Abstract
Varicocele can lead to impaired semen parameters and induce infertility. Varicocelectomy is considered the gold standard for varicocele treatment. However, its impact on improving assisted reproductive technologies (ARTs) outcomes remains contentious. This study seeks to compare reproductive outcomes between infertile men who underwent varicocelectomy and those who did not prior to ARTs. In addition, it evaluates the influence of types of fertilization, couples’ ages, and treatment years on clinical pregnancy rates. A comprehensive search was conducted through February 2023 across eight electronic databases using combinations of relevant keywords. Observational and randomized controlled trials (RCTs) were included. Fourteen eligible studies (n = 1,705) were included: 12 retrospective studies (n = 1,467) and 2 prospective studies (n = 238). Pooled results indicated that infertile men who received varicocelectomy had a significantly higher clinical pregnancy rate compared to the control group (OR: 1.38, 95% CI [1.05, 1.83]; I2 = 36%, p = .02). The live birth rate was also significantly higher in the treatment group (OR: 2.18, [1.58, 3.01]; p < .00001; I2 = 0%). However, miscarriage rates did not significantly differ (OR: 1.07; [0.57, 1.98]; I2 = 0%, p = .84). Varicocelectomy significantly improved sperm concentration and normal morphology rate, but did not result in a significant enhancement in sperm motility. Men who underwent varicocelectomy between 2012 and 2019 and subsequently received intracytoplasmic sperm injection exhibited improved outcomes, particularly when their female partners were under 30 years of age. Larger, high-quality RCTs are needed to clarify benefits or avoid unnecessary treatments.
Introduction
Varicocele, characterized by the abnormal dilation of the pampiniform plexus veins in the scrotum, is a prevalent condition associated with male infertility (Jellad et al., 2021; Santana et al., 2019; Y. Zhang et al., 2022). Early research indicated that men with varicocele had higher scrotal temperatures compared to those without varicocele (Garolla et al., 2015; Goldstein & Eid, 1989). It was previously hypothesized that the testicular hyperthermia caused by varicocele might be associated with impaired sperm parameters (Garolla et al., 2015; Goldstein & Eid, 1989). Past studies have supported that infertile men with varicocele have higher levels of reactive oxygen species (ROS), nitric oxide, and lipid peroxidation products (Cho et al., 2016; Mostafa et al., 2009). In addition, elevated ROS levels are related to impaired sperm concentration, motility, morphology, and sperm DNA fragmentation (SDF) (Agarwal et al., 2014).
Varicocelectomy has been considered the gold standard for varicocele therapy and offers a better prognosis when compared with other treatments (Baazeem et al., 2011; Birowo, Tendi et al., 2020; Johnson & Sandlow, 2017). Numerous studies and systematic reviews have demonstrated the benefits of varicocelectomy in reducing SDF and enhancing semen parameters such as sperm concentration, motility, and morphology (Al-Mohammady et al., 2019; Birowo, Rahendra Wijaya et al., 2020; Inci et al., 2009; Nakonechnyi et al., 2022; Soetandar et al., 2022). Varicocelectomy can also enhance the chances of spontaneous pregnancy (Peng et al., 2015). However, the development of advanced assisted reproductive technologies (ARTs) has enabled many infertile couples to achieve pregnancy (Inhorn & Patrizio, 2015). ARTs can also serve as an alternative to varicocelectomy for managing clinical varicocele with abnormal semen parameters (Sonmez & Haliloglu, 2018). Some reports suggested that despite improved semen quality post-surgery, a significant number of patients still require ARTs to achieve pregnancy (J. R. Kohn et al., 2020; T. P. Kohn et al., 2017). The reason might be that varicocelectomy typically does not fully restore normal sperm parameters in men. The procedure often improves semen parameters to a degree that allows men to attempt intrauterine insemination (IUI), whereas previously their sperm counts would have only qualified them for in vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI) (Dubin et al., 2018; Wang et al., 2019). Although the majority of patients opt for laparoscopic and microsurgical varicocelectomy due to its minimally invasive nature compared to open surgery, the risk of complications such as hematoma, wound infection, postoperative pain, and recurrence persists (Ameli et al., 2019). Therefore, it is critical to understand whether varicocelectomy can significantly improve ART outcomes, or whether it merely serves as an adjunct to these techniques.
Moreover, clinicians remain cautious regarding the potential impact of clinical parameters of varicocelectomy on reproductive outcomes until further evidence becomes available (Al-Mohammady et al., 2019; J. R. Kohn et al., 2020). Previous studies offered additional insights into optimizing outcomes through patient selection and individualized treatment approaches (Jensen et al., 2017; J. R. Kohn et al., 2020). Personalized treatment plans should consider the patient’s specific condition, including fertilization type and the age of the patient (Carson & Kallen, 2021; J. R. Kohn et al., 2020). Given the varying patient conditions, the substantial costs, and the potential risks associated with varicocelectomy, this understanding is critical for identifying which type of patients are most likely to benefit from varicocele treatment, particularly those who have already planned to seek the assistance of ART (Al-Mohammady et al., 2019; Inci et al., 2009; Pasqualotto et al., 2012).
Therefore, this meta-analysis aims to systematically evaluate the existing evidence on the impact of varicocelectomy on ART outcomes. By aggregating data from multiple studies, this analysis seeks to identify which patient populations are most likely to benefit from surgical intervention prior to ARTs. This comprehensive review will contribute to more informed clinical decision-making and optimize treatment strategies for couples facing infertility challenges.
Methods
Search Strategy
A comprehensive search was performed in the English databases of PubMed, Ovid, Medline, Embase, and Cochrane Library, as well as Chinese databases of China National Knowledge Infrastructure, and Cqvip and Wanfang. The date restriction was set up to February 2023. Medical Subject Headings terms included: “Assisted Reproductive Technique” and “Varicocelectomy.” The search strategy is detailed in Table 1.
Search Strategy

Inclusion and Exclusion Criteria
Inclusion criteria: (a) the case group was infertile male patients treated with varicocelectomy; (b) the control group was infertile male patients without treatment; (c) the original available data were provided in articles.
Exclusion criteria: (a) reviews, case reports, commentaries, and letters to the Editor article types; (b) duplicated publications; (c) studies without a control group; (d) subjects without ART utility data; (e) subjects who conceived naturally.
Data Extraction and Quality Assessment
Two researchers independently and carefully extracted available information from each eligible study: (a) author’s last name, (b) year of publication, (c) country, (d) study design, (e) sample size of cases and controls, (f) surgical approaches, (g) male age, (h) female age, (i) control, (j) fertilization type, (k) disease, (l) grade of varicocele, (m) semen characteristics, and (n) outcome measures. Quality assessment was independently conducted by two investigators, with discrepancies resolved by a third investigator. The risk of bias in randomized controlled trials (RCTs) was appraised using Cochrane’s tool. The quality of cohort studies was evaluated using the Newcastle-Ottawa Scale, with scores ranging from 0 to 9 points, encompassing three domains: selection, comparability, and exposure.
Statistical Analysis
The extracted data were analyzed with Review Manager 5.3 software (Cochrane Collaboration, Oxford, U.K.). The Mantel-Haenszel method and random-effects models were used for meta-analysis. The effect sizes were expressed as odds ratios (ORs) and calculated using their 95% confidence intervals (CIs). Summary ORs and 95% CIs were assessed graphically with forest plots. Heterogeneity was quantified by the I2 value. Sensitivity analysis was used to assess whether there was any study affecting the stability of the results. A p-value < .05 was considered statistically significant.
Results
Database Search and Characteristics of Included Studies
As shown in Figure 1, we first searched 838 publications (154 from CNKI, 49 from Cqvip, 142 from Wanfang, 154 from PubMed, 22 from Ovid, 141 from Medline, 140 from Embase, and 36 from Cochrane Library), of which 298 were removed as duplicates. After record screening, 519 studies were excluded for not fulfilling the experimental criteria. Among these, seven articles were excluded after reading the title and abstract and confirming that they did not meet the inclusion conditions were matched. We then downloaded 14 selected articles (13 English and 1 Chinese) and analyzed them carefully. The main characteristics of the included studies are shown in Table 2. The publication dates of the eligible studies ranged up to 2023. The number of patients ranged from 39 to 306. Two studies were prospective studies (Al-Mohammady et al., 2019; Ketabchi et al., 2018), and twelve studies were retrospective studies (Baazeem et al., 2009; Boman et al., 2008; Daitch et al., 2001; Esteves et al., 2010; Gokce et al., 2013; Haydardedeoglu et al., 2010; Huan et al., 2019; Inci et al., 2009; Marmar et al., 1992; O’Brien et al., 2004; Pasqualotto et al., 2012; Zini et al., 2008). RCTs were reported in one of the two prospective studies. The quality assessments for the RCTs and cohort studies were summarized in Tables 3 and 4.
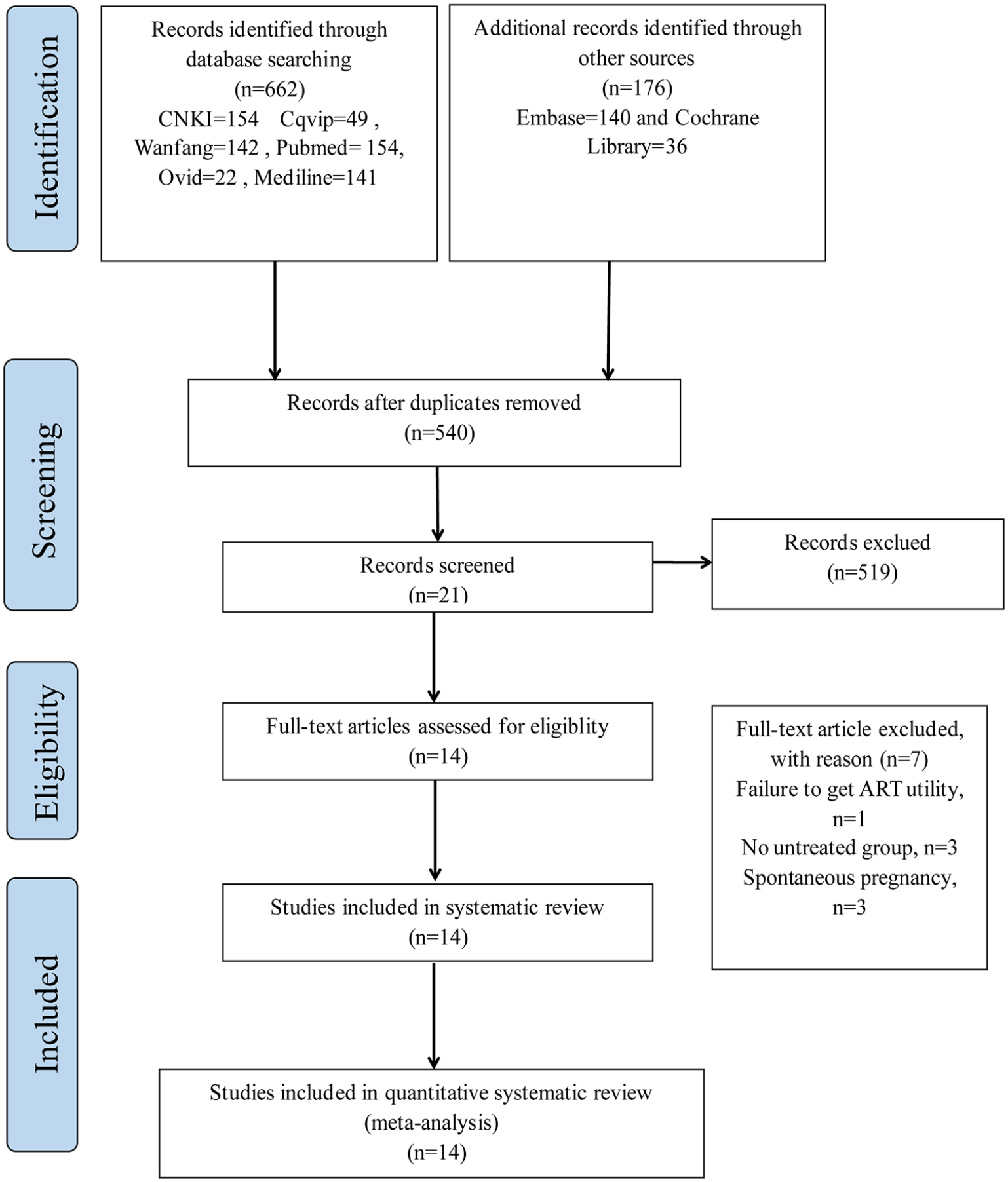
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Flowcharts
Main Characteristics of the Included Studies

Note. ICSI = intracytoplasmic sperm injection; IUI = intrauterine insemination; TESE = testicular epididymal sperm extraction; IVF = in vitro fertilization.
Risk of Bias Assessment of the Randomized Controlled Trials for Meta-Analysis Using the Cochrane Tool

Quality Assessment of the Cohort Studies for the Meta-Analysis Using the Newcastle-Ottawa Scale

Effects of Varicocelectomy on the Reproductive Outcomes of Infertile Patients
The clinical pregnancy rate outcomes were based on the data derived from 14 studies (872 cases and 833 controls). In retrospective studies, there was no significant difference between the two groups (OR: 1.32, 95% CI [0.95, 1.83]; p = .10; heterogeneity; I2 = 42%). In prospective studies, the clinical pregnancy rate was significantly higher in the case group than in the control group (OR: 1.79, [1.05, 3.06]; p = .03; heterogeneity; I2 = 0%). Overall, the clinical pregnancy rate was significantly higher in the case group than in the control group in a total of 14 studies (OR: 1.38, [1.05, 1.83]; p = .02; heterogeneity; I2 = 36%; Figure 2A).

Meta-Analyses of the Effect of Varicocelectomy on the Reproductive Outcomes of Infertile Patients with Varicocele. (A) Clinical Pregnancy Rate. (B) Live Birth Rate. (C) Miscarriage
The live birth rate outcomes were based on the data derived from five studies (348 cases and 398 controls). The live birth rate was significantly higher in the case group than in the control group (OR: 2.18, 95% CI [1.58, 3.01]; p < .00001; heterogeneity; I2 = 0%; Figure 2B).
However, the miscarriage rate outcomes were based on the data derived from three studies (284 cases and 250 controls). No difference was found in the miscarriage rate between the case and control groups (OR: 1.07, 95% CI [0.57, 1.98]; p = .84; heterogeneity; I2 = 0%; Figure 2C).
The Effects of Varicocelectomy on Sperm Parameters
The sperm concentration was significantly higher in the case group than in the control group (OR: 1.86, 95% CI [1.37, 2.35]; p < .00001; heterogeneity; I2 = 100%). The motility rate of sperm was significantly lower in the case group than in the control group (OR: −3.10, [−3.43, −2.76]; p < .00001; heterogeneity; I2 = 100%). The morphology rate of sperm was significantly higher in the case group than in the control group (OR: 0.32, [0.22, 0.43]; p < .00001; heterogeneity; I2 = 99%, Figure 3).
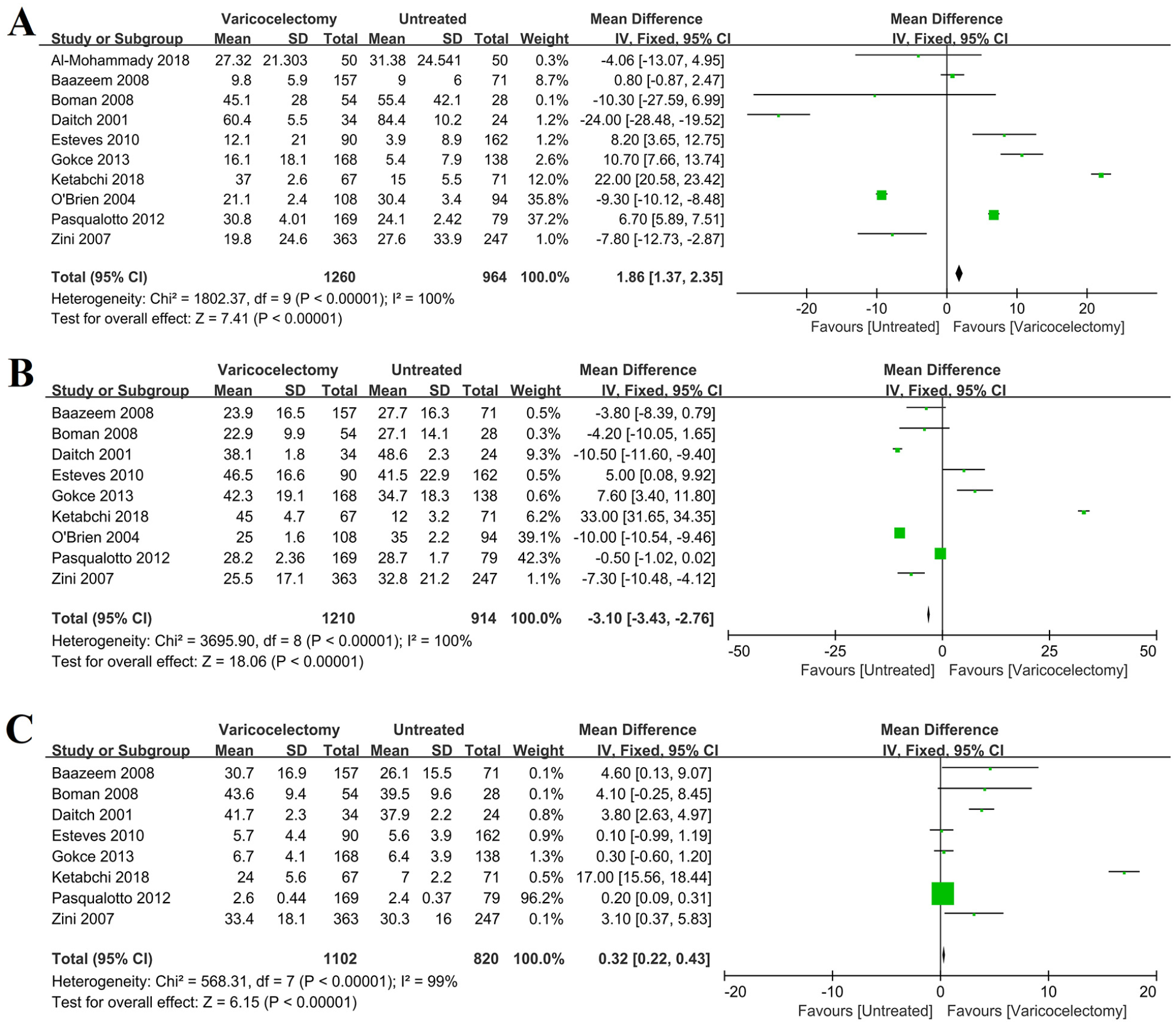
Forrest Plot for the Effect of Varicocelectomy on the Sperm Parameters. (A) sperm concentration. (B) motility rate of sperm. (C) morphology rate of sperm.
The Clinical Pregnancy Rate in Studies with Different Fertilization Types
No significant difference was found in the clinical pregnancy rate between the case and control groups when the fertilization type was IUI (OR: 1.65, 95% CI [0.65, 4.20]; p = .29; heterogeneity; I2 = 51%). When the infertile patients were treated with ICSI, the clinical pregnancy rate was significantly higher in the case group than in the control group (OR: 1.43, [1.01, 2.02]; p = .04; heterogeneity; I2 = 29%). No significant difference was found in the clinical pregnancy rate between the case and control groups when the fertilization type was ICSI with testicular epididymal sperm extraction (TESE) (OR: 2.20, [0.96, 5.04]; p = .06; heterogeneity; I2 = 0%; Figure 4).

Forrest Plot for the Effect of Varicocelectomy on the Clinical Pregnancy Rate in Studies with Different Fertilization Types
The Clinical Pregnancy Rate in Studies with Different Male and Female Ages
Regardless of whether the mean age of infertile male patients was less than 30, between 30 and 35, or more than 35 years, no significant difference in the clinical pregnancy rate was observed between the case and control groups (Not applicable, p = .13; OR: 1.34, 95% CI [0.63, 2.86]; p = .44; heterogeneity, I2 = 66%; OR: 1.22, [0.83, 1.79]; p = .31; heterogeneity, I2 = 29%; Figure 5A).
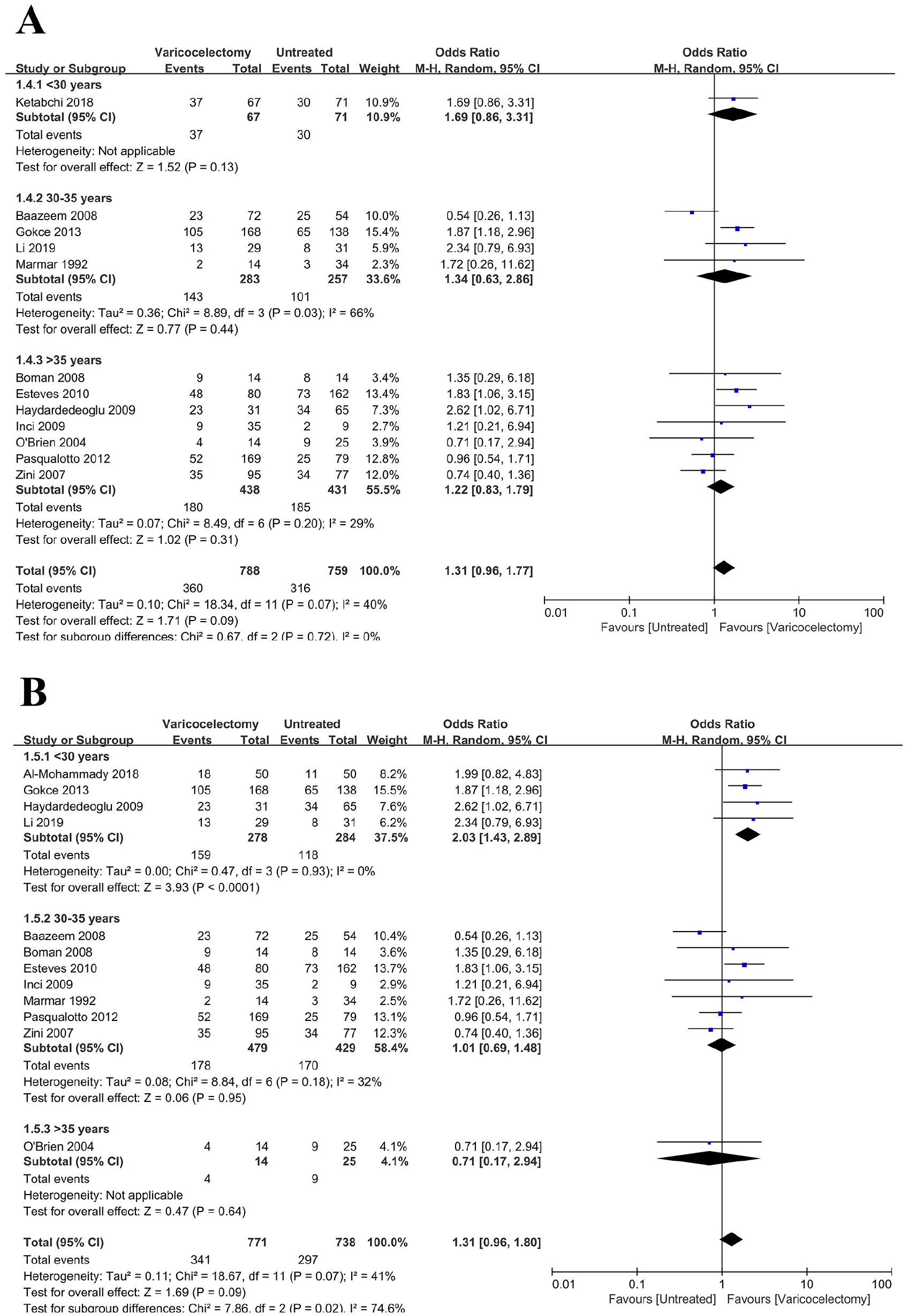
Forrest Plot for the Effect of Varicocelectomy on the Clinical Pregnancy Rate in Studies with Different Peer Review Mean Ages. (A) Male. (B) Female
When the mean age of female partners of infertile patients was less than 30 years, the clinical pregnancy rate was significantly higher in the case group than in the control group (OR: 2.03, 95% CI [1.43, 2.89]; p < .0001; heterogeneity, I2 = 0%). However, for female partners aged 30 to 35 years, there was no significant difference in the clinical pregnancy rate between the case and control groups (OR: 1.01, [0.69, 1.48]; p = .95; heterogeneity, I2 = 32%). Similarly, no significant difference was observed when the female partners were older than 35 years (Not applicable, p = .64; Figure 5B).
The Clinical Pregnancy Rate in Studies with Different Treatment Years
In infertile patients who underwent treatment from 1992 to 2001 and 2002 to 2011, no statistically significant difference in clinical pregnancy rates was observed between the case and control groups (OR: 2.36, 95% CI [0.81, 6.86]; p = .11; heterogeneity; I2 = 0%. OR: 1.11, [0.68, 1.82]; p = .67; heterogeneity; I2 = 51%). However, when patients underwent varicocelectomy between 2012 and 2019, the clinical pregnancy rate was significantly higher in the case group compared to the control group (OR: 1.59, [1.18, 2.14]; p = .002; heterogeneity; I2 = 5%; Figure 6).
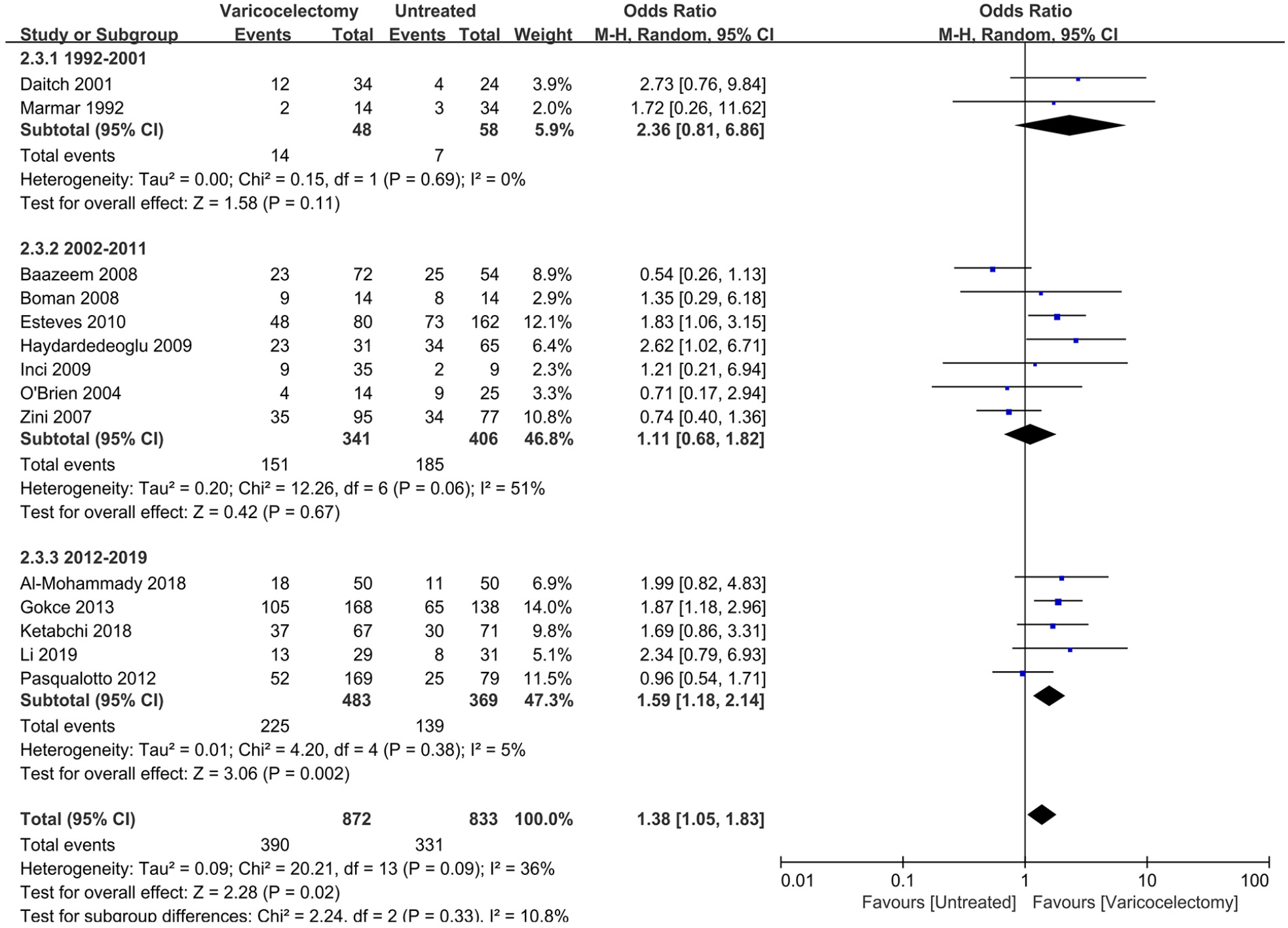
Forrest Plot for the Effect of Varicocelectomy on the Clinical Pregnancy Rate in Studies with Different Treatment Years
Sensitivity Analysis
The results of the sensitivity analysis are shown in Figure 7. It was suggested that the elimination of no single study had a remarkable effect on the merged results, and the conclusions of this study were statistically reliable.

Sensitivity Analysis for the Effect of Varicocelectomy on Clinical Pregnancy Rate of Infertile Patients with Varicocele Using Random Effect Model (Odds Ratio)
Discussion
Clinically, the results of this meta-analysis suggest that varicocelectomy can be considered a viable treatment option for improving clinical pregnancy and live rates in infertile patients with varicocele, but it fails to reduce the miscarriage rates. The overall I2 value is less than 50%, which indicates that the conclusion is reliable. These findings align with previous investigations that have reported improved reproductive outcomes following varicocelectomy (Baazeem et al., 2011; Kirby et al., 2016; Sonmez & Haliloglu, 2018). Compared to the control group, the case group exhibits a significant increase in sperm concentration and normal morphology rates; however, a similar improvement is not observed in sperm motility. The overall I2 value exceeds 50%, likely due to the inclusion of studies published between 1992 and 2019, during which the criteria for sperm selection underwent significant changes (Vasconcelos et al., 2022; X. Z. Zhang et al., 2011). In addition, various laboratories employ different selection criteria. Seventy percent of studies published in two leading reproduction journals (Human Reproduction and Fertility & Sterility) between 2011 and 2020 cited WHO5, while 10% referenced WHO4, 7% cited both WHO4 and WHO5, 1% cited WHO3, and 12% did not reference WHO at all (Vasconcelos et al., 2022).
Subgroup analysis of fertilization types reveals that a significantly increased pregnancy rate is observed exclusively in patients undergoing ICSI, with no such increase noted in patients undergoing IUI and ICSI-TESE. These results are in agreement with Esteves et al. and Gokce et al. (Esteves et al., 2010; Gokce et al., 2013). Our findings show that varicocelectomy improves normal sperm morphology rates. Furthermore, prior research has shown that morphology significantly influences blastocyst formation rates and the development of high-quality blastocysts by Day 6 in ICSI treatment (Jiang et al., 2024). Although sperm motility is significantly associated with IUI outcomes, the lack of improvement in sperm motility after varicocelectomy results in no significant difference in pregnancy rates following IUI (Taniguchi et al., 2024). Although sperm concentration significantly increased, the total progressive motile sperm count does not significantly influence the success rate of IUI (Lin et al., 2022; Lin et al., 2021). O’Brien et al. demonstrated the utility rate of IVF/ICSI collectively, rather than the utility rate of IVF alone; there is no precise data available for the IVF population (O’Brien et al., 2004). Only Ketabchi et al. provided pregnancy data comparing the IVF group with the control group (Ketabchi et al., 2018). Consequently, a subgroup analysis for IVF was not performed. TESE is frequently necessary due to a low sperm count in the ejaculate for some individuals and due to azoospermia recurrence following the recovery of spermatogenesis in others (F. Zhang et al., 2024). A high frequency of karyotype abnormalities or Y-chromosome deletions has been detected in azoospermic men (Cioppi et al., 2021; Lahoz Alonso et al., 2023). Men with nonobstructive azoospermia (NOA) have mutations in genes critical for spermatogenesis (AP1S2, AP1G2, and APOE) (Cheung et al., 2024). Individuals with NOA-related infertility possess mutations in genes related to spermatogenesis /spermiogenesis (ADAM29, SPATA31E1, MAK, POLG, IFT43, and ATG9B) and early embryonic development (MBD5, CCAR1, PMEPA1, POLK, REC8, REPIN1, MAPRE3, and ARL4C) (Cheung et al., 2024). Varicocelectomy does not alter genetic infertility, which might explain why ICSI-TESE men do not experience improved clinical pregnancy rates.
In recent years, there has been a global trend of women delaying childbearing, resulting in a rise in the average paternal and maternal age (Bhasin et al., 2019). Advanced paternal age is associated with a higher risk of spontaneous miscarriage (du Fossé et al., 2020). Maternal age is a crucial factor influencing oocyte quality, with advanced maternal age associated with higher risks of chromosome segregation errors and aneuploidy in oocytes (Charalambous et al., 2023; Mikwar et al., 2020). Given the potential influence of patient age at the time of varicocelectomy on ART outcomes, a detailed subgroup analysis was performed. The results indicate that male age does not significantly alter pregnancy outcomes. In contrast, female age has a significant impact on the clinical pregnancy rate. Notably, the results show that male patients significantly benefit from the varicocelectomy when the mean age of female partners is less than or equal to 30 years old. This observation is consistent with the established notion that the effect of paternal age is less pronounced than the impact of advanced maternal age on reproductive outcomes (du Fossé et al., 2020). Considering the high quality of oocytes from young women, the reproductive benefits are preserved due to the enhanced sperm quality following varicocelectomy. However, the adverse effects of advanced maternal age may counteract any positive impact of varicocelectomy. Therefore, it is plausible to observe no significant difference in clinical pregnancy rates between the two groups when the mean age of female patients exceeds 30 years in our study. In addition, previous research has shown that the recovery period following varicocelectomy varies (Al-Mohammady et al., 2019; Haydardedeoglu et al., 2010; Pasqualotto et al., 2003). Pasqualotto et al. observed that the duration from surgery to pregnancy in the varicocelectomy group was significantly extended, measuring 6.0 ± 0.49 months, compared to 2.7 ± 0.36 months in the untreated group (Pasqualotto et al., 2003). Al-Mohammady et al. demonstrated that the minimum time from varicocelectomy to ICSI was 12 months (Al-Mohammady et al., 2019). Haydardedeoglu et al. found a significant time interval between varicocele repair and TESE (Haydardedeoglu et al., 2010). In the pregnant varicocele repair group, this period was up to 42 months, while in the nonpregnant group, it reached 80 months (Haydardedeoglu et al., 2010). This delay in childbearing poses additional challenges for couples facing infertility, particularly when the male partner undergoes varicocelectomy. The recovery period after varicocelectomy can take approximately 6 to 80 months, which can be a significant duration for couples with advanced maternal age (J. R. Kohn et al., 2020; H. Li et al., 2022). The combined effect of advanced maternal age and the time required for postoperative recovery can result in heightened challenges for achieving successful pregnancies through ARTs (Gourinat et al., 2023; Ribeiro & Sousa, 2023). Given the critical importance of each reproductive cycle for patients of advanced maternal age, our findings indicate that correcting the varicocele prior to ARTs is unnecessary for female partners over the age of 30.
Due to the incomplete surgical information provided in the articles, a subgroup analysis was conducted based on treatment years rather than surgical methods, leading to the following conclusions. Patients treated between 2012 and 2019 experienced significant benefits, while those treated in the periods 1992 to 2001 and 2002 to 2011 did not enjoy the same advantages. Between 1992 and 2019, technological advancements drove changes in varicocelectomy techniques. For instance, minimally invasive procedures such as embolization and sclerotherapy reduced recovery times and complications, while open surgeries, including inguinal and high ligation techniques, were tailored to different patient needs (Binhazzaa et al., 2016; Motta et al., 2019; Nabi et al., 2004). The Palomo procedure, using a retroperitoneal approach, provided improved visibility and operational space (Chung et al., 2023; Sepúlveda et al., 2018). The introduction of microsurgical techniques greatly enhanced surgical precision, reducing recurrence rates, and laparoscopic methods further streamlined the process, shortening hospital stays and speeding up recovery (Motta et al., 2019; Sepúlveda et al., 2018). These advancements not only improved treatment outcomes but also enhanced the overall patient recovery experience. The time-based analysis effectively highlights the impact of evolving surgical techniques on treatment outcomes.
Our study has several limitations that should be addressed in future research. The heterogeneity in our analysis might be attributed to differences in study designs and varicocele grading. Among the fourteen included articles, only one was an RCT. Therefore, larger, well-designed RCTs are needed to confirm our findings. Previous studies have suggested that higher-grade varicoceles may have a more pronounced negative impact on fertility, and thus, the benefits of surgical intervention might vary accordingly (X. Li et al., 2020). The data based on the grade of varicocele in recruited articles were limited; thus, the potential impact of surgical intervention on ARTs in patients with various grades of varicocele has not been comprehensively analyzed. More detailed subgroup analyses associated with grades of varicocele are required. Future research should continue to elucidate the mechanisms through which varicocelectomy improves reproductive outcomes.
Conclusion
In summary, this meta-analysis indicates that varicocelectomy significantly improves clinical pregnancy and live birth rates in infertile patients, but does not prevent their female partners from experiencing miscarriages. Notably, clinical pregnancy rates can be influenced by fertilization type, female age, and treatment year. Further studies with larger sample sizes and RCTs are needed. Future research should focus on addressing these limitations and providing more robust evidence to effectively guide clinical practice.
Footnotes
Acknowledgements
All authors have no acknowledgments to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge financial support from the Sichuan Province (No. 2024NSFSC1676) and Special Support for Postdoctoral Research Project of Sichuan Province in 2024 (No. TB2024077).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
