Abstract
Many observational epidemiological studies have reported an association between matrix metalloproteinases (MMPs) and urologic cancers. However, the causal relationship between these two phenotypes remains uncertain. This study aims to examine the bidirectional causal relationship between serum MMPs and three urologic cancers: kidney, prostate, and bladder cancer. Using data from large-scale genome-wide association studies (GWAS), we employed two-sample Mendelian randomization (MR) methods to assess the causal relationship between serum MMPs and urologic cancers. We performed inverse variance-weighted MR as the primary method for calculating the overall effects of multiple instruments, while implementing additional MR methods and sensitivity analyses. Odds ratios (ORs) were employed to evaluate the causal relationship between serum MMPs and urologic cancers risk. Our findings indicated a causal relationship between serum MMP-3 levels and prostate cancer risk (OR = 1.07, 95% confidence interval [CI] = [1.02, 1.11], p = .003). There was a possible causal relationship between serum MMP-1 and prostate cancer (OR = 0.95, 95% CI = [0.92, 0.99], p = .02). Serum MMP-1 may also increase the risk of bladder cancer (OR = 1.24, 95% CI = [1.04, 1.49], p = .016). We did not find significant associations of the remaining MMPs with prostate, bladder, and kidney cancer. In reverse MR, no significant results were observed supporting the effect of urologic cancers on MMPs (p > .05). Our study provides evidence of a potential causal relationship between serum MMPs and both prostate cancer and bladder cancer. However, large-scale studies are necessary to confirm and reveal the underlying mechanisms of this association.
Introduction
Urologic cancers are a group of heterogeneous tumors with distinct clinical and molecular characteristics, affecting any part of the urinary system. According to the cancer statistics report of 2022, urinary system cancer accounted for three of the 10 most common cancers in men: prostate cancer, bladder cancer, and kidney cancer (Siegel et al., 2022). Among these, prostate cancer is the second leading cause of cancer death in men, while kidney cancer is one of the most common malignant tumors in women, ranking ninth in incidence (Siegel et al., 2022). The annual economic burden of urinary cancer in Europe is estimated to be over 13 billion euros (Leal et al., 2016; Luengo-Fernandez et al., 2013). With the rising incidence, urinary system cancer is expected to place an increasing burden on patients and society.
Matrix metalloproteinases (MMPs) are a family of zinc-dependent neutral endopeptidases that can degrade extracellular matrix components (Qin et al., 2024). The MMP family includes 26 zinc-dependent endopeptidases, of which 23 MMP genes have been identified in humans (Z. Zhao et al., 2023). Functionally, MMPs are known for their prominent role in tissue remodeling, promoting cancer invasion and metastasis, and angiogenesis (Zou et al., 2023). In addition, some MMPs have activity that inhibits tumor progression (Dong et al., 2024).
Previous studies have shown that several types of MMPs are associated with urologic cancers. Extracellular matrix (ECM) remodeling is present in anatomically different regions of the renal cortex and has different functions depending on its molecular components: glomeruli, tubule interstitium, and blood vessels. MMPs are widely involved in the ECM of urinary tract and kidney cells, which enables MMPs to regulate basement membrane destruction, angiogenesis, cell migration, and apoptosis, thus promoting the occurrence of urinary tract tumors (Młynarczyk et al., 2024). Another study confirmed that induction of MMP activity contributes to the disintegration of intercellular connections and degradation of extracellular matrix, thereby overcoming physical limitations on cell movement and participating in tumor invasion (K. Wang et al., 2024). The study by Hsieh et al. (2017) found that MMP-3 was elevated in anterior adenocarcinoma cells compared with their normal counterparts. This is supported by research by Jeremy et al., who found that tumor-derived MMP-3 contributes to the growth of prostate cancer in bones (Frieling et al., 2020). In addition, Murray et al. (2012) reported that increased expression of MMP-2 is related to the progression and metastasis of prostate cancer. The study by Schveigert et al. (2013) found that the positive expression of the MMP-9 gene was associated with advanced prostate cancer, suggesting that the expression of the MMP-9 gene is the only prognostic factor for the survival of patients with prostate cancer. In bladder cancer, the study by Fouad et al. (2019) found that the expression levels of MMP-2 and MMP-9 in the blood and urine of bladder cancer patients were significantly higher than those of healthy subjects. This is consistent with the results of Jacek Kudelski et al. (2023). T. Zhao et al. (2023) found that the expression of mRNA and protein of four members of the MMP gene family, MMP-2, MMP-9, MMP-14, and MMP-16, were significantly increased in renal clear cell carcinoma samples. These studies strongly suggest that MMPs may be associated with urologic cancers. However, due to the current randomized controlled trials based on small samples or observational epidemiological studies, the relationship between changes in serum MMP levels and urinary system cancer remains controversial.
Genome-wide association studies (GWAS) provide a new perspective for understanding the genetic determinants behind complex diseases. The Mendelian randomization (MR) technique uses single-nucleotide polymorphisms (SNPs) as instrumental variables (IVs) to identify the causal relationship between various risk factors and complex diseases. Unlike traditional observational studies, this analytical tool is less vulnerable to confounding and reverse causality (Davey Smith & Hemani, 2014). Given the hypothesis that MMP is involved in the development of urinary system cancer, we conducted an MR study to determine whether there is a causal relationship between serum MMP and urinary system cancer.
Materials and Methods
Study Design
In this study, a double-sample MR design was used to explore the effect of serum MMP levels, determined by genetic factors, on the risk of urinary system cancer using published GWAS data. Univariate MR was employed to analyze the causal relationship between serum MMP levels (including MMP-1, MMP-3, MMP-7, MMP-10, and MMP-12) and common urologic cancers, such as kidney cancer, bladder cancer, and prostate cancer. Subsequently, we collected genetic variations closely related to urologic cancers to verify the reverse causal relationship between urologic cancers and serum MMP levels. This study was conducted following the Mendelian Randomization (STROBE-MR) guide, an observational study of enhanced epidemiology (Supplementary Table 5). An overview of this bidirectional MR research design is shown in Figure 1.

Procedure for MR Analysis of Causal Associations Between Serum MMP and Urologic Cancers
Data Sources
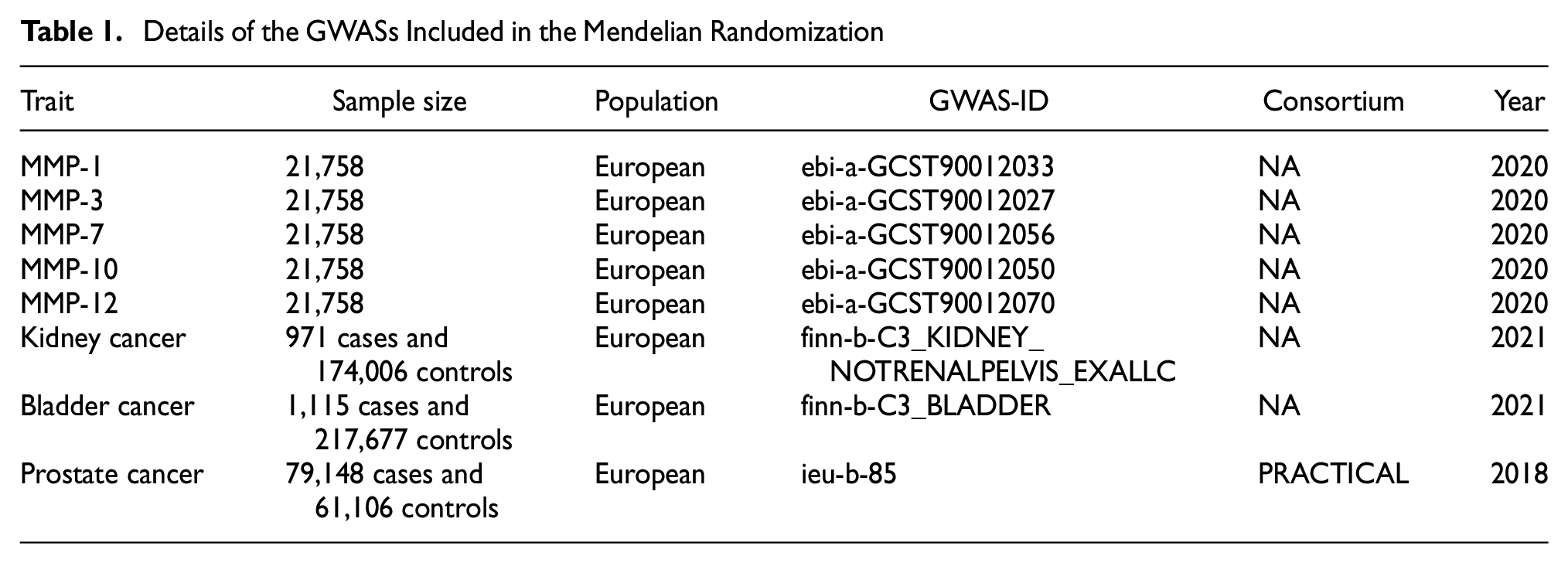
The GWAS summary statistics of five kinds of serum MMP levels (including MMP-1, MMP-3, MMP-7, MMP-10, and MMP-12) were obtained from the IEU OpenGWAS database. These data include a meta-analysis of 21,758 individuals of European descent by Lasse Folkersen et al. (2020). GWAS summary statistics for kidney and bladder cancer were also sourced from the IEU OpenGWAS database. The sample sizes were 174,977 individuals of European origin (971 cases and 174,006 controls) for kidney cancer and 218,792 individuals of European origin (1,115 cases and 217,677 controls) for bladder cancer. Abstract-level data on prostate cancer were extracted from the GWAS of 140,254 individuals of European origin in The Prostate Cancer Association Group to Investigate Cancer Associated Alterations in the Genome (PRACTICAL) consortium (Schumacher et al., 2018). See Table 1 for details of all data sources.
Details of the GWASs Included in the Mendelian Randomization
Selection of Instrumental Variables
The SNPs used in this MR study must meet three assumptions: (1) the SNP needs to be strongly associated with the exposure, (2) the SNP should be independent of confounders, and (3) the SNP should be linked to the outcome solely through the exposure, without a direct association with the outcome (Burgess et al., 2019; Davey Smith & Hemani, 2014; Rasooly & Patel, 2019). To satisfy the first hypothesis, we need to select p-values that are closely related to exposure factors, and to avoid accidental errors, we also need to include as many SNPs related to MMPs as possible, and finally p < 5 × 10−6 is used as our threshold. This threshold has also been reported in other studies (Chen et al., 2020; Kwok & Schooling, 2021). Second, the existence of linkage disequilibrium (LD) between SNPs may lead to biased results. Therefore, we ensured that the LD of SNPs significantly related to exposure met R2 = .001 and KB > 10,000 to maintain independence. We then adjusted the exposure and outcome datasets, eliminating SNPs with different alleles and those with intermediate allele frequencies (Yin et al., 2022). We used Mendelian Randomization Pleiotropy Residual Sum and Outlier (MR-PRESSO) algorithm to detect and eliminate outliers with significant differences (Verbanck et al., 2018). In addition, we used F statistics to ensure the correlation strength between IVs and exposure. It is generally accepted that F statistics greater than 10 satisfy the strong correlation requirements (Pierce et al., 2011). The equation for the F-value used in this study is F = Beta2/Se2, where Beta is the allele effect value and Se is the estimated standard error of Beta. Finally, we uploaded the SNPs that met the above conditions to the Phenoscanner database to ensure that the selected SNPs were not associated with confounding factors (Kamat et al., 2019).
Statistical Analysis
In this MR analysis, MR-Egger, weighted median (WM), and inverse variance weighted (IVW) methods were used to investigate the genetic association between serum MMP levels and urologic cancers. Among them, IVW is an extension of the Wolde ratio estimation method based on the principle of meta-analysis (Burgess et al., 2015). The inverse variance of SNPs can be used as a weight to evaluate the comprehensive causal effect in meta-analysis. In MR analysis, IVW is the most effective method to detect causality (Hartwig et al., 2017). Therefore, this study uses IVW as the primary analysis method, supplemented by MR-Egger and WM to complement the MR results. The MR-Egger method assumes that all genetic variations are invalid IVs, providing an effective test for the invalid causality hypothesis (Bowden et al., 2015). This method uses intercept terms to perform weighted regression, which can evaluate the multiplicity between IVs, with the slope representing the estimated value of causal effect (Burgess & Thompson, 2017). The WM method calculates the causal effect by selecting the median MR estimation under conditions where genetic variation is invalid or multiple (Bowden et al., 2016). In addition, we used Bayesian model averaging (BMA) to further verify the IVW results. BMA analysis can account for the uncertainty of estimated weak effects and weak horizontal pleiotropic effects while adaptively detecting outliers due to a few large horizontal pleiotropic effects (J. Zhao et al., 2020).
To confirm the stability and reliability of the results, we conducted separate tests on heterogeneity and multiplicity. First, we used Cochran’s Q test to determine whether there is heterogeneity (Kulinskaya & Dollinger, 2015). It is worth noting that when the overall heterogeneity is balanced, its presence does not affect the results of IVW estimation of random effects (Cai et al., 2022). Second, the MR-Egger method, which allows for a non-zero intercept, was used to detect horizontal multiplicity (Bowden et al., 2015). In addition, we conducted a leave-one-out analysis to determine whether the removal of a single SNP significantly affected the results, providing a comprehensive evaluation of the stability of MR results.
All analyses were performed in R software (version 4.3.1) using the “TwoSampleMR” and “MRPRESSO” packages for MR analysis. p < .05 was considered statistically significant. This study is a secondary analysis of public data, so ethical approval was not required.
Results
Causal Effect of Serum MMP Levels on Prostate Cancer
In the MR study evaluating serum MMP levels and prostate cancer, we included 23, 19, 20, 20, and 22 SNPs (MMP-1, MMP-3, MMP-7, MMP-10, and MMP-12) for analysis. Supplementary Table 1 shows the details of all SNPs. The univariate MR study assessing the effect of serum MMP levels on prostate cancer risk found a causal relationship between genetically predicted serum MMP-3 levels and prostate cancer risk (IVW: odds ratio [OR] = 1.07, 95% confidence interval [CI] = [1.02, 1.11], p = .003). See the forest plot (Figure 2) for details. WM (OR = 1.05, 95% CI = [1.00, 1.09], p = .03) and BMA (OR = 1.07, 95% CI = [1.02, 1.15], p = .004) showed consistent results (Supplementary Table 2 and Supplementary Table 4). In addition, our study found a potential causal relationship between serum MMP-1 and prostate cancer (IVW: OR = 0.95, 95% CI = [0.92, 0.99], p = .021; BMA: OR = 0.95, 95% CI = [0.91, 0.99], p = .02). However, the results of WM and MR-Egger were not statistically significant (p > .05). Although heterogeneity was observed in some results after Cochran’s Q test, the heterogeneity based on random effect IVW analysis was acceptable. As shown in Supplementary Table 3, MR-Egger regression intercept did not indicate pleiotropy (p > .05). This MR study found a causal relationship between serum MMP-3 levels and an increased risk of prostate cancer, while serum MMP-1 levels may be a protective factor for prostate cancer prevention.

The Risk Association Between Serum MMPs and Urologic Cancers in a Forest Plot
Causal Effect of Serum MMP Levels on Bladder Cancer
In the MR study evaluating serum MMP levels and bladder cancer, we included 23, 18, 22, 22, and 21 SNPs (MMP-1, MMP-3, MMP-7, MMP-10, and MMP-12). The results of IVW showed a causal relationship between serum MMP-1 and an increased risk of bladder cancer (OR = 1.24, 95% CI = [1.04, 1.49], p = .016). The results of BMA were consistent (OR = 1.25, 95% CI = [1.04, 1.49], p = .015). However, the two supplementary methods, WM and MR-Egger, did not support the IVW results (p > .05). Therefore, serum MMP-1 may be a risk factor for bladder cancer. We found no association between other serum MMP levels and bladder cancer. Detailed results can be found in Figure 2. In addition, in the sensitivity analysis, there was no evidence of pleiotropy or heterogeneity (Supplementary Table 3). The leave-one-out method did not find that any single SNP had a significant effect on the results (Supplementary Figure).
Causal Effect of Serum MMP Levels on Kidney Cancer
In the MR study evaluating serum MMP levels and kidney cancer, we included 23, 18, 22, 22, and 21 SNPs (MMP-1, MMP-3, MMP-7, MMP-10, and MMP-12). In the univariate MR study assessing the effect of serum MMP levels on the risk of kidney cancer (Figure 2), the IVW results suggested that genetically predicted serum MMP levels are not associated with the risk of kidney cancer (p > .05). Although the p value of WM between serum MMP-7 and kidney cancer was less than .05 (p = .024), the results of IVW, MR-Egger, and BMA were not consistent(Supplementary Table 2). Therefore, we conclude that there is no causal relationship between serum MMP-7 levels and kidney cancer. In the sensitivity analysis test, no heterogeneity was detected by Cochran’s Q test. The MR-Egger test did not detect pleiotropy (Supplementary Table 3).
Reverse Two-Sample MR Analysis
In reverse two-sample Mendelian randomization (TSMR), urologic cancers were the exposure factor, while serum MMP levels were the outcome factor. The reverse MR results did not support a causal relationship between genetic susceptibility to urologic cancers and serum MMP levels. Figure 2 shows the main results of this MR analysis. The estimated effect sizes for the gene predictions are shown as scatter plots (Supplementary Figure).
Discussion
In this study, we found a causal relationship between serum MMP-3 levels and prostate cancer. Elevated MMP-3 serum levels increase the risk of prostate cancer, indicating that serum MMP-3 is a risk factor for prostate cancer. In addition, we found that at the genetic level, increased serum MMP-1 may be associated with a higher risk of bladder cancer and a lower risk of anterior adenocarcinoma. We did not find statistically significant associations between other MMPs and urinary tumors.
To the best of our knowledge, our study is the first to report a bidirectional causal relationship between serum MMP levels and urologic cancers. Numerous previous studies have confirmed that MMPs, as mediators of tumor progression, can degrade fine extracellular matrix components and regulate the biological activity of growth factors and cytokines. Depending on the tissue environment, MMPs can either promote or inhibit tumorigenesis (Lin et al., 2022; Serra, 2022). Our study found that elevated serum MMP-3 levels may increase the risk of prostate cancer, which aligns with previous clinical studies.
An early cross-sectional study showed that serum MMP-3 levels in patients with metastatic prostate cancer were significantly higher than those in normal controls (Wei, 2023). Another study confirmed the trend of elevated serum MMP-3 levels in patients with prostate cancer across pathological grades (Hsieh et al., 2017). MMP-3 is an important member of the MMP family. Numerous studies have confirmed that MMP-3 plays a significant role in the growth and metastasis of cancer cells (Frieling et al., 2020; Ganguly et al., 2020). Recent research indicates that MMP-3 can control cell growth and external angiogenesis in prostate cancer. Jeremy et al. found that MMP-3 can promote the growth of prostate cancer cells in vitro and tumor growth in vivo in a tibial model of bone metastasis of anterior adenocarcinoma (Frieling et al., 2020). This study found that silencing MMP-3 can inhibit the growth of prostate tumors in bone and down-regulate the transcription of angiogenic genes. MMP-3 ablation can inhibit the angiogenesis of prostate tumors in bone (Frieling et al., 2020). Gakuji et al. discovered that MMP-3 can activate the angiogenic activity of vascular endothelial growth factor (Hashimoto et al., 2002). In addition, some studies have found that MMP-3 can promote angiogenesis by cleaving and secreting matrix-related proteins that are acidic and rich in cysteine to produce novel peptides that regulate the proliferation and migration of endothelial cells (Sage et al., 2003). Several studies have confirmed the association between MMP-3 and prostate cancer on the other hand. Deb et al. (2019) found that green tea polyphenols can reduce the growth and metastasis of prostate cancer cells by inhibiting the secretion of MMP-3. Qu et al. (2016) found that the IL-7/IL-7R axis is involved in the invasion and migration of prostate cancer cells by up-regulating MMP-3. However, although previous clinical studies have shown an association between prostate cancer and MMP-3, they have not demonstrated a bidirectional causal relationship. Therefore, our study not only confirmed that elevated serum MMP-3 levels are a risk factor for prostate cancer but suggested a new potential relationship between serum MMP-3 levels and prostate cancer. However, MR analysis uses genetic variation as an IV to infer causality between exposure factors and outcomes. When exposure is expressed as a binary result, the limitations of MR analysis become apparent. In MR analyses, we generally assume that the association between genetic variation and exposure is causal, and that genetic variation and confounders are independent of each other. Nevertheless, these hypotheses may be more difficult to test in the context of binary exposure, as the binary results are more susceptible to many unobtrusive confounding factors. Therefore, even if MR analyses provide strong evidence for a relationship between exposure and outcomes, these results should be interpreted as associations rather than definitive causation. In particular, for binary outcomes, estimates from MR analyses can be influenced by a combination of sample size, effect size of genetic variation, and potential confounders that can lead to overestimating or underestimating causal effects. Thus, while MR analysis can reveal associations between exposure and outcomes, the interpretation of these relationships should avoid over-extrapolating to true causation (Burgess & Labrecque, 2018). In addition, research on the relationship between MMP-3 levels and the development of prostate cancer is still incomplete. Especially, in terms of potential mechanisms and targets of action, further exploration is needed to verify these findings.
In addition, our results suggest a causal relationship between serum MMP-1 levels and both bladder cancer and prostate cancer. Although the sensitivity test indicates that the IVW method results are robust, the WM and MR-Egger methods do not provide consistent results. Therefore, we consider the causal relationship between serum MMP-1 levels and these cancers to be potential. Numerous studies have shown that MMP-1 content in the urine of bladder cancer patients plays an important role in prognosis and serves as a unique biomarker (Jin et al., 2015; Zheng et al., 2019). One study found that the MMP1-1607 polymorphism is associated with a significant increase in MMP1 transcription (Li et al., 2015). A recent large meta-analysis found that increased MMP1 transcription due to the MMP-1-1607 polymorphism was significantly associated with an increased risk of bladder cancer (Zhou et al., 2018). Another meta-analysis indicates that the MMP-1-1607 1G/2G polymorphism may inhibit the development of prostate cancer in Caucasians (Weng et al., 2017). These observational studies align with our findings.
The advantages of this study are as follows: First, to our knowledge, this is the first study to report a bidirectional causal relationship between serum MMP levels and urinary cancer. Second, we employed multiple MR methods to improve reliability and reduce potential bias. In addition, due to the large sample size of the GWAS aggregate data for MMPs and urinary cancer, our results are considered robust. Various types of sensitivity analyses further confirmed the strength and efficiency of our findings.
Environmental and lifestyle factors can directly or indirectly alter gene expression, including changes in chromosome structure, changes in DNA methylation, and adjustment of transcriptional regulation. MR is a statistical method that uses genetic variation as an IV to assess the causal relationship between exposure factors (such as lifestyle or environmental factors) and health outcomes. It is based on several key principles: At fertilization, a random combination of genes from the parents is assigned to the offspring. This means that certain genetic variants are not associated with the influence of environmental and lifestyle factors, and can be used as a natural “randomization” tool for exposure. On the other hand, through association studies, such as GWAS, scientists can identify specific genetic variants that are associated with a particular disease or health condition. MR uses these genetic variants as IVs to estimate the effect of a particular exposure on health outcomes. This allows MR to avoid confounding genetic associations caused by environmental or lifestyle factors (H. Wang et al., 2024).
There are some limitations in our experiment. First, the estimated value of MR represents the long-term regulation of serum MMP levels on disease risk. This implies that when serum MMP concentration increases over a short period, the effect might be smaller than the experiment’s estimate. Therefore, in clinical observation of the relationship between specific MMP concentration and urinary cancer, the actual increase in risk may not align with the effect observed in this MR study. However, we believe it can still guide potential targets for cancer treatment. Second, the current GWAS summary data do not contain enough IVs to complete the analysis of other types of MMPs, preventing MR analysis between these MMPs and urinary cancer. Third, the focus on European variables limits the applicability to other populations. Future research should verify these findings by considering different ethnic groups, such as Asians. Fourth, the GWAS data used in this analysis lack stratified analysis based on gender, age, disease course, and other factors, limiting the ability to research-specific details. Therefore, future studies should include more types of serum matrix proteins for analysis. On the other hand, we need to further expand the analysis population, such as using GWAS data from both Asian and African populations. In addition, it is also necessary to conduct stratified analysis for people of different ages, genders, and tumor stages. In summary, we look forward to more complete GWAS data in the future to help us conduct more comprehensive and larger analysis.
Conclusion
In summary, this MR study found that high levels of serum MMP-3 have a causal relationship with a high risk of prostate cancer in the European population, while an increase in serum MMP-1 may be associated with an increased risk of bladder cancer and a reduced risk of prostate cancer. Our results provide new insights for exploring potential biomarkers and therapeutic targets for urinary cancer in clinical settings.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883241311229 – Supplemental material for Serum Matrix Metalloproteinases and Risk of Urologic Cancers
Supplemental material, sj-docx-1-jmh-10.1177_15579883241311229 for Serum Matrix Metalloproteinases and Risk of Urologic Cancers by BoWen Yang, XiaoYu Zeng, HanYu Wang, JiuHuan Feng and ShuFang Hou in American Journal of Men's Health
Supplemental Material
sj-xlsx-2-jmh-10.1177_15579883241311229 – Supplemental material for Serum Matrix Metalloproteinases and Risk of Urologic Cancers
Supplemental material, sj-xlsx-2-jmh-10.1177_15579883241311229 for Serum Matrix Metalloproteinases and Risk of Urologic Cancers by BoWen Yang, XiaoYu Zeng, HanYu Wang, JiuHuan Feng and ShuFang Hou in American Journal of Men's Health
Supplemental Material
sj-xlsx-3-jmh-10.1177_15579883241311229 – Supplemental material for Serum Matrix Metalloproteinases and Risk of Urologic Cancers
Supplemental material, sj-xlsx-3-jmh-10.1177_15579883241311229 for Serum Matrix Metalloproteinases and Risk of Urologic Cancers by BoWen Yang, XiaoYu Zeng, HanYu Wang, JiuHuan Feng and ShuFang Hou in American Journal of Men's Health
Supplemental Material
sj-xlsx-4-jmh-10.1177_15579883241311229 – Supplemental material for Serum Matrix Metalloproteinases and Risk of Urologic Cancers
Supplemental material, sj-xlsx-4-jmh-10.1177_15579883241311229 for Serum Matrix Metalloproteinases and Risk of Urologic Cancers by BoWen Yang, XiaoYu Zeng, HanYu Wang, JiuHuan Feng and ShuFang Hou in American Journal of Men's Health
Supplemental Material
sj-xlsx-5-jmh-10.1177_15579883241311229 – Supplemental material for Serum Matrix Metalloproteinases and Risk of Urologic Cancers
Supplemental material, sj-xlsx-5-jmh-10.1177_15579883241311229 for Serum Matrix Metalloproteinases and Risk of Urologic Cancers by BoWen Yang, XiaoYu Zeng, HanYu Wang, JiuHuan Feng and ShuFang Hou in American Journal of Men's Health
Supplemental Material
sj-xlsx-6-jmh-10.1177_15579883241311229 – Supplemental material for Serum Matrix Metalloproteinases and Risk of Urologic Cancers
Supplemental material, sj-xlsx-6-jmh-10.1177_15579883241311229 for Serum Matrix Metalloproteinases and Risk of Urologic Cancers by BoWen Yang, XiaoYu Zeng, HanYu Wang, JiuHuan Feng and ShuFang Hou in American Journal of Men's Health
Footnotes
Acknowledgements
This study was conducted by using GWAS data from the IEU OpenGWAS database and the PRACTICAL database. We would like to thank all participants and the abovementioned consortiums for their contribution.
Author Contributions
All authors contributed to the conception and design of the study. XiaoYu Zeng, HanYu Wang, and BoWen Yang designed the research. HanYu Wang collected the data. XiaoYu Zeng and BoWen Yang analyzed the data. XiaoYu Zeng, HanYu Wang, and BoWen Yang drafted the manuscript. ShuFang Hou revised the manuscript. ShuFang Hou jointly supervised the study. All the authors were involved in writing the paper. All the authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Clinical efficacy evaluation of Dongguan Science and Technology Bureau’s high-level special program “Zhixueshengjifang” for the treatment of chronic radiation proctitis and Hou ShuFang Dongguan Famous Traditional Chinese Medicine Expert Inheritance Studio.
Ethics Approval and Consent to Participate
All analyses were based on publicly available summary statistics and did not require ethical approval or consent.
Consent for Publication
This manuscript does not include details, images, or videos relating to an individual person; therefore, consent for publication is not required, beyond the informed consent provided by all study participants as described above.
Data Availability
All the data used in the current study are publicly available in GWAS summary data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
