Abstract
Objective
This study aimed to compare the pregnancy outcomes of Day 2 (D2) fresh embryo transfer and D3 fresh embryo transfer in women with only one zygote with two pronuclei (2PN).
Methods
Data on 432 in vitro fertilization-embryo transfer cycles with only one 2PN zygote from January 2016 to January 2022 were retrospectively collected. A total of 302 fresh embryo transfers on D2 (n = 193) and D3 (n = 109) were analyzed, and pregnancy outcomes were compared.
Results
The patients’ characteristics were not different between D2 and D3 embryo transfer. There were no significant differences in the rates of clinical pregnancy, early abortion, or live birth between D2 and D3 embryo transfer. A multivariate logistic regression model controlling for age, the fertilization method, the number of oocytes harvested, and the number of high-quality embryos transferred showed that the live birth rate was similar between D2 and D3 embryo transfer.
Conclusion
In in vitro fertilization-embryo transfer cycles with only one 2PN zygote, D2 fresh embryo transfer may provide similar pregnancy outcomes to those of D3 embryo transfer. D2 embryo transfer may be an option because of the risk of cycle cancellation due to the absence of viable embryos on D3.
Introduction
Embryos are conventionally transferred on day 2 (D2) or D3 because the uterus provides the ideal environment for survival of the embryo. 1 Over the past decade, there has been a growing tendency towards embryo transfer on day 5 or 6 when the embryos are blastocysts. Delaying embryo transfer allows selection of the most promising embryos for transfer, which may result in a positive effect on the pregnancy outcome. 2
We inevitably encounter patients in whom the number of oocytes harvested is low and only a few oocytes are normally fertilized. Patients then have a low number of zygotes with two pronuclei (2PN). With extension of the length of in vitro culture, the zygotes may be further blocked, leading to a decreased number of viable embryos available for transfer. 3 In this group of people, shortening the duration of in vitro culture of embryos to D2 may result in more chances for embryo transfer.
The rates of clinical pregnancy, abortion, implantation, and live birth of D2 embryo transfer are similar to those of D3 embryo transfer in women with a normal ovarian reserve.4,5 However, controversy remains regarding whether the timing of embryo transfer affects the outcomes of poor responders. Xin et al. showed that patients younger than 35 years with a poor ovarian response (<6 oocytes retrieved) may have better pregnancy outcomes from D3 embryo transfer. 6 However, Bahceci et al. found that poor responders may benefit from D2 embryo transfer in terms of the clinical pregnancy rate. 3 Sacha et al. suggested that transferring embryos on D2 versus D3 in women with a low number of fertilized oocytes (≤two 2PN-stage zygotes) does not affect early pregnancy outcomes. 7 A prospective, randomized trial conducted by Shahine et al. did not show a difference in the pregnancy outcome between D2 and D3 embryo transfer in poor responders. 8 However, their definition of poor responders was vague.
A poor ovarian response was defined differently in the studies mentioned above. Therefore, whether D2 embryo transfer generates better clinical outcomes than D3 embryo transfer in patients with only one available embryo remains controversial. To the best of our knowledge, no studies have examined whether the timing of cleavage-embryo transfer is important in patients with only one fertilized oocyte. Therefore, this study aimed to investigate whether similar or even better pregnancy outcomes can be achieved in D2 embryo transfer compared with D3 embryo transfer in patients with only one 2PN zygote.
Materials and methods
Study design and population
This was a retrospective cohort study. We used the assisted reproductive technology (ART) databases from the Department of Reproductive Medicine at the Women and Children’s Hospital, School of Medicine, Xiamen University. In vitro fertilization (IVF)/intracytoplasmic sperm injection (ICSI) cycles between January 2016 and January 2022 that were intended for fresh embryo transfer with only one 2PN formed 16 to 18 hours after fertilization were included. D2 or D3 embryo transfer was performed according to the convenience and preference of patients. The exclusion criteria were as follows: (i) freeze-all cycles or cycles using donor oocytes; (ii) recurrent implantation failure; and (iii) the presence of uterine anomalies, such as endometrial polyps, uterine cavity effusion, and intrauterine adhesion. This study was conducted in accordance with the tenets of the Declaration of Helsinki. This study was approved by the ethics committee of the Women and Children’s Hospital, School of Medicine, Xiamen University (KY-2020-046). All patients participating in this study signed written informed consent. To protect patient privacy, all patients’ details were de-identified. The reporting of this study conforms to the STROBE guidelines. 9
Ovarian stimulation and IVF/ICSI procedures
All patients received the routine ovulation stimulation programs offered at our center. These programs included the long gonadotropin-releasing hormone agonist protocol, the gonadotropin-releasing hormone antagonist protocol, and other regimens (natural cycle, mild stimulation). Recombinant follicle-stimulating hormone ([FSH] Gonal-F; Merck Serono, Darmstadt, Germany), urinary FSH (Livzon, Guangzhou, China), or human menopausal gonadotropin (Livzon) was used in controlled ovarian stimulation. The doses ranged from 150 to 300 IU and were adjusted depending on the individual ovarian response. When there was at least one follicle with a diameter ≥18 mm, a subcutaneous injection of 250 µg of recombinant human chorionic gonadotrophin (hCG) (Ovidrel; Merck Serono) was performed. Oocytes were retrieved at approximately 34 to 36.5 hours after the injection. IVF or ICSI was performed according to the couple's indications for assisted reproduction.
Embryo scores
The formation of pronuclei was observed 16 to 18 hours after insemination, and the number of 2PN zygotes was recorded. All obtained embryos were evaluated according to the embryo grading criteria modified from Scott et al. 10 In grade 1, there were four cells on D2 and eight cells on D3. The cells were uniform in size, arrangement, and cytoplasm, there were no vacuoles or polynuclear phenomenon, and there were ≤5% of blastomere fragments. In grade 2, there were three to five cells on D2 (mostly 4 cells) and seven to nine cells on D3 (mostly 8 cells). The blastomeres were uniform or approximately uniform and had a uniform cytoplasm, without large vacuoles and multinucleation. There were 10% to 20% of blastomere fragments. In grade 3, there were 2 to 6 cells on D2 and 6 to 12 cells on day 3. Blastomeres were uniform or uneven, the cytoplasm contained more vacuoles than the lower grades, there was no polynuclear appearance, and blastomere fragments accounted for 20% to 30%. In grade 4, there were >6 embryonic cells or no cleavage on D2 and <6 or >12 embryonic cells on D3. Blastomeres and the cytoplasm were not uniform and contained many vacuoles, and there was multinucleation and a blastomere fragment content >30%. Embryos classified as grade 4 were not usable. Grade 1 and 2 embryos were classified as high-quality embryos. 11
Embryo transfer and pregnancy outcomes
Abdominal ultrasound-guided delivery of D2 or D3 embryos into the uterine cavity was performed using a dedicated transfer catheter. Luteal support included 90 mg vaginal progesterone and 20 mg dydrogesterone supplementation once a day after oocyte retrieval. This support was maintained until 10 weeks of pregnancy if pregnancy was established.
The primary outcome was the live birth rate, which was defined as delivery at least by 28 weeks. Secondary outcome measures were the rates of positive hCG, clinical pregnancy, and miscarriage. Positive hCG was defined as a β-hCG concentration >5 mIU/mL 14 days after embryo transfer. A clinical pregnancy was established if the gestational sac and fetal heartbeat were observed in a transvaginal ultrasound examination 4 to 5 weeks after embryo transfer. Miscarriage was defined as spontaneous abortion before 12 weeks of gestation.
Statistical analysis
SPSS 20.0 software (IBM Corp., Armonk, NY, USA) was used for data analysis. Continuous variables with a normal distribution are expressed as the mean ± standard deviation and were analyzed using Student’s t-test. Continuous variables that were not normally distributed are shown as the median (25th–75th percentiles) and were compared using the Mann–Whitney U test. Categorical variables were compared using the chi-square test. A multivariate logistic regression analysis was performed to estimate the odds ratio (OR) and 95% confidence interval (CI) for live birth across embryo transfer stages and other possible confounders. Differences were considered statistically significant at P < 0.05. A post hoc power analysis was carried out using G-power software (G-power v3.1; Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany; http://www.gpower.hhu.de/).
Results
Basic characteristics
Data of 445 cycles from 421 patients with only one 2PN zygote were collected. Of these, 193 cycles with D2 embryo transfer and 109 cycles with D3 embryo transfer were analyzed. A total of 105 cycles were cancelled owing to the absence of viable embryos on D3 in delayed embryo transfer cycles (Figure 1).

Flow chart showing the data selection process for analysis in this study. D2, day 2; D3, day 3; D5, day 5; IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection.
There were no significant differences in age, years of infertility, body mass index, anti-Mullerian hormone concentrations, the antral follicle count, basal bFSH concentrations, average gonadotropin dose, days of gonadotropin use, endometrial thickness, number of oocytes harvested, fertilization method, or high-quality embryo rate between the D2 and D3 embryo transfer groups (Table 1).
Comparison of the general clinical characteristics of patients in the D2 and D3 embryo transfer groups.

Data are the mean ± standard deviation, n (%), or median (25th–75th percentiles).
D2, day 2; D3, day 3; BMI, body mass index; AMH, anti-Mullerian hormone; AFC, antral follicle count; FSH, follicle-stimulating hormone; IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection; NC, natural cycle; Gn, gonadotropin.
Comparison of clinical outcome indicators
The rates of positive hCG, clinical pregnancy, miscarriage, and live birth were not significantly different between the D2 and D3 embryo transfer groups (Table 2). Thirty-nine newborns from D2 embryo transfer cycles and 25 newborns from D3 embryo transfer cycles were delivered without any major structural malformations. There was no significant difference in the live birth rate between the two groups (Table 2).
Comparison of pregnancy outcomes between the D2 and D3 embryo transfer groups.

D2, day 2; D3, day 3; hCG, human chorionic gonadotrophin.
According to the post hoc power analysis, we achieved a power of 0.80 with a 5% level of significance and a 0.3 effect size using the chi-square test.
Multiple logistic regression analysis of live birth
The multivariate logistic regression model was adjusted for age, the fertilization method, the number of oocytes harvested, and the number of high-quality embryos transferred. This analysis showed that the live birth rate was similar between the D2 and D3 embryo transfer groups (OR = 1.23, 95% CI: 0.68–2.22) (Table 3). Maternal age and the number of high-quality embryos transferred were independently associated with the live birth rate. A maternal age older than 35 years was negatively associated with live birth (OR = 0.50, 95% CI: 0.27–0.90, P = 0.022) and the number of high-quality embryos transferred was positively associated with live birth (OR = 2.26, 95% CI: 1.26–4.02, P = 0.006).
Multiple logistic regression analysis of live birth.
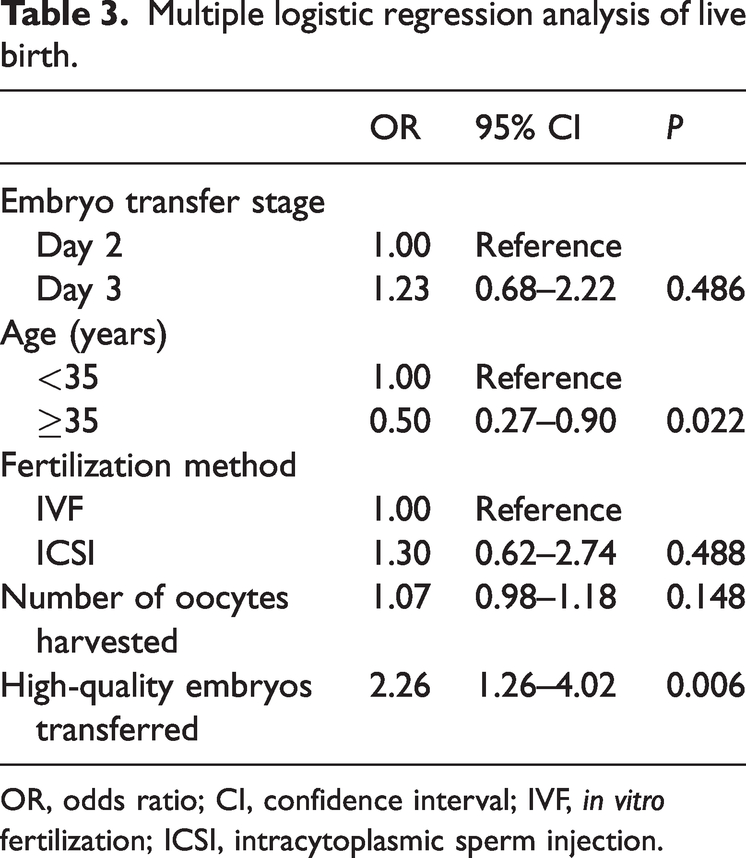
OR, odds ratio; CI, confidence interval; IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection.
Discussion
This retrospective study suggested that D2 fresh embryo transfer provided similar pregnancy outcomes to those with D3 embryo transfer in patients with IVF/ICSI-ET with only one 2PN zygote.
The normal 2PN state is usually formed 16 to 18 hours after fertilization, accompanied by the ejection of two polar bodies, which is a sign of normal fertilization. 12 The number of 2PN zygotes is correlated with the age of the woman, ovarian reserve, ovarian reactivity, ovulation promotion program, number of oocytes harvested, semen quality of the male partner, and pregnancy outcome.13,14 Some studies have shown that the process of in vitro culture may have adverse effects on embryonic development, 15 leading to a further decrease in the number of available embryos from D2 to D3. If there is only one 2PN zygote after fertilization in a cycle, the risk of having no available embryos is considerably increased. Prospectively selecting suitable embryo transfer strategies for patients with fewer oocytes and fewer 2PN zygotes is necessary.
Of the 239 cycles that continued to be cultured to D3, 105 (43.9%) were cancelled owing to the absence of viable embryos on D3 (Figure 1). In patients with only one 2PN embryo, D2 embryo transfer can effectively reduce the risk of cycle cancellation owing to a lack of available embryos, providing patients with the opportunity of transfer in the current cycle. However, the overall effect of embryos that were transferred on D2 that would have arrested in vitro compared with those that were transferred on D3 remains unknown. More attention needs to be paid to the safety of embryos that would have arrested on D3, but were transferred on D2, especially the long-term follow-up health data of the offspring.
Currently, D2 embryo transfer is less frequently used in most assisted reproductive centers. Therefore, unsurprisingly, the clinical outcomes of D2 embryo transfer are unclear. Two previously published studies showed a benefit of D2 embryo transfer in poor responders. In a retrospective study, Shen et al. showed that the ongoing pregnancy rate of D2 embryo transfer was higher than that of D3 embryo transfer in patients <40 years of age with few available embryos. The IVF outcomes were not different between these two groups in patients ≥40 years of age. 16 The other prospective, randomized, clinical study showed that D2 embryo transfer had a higher chance of ongoing pregnancy per oocyte retrieval than D3 embryo transfer in patients with a poor response. 3 The poor responders were defined as those with five or fewer follicles (>13 mm) at the end of ovarian stimulation. However, Zhu et al. showed that the clinical pregnancy rate of D3 embryo transplantation was higher than that of D2 embryo transplantation in patients younger than 35 years of age with a normal or poor ovarian response. 6
Unlike the studies mentioned above, the present study showed that D2 embryo transfer had similar rates of positive hCG, clinical pregnancy, miscarriage, and live birth to D3 embryo transfer in patients with only one 2PN, regardless of the patient’s age. This discrepancy between studies may be due to differences in the study design and grouping criteria. Similar to our results, Lora et al. and Dayal et al. found no difference in the ongoing pregnancy or live birth rate if embryos were transferred on D2 or D3 in patients with limited available embryos (12.3% vs. 12.7% and 10.0% vs. 16.0%, respectively).8,17 The live birth rate of D2 embryo transfer vs. D3 embryo transfer was 20.2% vs. 22.9% in our study. The higher live birth rate in our study than in these two studies may be because our patient population was an average of 3 years younger.
To analyze the confounding effects of age on live birth, the patients were stratified by the ages of <35 years and ≥35 years. The multivariate regression analysis showed that patients with D2 embryo transfer had a similar live birth rate to patients who had D3 embryo transfer after adjustment for age. Therefore, we suggest that age (<35 years vs. ≥35 years) has similar effects on the live birth rate of D2 and D3 embryo transfer in patients with only one 2PN. These findings are in line with those of Lee et al. 18 who found no difference in outcomes between D2 and D3 embryo transfer in young and old age patient groups.
This study has some limitations. This was a retrospective study with a limited sample and it lacked randomization. Whether embryos were transferred on D2 or D3 depended on the convenience and preference of the patients. Additionally, the comparisons were not paired. The same woman did not undergo D2 embryo transfer vs. D3 embryo transfer to determine the true effect of each transfer. Furthermore, the mean age of the patients included in this study was younger than that in previous studies.16–18 Whether the conclusions of this study are applicable to elderly women (i.e., ≥40 years) needs to be determined.
We conclude that the live birth rate of D2 fresh embryo transfer is similar to that of D3 fresh embryo transfer in patients with only one 2PN zygote. D2 embryo transfer may be an option when there is the risk of cycle cancellation owing to the absence of viable embryos on D3. Further studies with larger sample sizes and a randomized, controlled design are required to confirm our findings.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241233985 - Supplemental material for Day 2 versus day 3 embryo transfer in patients with in vitro fertilization and only one zygote with two pronuclei
Supplemental material, sj-pdf-1-imr-10.1177_03000605241233985 for Day 2 versus day 3 embryo transfer in patients with in vitro fertilization and only one zygote with two pronuclei by Yaping Ye, Hui Huang, Longmei Wang, Lu Ding, Libin Mei and Ping Li in Journal of International Medical Research
Footnotes
Acknowledgement
We thank Professor Yuhua Shi from Guangdong Province People’s Hospital for assistance with the research design and discussion.
Author contributions
Yaping Ye: study design and original draft preparation; Longmei Wang: study design and acquisition of data; Lu Ding and Libin Mei: data collection and data analysis; Ping Li and Hui Huang: conceptualization, writing-reviewing, and editing. All authors reviewed and approved the final version of the manuscript.
Data availability statement
The dataset supporting the conclusions of this article is included within the article. The source dataset is available from the authors upon request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Health Youth Project of Fujian Province (2020QNB068), the General Program of Nature Science Foundation of Fujian Province (2023J011611), and the Bethune Medical Science Research Foundation (QL002DS).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
