Abstract
Mycelium-bound composites are new environmentally friendly, cost-effective and sustainable materials, enable energy-saving bio-composite fabrication, and provide an alternative to synthetic materials. Current research on mycelium-based composites reports on relatively coarse material compositions such as rice husks, cotton residues, sawdust, leaves and bio-waste, etc. According to research, very few publications report on mycelium-reinforced composites with the use of nanomaterials and this topic is under-researched and this study helps to fill this gap. The focus of this study deals with the preparation of mycelium-reinforced nanocomposites including nanofiber mats and the investigation of the different nanofiber mat morphologies on the growth of fungal mycelium. The mycelium macrofibers from Pleurotus ostreatus fungi were grown on polyacrylonitrile (PAN) nanofiber mats. Different morphologies of nanofiber mats such as fibrous and non-fibrous membrane areas or a mixture of both were used for mycelial growth with an additional nutrient. Moreover, mycelium/PAN nanocomposites were oxidative stabilized and carbonized and mycelium retains its morphology. For faster color differentiation between mycelium and nanofibers, PAN nanofiber mats were dyed in a one-step process by adding dye powder to the electrospinning solution as an additional tool. No significant differences in mycelial growth and morphology were observed regarding the different nanofiber mat types and the use of dye. These mycelium-reinforced nanocomposites are promising for many applications such as medicine and biotechnology, air and water purification and filtration, vertical farming, architecture, etc., and enable energy-saving bio-composite fabrication.
Keywords
Introduction
Mycelium-bound composites are promising materials for sustainable products and can be used in many fields such as packaging, insulation, air and water filtration, clothing, fashion, for vertical farming, furniture, architecture, medical, and biotechnological fields.1–6
Pleurotus ostreatus is a well-known edible mushroom that, due to its shorter growth period compared to most other mushrooms, is the second most cultivated mushroom in the world. 7 The fungus achieves very high profitability by converting a high proportion of the substrate into fruiting bodies. In nature, the fungus grows on diseased and dead trees and rarely on coniferous trees and readily uses many other different substrates as nutrients for its growth. 8 Preferably, Pleurotus ostreatus grows on lignin-containing substrates and is harvested faster on substrates with higher lignin content. In addition, cellulose, minerals, and nitrogen are also essential for the fungal fruit. 9 The mycelium of these fungi is the entirety of the vegetative fungal fibers, called hyphae, which consist of filamentous cells of a fungus and grow radially in an undefined manner by tip extension of individual hyphae.10,11 The mushrooms are rich in protein, possess antimicrobial properties, considered antioxidant, used for food production and treatment of oil-contaminated soils, and are thus promising for many technical applications.12–14 In the production of bio-composites, the fungal mycelium serves as a natural adhesive and as a connecting element, holding all the layers together. As layers, materials of synthetic and bio-based origin are suitable for mycelium composite formation, whereby the variety in the macro and nano range is almost unlimited.15,16
Needleless electrospinning is a process in which polymer fibers with diameters in the range of ten to several hundred nanometers are produced.17,18 In recent years, electrospinning has received increasing attention in scientific research and has found several technical applications. 19 The nanofiber mats can be formed from a wide range of synthetic and natural polymers as well as through the insertion of particles, numerous new variations of nanofibers can be produced in a simple electrospinning process. 20 Nanofiber mats, because of their large specific surface area, are also used for cultivating fibroblasts and microalgae. 21 In addition, nanofiber-based materials promote nutrient diffusion in culture media as well as the adhesion of the cultured cell due to their unique property, the large surface area compared to the volume, and the spatially complex structure. 22 For this reason, nanofiber mats are often used as substrates for various biological materials such as Chinese hamster ovary cells growing on PAN/gelatin nanofiber mats or nanofiber composite yarns with an immobilized antibacterial agent.23–26
Due to their unique properties, polyacrylonitrile (PAN) nanofibers are used in various fields, such as in air filtration as a precursor for carbon fiber, in the removal of heavy metals from contaminated water or in dye-sensitized solar cells.27–31 PAN is one of the few waterproof polymers that can be electrospun from relatively physiologically harmless solvents like dimethyl sulfoxide (DMSO) which enables its use in diverse medical and biological applications such as cell growth promotion.25,32,33 In addition, DMSO is a low-toxic solvent unlike the more common toxic solvents such as dimethylacetamide (DMAc), dimethylformamide (DMF) used in electrospinning, and DMSO solvent does not cause disposal problems.34–36 DMSO has the ability to increase penetration of the skin and to penetrate cell membranes and is therefore often used in drugs as a carrier substance for the introduction of active ingredients. 36 PAN is a commonly used polymer in electrospinning and a cost-effective and popular engineering polymer, which is well known due to its good chemical resistance, lightfastness, high dry and wet strength, bulk strength, elasticity, and good mechanical strength.37–41 Moreover, PAN is often used as a precursor in the manufacturing of carbon nanofibers and belongs to the most often used precursors for the production of carbon fibers due to its high carbon material yield.42–45 The precursors for the production of carbon fibers are based on fossil materials and this accounts for the high prices. Carbon can be produced from many biomaterials such as lignin, bio-waste, or agricultural residues but PAN is preferred because of the high yield of carbon material.46,47 Mycelium can also be regarded as a cost-effective precursor for the production of carbon material from biological sources and does not require energy to form mycelium fibers because natural fungal growth takes place. 48 Using electrospinning, polyacrylonitrile (PAN) nanofiber mats can be prepared and afterwards stabilized and carbonized to prepare carbon nanofiber mats which, by adding other materials, will be useful for several applications in biotechnological and industrial fields.49–52 The carbonization of PAN nanofibers with organic materials such as fungi can be used to expand the applications of mycelium-reinforced nanocomposites. 53 In the previous study, we found that the growth process of the mycelium on nanofiber mats is difficult to detect visually or with optical measurement methods, such as microscope, because the fungal mycelium has white color like the white nanofiber mat. 53 In general, PAN nanofibers are stained subsequently with basic dyes under acidic conditions, which frequently results in a limitation of the medical application. 54 In this study, nanofibers were manufactured in one step by adding dye to electrospinning solution and avoiding these limitation. As an additional tool, the nanofibers were dyed to allow better color differentiation between mycelium and nanofibers. The focus of this study consists in investigating the influence of different morphologies of nanofiber mats such as fibrous and non-fibrous membrane areas or a mixture of both on growth of mycelium. Furthermore, mycelium-enhanced nanocomposites were oxidatively stabilized and carbonized.
Overview of nanomaterials used in mycelium-reinforced nanocomposites
The mycelium-reinforced composites form a new class of low-cost and environmentally friendly materials in which natural fungal growth serves as an energy-efficient and biological method for producing composites, providing an alternative to man-made materials. 55 Current research on mycelium-based materials lacks essential details on manufacturing processes, material compositions, incubation, and growth conditions because fungal mycelium is a living organism and requires intensive research.56,57 In contrast to well researched and standardized processes for engineering composites, mycelium-bound composites represent a nowum. According to research, publications report on relatively coarse material compositions for mycelium-bound composites such as rice husks, oat hulls, rapeseed cake, cotton residues, sawdust, leaves and bio-waste, etc. and only a few on sensitive materials such as nanomaterials.3,58–62
In the study from Zhou et al., 63 novel self-assembled polycyclic aromatic hydrocarbons (PAH)-degrading fungal mycelia of the fungus (Penicillium oxalicum SYJ) and carbon nanotube (CNT) composites for pyrene removal were prepared and investigated. Jones et al. 64 upcycled agricultural by-products to chitin by enzymatic fungal growth and produced fungal microfibers with chitin-glucan nanofibers under ambient conditions. A new bio-ceramic hybrid for improved water treatment was developed by Ahn et al. 65 by functionalizing fungal mycelia with halloysite nanotubes to regulate hyphal dispersal and sorption behavior. Moreover, a scalable and simple method for structural modification of fungal hyphae was proposed that allows maximization of contaminant sorption as well as self-regulation of fungal pellet morphology. A Halloysite-doped mycelial architectures were formed by immersing Aspergillus fumigatus pellets in halloysite nanotube-dispersed water. Campaña and Arias 66 investigated the use of nanofibers of poly(ethylene oxide) (PEO) as a transport system for arbuscular mycorrhizal fungi (AMF) through the production of coated bean seeds. The results of the study showed that PEO nanofibers have no negative effect on the infectivity of AMF. As a promising alternative for seed coating material, nanofibers can be used for the development of easy-to-apply technologies for the application of AMF inoculants in agriculture, horticulture, ecological restoration, forestry, etc. In the study by Park et al., 67 manganese-oxidizing fungi were immobilized on nanofiber surfaces for the purpose of supporting remediation of heavy metal-contaminated water. Coniothyrium sp. and Coprinellus sp. fungal isolates from a superfund site (Lot 86, Farm Unit #1) water treatment system were incubated in nanofibers. It was found that these hybrid materials of fungal hyphae and manganese oxides enclosed in nanofibers adsorbed heavy metal ions from the water. Our previous study has shown that cultivation of the fungus Pleurotus ostreatus on electrospun nanofiber mats with additional nutrient substrates leads to different morphologies depending on the substrate and forms mycelium-reinforced nanocomposites. 53 According to research, the field of nanomaterials in the production of bio-mycelium composites is still limited, but the interest is steadily increasing in recent years.
Materials and methods
The materials and methods used in the following sections are described in more detail.
Electrospinning parameters
The needleless electrospinning machine “Nanospider Lab” (Elmarco Ltd., Liberec, Czech Republic) was implemented for the production of nanofiber mats. Polypropylene (PP) nonwovens (Elmarco Ltd.) were used as substrates for the nanofibers mats. The following spinning parameters were selected (Table 1):
Electrospinning parameters.

Electrospinning solution
The spinning solution for the production of nanofiber mats contained 16% polyacrylonitrile (PAN) (Woolworth, Unna, Germany). PAN was dissolved in DMSO (dimethyl sulfoxide, at least 99.9%, S3 Chemicals, Bad Oeynhausen, Germany) and electrospinning solution was stirred for 1 h at room temperature.
After the PAN has completely dissolved, the dye (Funfood4you, color: yellow-orange E110, fat-soluble) was added in small portions to the solution and then stirred for 2 h on a magnetic stirrer at room temperature. Table 2 gives a sample overview.
Sample overview.

Sample overview
Depending on the spinning parameters, different morphologies can be produced such as, that is non-fibrous membranes, nanofiber mats, or mixtures of both morphologies and have been reported in detail by Sabantina et al. 68 The distance between the high-voltage electrode and the substrate allows adjusting the ratio of membrane to fibrous areas. 68 The smaller distance leads to the formation of membrane areas, as the solvent cannot evaporate completely and the polymerization process is not finished when nanofibers already adhere to the substrate. In this study, different morphologies of nanofibers were attempted by altering electrospinning parameters. After electrospinning, the dyed nanofibers were placed in water for 30 min, dried and then showed no visible color changes compared to untreated reference samples. The water in which the samples were placed was also clear and no color residues could be identified.
Sample preparation
The samples were cut to the size of 5 cm × 5 cm to allow fitting in petri dishes. The PP nonwoven was cut together with nanofiber mats and placed in petri dishes for better handling. In addition, it facilitated the later removal of nanofiber mats from malt extract agar medium after mycelium growth. The pure nanofiber mat is too thin and would tear when removed from the petri dishes and malt extract agar.
Sterilization of nanofiber mats
Double-zip-closure freezer bags were obtained from Toppits, Cofresco Frischhalteprodukte GmbH, Minden, Germany. A closed box with an ozone generator (CHM GmbH, Amberg, Germany, operating according to ISO 105-G03) was used for sterilization of nanofiber mats for 40 min. Samples were then sealed inside the box in the double-zip freezer bag after the sterilization process to avoid contamination. Petri dishes were autoclaved at 121 °C for 40 min in a Systec-VX75 autoclave (Systec, Linden, Germany).
Inoculation of nanofiber mats with liquid mycelium
The nutrient medium for mycelial growth was malt extract agar with the following composition: 1 l deionised water, 24 g agar (Agar-Agar Kobe I, Roth, Karlsruhe, Germany), 20 g barley malt extract (Lindenmeyer GmbH & Co. KG, Weinsberg, Germany), and 1 g peptone (Peptone Water 77185, Sigma-Aldrich GmbH, Steinheim, Germany). After preparation of the liquid malt extract agar solution was autoclaved and placed in petri dishes with a diameter of 8.5 cm (VWR, Darmstadt, Germany). After this process, petri dishes were inoculated with the sterile liquid mycelium “Oyster Mushroom Culture XXL, BIO” (Pilz and Zubehör Shop, Münster, Germany) with 0.2 ml of the liquid mycelial culture on a nanofiber mat in the middle of the petri dish. To prevent sample drying and contamination, all petri dishes were sealed with parafilm (Pechiney Plastic Packaging, Chicago, IL, USA). Mycelial growth was performed and recorded at a temperature of 28 °C and a relative humidity of 30%–34% in almost complete darkness in a UN 75 universal oven (Memmert, Schwabach, Germany).
Characterization techniques
A confocal laser scanning microscope (CLSM) VK-8710 (Keyence, Neu-Isenburg, Germany) and a scanning electron microscope (SEM) Zeiss 1450VPSE (Oberkochen, Germany) with nominal magnification of 5000x were used for more detailed investigations. An Excalibur 3100 (Varian, Inc., USA) was applied for fourier transform infrared (FTIR) spectroscopy. Absorption spectra with a resolution of one data point per 2 cm−1 were recorded in the range between 4000 and 650 cm−1 and 32 scans were performed for each sample. For the investigation of the mycelium growth on dyed nanofiber mats, photographic images were taken for 29 days with an iPhone 6 (Apple, California, America) from a constant distance of 15 cm.
Oxidative stabilization parameters
To examine the possibility of carbonizing nanofiber mats and mycelium together into carbon composites, some of the PAN/mycelium composites were chemically stabilized and carbonized. Oxidative stabilization was performed in a muffle furnace B150 (Nabertherm, Lilienthal, Germany) at a typical stabilization temperature of 280 °C using a heating rate of 1 °C/min, followed by isothermal treatment for 1 h.
Carbonization parameters
For carbonization, a furnace (Carbolite Gero, Neuhausen, Germany) was used which approached a temperature of 500 °C for mycelium/PAN nanocomposites and 800 °C for PAN nanofiber mats with a heating rate of 10°C/min in a nitrogen flow of 150 ml/min (STP), followed by isothermal treatment for 1 h.
Results and discussions
The dyeing of PAN with yellow color powder was successful in first attempt, it served as an auxiliary tool for the differentiation of white mycelium and therefore the further dyeing attempts were not undertaken. Figure 1(a) demonstrates the covering of nanofiber mat with mycelium and difficulty of visual detection of the growth of mycelium on nanofiber mat. This color difference is important to distinguish when the required formation of defined nanocomposites is reached. Furthermore, this difference in color makes it possible to determine whether the mycelium covers the nanofiber mat relatively evenly and when the mycelium growth process can be stopped. Figure 1(b) shows the cross-section of mycelium on nanofiber mat. The white mycelium on white nanofiber mat makes it impossible to see the border between both components. Here it could be assumed that mycelium not only covers the surface of the nanofiber mat but also grows through the nanofiber mat and therefore it is reasonable to create a color difference between the composite components.

The mycelium on PAN nanofiber mat (a) and the cross section of mycelium on PAN and polypropylene substrate (PP) (blue color) on malt extract agar medium (b).
In the following Figures 2 to 4, the mycelium growth on the PAN nanofiber mat types 1–3 after 1 day (a), 8 days (b), 15 days (c), 21 days (d), 26 days (e), and 29 days (f) is shown more detailed. Figure 2 shows a fungal growth on nanofiber mats type 1 with predominantly fibrous areas. After 8 days, the fungal mycelium is very well identifiable and almost reaches the border of the petri dish.

The mycelium growth on PAN nanofiber mats type 1 with predominantly fibrous areas after 1 day (a), 8 days (b), 15 days (c), 21 days (d), 26 days (e), and 29 days (f).

The mycelium growth on PAN nanofiber mats type 2 with the mix of fibrous and non-fibrous membrane areas after 1 day (a), 8 days (b), 15 days (c), 21 days (d), 26 days (e), and 29 days (f).

The mycelium growth on PAN nanofiber mat type 3 with predominantly non-fibrous membrane areas after 1 day (a), 8 days (b), 15 days (c), 21 days (d), 26 days (e), and 29 days (f).
The mycelium on nanofiber mats type 2 with the mixture of fibrous and non-fibrous membrane areas does not grow as prolifically as on type 1 after 8 days (see Figure 3). However, after 15 days of growth, the mycelium also reaches the borders of the petri dish and does not differ from nanofiber mats type 1.
The mycelium on the nanofiber mat type 3 with predominantly non-fibrous membrane areas in Figure 4 appears to spread on malt extract agar rather than on the nanofiber mats, although all samples in the middle were seeded with liquid fungal mycelium. However, even here after 21 days, the growth of fungal mycelium does not differ from other nanofiber mate types.
After evaluation of the growth attempts, no substantial differences in mycelium growth and morphology were observed for these three different nanofiber mat types.
According to this study the mycelium usually grows star-shaped in all directions and tries to cover the surface extensively to counteract other fungi. Sometimes it occurs that the Pleurotus ostreatus fungus and other kind of fungi, for example mold, are to be found in a petri dish at the beginning of the growth investigation. Sometimes, despite careful sterilization of work surfaces and instruments, some petri dishes are contaminated because malt extract agar medium is attractive for a wide range of species such as mold, other fungi, and bacteria. After some time the foreign fungus is completely covered by Pleurotus ostreatus and in some cases it disappears completely or cannot develop further and remains only on a small spot. As can be seen in Figure 5, Pleurotus ostreatus fungus has successfully overgrown another fungus and conquered the area.

The mycelium growth on PAN nanofiber mats after 3 days (a), 13 days (b), and 25 days (c).
Figure 6 presents the growth of mycelium on yellow dyed PAN nanofiber mat type 1 with predominantly fiber areas (6a) and for comparison the mycelium is shown on non-dyed PAN nanofiber mat (6b) after 1 h drying at 60 °C.

The CLSM images of the mycelium on yellow dyed PAN nanofiber mat (type 1 with predominantly fiber areas (a) and not dyed PAN nanofiber mat (b)) after 1 h drying at 60 °C. Scale bars indicate 10 μm.
No significant differences in mycelial growth and morphology were observed on dyed and non-dyed nanofiber mats.
Figure 7 shows CLSM images of the mycelium grown on the PAN nanofiber mat type 2 with the mix of fibrous and non-fibrous membrane areas (Figure 2(a)). In some parts it is visible that mycelium grows through the nanofiber mat, unambiguously forming a composite. The thicker mycelium fibers can be clearly distinguished from the thinner nanofibers in the mat. The growth of mycelium on all three types of nanofiber mats produced in this study showed no significant differences and therefore only one type of nanofiber mat is shown here. These areas shown here on CLSM images were explicitly selected to show nanofibers and mycelium fibers on one image. This occurred when the mycelium has not yet covered the entire nanofiber mats area and has not yet reached the borders of the petri dish.

The CLSM images of mycelium on PAN nanofiber mats with the mix of fibrous and non-fibrous membrane before stabilization (a), after stabilization (b), and after carbonization at 500 °C (c). Scale bars indicate 10 µm.
In all test series and all nanofiber types from 1 to 3, the mycelium has well colonized the nanofiber mats surface, which is illustrated in Figures 2 to 4. The mycelium remains very well preserved after oxidative stabilization and carbonization as observed in previous studies. 53 Apparently, the stabilization process changes the mycelium morphology, similar to the well-known change of the nanofiber structure during stabilization. 69 These findings will have to be investigated in more detail in a future study.
Figure 8 shows the morphology change after stabilization and carbonization processes. In order to investigate dimensional stability of dyed nanofibers by changing electrospinning parameters, they were investigated without mycelium.
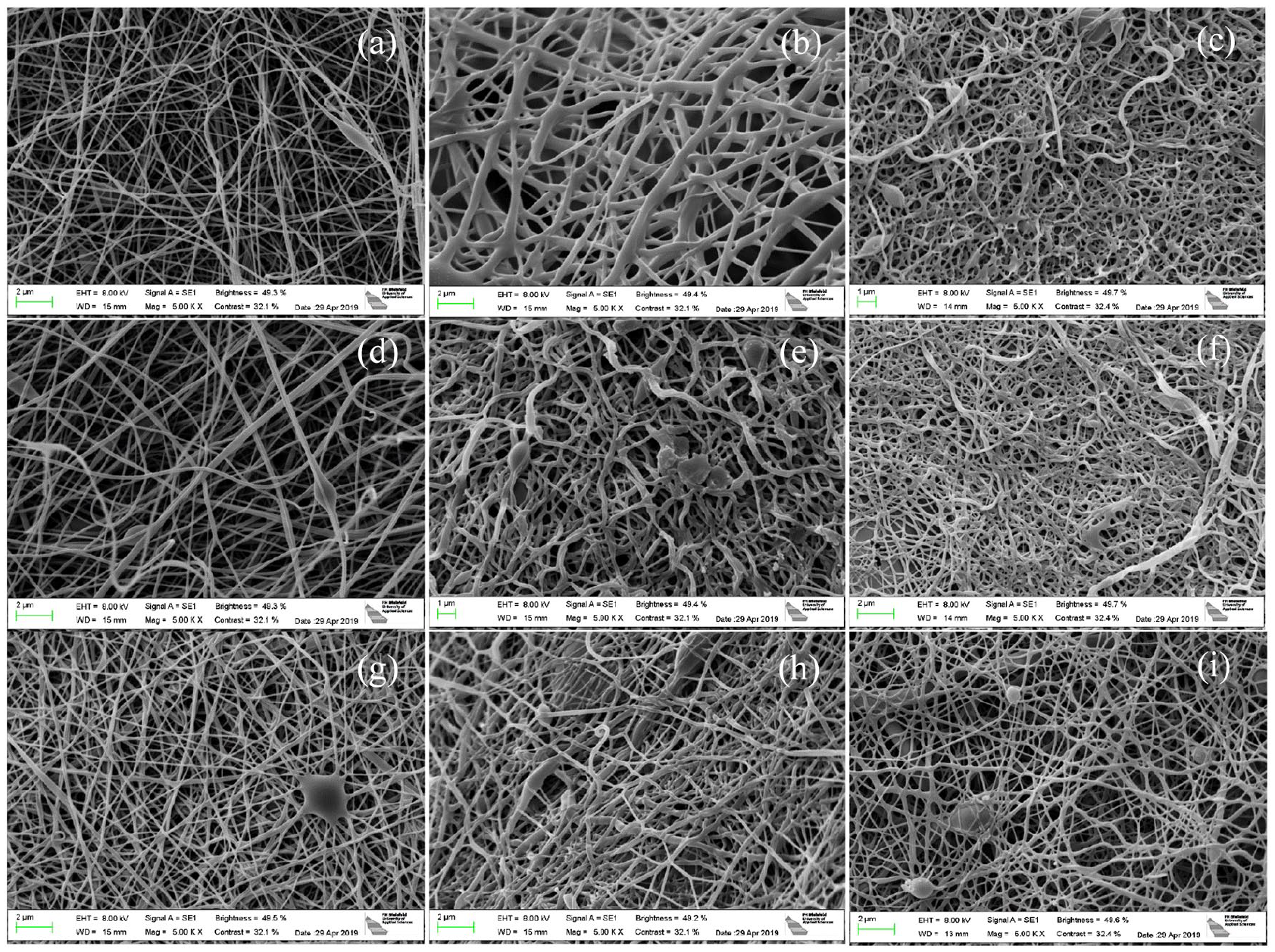
The SEM images of PAN nanofiber mats without mycelium. PAN nanofiber mats type 1 with predominantly fibrous areas after electrospinning (a), stabilized (b), and carbonized (c); PAN nanofiber mats type 2 with the mix of fibrous and non-fibrous membrane areas after electrospinning (d), stabilized (e), and carbonized (f), PAN nanofiber mat type 3 with predominantly non-fibrous membrane areas after electrospinning (g), stabilized (h), and carbonized (i).
The nanofiber mats shown in Figure 8 were not fixed which usually leads to a change in morphology toward meandering fibers with adhesions along the crossing points that also leads to more visible beads per displayed area. 69 Interestingly, all samples do not show these typical observations, only a few beads are visible and the fibers remain relatively straight after stabilization and carbonization. The samples with membrane content show nearly no beads and do not form meandering fibers with adhesions at the crossing points. Probably the dye has an influence here and gives the dimensional stability of the samples. In addition, the areas with membranes (not depicted here) should offer additional stability.
The results of the FTIR measurements for chemical analysis are shown in Figure 9 after electrospinning, oxidative stabilization, and carbonization processes. All FTIR results show the same effects since the samples were prepared with the same polymer solution, but only the electrospinning parameters differ.

FTIR measurements after electrospinning, stabilization, and carbonization of PAN nanofiber mats type 2 with the mix of fibrous and non-fibrous membrane areas. The graphs are vertically offset for clarity.
All samples show the typical PAN absorption peaks. A stretching vibration of the nitrile functional group C≡N at 2240 cm−1, a carbonyl (C=O) stretching peak at 1732 cm−1, esters (C-O and C-O-C) vibrations of the co-monomers such as itaconic acid or methyl acrylate in the ranges of 1230–1250 cm−1 and 1050–1090 cm−1 as well as bending and stretching vibrations of CH2 at 2938, 1452, and 1380 cm−1 additionally. 70
During stabilization, most of these peaks are replaced by new ones: large peaks of C=N stretching vibrations can be seen at 1582 cm−1 and C=C stretching vibrations at 1660 cm−1 as well as C-H bending and C-H2 wagging at 1360 cm−1. 71 The peak around 800 cm−1 belongs to the aromatic C-H oscillations, which are caused by oxidative dehydrogenation in the presence of oxygen. 72 After carbonization, most of the high peaks observed during oxidative stabilization either decreased or were replaced by other peaks (Figure 9).
Figure 10 shows the results of mass yield after temperature treatment of pure PAN nanofiber mats and mycelium/PAN nanocomposites.

The comparison of mass yield between PAN nanofiber mats and mycelium/PAN nanocomposites.
After oxidative stabilization at 280 °C, a material yield of PAN nanofiber mats of approximately 58% was achieved compared to the samples after electrospinning, while carbonization at 800 °C resulted in a material yield of approximately 42% compared to the stabilized samples.
For mycelium/PAN nanocomposites, a much higher material yield of approximately 72% was achieved after stabilization at 280 °C compared to pure PAN nanofiber mats. After carbonization, however, the material yield was much lower and amounted to approximately 40%. Overall, according to these results, the final material yields of pure PAN nanofiber mats and mycelium/PAN nanocomposites after carbonization do not differ significantly and are of approximately the same order of magnitude. It could be concluded that in this case the mycelium does not have a significant influence on the final material yield and mass loss of the whole composite.
Conclusions
The majority of publications report relatively coarse material compositions for macroscale mycelium-bound composites and the use of nanomaterials is under-researched and relatively new. The present study deals with the use of nanofibers in mycelium-reinforced bio-composites and contributes to closing this gap. The focus of this work concentrates on the reinforcement of sensitive PAN nanofiber mats with mycelium from Pleurotus ostreatus and the formation of mycelium-reinforced nanocomposites. The three different types of nanofiber mats such as predominantly nanofiber areas, the mix of fibrous and non-fibrous membrane areas, and predominantly membrane areas were produced and, according to this study, do not exert a direct influence on mycelium growth or morphology.
The nanofibers were dyed in a one-step process by adding dye powder to the electrospinning solution and serves as an additional benefit. According to this study, the dye does not appear to affect either the morphology of the nanofiber mat or the growth of the mycelium of Pleurotus ostreatus.
Furthermore, this mycelium/PAN nanocomposite can be used as a cheap, bio-manufactured precursor for the production of carbon materials. In addition, the mycelium can mechanically strengthen PAN nanofiber mats by forming mycelium on the surface of the nanofiber mats and the mechanical properties of such composites will be investigated in further studies in the near future. The mycelium-reinforced nanocomposites open up many industrial fields of application and enable energy-saving, environmentally friendly bio-fabrication.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partly funded by the European Regional Development Fund (EFRE), Start-Up project “Tredico”. We acknowledge support by the Open Access Publication Fund of the University of Applied Sciences Bielefeld.
