Abstract
Objective
Endometrial injury (EI) is a simple procedure that may improve endometrial receptivity. The purpose of our study was to investigate whether EI improves reproductive outcomes of frozen-thawed embryo transfer (FET) cycles in patients with one implantation failure.
Methods
The EI group comprised 258 selected patients, and the control group comprised 258 patients retrospectively matched by age, number of transferred embryos, and stage of transferred embryos. The EI group underwent EI in the proliferative phase of FET cycles, and the control group did not undergo EI. The primary outcome was clinical pregnancy rate.
Results
We detected significant differences between the EI and control groups in rates of biochemical pregnancy (5.0% vs. 10.1%), implantation (31.7% vs. 25.3%), clinical pregnancy (47.3% vs. 37.6%), and live birth (39.1% vs. 30.2%). Moreover, EI was found to be an independent factor that affected clinical pregnancy rate, as assessed by logistic regression analysis.
Conclusion
EI in the proliferative phase of FET cycles improves rates of implantation, clinical pregnancy, and live birth in women with one implantation failure.
Keywords
Introduction
Over the last decade, infertility has remained widespread worldwide and has become a major issue affecting human reproduction. 1 Despite the continuous efforts of clinicians to improve assisted reproductive technologies, the clinical pregnancy rate per transfer after in vitro fertilization (IVF) remains stable at 33.2% based on the ESHRE report. 2
Implantation plays a vital role in IVF cycle success and it reflects the effective communication between an embryo and the endometrium. 3 Most implantation studies have focused on factors related to the embryo, and the frequent occurrence of implantation failure after transferring high-grade embryos is frustrating to women, both physiologically and psychologically. The literature suggests that endometrial receptivity is one cause of implantation failure, accounting for approximately two-thirds of these cases.
Endometrial injury (EI) is a potential strategy to improve embryo implantation by enabling endometrial receptivity. 4 Based on the discovery of EI-induced decidualization of guinea pigs in 2003, Barash et al. 5 initially reported that the implantation rate increased twofold when EI preceded IVF treatment. Many studies regarding the effect of EI on implantation during a fresh embryo transfer (ET) cycle have been conducted.6–9 A recent Cochrane review of fresh ET cycles revealed that EI is associated with increases in clinical pregnancy rate and live birth rate. 4 However, few studies have examined the effect of EI on frozen–thawed embryo transfer (FET) cycles. Accordingly, in 2014, Dunne and Taylor performed EI before the FET cycle, and found no significant difference in clinical pregnancy rate between the EI group and the control group. 10 Furthermore, the merits of EI in the FET cycle were not confirmed in several subsequent studies.11–13 Several questions remain regarding the optimal effects of the procedure, and three important factors are noted: the subgroup of patients and the timing and type of the ET cycles. The effect of limiting the population to individuals who have undergone one previous implantation failure has not yet been examined, and the present study is the first to investigate the effect of EI during the proliferative phase of an artificial FET cycle in women with one failed implantation.
Materials and methods
Study design and participants
This was a retrospective study, including 516 patients who underwent FET between January 2014 and December 2016 at the Reproductive Center of The First Affiliated Hospital of Zheng Zhou University. All infertile patients had experienced one previous implantation failure. The inclusion criteria were as follows: (1) eligible to undergo an artificial FET cycle; (2) at least one good-quality embryo; (3) age ≤40 years; (4) body mass index (BMI) ≤30 kg/m2; (5) basal follicle-stimulating hormone (FSH) level ≤12 mIU/mL; and (6) anatomically normal uterine cavity determined by hysteroscopy or pathological examination. The exclusion criteria were as follows: (1) chromosomal anomaly; (2) pelvic abnormality; (3) endometriosis or adenomyosis; (4) autoimmune diseases; and (5) intrauterine manipulation within the previous 3 months. The experimental group comprised 258 selected patients, and the control group comprised 258 patients matched by age, number of transferred embryos, and stage of embryos transferred at the same study stage.
Ethical approval
This study was approved by the Institutional Ethics Committee Review Board of the First Affiliated Hospital of Zheng Zhou University (Science-2017-LW-91) on 1 September 2017. Informed consent was obtained from all the patients in the experimental group prior to EI.
EI
All patients in the experimental group underwent EI during the proliferative phase (day 5 of menstruation) of the FET cycle. Clinicians performed EI by inserting the biopsy catheter S-3.2 (Jiang Xi Nord Medical Device Company, Jiang Xi, China) upward through the cervix into the uterine fundus under ultrasound guidance. The clinicians performing EI paid attention to the pain level of patients and ensured that the patients did not have active bleeding at the end of the procedure. No adverse events occurred in this study.
Embryo cryopreservation
The Vitrification Freeze Kit was used to cryopreserve embryos and the Vitrification Thaw Kit (both from Kitazato Biophama Co. Ltd, Shizuoka, Japan) was used to warm embryos according to standard protocols.
Artificial-cycle FET
In the FET cycle, all patients were subjected to our standard endometrial preparation protocol of daily administration of 2 to 4 mg of estradiol valerate tablets (Delpharm Lille S. A. S, Rue de Toufflers, France) to induce endometrial proliferation and suppress the development of a dominant follicle. Endometrial thickness was measured by transvaginal ultrasound on day 3 of the menstrual cycle and was monitored by ultrasound over the course of 12 days to adjust the estrogen dosage as needed. The hormone levels of progesterone, luteinizing hormone (LH), and estradiol (E2) were measured and transvaginal ultrasound conducted on day 12 after estrogen administration to rule out premature ovulation. Patients began daily intramuscular injections of 40 or 60 mg of progesterone (Zhe Jiang Xian Ju Pharmaceutical Company, Zhe Jiang, China), once the endometrial thickness reached ≥7 mm with a triple-line appearance on that day; if the endometrial thickness was <7 mm, we prolonged the days of estrogen to a certain extent, until endometrial thickness reached 7 mm; patients were not injected with progesterone until endometrial thickness was ≥7 mm. A good-quality embryo was transferred 3 or 5 days later, and cleavage-stage embryos were graded according to Peter’s classification; embryos of grade 1 or 2 were considered good quality. Blastocysts were graded according to Gardner’s classification, and an assessment of ≥3BB was indicative of a good-quality blastocyst. Micronized progesterone (Fleet Laboratories Ltd., Watford, UK) was then administered vaginally to replace intramuscular progesterone for luteal support.
Outcome measures
The primary outcome measure was clinical pregnancy, which was defined as one or more intrauterine gestational sacs detected by ultrasound 5 or 6 weeks after ET. The secondary outcome measure included the rates of positive human chorionic gonadotropin (hCG), biochemical pregnancy, implantation, multiple pregnancy, miscarriage, and live birth. Serum β-hCG level was measured within 14 and 18 days after ET. Biochemical pregnancy was defined as positive hCG without visualization of a gestational sac. Implantation rate referred to the number of visible gestational sacs on transvaginal ultrasound divided by the number of transferred embryos. Live birth rate was defined as the number of liveborn-infant cycles divided by the number of transferred cycles.
Statistical analysis
SPSS software (version 23.0; IBM Corp., Armonk, NY, USA) was utilized for all statistical analyses. The normal distribution of continuous data was verified with the Shapiro–Wilk test. The partial baseline characteristics were compared by the Kruskal–Wallis test. Qualitative variables were expressed as frequency, and outcome measures were compared using the chi-square test and evaluated by univariate logistic regression. Characteristics associated with pregnancy cycles were demonstrated by use in the multiple logistic regression model. A
Results
Baseline characteristics of the EI and control groups
The baseline characteristics are illustrated in Table 1. There were no significant differences in age, BMI, duration of infertility, type and cause of infertility, gonadotropin (Gn) days and dosage in the IVF cycle, number of oocytes retrieved and metaphase II (MII) oocytes, number of embryos transferred in IVF cycle, total number of good-quality embryos in IVF cycle, basal FSH, basal LH, day-3 endometrial thickness, estrogen days, LH on day 12 after estrogen administration, number of transferred embryos, or stage of embryos transferred. The levels of E2 and progesterone on day 12 after estrogen and the dosage of estrogen were significantly lower (
Baseline characteristics in patients between endometrial injury (EI) and control groups.
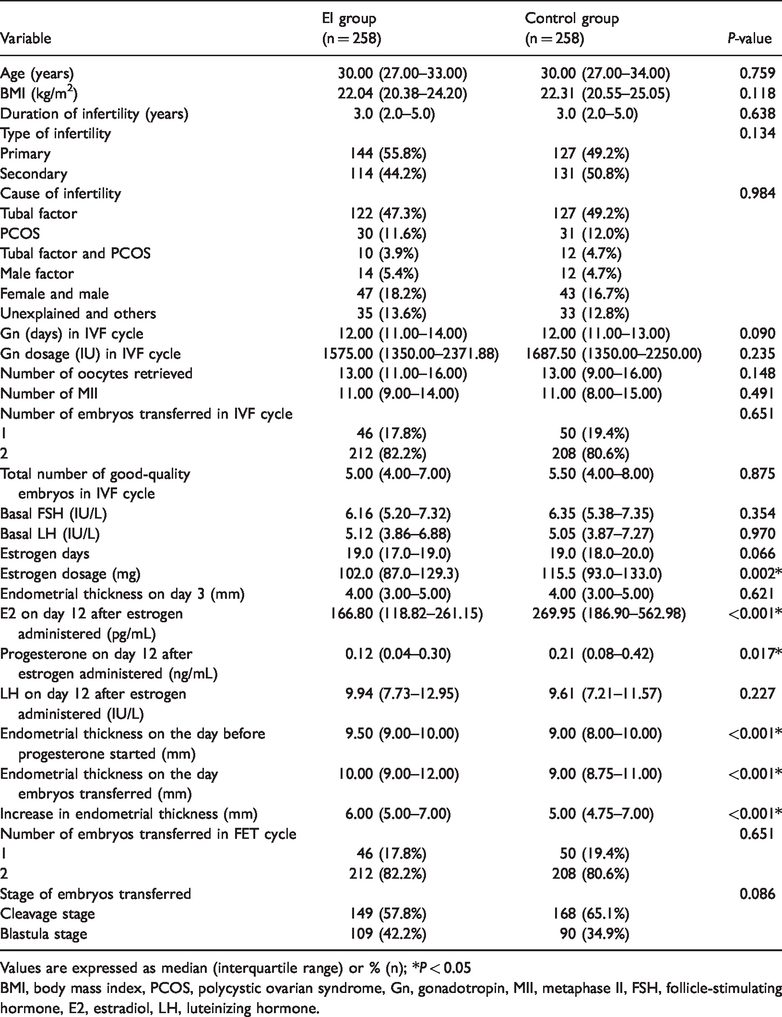
Values are expressed as median (interquartile range) or % (n); *
BMI, body mass index, PCOS, polycystic ovarian syndrome, Gn, gonadotropin, MII, metaphase II, FSH, follicle-stimulating hormone, E2, estradiol, LH, luteinizing hormone.
Pregnancy outcomes in the EI and control groups
According to the study aims, we compared pregnancy outcomes between the two groups (Table 2). We found significant differences in the rates of biochemical pregnancy [5.0% vs. 10.1%; odds ratio (OR) 0.473, 95% confidence interval (CI): 0.238–0.994,
Pregnancy outcomes between endometrial injury (EI) and control groups

Values are expressed as % (n); *
hCG, human chorionic gonadotropin.
Multivariate logistic regression analysis to assess predictors of clinical pregnancy
We assessed the predictors of clinical pregnancy by multivariate logistic regression analysis. Significant differences in age (
Baseline characteristics between pregnancy and non-pregnancy cycles of pregnancies in endometrial injury (EI) and control groups
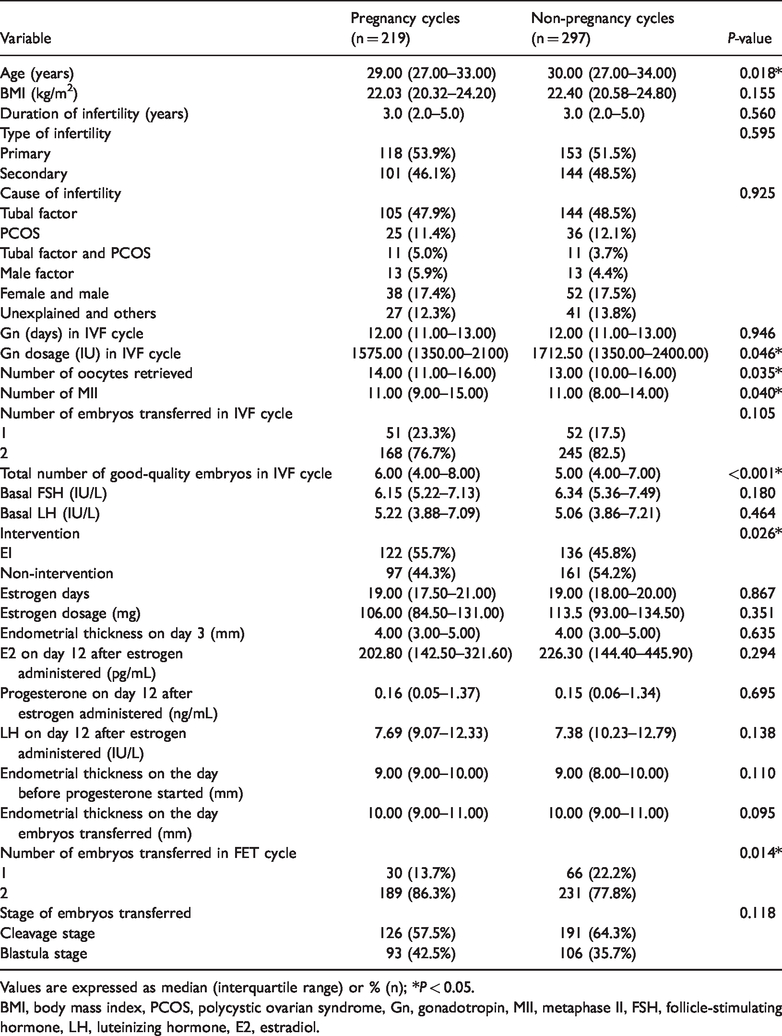
Values are expressed as median (interquartile range) or % (n); *
BMI, body mass index, PCOS, polycystic ovarian syndrome, Gn, gonadotropin, MII, metaphase II, FSH, follicle-stimulating hormone, LH, luteinizing hormone, E2, estradiol.
Multivariate logistic regression analysis assessing predictors of pregnancy in endometrial injury (EI) and control groups

Values are expressed as means (95% CI).
Gn, gonadotropin, MII, metaphase II.
Discussion
Published studies have provided insufficient evidence to confirm that EI is effective in improving pregnancy outcomes or potentially even detrimental. To our knowledge, this study is the first to limit the population to women who have undergone one implantation failure, and we explored the effects of EI on the pregnancy outcome of FET cycles. We aimed to find an approach that improved reproductive outcomes and decreased the number of ET procedures. We demonstrated that EI in the proliferative phase could improve the rates of implantation and clinical pregnancy in the FET cycle compared with the control group.
EI is an inexpensive and time-saving intervention that aims to improve reproductive outcomes of infertile couples. In 2003, Barash et al. 5 first demonstrated that performing EI with a biopsy catheter before the ET cycle in guinea pigs yielded rates of implantation, clinical pregnancy, and live birth approximately twofold higher than those of controls. Thereafter, researchers conducted numerous studies of EI in IVF/intracytoplasmic sperm injection cycles. In 2008, Zhou et al. 6 showed that use of a biopsy catheter to scrape the endometrium in the proliferative phase during a controlled ovarian hyperstimulation (COH) cycle improved rates of implantation, clinical pregnancy, and live birth in women inhomogeneous echo by B-mode ultrasound of endometrium. In 2009, Karimzadeh et al. 7 suggested that performing EI by biopsy catheter on days 21 to 26 of the menstrual cycle improved pregnancy outcomes among patients with recurrent implantation failure (RIF). In 2017, Reljič et al. 8 found that hysteroscopy with EI preceding ovarian stimulation could improve rates of implantation and pregnancy in women with RIF. Furthermore, in 2012, a meta-analysis by El Toukhy et al. 9 demonstrated that performing EI between the proliferative and luteal phases before starting IVF treatment was beneficial in improving embryo implantation in an unselected infertile population. Similarly, in 2015, a systematic review by Nastri et al. 4 claimed that performing EI between day 7 of the previous cycle and day 7 of the ET cycle improved rates of clinical pregnancy and live birth in women with two or more previous ET failures. In 2018, Vitagliano et al. 14 conducted a systematic review and meta-analysis and found that EI may improve live birth rate and clinical pregnancy rate in women with two or more previous ET failures undergoing fresh cycles; however, the timing and technique of EI may play a crucial role in embryo implantation. However, results of other studies did not replicate or, in some cases, were opposed to the above findings.15–17 In 2015, Panagiotopoulou et al. 18 carried out a systematic literature review and found that there was insufficient evidence to support the use of EI in the proliferative or luteal phase, or both, in women with RIF. In 2019, an updated meta-analysis by Vitagliano et al. 19 indicated that current evidence does not support the use of EI to improve the success of a first ET attempt.
Clinicians have gradually begun to focus on the effects of EI on the reproductive outcomes of FET cycles, although there is still no consensus. In 2014, Dunne and Taylor 10 first reported that EI preceding artificial-cycle FET did not appear to significantly improve the rates of implantation or clinical pregnancy in women with at least one failed implantation. In 2016, Shahrokh-Tehraninejad et al. 11 noted that performing EI before artificial-cycle FET did not increase the clinical pregnancy rate of patients with RIF. In 2017, Mak et al. 12 suggested that EI before a natural FET cycle had no benefits in women with at least one IVF transfer cycle. In the same year, Kanazawa et al. 20 found that EI preceding artificial-cycle FET had a positive effect on pregnancy in patients with RIF. Furthermore, Matsumoto et al. 21 suggested that EI before frozen–thawed blastocyst transfer might increase the pregnancy rate and decrease the risk of miscarriage in patients with at least three unsuccessful embryo transfers. In all of the above studies, EI was performed with a pipelle biopsy once during the luteal phase of the menstrual cycle preceding the FET cycle.
The effects of certain variables, such as the population and the timing, frequency, degree, and type of ET cycle, on optimizing EI remain unknown. Our results showed that EI was beneficial for improving implantation and clinical pregnancy rates. The main differences between the current study and previous reports on EI were that previous studies mainly examined fresh ET cycles, whereas we observed FET cycles. The “backwards development theory” proposed by Zhou et al. 6 is a plausible explanation for this difference. They hypothesized that COH negatively regulated embryo implantation through the advancement of endometrial histological development as well as pinopode maturation. However, the FET cycle could not achieve an effect similar to that of the COH cycle. The proliferative phase and luteal phase are two important phases of the menstrual cycle that are typified by their own histological features. Li et al. 22 demonstrated, using an animal model, that complete repair of the endometrium required more than 2 weeks following mechanical injury. In our study, EI was performed in the proliferative phase of the FET cycle. EI performed in different stages of the cycle may explain the diverse pregnancy outcomes reported. At present, there is no consensus regarding the frequency (1 or 2) of EI to achieve optimal effects; in our study, EI was performed once per FET cycle. 14 Although previous studies may not indicate that EI is suitable for all infertile women, EI still appears likely to be of benefit in women with two or more prior ET failures. 4 We focused on women with one implantation failure and suspect that the specific subpopulation of EI might influence the outcomes.
At present, the potential mechanism underlying how EI improves the embryo implantation rate remains unclear. The possible histopathological hypotheses are summarized as follows. First, EI may increase endometrial receptivity by inducing decidualization of the endometrium.5,23 Second, a series of inflammatory responses, including those involving macrophages/dendritic cells and tumor necrosis factor-α, are provoked after EI; these responses facilitate the preparation of a receptive endometrium. 24 Finally, EI induces a rapid increase in side population progenitor cells. These cells contribute to endometrial regeneration, which generates a lag of disproportionate endometrial development during COH cycles, which promotes synchronization between endometrial and embryo development. 25 Kalma et al. 26 analyzed gene expression by microarray analysis of patients who underwent endometrial biopsy; in total, 183 genes were upregulated and 39 were downregulated in the subsequent IVF cycle. The highest increase in expression was shown for endometrial bladder transmembrane I protein (UPIb). In the current study, women in the EI group had significantly higher implantation and clinical pregnancy rates than did women in the control group. Endometrial thickness on day 3 and number of estrogen days did not differ between the EI group and control group. Additionally, a significantly lower dosage of estrogen and a thicker endometrium on the day of ET were observed in the EI group than in the control group. We suggest that EI in the proliferative phase of the FET cycle may decrease the dosage of estrogen and promote development of the endometrium.
Our study has several limitations because of its retrospective, observational design. This meant we were unable to analyze morphological and histological changes in endometrial samples after EI. Further randomized controlled trials are needed to verify our findings and uncover the mechanism by which EI helps increase the rates of implantation and clinical pregnancy. A prospective, randomized controlled study of EI is underway in our center.
It has been speculated that EI may have adverse effects, 13 although we found that EI improved the rates of implantation, clinical pregnancy, and live birth. Similarly, Vitagliano et al. 14 found no evidence of an adverse impact of EI on reproductive outcomes or complications associated with EI.
The current study adds to the set of existing data by examining the effects of EI. In this study, EI in the proliferative phase of the FET cycle improved the rates of implantation, clinical pregnancy, and live birth in women who have undergone one implantation failure. It may decrease the number of ET procedures for patients with one implantation failure to obtain better outcomes.
Conclusions
These results reveal that EI in the proliferative phase improves the rates of implantation, clinical pregnancy, and live birth associated with frozen–thawed ET cycles in women with one implantation failure.
Footnotes
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
