Abstract
Objective
This study employed a bidirectional two-sample Mendelian randomization approach to investigate the causal links between Alzheimer’s disease, Lewy body dementia, vascular dementia, and various retinal diseases.
Methods
Summary data from large-scale genome-wide association studies of European ancestry were used to select genetic variants as instrumental variables. Causal estimates were derived using the inverse variance–weighted method, complemented by Mendelian randomization–Egger, weighted median, and weighted mode analyses to ensure robustness.
Results
Genetically predicted Alzheimer’s disease was associated with a reduced risk of disorders of the choroid and retina (odds ratio = 0.93, 95% confidence interval: 0.88–0.98), retinal detachments and breaks (odds ratio = 0.90, 95% confidence interval: 0.84–0.97), and retinal detachment with retinal break (odds ratio = 0.84, 95% confidence interval: 0.74–0.95). Lewy body dementia was negatively associated with age-related macular degeneration (odds ratio = 0.88, 95% confidence interval: 0.79–0.98), disorders of the choroid and retina (odds ratio = 0.96, 95% confidence interval: 0.93–0.99), and degeneration of the macula (odds ratio = 0.93, 95% confidence interval: 0.88–0.98). Vascular dementia showed negative associations with age-related macular degeneration (odds ratio = 0.93, 95% confidence interval: 0.87–0.99) and degeneration of the macula (odds ratio = 0.96, 95% confidence interval: 0.93–0.99). Conversely, reverse Mendelian randomization indicated that genetic liability to macular degeneration and choroidal/retinal disorders was causally associated with cognitive performance and a reduced risk of Alzheimer’s disease.
Conclusions
Findings support inverse causal relationships in which specific dementias may reduce retinal disease risk and vice versa, suggesting complex shared biological mechanisms.
Keywords
Introduction
Retinal diseases are major ophthalmological conditions that encompass a range of disorders, such as retinal vascular diseases, age-related macular degeneration (AMD), chorioretinitis, and retinal detachment, which are major contributors to blindness worldwide.1,2 Clinically, these conditions primarily manifest as severe visual impairments that often lead to blindness and substantially affect patient quality of life. 3 Epidemiological data from Europe indicate that the prevalence of age-related retinal diseases is likely to increase with the aging population. 4 Although previous research has linked age, genetics, and specific lifestyle factors to retinal diseases, 5 the precise etiology of many cases remains uncertain.
In recent years, cognitive impairments associated with retinal diseases have been widely reported. 6 However, the potential causal link between cognitive impairments and retinal diseases has not been sufficiently explored. Cognitive impairments, including Alzheimer’s disease (AD), Lewy body dementia (LBD), and vascular dementia, affect memory and executive functions and may also contribute to visual impairments. 7 Global epidemiological data indicate that the prevalence of cognitive impairments ranges from 5.1% to 41%, with an incidence rate of 22–76.8 per 1000 person-years. 8 Recent studies suggest that visual attention impairments, reduced visual processing speed, and altered visual perception in patients with cognitive impairments might be related to visual function impairments, 9 pointing to a possible biological link between these conditions.
However, the relationship between cognitive impairments and retinal diseases remains contentious, as shown by inconsistent and even contradictory findings across observational studies. Some studies report a higher incidence of retinal diseases among patients with cognitive impairments, 10 while others comparing patients with mild cognitive impairment (MCI) to control groups have observed significant thinning of the retinal inner layer in the central macular region. 11 In contrast, a cohort study from Korea reported an inverse association between retinal thickness and LBD. 12 In addition, several investigations have not yet identified significant correlations. For instance, a clinical study of 165 participants observed no retinal thickness differences between preclinical AD patients and healthy controls; 13 a large-scale population cohort reported that retinal diseases were not significantly linked to subsequent risk of AD or vascular dementia during follow-up; 14 and another study noted that although both AMD and AD are age-related conditions, there was no clear evidence of correlation between them. 15 These conflicting results are likely driven by residual confounding and reverse causality that are difficult to eliminate in observational designs.16,17 Clarifying this uncertainty is crucial for determining whether liability to cognitive impairment alters retinal disease risk and for resolving the current debate in the literature.
Mendelian randomization (MR) provides a robust method to assess the causal relationships between exposure factors and disease outcomes by using genetic variants as instrumental variables (IVs). This approach minimizes confounding and precludes reverse causation, approximating the conditions of a randomized controlled trial and thereby strengthening causal inference. This study uses a two-sample MR approach, selecting genetic variants associated with cognitive impairments, including AD, LBD, and vascular dementia, to investigate their potential impact on the risk of developing retinal diseases such as AMD, chorioretinitis, and retinal detachment. Recent applications of MR in studies examining the causal links between cognitive impairments and conditions such as rheumatoid arthritis 18 and psychiatric disorders 19 demonstrate that MR is a reliable tool for investigating associations between cognitive impairments and the risk of retinal diseases. By providing subtype-specific, genetically anchored effect estimates across multiple retinal phenotypes, this work addresses contradictions in the observational literature and contributes higher-level causal evidence to the field.
Clinically, elucidating the causal relationship between cognitive impairments and retinal diseases is important for the early prevention, diagnosis, and integrated management of these two prevalent age-related disorders. For instance, if a causal association exists, routine retinal imaging could serve as an accessible window for early detection of cognitive decline, while cognitive screening might help identify individuals at increased risk of retinal disease, enabling timely lifestyle or therapeutic interventions. 20 Such bidirectional clinical insights could inform risk-stratification strategies and promote multidisciplinary care models.
In summary, through two-sample MR analysis, this study selected known genetic variants associated with cognitive impairments as IVs to explore their potential impact on the risk of retinal diseases. This methodology clarifies the connection between these two conditions, provides a framework to address inconsistent epidemiological findings, and supports new approaches for the early diagnosis and treatment of retinal disease-related conditions, thereby improving the specificity and effectiveness of disease management.
Methods
Study design
As shown in Figure 1, this study used a two-sample MR approach to explore the causal relationship between cognitive disorders and retinal diseases. Four cognitive disorders—AD, LBD, frontotemporal dementia, and vascular dementia—were selected as exposure factors. Fifteen retinal conditions, including retinal vascular diseases, AMD, chorioretinal inflammation, and retinal detachment, were analyzed as outcome variables to determine causality. The MR analysis was guided by three core assumptions essential for validity: the relevance, independence, and exclusion restriction assumptions. 21 The genome-wide association study (GWAS) summary data required for the analysis were obtained from public databases and previously published literature, and no additional ethical approval was required.

Study design and analytic workflow of the bidirectional two-sample MR analysis. This flowchart illustrates the bidirectional MR framework used to investigate the causal relationship between cognitive impairment and retinal diseases. The solid arrow from cognitive impairment to retinal diseases represents the forward MR analysis, in which genetic variants associated with cognitive impairment were used as instrumental variables. The dashed arrow in the reverse direction represents the reverse MR analysis, testing the causal effect of retinal diseases on cognitive impairment. The study design adheres to three key MR assumptions: (a) relevance (genetic variants are robustly associated with the exposure, indicated by “√”); (b) independence (variants are not associated with confounders, indicated by the crossed-out path “x”); and (c) exclusion restriction (variants affect the outcome only through the exposure, also indicated by “x”). Primary causal estimates were derived using the IVW method, with MR–Egger, weighted median, and weighted mode as supplementary methods. A comprehensive suite of sensitivity analyses was performed to assess the robustness of the findings in both directions. MR: Mendelian randomization; IVW: inverse variance–weighted.
Data sources
GWAS summary data for the cognitive disorders were extracted from multiple sources. Data for AD included 39,106 cases and 401,577 European controls, sourced from a previous study. 22 LBD data comprised 2591 cases and 4027 European controls, obtained from previously published literature. 23 Data for frontotemporal dementia included 515 cases and 49,680 European controls, sourced from previously published literature. 24 Vascular dementia data, comprising 201 patients and 400,598 European controls, were sourced from the UK Biobank. GWAS summary data for the 15 types of retinal diseases were obtained from the FinnGen database, 6 with detailed case compositions and GWAS information provided in Table S1.
IV selection
IVs for the study were selected based on specific inclusion criteria. For AD and LBD, single-nucleotide polymorphisms (SNPs) were selected at genome-wide significance levels, with a P-value threshold of less than 5 × 10−8. For frontotemporal dementia and vascular dementia, a relaxed threshold of P-value less than 5 × 10−6 was applied due to fewer available SNPs. 25 This less stringent threshold (P < 5 × 10−6) was used for exposures with smaller GWAS sample sizes to ensure a sufficient number of IVs for a robust MR analysis, consistent with common practice in the field. To address the potential for weak instrument bias, the strength of each selected SNP was assessed using the F-statistic, calculated as F = (R2 × (N−2))/(1−R2). 26 To further confirm instrument robustness, we summarized the F-statistics for all selected SNPs in Table S2, demonstrating that each instrument exceeded the conventional F > 10 threshold, supporting the adequacy of instrument strength and minimizing potential weak-instrument bias.
Additionally, only SNPs with a minor allele frequency (MAF) greater than 0.01 were considered. To mitigate linkage disequilibrium (LD) among SNPs, those with R2 less than 0.001 within a window of 10,000 kb were selected. 27 When selected IVs were not available in the summary statistics for outcomes, SNPs in high LD (R2 > 0.8) with the IV were used as proxies. 28 Dataset harmonization was performed to ensure consistency of IVs across datasets by correcting for inconsistent alleles and intermediate allele frequencies.
MR analysis
Primary analysis using the inverse variance–weighted (IVW) method was performed to assess the causal relationships between cognitive impairments and retinal diseases. This method calculates the odds ratios (ORs) and their 95% confidence intervals (CIs) for each exposure–outcome pair. By employing the inverse variance of each SNP as weights, the IVW method provides a weighted average of effect sizes and serves as the main approach for interpreting MR results. 29 In addition to the IVW method, this study implemented MR–Egger regression, 30 weighted median, 31 and weighted mode 32 methods to test the robustness of the findings. The MR–Egger method adjusts for pleiotropy by incorporating an intercept term, providing a more accurate estimate of causal effects when pleiotropic effects are suspected. The weighted median method, based on the assumption that at least half of the IVs are valid, offers a robust assessment of the causal relationship. All statistical analyses were conducted using the ‘TwoSampleMR’ software package, 33 with results visualized through scatter plots and sensitivity analysis graphs. To assess the directionality of the causal relationship and test for reverse causation, we performed a bidirectional two-sample MR analysis. In this analysis, retinal diseases that showed a significant association in the primary analysis were treated as exposures, and the cognitive impairment phenotypes were treated as outcomes.
Sensitivity analysis
Sensitivity analyses were performed to ensure the reliability of the findings. Heterogeneity among IVs was assessed using Cochran’s Q test, where a P-value greater than 0.05 indicates low heterogeneity, suggesting that the variations among the IV estimates are due to random variation rather than systematic error, thereby supporting the reliability of the IVW results. 34 To address potential pleiotropy, which can influence the estimation of association effects, MR–Egger regression was employed. 30 An intercept close to zero or not statistically significant in MR–Egger regression indicates an absence of horizontal pleiotropy. Furthermore, the MR–PRESSO method was used to identify and exclude potential outliers—SNPs with significant pleiotropic effects detected at P < 0.05. 35 Recalculation of causal estimates after removing these outliers helps to correct for any distortion due to horizontal pleiotropy. In addition, a leave-one-out analysis was conducted to ensure the robustness and consistency of the results, confirming the reliability of the causal inferences drawn from the study.
Results
Selection of IVs
For AD as the exposure, 59 related IVs were selected. Among these, rs10769263 served as a proxy SNP for rs11500477, and rs35118762 served as a proxy for rs35472547; one SNP did not have a proxy available. For LBD, five IVs were selected; for frontotemporal dementia (TDP subtype), seven were selected; and for vascular dementia, six were selected. All IVs had an F-statistic greater than 10 (Table S3).
Preliminary sensitivity analysis led to the exclusion of outliers, followed by MR analysis. Specifically, MR–Egger regression indicated pleiotropic influences in the analysis of AD with chorioretinal inflammation as the outcome. Outliers (“rs6733839”) were excluded based on the results of leave-one-out test. MR–PRESSO analysis led to the exclusion of SNPs, indicating potential pleiotropy between AD and traction detachment of the retina as well as disorders of the choroid and retina (“rs35472547”) and between vascular dementia and disorders of the choroid and retina (“rs429358” and “rs9986991”). The analysis was then rerun after removing these SNPs to ensure the robustness of the MR results.
MR analysis
As shown in Table 1, IVW analysis revealed significant negative causal associations between AD and disorders of the choroid and retina (OR = 0.93, 95% CI: 0.88–0.98, P = 0.007), retinal detachments and breaks (OR = 0.90, 95% CI: 0.84–0.97, P = 0.007), and retinal detachment with retinal break (OR = 0.84, 95% CI: 0.74–0.95, P = 0.007). LBD was significantly associated with AMD (both dry and wet) (OR = 0.88, 95% CI: 0.79–0.98, P = 0.018), disorders of the choroid and retina (OR = 0.96, 95% CI: 0.93–0.99, P = 0.005), and degeneration of the macula and posterior pole (OR = 0.93, 95% CI: 0.88–0.98, P = 0.005). Vascular dementia also showed significant associations with AMD (both dry and wet) (OR = 0.93, 95% CI: 0.87–0.99, P = 0.034) and degeneration of the macula and posterior pole (OR = 0.96, 95% CI: 0.93–0.99, P = 0.006). Complete results are provided in Table S4. These findings were visualized through scatter plots (Figure S1, left panels).
Causal effects of cognitive impairments on retinal diseases (forward MR analysis).

N.SNPs: number of single-nucleotide polymorphisms; OR: odds ratio; CI: confidence interval; MR: Mendelian randomization; AMD: age-related macular degeneration.
All P-values are two-sided. Associations are presented when at least one MR method reached P < 0.05, after correction for heterogeneity and pleiotropy where applicable.
Furthermore, the weighted median and weighted mode methods supported the causal associations between AD and disorders of the choroid and retina, LBD and AMD (both dry and wet), and degeneration of the macula and posterior pole. The weighted median and weighted mode methods, together with MR–Egger regression, also supported the causal association between vascular dementia and degeneration of the macula and posterior pole, with MR–Egger further confirming the association between AD and retinal detachments and breaks. These results reinforce the robustness of the causal inferences.
Sensitivity analysis
Sensitivity analyses were conducted on the previously identified positive results, using Cochran’s Q test to assess heterogeneity. As shown in Table 2 and Table S5, heterogeneity was observed in the analyses of AD and disorders of the choroid and retina (P = 0.007), LBD and AMD (both dry and wet) (P = 0.021), and vascular dementia and AMD (both dry and wet) (P = 0.002). Given that this study primarily utilized random-effect IVW analysis, this level of heterogeneity is considered somewhat acceptable. Results from MR–Egger regression analysis indicated that the analysis is not affected by horizontal pleiotropy. However, the MR–PRESSO analysis showed that the analysis of AD and disorders of the choroid and retina was affected by pleiotropy (P = 0.006), as shown in Table 3 and Table S6. The leave-one-out test, illustrated in Figure S1 (right panels), did not reveal any outliers, further affirming the robustness of the MR results.
Heterogeneity and pleiotropy between cognitive impairments and retinal diseases assessed by MR.

MR: Mendelian randomization; IVW: inverse variance–weighted; AMD: age-related macular degeneration.
Values are reported as P-values. Nonsignificant results (P ≥ 0.05) indicate no evidence of substantial heterogeneity or directional pleiotropy.
Assessment of pleiotropy of cognitive impairments and retinal diseases using MR–PRESSO.
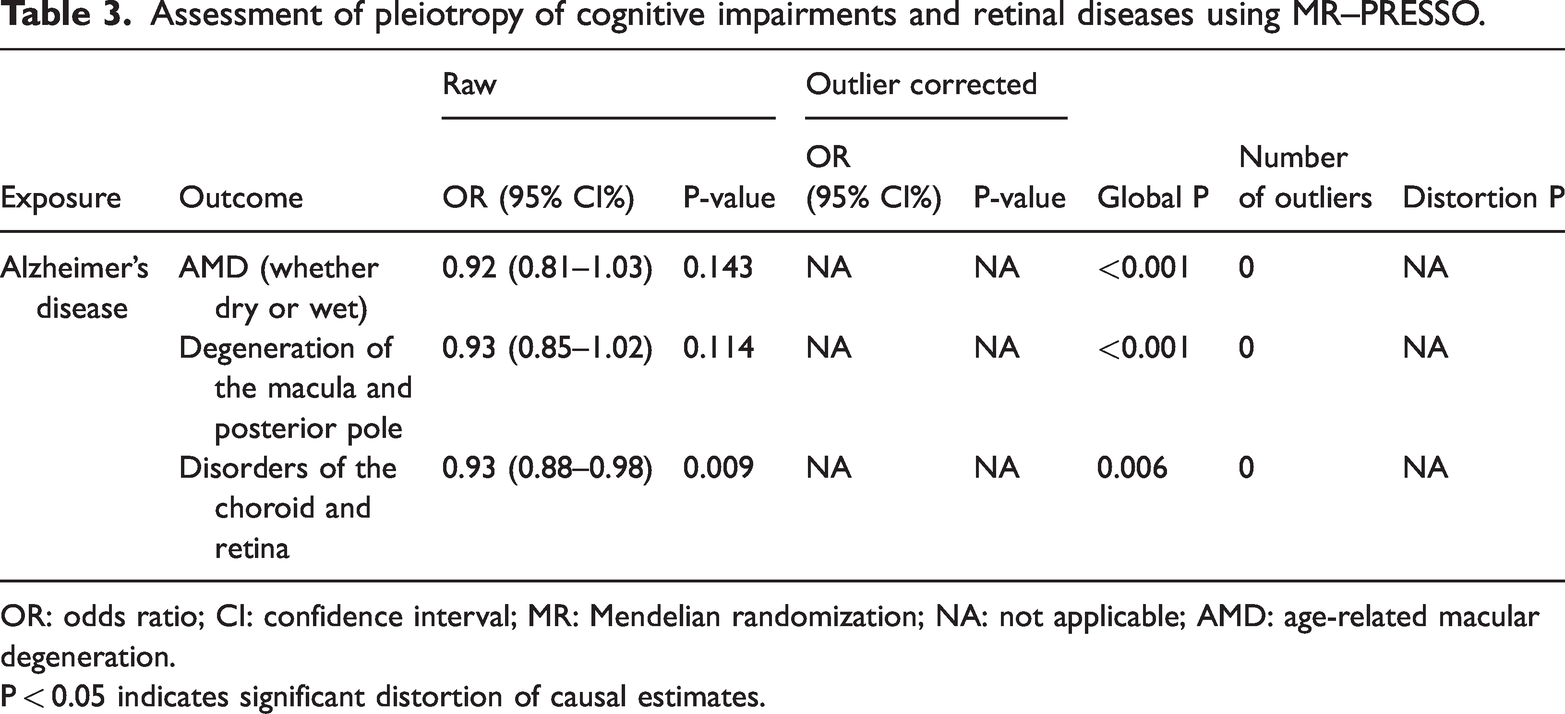
OR: odds ratio; CI: confidence interval; MR: Mendelian randomization; NA: not applicable; AMD: age-related macular degeneration.
P < 0.05 indicates significant distortion of causal estimates.
For other exposure–outcome pairs where positive results were not initially detected, Cochran’s Q test indicated heterogeneity in the analyses of AD and degeneration of the macula and posterior pole (P < 0.001), LBD with retinal detachments and breaks (P = 0.032), retinal detachment with retinal break (P = 0.022), retinal vascular disorders (P = 0.009), retinoschisis and retinal cysts (P = 0.049), retinal vascular occlusion (P = 0.012), and vascular dementia with traction detachment of the retina (P = 0.032), as detailed in Table 2. MR–Egger regression analysis indicated that these analyses were not influenced by horizontal pleiotropy. However, MR–PRESSO analysis showed that the analyses of AD with AMD (both dry and wet) and degeneration of the macula and posterior pole were affected by pleiotropy (P < 0.001), as listed in Table 3. Despite these findings, the leave-one-out test and funnel plots did not reveal any outliers, supporting the robustness of the MR results across all tested exposure–outcome pairs.
Bidirectional MR and reverse causality analysis
Based on the initial sensitivity analyses, outlier variants were removed before conducting the reverse-direction MR analyses. In these analyses, we investigated whether genetic liability to retinal diseases had a causal effect on cognitive impairment phenotypes (Table S7). After correcting for multiple testing (false discovery rate (FDR) < 0.05), significant associations were observed for several retinal traits. Specifically, genetically predicted degeneration of the macula and posterior pole was positively associated with cognitive function (OR = 1.1677, 95% CI: 1.0872–1.2542, P = 2.10 × 10−5, FDR = 0.003) and cognitive performance (OR = 1.0252, 95% CI: 1.0125–1.038, P = 8.90 × 10−5, FDR = 0.007). Disorders of the choroid and retina were associated with a higher risk of impaired cognitive performance (OR = 1.0488, 95% CI: 1.0215–1.0767, P = 0.00039, FDR = 0.021), whereas an inverse association was detected with AD (OR = 0.7692, 95% CI: 0.6568–0.9009, P = 0.001, FDR = 0.041). These findings are illustrated in Figure S2 (left panels).
Sensitivity analyses confirmed the robustness of these reverse-direction findings. Outliers identified by MR–PRESSO and leave-one-out tests, including several APOE variants (Table S8), were removed, after which the key associations—degeneration of the macula and posterior pole with cognitive function and cognitive performance and disorders of the choroid and retina with cognitive performance and AD—remained directionally consistent and statistically significant. MR–Egger intercept tests showed no evidence of directional pleiotropy. Heterogeneity was detected in a few additional pairs, including chorioretinal inflammation with AD, degeneration of the macula and posterior pole with fluid-intelligence score, retinal vascular disorders with cognitive function, AMD with reaction time, and central retinal artery occlusion with cognitive performance (all Q-test P < 0.05; Table S9). MR–PRESSO global tests were also positive for AMD with reaction time (global P = 0.039), central retinal artery occlusion with cognitive performance (P = 0.035), retinal vascular disorders with cognitive function (P = 0.043), and retinoschisis and retinal cysts with cognitive performance (P = 0.039) (Table S10). Importantly, exclusion of these outliers did not materially alter the effect estimates. Leave-one-out analyses (Figure S2, right panels) similarly revealed no influential SNPs (Figures S3 and S4), further supporting the stability of the reverse-direction MR findings.
Discussion
This study used a two-sample, bidirectional MR design to systematically examine the potential causal relationships and directionality between cognitive impairments and a range of retinal diseases. In the forward MR analysis, we observed that genetically predicted AD, LBD, and vascular dementia were associated with a lower risk of specific retinal disorders, suggesting a potential inverse causal effect. For example, AD liability was associated with a reduced risk of choroidal and retinal disorders (OR = 0.93, P = 0.007) and retinal detachment (OR = 0.90, P = 0.007). To confirm the causal direction, we further performed reverse MR analysis, which showed no evidence that retinal diseases are causal risk factors for cognitive impairments. Collectively, these bidirectional analyses indicate a likely causal direction from cognitive impairments toward a reduced risk of retinal diseases.
Interestingly, the bidirectional MR analysis provided a more nuanced perspective. Although the primary analysis suggested a potential protective effect of dementia on retinal diseases, the reverse analysis revealed a significant causal effect of retinal degenerative diseases on cognitive performance and AD. This bidirectional association indicates a complex shared pathophysiology rather than a simple unidirectional causal relationship. For example, neurodegenerative processes, such as abnormal protein aggregation or chronic inflammation, may occur in parallel in both the brain and the retina, leading to these observed bidirectional effects. These findings extend beyond a simple cause-and-effect relationship and point to shared biological mechanisms or pathways underlying both neurodegeneration and retinal degeneration.
Our findings both contrast with and complement prior work, in which observational results have been inconsistent and sometimes contradictory. Several studies reported positive associations—for example, macular retinal nerve fiber layer thinning correlating with increased dementia incidence,36,37 or reduced retinal blood flow in patients with MCI and AD 38 —while others, such as a Korean cohort study, observed negative associations between retinal thickness and LBD.12,39 Additional investigations reported null results, including a clinical study of 165 participants showing no retinal thickness differences between preclinical AD and controls, 13 a large population-based cohort indicating no association between retinal disease and AD or vascular dementia risk, 14 and a study finding no clear correlation between AMD and AD. 15 Such divergence likely arises from unmeasured confounding factors (e.g. lifestyle, comorbidities) and reverse causality that cannot be fully addressed in conventional epidemiology. By leveraging germline genetic variants as IVs, our bidirectional MR framework minimizes these biases and formally tests causal directionality, thereby providing higher-level genetic evidence to resolve these long-standing inconsistencies.
The inverse causal associations observed in this study raise important questions about the biological pathways through which cognitive impairment-related processes might reduce the risk of retinal disease. One plausible mechanism involves enhanced neuroprotection. In the prodromal or early stages of neurodegenerative disorders, compensatory upregulation of endogenous neurotrophic and synaptic maintenance factors—such as brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), and glial cell line-derived neurotrophic factor (GDNF)—has been reported. 40 These factors can cross the blood–brain barrier or enter the systemic circulation, potentially reaching the retina, an anatomic extension of the central nervous system. Once in the ocular microenvironment, they may promote retinal ganglion cell survival, support synaptic remodeling, and attenuate oxidative stress and mitochondrial dysfunction, all of which are implicated in chorioretinal degeneration and AMD. 41 A second, nonexclusive mechanism involves systemic immune modulation. Neurodegenerative disorders such as AD and LBD are known to reshape peripheral immune profiles, shifting cytokine balances (e.g. interleukin-10 upregulation and tumor necrosis factor-α downregulation) and altering microglial and T-cell activation states. 42 Such systemic changes may reduce chronic low-grade inflammation within the retina, stabilize the blood–retina barrier, and mitigate complement-mediated or microglia-driven retinal injury. 43
Key strengths of this study include the two-sample bidirectional MR design, which allowed simultaneous assessment of causal effects and their directionality, and the large-scale GWAS datasets, which enhanced statistical power. Multiple sensitivity analyses (MR–Egger regression, MR–PRESSO, leave-one-out) confirmed the robustness of the results. Nevertheless, several limitations should be considered. First, the predominantly European ancestry of the GWAS datasets may limit the generalizability of the findings to other populations. Second, although tests for pleiotropy and heterogeneity were conducted, residual unrecognized pleiotropy cannot be entirely excluded. Third, for some dementia subtypes with relatively small numbers of instrumental SNPs, such as frontotemporal dementia, null findings should be interpreted with caution due to limited power. Fourth, formal statistical power calculations were not performed, particularly for dementia subtypes with smaller sample sizes. Therefore, while the absence of significant associations may reflect a true lack of causal effect, the possibility that insufficient statistical power masked weak associations cannot be entirely ruled out. Future studies with larger GWAS datasets and explicit power calculations are warranted to validate these null findings.
In summary, this bidirectional MR study provides genetic evidence that certain neurodegenerative cognitive disorders may causally reduce the risk of specific retinal diseases, whereas no evidence supports retinal disease as a causal risk factor for cognitive impairment. These results offer a novel perspective on brain–eye connections. Future studies should incorporate functional experiments to validate the proposed neuroprotective and immune-modulatory pathways and replicate these analyses in diverse populations to strengthen external validity. Ultimately, a deeper understanding of these intrinsic protective mechanisms may identify new therapeutic targets for simultaneous neuroprotection in both the brain and retina.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251404265 - Supplemental material for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251404265 for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study by Qin Shi, Dongmei Zhan, Weiling Wang, Xinxin Wu, Siqi Sheng and Yifan Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251404265 - Supplemental material for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study
Supplemental material, sj-pdf-2-imr-10.1177_03000605251404265 for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study by Qin Shi, Dongmei Zhan, Weiling Wang, Xinxin Wu, Siqi Sheng and Yifan Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251404265 - Supplemental material for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study
Supplemental material, sj-pdf-3-imr-10.1177_03000605251404265 for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study by Qin Shi, Dongmei Zhan, Weiling Wang, Xinxin Wu, Siqi Sheng and Yifan Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-4-imr-10.1177_03000605251404265 - Supplemental material for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study
Supplemental material, sj-pdf-4-imr-10.1177_03000605251404265 for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study by Qin Shi, Dongmei Zhan, Weiling Wang, Xinxin Wu, Siqi Sheng and Yifan Wang in Journal of International Medical Research
Supplemental Material
sj-xlsx-5-imr-10.1177_03000605251404265 - Supplemental material for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study
Supplemental material, sj-xlsx-5-imr-10.1177_03000605251404265 for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study by Qin Shi, Dongmei Zhan, Weiling Wang, Xinxin Wu, Siqi Sheng and Yifan Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-6-imr-10.1177_03000605251404265 - Supplemental material for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study
Supplemental material, sj-pdf-6-imr-10.1177_03000605251404265 for Causal associations between cognitive impairments and retinal diseases: A two-sample Mendelian randomization study by Qin Shi, Dongmei Zhan, Weiling Wang, Xinxin Wu, Siqi Sheng and Yifan Wang in Journal of International Medical Research
Footnotes
Acknowledgments
None.
Authors’ contributions
Qin Shi and Yifan Wang conducted the study, participated in data collection, and drafted the manuscript. Dongmei Zhan and Weiling Wang performed the statistical analysis and contributed to the study design. Xinxin Wu and Siqi Sheng participated in data acquisition, analysis, or interpretation and contributed to drafting the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this article and supplementary information files.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
This article is a Mendelian randomization study. The data were obtained from publicly available databases and published literature; therefore, ethical approval and the need for written informed consent were waived.
Funding
This study was supported by the Key Research and Development Program of Ningxia (No. 2022BEG02046).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
