Abstract
Objective
In this study, we aimed to explore the potential association between COVID-19 infection, hospitalization, severe COVID-19, and erection dysfunction (ED) using the two-sample Mendelian randomization (MR) method.
Methods
Data pertaining to COVID-19 were extracted from the latest version of the COVID-19 Host Genetics Initiative genome-wide association study (GWAS) meta-analyses (Round 7, April 2022), and outcome data were obtained from the Open GWAS database. We applied various MR analysis methods, including the inverse variance weighted method, weighted median method, and MR-Egger regression.
Results
Our investigation revealed a negative causal association between COVID-19 hospitalization and ED (total testosterone levels: beta = −0.026; 95% confidence interval: −0.049 to −0.001). However, no evidence supported causal relationships between COVID-19 infection, hospitalization for COVID-19, or severe COVID-19 and other ED risk factors.
Conclusion
The results of this comprehensive MR analysis suggest a negative causal link between COVID-19 hospitalization and total testosterone levels. Nonetheless, COVID-19 (comprising infection, hospitalization, and severe illness) may not directly correlate with an increased risk of ED. These findings imply that COVID-19 may exert a distinct impact on ED through indirect pathways.
Keywords
Introduction
Erectile dysfunction (ED) is defined as the inability of men to achieve or maintain sufficient penile erection for satisfactory sexual function. 1 ED is prevalent in 10% of men over age 40 years and surpasses 50% among men over 70 years old.2,3 The worldwide prevalence of ED is on the rise, making it a major health concern in a growing aging population.4,5 Many risk factors can decrease the level of testosterone and impair endothelial function, thereby leading to ED. 6 COVID-19, an acute infectious disease, has infected nearly 250 million people and resulted in millions of deaths since the start of the pandemic in 2019. 7 Studies show that the virus that causes the disease, SARS-CoV-2, can damage endothelial function, leading to systemic disorders. 8 Vascular endothelial cell dysfunction serves as a pathological basis for ED.9,10 Since the onset of the COVID-19 pandemic, several studies have investigated the impact of COVID-19 infection on male reproductive health. 11 Dubin et al. reported that COVID-19 infection can impair sexual function. 11 Another study suggested that SARS-CoV-2 can persist in the penis for a long time, implying that endothelial dysfunction resulting from COVID-19 infection may lead to ED. 12 Additionally, research has revealed that COVID-19 has a detrimental effect on the testis and sperm, contributing to ED.13,14 Katz et al. found that ED is more likely to occur in patients with COVID-19, even after adjusting for confounding factors. 15 Some studies have reported a decrease in erectile function during the epidemic period of COVID-19.16,17 However, Gutzeit et al. demonstrated that COVID-19 infection has no significant effect on male sexual function. 18 It is important to note that observational studies face limitations such as confounding factors and reverse causality, necessitating further research to fully understand the relationship between COVID-19 and ED. Moreover, randomized controlled studies are unsuitable for investigating ED. 19 In the context of assessing the relationship between COVID-19 and ED, using a Mendelian randomization (MR) design could provide crucial insights into the potential causal nature of this association. MR analysis is a powerful method used to generate causal evidence and can circumvent many of the limitations of observational studies.
In this study, we aimed to investigate the association between COVID-19 and the risk of ED and provide valuable insight into the risks associated with ED. To determine whether COVID-19 affects erectile function via indirect factors, we also examined the potential causal effects of COVID-19 on ED risk factors.
Methods
Ethics approval and informed patient consent were not required for the present study because we used data from the Open GWAS database. In the MR analysis, we relied on three assumptions: (1) the genetic variant is associated with the exposure; (2) the genetic variant is not associated with the outcome; (3) the genetic variant influences the outcome only through the exposure. 20 The study flow chart is presented in Figure 1.

Flow chart of Mendelian randomization (MR) study on the causal relationship of COVID-19 infection, hospitalization, and severe COVID-19 with ED. ED, erectile dysfunction; SNP, single nucleotide polymorphism; GWAS, genome-wide association study.
Genetic variants associated with COVID-19
We retrieved the latest version of COVID-19 data from the COVID-19 Host Genetics Initiative genome-wide association study (GWAS) database (Round 7, April 2022).
21
The data comprised 122,616 COVID-19 cases and 2,475,240 controls. Hospitalization data included 32,519 cases and 2,062,805 controls, and severity data involved hospitalized patients who either died or required respiratory support owing to COVID-19 (cases = 13,769, controls =1,072,442). Single nucleotide polymorphisms (SNPs) significantly associated with COVID-19 at a genome-wide significance level (
Outcome data source
The summary data for ED were obtained from the Open GWAS database (https://gwas.mrcieu.ac.uk/). This dataset comprised 6175 cases and 217,630 controls. Summary-level statistics for ED risk factors were also extracted from the Open GWAS database. Common risk factors for ED, such as type 2 diabetes, primary hypertension, low-density lipoprotein cholesterol (LDL), testosterone level, and body mass index (BMI) were collected. 22 All data for the analysis are based on the European population. Details of all data included in our study are listed in Table 1.
Details of GWAS data included in the Mendelian randomization.
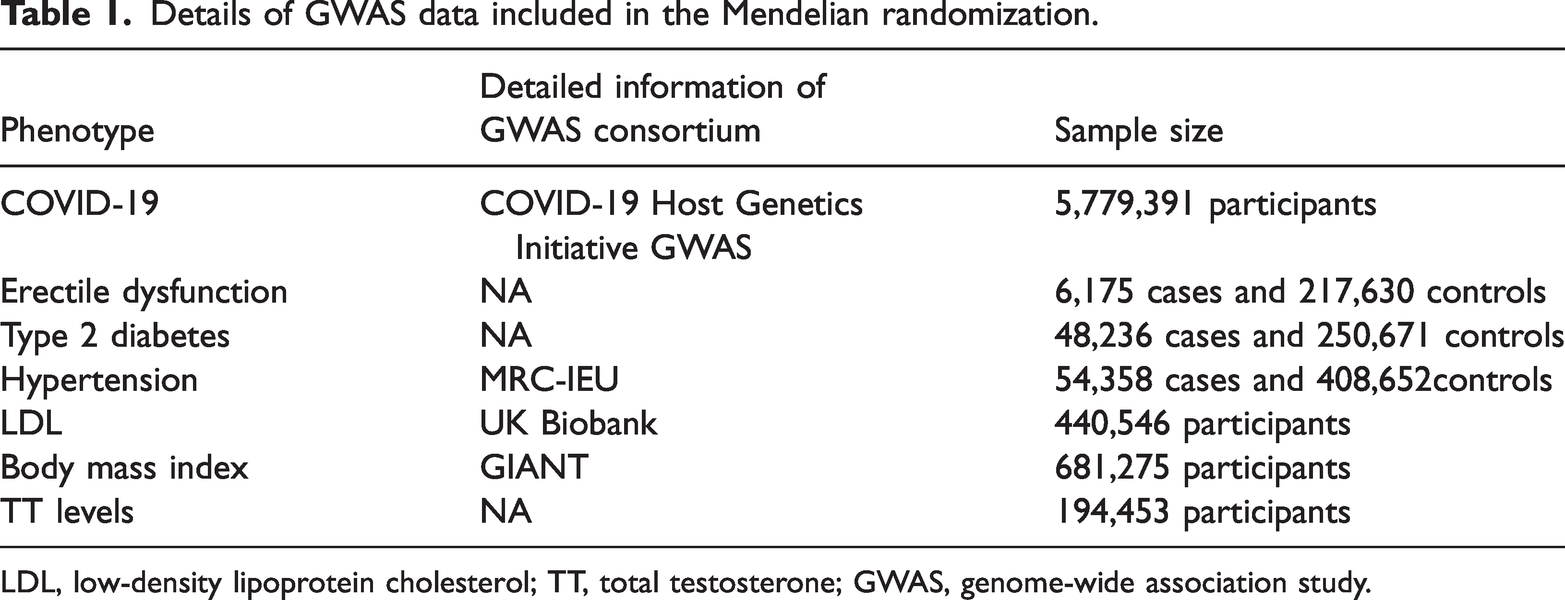
LDL, low-density lipoprotein cholesterol; TT, total testosterone; GWAS, genome-wide association study.
Mendelian randomization (MR) analyses
In this MR study, we applied three methods including inverse variance weighting (IVW), MR-Egger, and weighted median. The IVW method estimates the Wald ratio of each SNP between the exposure and the outcome, serving as the primary method. 23 The MR-Egger method can achieve reliable causal effects when SNPs are pleiotropic. 23 The weighted median method accurately estimates the causal relationship when genetic variants are invalid. 24
The results are presented using odds ratio (OR) and 95% confidence interval (CI), with
Results
Association between COVID-19 and ED
We identified 11, 25, and 21 genetic instruments with a significance of

Forest plots for the association of ED risk with COVID-19 infection, hospitalization, and severe COVID-19 SNPs. ED, erectile dysfunction; SNP, single nucleotide polymorphism; MR, Mendelian randomization, IVW, inverse variance weighted; OR, odds ratio; CI, confidence interval.
Association between COVID-19 and ED risk factors
We further investigated the causal effects between COVID-19 and ED risk factors, including type 2 diabetes, primary hypertension, LDL cholesterol, testosterone level, and BMI. The results are presented in Figure 3. Among these factors, we identified a potential causal association solely between COVID-19 hospitalization and total testosterone levels. The result of the IVW method revealed that COVID-19 hospitalization was associated with a reduction in total testosterone levels (beta = −0.026; 95% CI: −0.049 to −0.001;

Causal effect of COVID-19 infection, hospitalization, and severe COVID-19 on ED risk factors. Forest plots showing the range of OR values for different risk factors. The black point (
Discussion
In this study, we did not find evidence supporting the causal effects of COVID-19 infection, hospitalization, or severity on the risk of ED. Some recent studies have shown an increased prevalence of ED linked to COVID-19 infection9,15 whereas other studies have reported higher rates of both premature ejaculation and ED in the post-pandemic era, as compared with the pre-pandemic period.26,27 However, confounding factors such as diabetes, coronary artery disease, and chronic obstructive pulmonary disease were included in those studies, making it unclear whether COVID-19 contributes directly to the occurrence of ED. To address this uncertainty, we leveraged genetic instruments to overcome the limitations inherent in observational studies, aiming to provide more reliable evidence concerning COVID-19's potential impact on the development of ED. Our findings did not indicate a causal impact of COVID-19 infection, COVID-19 hospitalization, or COVID-19 severity on ED. Additional investigation revealed a potential causal association between COVID-19 hospitalization and total testosterone levels.
To our knowledge, only one prior study has delved into the potential causal effect between COVID-19 and ED within an MR framework. That study only included six SNPs and reported a causal link between COVID-19 susceptibility and an elevated risk of ED. 28 Notably, we found that one of the six SNPs included in that study was associated with coronary artery disease and myocardial infarction. However, existing research has established that coronary artery disease and metabolic disorders can induce ED by affecting both pituitary and penile function. 29 Furthermore, all instrumental variables in that prior study had linkage disequilibrium r2 < 0.3, which failed to confirm the independence of the selected genetic variants. Compared with that study, we used a larger sample size and more genetic variants and excluded confounding factors associated with coronary artery disease and myocardial infarction. We also conducted subgroup analysis based on the severity of COVID-19. The study results did not support a causal association between COVID-19 and ED. It is noteworthy that our findings implied that enhancing ED screening in patients with COVID-19 may not be useful. Moreover, our results can alleviate worries among men regarding ED during the COVID-19 pandemic. Earlier studies have highlighted that anxiety and depression associated with COVID-19 contribute to the development ED.30–32 In addition, some studies have shown that COVID-19 and ED share common risk factors including obesity, cardiovascular disease, diabetes, and chronic kidney disease. 15 Here, we identified associations between certain SNPs (rs1128175 and rs550057 with diabetes mellitus, rs1498399 and rs1634761 with BMI) and specific risk factors. Recent research has demonstrated that both diabetes and obesity have detrimental effects on both COVID-19 infection and ED. 33 The pro-inflammatory state induced by diabetes and obesity can decrease nitrous oxide biological activity, contributing to ED.34,35 Furthermore, SNP rs11264339 is associated with smoking, which has been reported to damage erectile function in a dose-dependent manner. 36 The SNPs rs368565, rs492602, and rs679574 are related to alcohol. Wang et al. demonstrated that alcohol abuse can lead to ED. 37 In addition, the SNPs rs62056905 and rs63750417 are associated with mental disorders, such as anxiety and depression. Mental disorders and ED mutually influence and reinforce each other. 38 In our study, we excluded the abovementioned SNPs associated with diabetes, hypertension, smoking, alcohol, and obesity. This ensured that our results would be less likely to be biased by these factors, yielding a more focused and specific analysis.
To verify our results and further explore the role of COVID-19 in ED, we added ED risk factors as outcomes and scrutinized the causal associations. Previous studies have revealed a significant increase in the rates of hypertension, hyperlipidemia, and diabetes among men with ED.39,40 Testosterone, recognized as the primary regulatory factor, plays a crucial role in male erection. 41 Lower testosterone levels may not only exacerbate ED but may also cause it. 42 After analyzing the effect of COVID-19 on ED risk factors including type 2 diabetes, hypertension, LDL, total testosterone levels, and BMI, we discovered potential negative causal evidence between COVID-19 hospitalization and total testosterone levels. However, no significant causal association was found between COVID-19 and other risk factors. The mechanisms underlying the observed association between COVID-19 and testosterone levels remain unclear. Studies have revealed that COVID-19 pneumonia leads to a decrease in testosterone levels, and this decrease is associated with severity of the disease. 43 Approximately 30% of men who had recovered from COVID-19 exhibited low circulating testosterone levels, indicating a potential condition of hypogonadism, even after a minimum 12-month follow-up. 44 One study reported that COVID-19 has a notable impact on erectile function, particularly among older patients with underlying health conditions. 45 Studies examining the effects of SARS-CoV-2 on gonadal function have shown that the virus lowers testosterone levels through suppression of the pituitary gland.46,47 In summary, our results suggested that only COVID-19 hospitalization was causally associated with decreased total testosterone levels.
Exposure to the COVID-19 pandemic is accompanied by increased anxiety, depression, and stress disorders, potentially leading to an increase in ED cases. 48 Moreover, a pilot study revealed that COVID-19 may lead to ED owing to damaged cavernosal smooth muscle. 49 In addition, COVID-19 may impact erectile function through indirect impacts on the cardiovascular system and as a consequence of treatments for COVID-19. 41 In summary, COVID-19 may harm men’s health and erectile function by influencing metabolic disorders, mental health, and via other mechanisms. Focusing on mental disorders in individuals with COVID-19 after the pandemic may be necessary. However, in our study, we did not explore the effect of COVID-19 on cardiovascular disease, mental disorders, socioeconomic issues, and other risk factors.
Our study possesses several strengths. MR analysis is a method for establishing potential causal relationships. By leveraging genetic variants as instrumental variables, MR analysis can overcome limitations associated with observational studies, such as confounding factors and reverse causality. The present results can contribute valuable insights to the existing literature on the association between COVID-19 and the risk of ED. Specifically, our findings provide robust evidence of decreased total testosterone levels associated with COVID-19 hospitalization, suggesting a potential adverse effect of COVID-19 hospitalization on testosterone levels. Such insight is particularly pertinent given the worldwide pandemic of COVID-19. However, our study has some limitations. The prevalence of ED among different racial and ethnic groups is the result of complex phenomena and may be dependent upon the interplay of socioeconomic, demographic, medical, cultural, and lifestyle factors, which we did not explore deeply in this research. In addition, generalizability may be restricted as our study predominantly included individuals of European ancestry, leaving uncertainties about the consistency of our findings in other populations. A sex bias may be present given that the COVID-19 data encompassed a mixed-sex database, and the ED data exclusively involved male individuals. This may have influenced the results to some extent, but it is difficult to isolate male COVID-19 data. Furthermore, although MR analysis can enable genetic causal inference, it cannot be used to directly elucidate the clinical impact of COVID-19 hospitalization on total testosterone levels.
Conclusion
Our results indicated that COVID-19 infection, hospitalization, and severity may not be causally associated with ED. Consequently, our findings do not support routine screening for ED after the pandemic. However, our results demonstrated a potential causal effect of COVID-19 hospitalization on reduced total testosterone levels. The examination of total testosterone levels may be useful, and screening for mental disorders is recommended among individuals with COVID-19 and ED. Further investigation is warranted to elucidate the underlying mechanisms and assess the clinical implications of these findings. The evidence presented here calls for a greater focus on men’s health and highlights the adverse effect of COVID-19 hospitalization on testosterone levels. Owing to the prolonged presence of COVID-19, identifying mental effects and providing additional health care resources may be advisable to alleviate the impact of COVID-19 on men’s health.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241274236 - Supplemental material for Causal associations between COVID-19 and erectile dysfunction: a Mendelian randomization study
Supplemental material, sj-pdf-1-imr-10.1177_03000605241274236 for Causal associations between COVID-19 and erectile dysfunction: a Mendelian randomization study by Chao Ma, Li Ma and Pu Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605241274236 - Supplemental material for Causal associations between COVID-19 and erectile dysfunction: a Mendelian randomization study
Supplemental material, sj-pdf-2-imr-10.1177_03000605241274236 for Causal associations between COVID-19 and erectile dysfunction: a Mendelian randomization study by Chao Ma, Li Ma and Pu Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605241274236 - Supplemental material for Causal associations between COVID-19 and erectile dysfunction: a Mendelian randomization study
Supplemental material, sj-pdf-3-imr-10.1177_03000605241274236 for Causal associations between COVID-19 and erectile dysfunction: a Mendelian randomization study by Chao Ma, Li Ma and Pu Wang in Journal of International Medical Research
Footnotes
Acknowledgements
We sincerely appreciate the great work of the COVID-19 Host Genetics Initiative Consortium and all GWAS consortiums and authors for making the summary statistics publicly available.
Author contributions
Chao Ma and Pu Wang conceived the study and analyzed the data. Chao Ma wrote the manuscript and Li Ma revised the manuscript. Both authors reviewed the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
