be readily adapted to add durable functions to various textiles with manufacturing of textiles with multiple uses.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data are within the manuscript and available from the corresponding author upon request.
