Abstract
Objective
Osteoarthritis (OA) and asthma are two common chronic diseases with increasing incidence and prevalence, whereas there has been rare evidence to suggest the relationship between OA and asthma. This study aimed to analyze the causal relationship between OA and asthma.
Methods
Existing studies of the relationship between asthma and OA published till July 18, 2023, were identified from PubMed and Web of Science databases for meta-analysis. Subsequently, the causal relationship of all and site-specific OA with asthma was explored through a bidirectional two-sample Mendelian randomization (MR) analysis.
Results
A total of four eligible studies were included in the meta-analysis. In these studies, 80,550 participants were recruited, of whom 13,781 patients had OA. The asthma group had a significantly higher prevalence of OA than the control group (odds ratio (OR) = 2.08; 95% confidence intervals (CI): 1.42–3.03). However, MR analysis did not support a causal relationship between asthma and all OA and site-specific OA: knee and hip OA (OR = 1.03; 95% CI: 0.98–1.09), knee OA (OR = 1.02; 95% CI:0.96–1.08), and hip OA (OR = 1.04; 95% CI: 0.97–1.12). No causal relationship between OA and asthma was found through reverse MR analysis.
Conclusions
This meta-analysis suggests that patients with asthma are likely to have a greater prevalence of OA. However, the result of MR analysis reveals that asthma does not have a causal relationship to all OA or site-specific OA.
Introduction
Osteoarthritis (OA) refers to a degenerative injury of articular cartilage, and its risk factors are complex (e.g. aging, obesity, strain, and trauma). 1 OA has been ranked the fourth most disabling disease worldwide in 2020, with over 527 million people currently subjected to OA.2,3 It is noteworthy that the development of OA is irreversible, although it is being actively investigated for, e.g. stem cell and exosome therapies with significant promise. Accordingly, it is imperative to explore the etiology and development mechanism of OA and to find the risk factors closely related to the development of OA. Asthma also is one of the most common chronic diseases that has an effect on the respiratory tract of the lungs, which has affected an estimated 339 million people worldwide. 4 It is characterized by a range of respiratory symptoms and a limitation of airflow to varied degrees. 5 In general, the causative factors of asthma comprise allergy, respiratory infection, stress, exercise, and drugs (such as aspirin, beta-blockers, or nonsteroidal anti-inflammatory drugs). However, its etiology is complex, and it is primarily considered to bethe result of the interaction of genes and environment. 6
Asthma and OA are common chronic diseases that inhibit physical activity and cause a poor quality of life. Considerable studies of comorbidities have suggested that OA appears to increase the hospitalization rate of asthma and even exacerbate asthma. 7 Asthma and OA also interact in multiple ways at the level of molecular mechanisms. For instance, reactive oxygen species (ROS) play an important role in the occurrence and development of OA, whereas asthma will develop oxidative stress due to excessive production of ROS and the corresponding reduction of antioxidant response.8–11 As a result, oxidative stress worsens asthma attacks and exacerbations and causes tissue oxidative damage. 10 Genome-wide association studies (GWAS) have suggested that numerous single nucleotide polymorphisms (SNPs) in the SMAD family member 3 (SMAD3) gene were correlated with hip and knee OA.12,13 Additionally, SMAD3 methylation was significantly correlated with the generation of IL-1β, an inflammatory mediator intimately related to OA, in neonates of asthmatic mothers. 14 However, the evidence from observational research for a relationship between asthma and OA risk has been inconsistent. For instance, a study that examined the relationship between asthma and other chronic conditions has suggested that those with asthma have a significantly higher incidence of OA than people without asthma. 15 Furthermore, the incidence of OA in asthma patients is significantly higher when chronic obstructive pulmonary disease (COPD) patients are assigned to controls.16,17 Collectively, these findings suggest a strong association between OA and asthma. However, the study by Choi et al. 18 has not shown evidence of a strong relationship between OA and asthma.
Mendelian randomization (MR) is a novel method of determining causal relationships using genetic variation as an instrumental variable (IV). 19 Extensive studies have recognized that conventional observational research cannot determine the causal sequence with accuracy. Genotype can serve as a powerful tool to investigate the causal relationship between exposure and outcome since it is intrinsic, must occur before the outcome, and is unaffected by numerous confounding factors (e.g. acquired environment). 20 MR is a valuable way to push epidemiology beyond its limits. 21 Based on the ambiguous causal relationship, we aimed to conduct a bidirectional MR to determine the direction of the causal effect of OA on asthma. 22 Currently, there is no study focusing on MR analysis of asthma and OA.
Accordingly, to explore whether the relationship between OA and asthma exists, we conducted a meta-analysis of observational studies. Meanwhile, A two-sample bidirectional MR was further conducted to examine whether this relationship reflects causal relationship.
Methods
Meta-analysis
Research search and screen
Our systematic review and meta-analysis were conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guideline. 23 All studies in PubMed and Web of Science databases studying the correlation between asthma and OA by 18 July 2023, were searched. The following search terms were employed: (“asthma” AND (“Osteoarthritis” OR “Osteoarthritides” OR “Osteoarthrosis” OR “Osteoarthroses”)) The language was restricted to English. We developed a number of criteria to screen the retrieved studies. (i) Observational studies examine the link between asthma and OA (e.g. cross-sectional, case–control, and cohort studies). (ii) There were no restrictions on the age and sex of the participants and other basic characteristics. (iii) Asthma and OA were diagnosed through self-report or a medical certificate. If multiple studies use data from the same population, then select the studies with the largest sample size or the most detailed analysis. Information for the respective study was extracted and recorded by two investigators (Yaoyao Nie and Houpu Liu), including the first author, year of publication, study design and name, country, sample description (e.g. age, sex, total sample size, and number of cases), OA and asthma assessment methods, and corresponding risk estimates with 95% confidence intervals (CIs). The quality of the included studies was evaluated using the scale recommended by the American Agency for Healthcare Research and Quality (AHRQ). 24 The quality assessment items are presented in Figure 1.

Agency for Healthcare Research and Quality (AHRQ) Quality Assessment Scale for cross-sectional studies.
Statistical analyses
The relationship between OA and asthma was evaluated through meta-analysis (Figure 2). The data from the original text were extracted (e.g. total sample size, number of asthma, number of OA, and incidence of OA in asthma). The effective data were manually calculated if the original publication only presented the prevalence and odds ratio (OR) values, or if the original data cannot be directly obtained. The OR and 95% CI for asthma in participants with and without OA were obtained. Statistical heterogeneity was examined through the

Flow diagram of study identification and screening.
Mendelian randomization analysis
The MR analyses of this study were based on the STROBE-MR guideline.
25
The two-sample MR analyses were conducted in accordance with two nonoverlapping summary GWAS data. All summary data were originated from European ancestry. The UK Biobank presented the GWAS summary statistics of OA (77,052 OA cases and 378,169 controls) based on 455,211 people of European ancestry.
26
Asthma GWAS data were gathered from the FinnGen cohort in Finland. FinnGen (https://www.finngen.fi/en) refers to a large biological library research with an aim to classify the genotypes of 500,000 Finns, which involved prospective and retrospective epidemiological cohorts based on disease and hospital library samples. The diagnosis of asthma was identified in accordance with the J10 code in the International Classification of Diseases ICD10. The genetic predictors of asthma originated from a published GWAS involving 20,629 asthma cases and 135,449 controls.
27
SNPs served as the IVs for MR analysis. The selected IVs should conform to three conditions as follows. (i) SNPs should be significantly correlated with exposure. (ii) The selected SNPs are not related to confounding factors. (iii) SNPs should not be correlated with outcomes and affect outcomes only by exposure.
20
To conform to the above criteria, genome-wide significant (
Consent statement
Since our data are publicly accessible aggregated data, consent is not applicable.
Results
Meta-analysis of observational studies
Screening and data extraction were incorporated into the meta-analysis.15,16,33,34 One of the studies used data from the Korean National Health and Nutrition Examination Survey (KNHANES), one from German Health Update (GEDA), and one from National Health and Nutrition Examination Survey (NHANES), and the last research's participants were enrolled in The Mayo Clinic Bipolar Disorder Biobank. Steppuhn et al.
15
studied comorbidities of asthma including the prevalence of OA. The study by Mendy and Mersha
33
sought to uncover comorbidities related to adult and pediatric asthma and comorbidities related to age at asthma diagnosis. Only Koo et al.
16
studied the correlation between asthma and OA using chronic obstructive pulmonary disease as a control. Romo-Nava et al.
34
found individuals with asthma had higher odds of having other diseases after adjusting for age, sex, and site, such as OA (OR = 2.08, 95% CI: 1.20–3.61). Subsequently, we evaluated the quality of the literature according to the AHRQ Quality Assessment Scale (Figure 1), and the results were determined according to Hu et al.
35
All four studies were assessed as of medium quality, as shown in Table 1. There were 80,550 individuals in all, 13,781 of whom had OA, and 1537 of them also had asthma. Asthma detrimentally affected OA (OR = 2.08; 95% CIs 1.42–3.03), as indicated by the results of this meta-analysis (Figure 3(a)). The random-effects model was applied since the
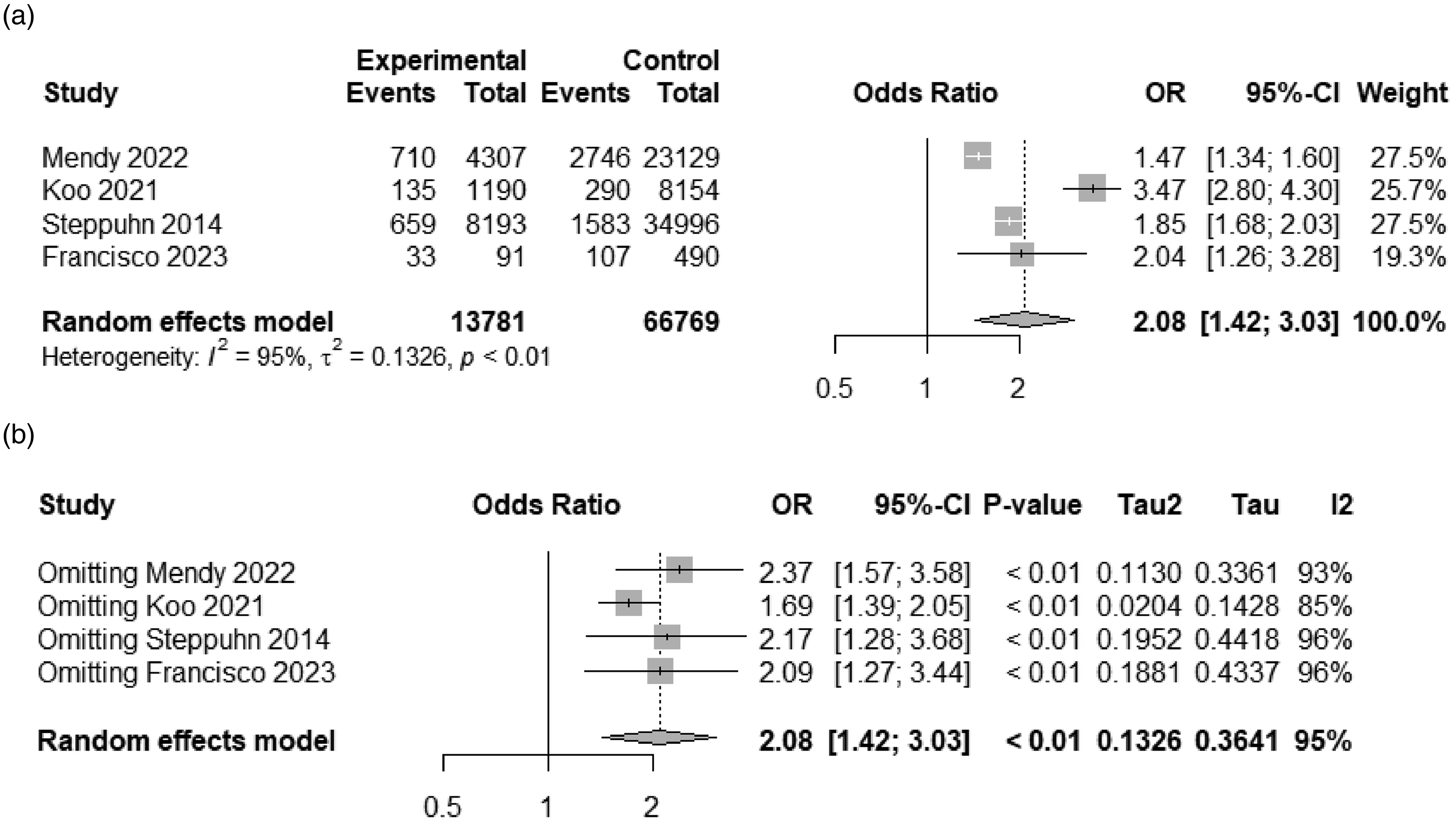
Forest plot of meta-analysis (a) and sensitivity analysis (b).

Funnel plot of meta-analysis (a) and after rectification (b).
Results of literature quality assessment.

Mendelian randomization analysis
The information on the two diseases is listed in Table 2. The IVs of asthma and OA at the respective site (mostly picked the weight-bearing joint site) conformed to the three assumptions of MR, as indicated by the results of the MR analysis. And three confounding factors were commonly adjusted in epidemiological studies, namely body mass index (BMI), educational attainment, and smoking status. Specifically, we excluded SNPs (rs143384 and rs6499244 for knee OA and rs3774354 and rs5611684 for all OA) linked to BMI; additionally, the SNP of rs6206328, which was associated with educational attainment, was excluded in the analysis of hip OA. Notably, none of the asthma IVs was found related to these factors.In addition, rs28453249, rs35656734 (IVs as asthma), and rs9277552 (significantly associated with hip OA and all OA) were located in the MHC region, so we removed these SNPs in our two sample Mendelian randomization analysis. Detailed information on included and excluded SNPs is provided in Supplemental Tables S1–S5.
Instrumental variable sources.

The findings of the main analysis (IVW method) indicated that genetic liability to asthma was not significantly correlated with OA or OA in the respective site, knee and hip OA (OR
The Mendelian randomization analysis results with regard to causal effect of asthma on OA.

OA: osteoarthritis; MR: Mendelian randomization; SNP: single nucleotide polymorphism; IVW: inverse-variance weighting; OR: odds ratio; CI: confidence interval;
The Mendelian randomization analysis results with regard to causal effect of OA on asthma.

OA: osteoarthritis; MR: Mendelian randomization; SNP: single nucleotide polymorphism; IVW: inverse-variance weighting; OR: odds ratio; CI: confidence interval;
The sensitivity analyses presented in Supplemental Figure S6 indicated that our results might be affected by the exclusion of rs1837253. However, upon conducting a supplementary analysis using the two-sample MR, wherein we deliberately excluded rs1837253 from our IVs, we observed that our findings remained unchanged and exhibited robustness (Supplemental Table S6).
Discussion
Findings from meta-analysis confirmed a significant correlation between asthma and OA, both in terms of the raw data and after sensitivity and adjustment studies. Using bidirectional two-sample MR analysis, we did not find evidence for a casual association between the two diseases; however, the LDSC analysis showed that asthma and knee and hip OA were highly correlated.
To the best of our knowledge, this study has been the initial comprehensive study of the causal relationship between asthma and OA risk through meta-analysis and MR analysis. Moreover, the correlation between asthma and OA, two common chronic diseases, was comprehensively evaluated through meta-analysis and bidirectional two-sample MR analysis. Compared with observational studies, MR analyses are less susceptible to confounding, reverse causal relationship, and nondifferential exposures measured by error. Furthermore, sensitivity analyses were also conducted to ensure the consistency and robustness of the results. Thus, the reliability of the IVs of this study was ensured (Table 3).
The possible reason for the inconsistent results between meta-analysis and MR analysis is the limitation of observational research. 36 It is noteworthy that two of the four included studies focused on the relationship between different comorbidities, and some confounding factors were not adjusted.15,18 For instance, Zhu et al. 37 highlight a positive association between smoking and OA prevalence in the general US population, while the association between smoking and asthma was unquestionable. However, smoking was not taken into account in the study by Romo-Nava et al. 34 Choi et al. concluded that OA was not associated with asthma, which might be because different classification criteria were used. Kellgren-Lawrence (KL) scale ≥1 were considered as OA according to Osteoarthritis Research Society International (OARSI). However, Choi et al. used KL scale ≥2 as criteria in their study. Although we did not find evidence for a causal association between OA and asthma, the LDSC analysis showed that these two diseases were highly correlated. OA and asthma interact through a variety of mechanisms. As mentioned in the introduction, several SNPs in the gene encoding SMAD3 are associated with knee or hip OA in populations.12,13 In addition, SMAD3 methylation was significantly associated with the production of IL-1β, an inflammatory mediator strongly associated with OA. 14 This may lead to a genetic link between the two diseases. Since the two diseases have diverse etiologies and are susceptible to misdiagnosis, like asthma and COPD, OA, and rheumatoid arthritis, the estimation of prevalence based on patient-reported questionnaire data is inherently flawed, which were subjected to selection bias, information bias, and social expectation bias.38–40 Patients cannot be induced to abandon treatment in order to study disease associations, and the treatment after the disease may introduce some bias since the subjects of observational research are often people with diseases. For instance, a few nonsteroidal anti-inflammatory drugs (e.g. aspirin and ibuprofen) have been extensively employed in OA and have been shown to may exacerbate asthma.41–45 Glucocorticoids are used in the treatment of both and may also be an interfering factor.46,47 As Mendy and Mersha pointed out, adult-onset asthma is more significantly associated with the prevalence of obesity, dyslipidemia, and OA than childhood-onset asthma, suggesting that age may be a confounding factor. However, age-stratified GWAS with OA is lacking at present, and the association between adult or pediatric asthma and OA needs to be explored in the future.
However, there are some limitations to this study. All four included studies had low scores after evaluation. In addition, the control group in the study of Koo et al. was not healthy people but COPD patients, which may cause bias and reduce the credibility of the results. Subgroup analysis was not conducted in the meta-analysis due to the heterogeneous and complex data sources. For the MR analysis, first, even though the largest GWAS available were used, some only discovered a small number of significant SNPs at the genome-wide level, thus leading to relatively flimsy genetic instruments. Furthermore, even though an updated array of sensitivity tests was performed to rule out horizontal pleiotropy, it is not possible to entirely exclude pleiotropic mechanisms without a thorough functional follow-up of these loci since we are unsure of the biological function of these SNPs. Although horizontal pleiotropy is a concern in MR reasoning, vertical pleiotropy—exposure acting on an outcome through other variables along the same causal path—is acceptable. For instance, if an exacerbation of OA leads to a decrease in physical activity and an increase in obesity, it may further lead to an exacerbation of asthma. Second, although meta-analyses have shown a correlation between asthma and the risk of OA, this study was not designed to address this issue. In addition, since fewer valid SNPs are obtained from other sites (such as hand, thumb, and ankle) of OA, the correlation of knee OA and hip OA with asthma was only considered in our study, whereas whether asthma also affects OA in other parts of the body was ignored, and there are complex relationships between recent medication, rest status, and work intensity. This may have affected the magnitude and direction of the observed MR estimates, whereas it cannot be evaluated in this study. Lastly, the study population included only individuals of European ancestry. Although the limitation of the study to ethnically homogeneous populations reduces the bias of population stratification, the results of this study cannot be generalized to other populations with different genetic backgrounds.
Conclusion
In brief, at present, no evidence was presented to support a direct causal relationship between asthma and OA. However, as the two most common chronic diseases, their effect on health is rising in scope. Given the limitations of this study, larger sample sizes and more ethnically diverse populations are required to analyze the relationship and underlying mechanisms between OA and asthma in the future.
Supplemental Material
sj-docx-7-dhj-10.1177_20552076231203648 - Supplemental material for Systemic evaluation of the relationship between asthma and osteoarthritis: Evidence from a meta-analysis and Mendelian randomization study
Supplemental material, sj-docx-7-dhj-10.1177_20552076231203648 for Systemic evaluation of the relationship between asthma and osteoarthritis: Evidence from a meta-analysis and Mendelian randomization study by Yaoyao Nie, Houpu Liu, Jing Wang, Ye Yang, Wenxia Zhao, Dingwan Chen and Yingjun Li in DIGITAL HEALTH
Supplemental Material
sj-docx-8-dhj-10.1177_20552076231203648 - Supplemental material for Systemic evaluation of the relationship between asthma and osteoarthritis: Evidence from a meta-analysis and Mendelian randomization study
Supplemental material, sj-docx-8-dhj-10.1177_20552076231203648 for Systemic evaluation of the relationship between asthma and osteoarthritis: Evidence from a meta-analysis and Mendelian randomization study by Yaoyao Nie, Houpu Liu, Jing Wang, Ye Yang, Wenxia Zhao, Dingwan Chen and Yingjun Li in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors deeply appreciate the initial GWAS and the associated consortiums for managing and disseminating the summary statistics.
Contributorship
YN: preparation, visualization, and writing—original draft. HL: data curation and investigation. JW: formal analysis and methodology. YY: project administration. WZ: data curation and validation. DC: supervision and validation. YL: funding acquisition and writing—review and editing. All authors have read and approved the final version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
In this study, publicly available data sets without any personal information were analyzed. There was no need for extra ethical approval because the initial GWAS already had the approval of the appropriate ethics review committees.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81703289).
Guarantor
YL.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
