Abstract
Antioxidants act by preventing excessive oxidative stress within the developing sperm cells. Hookah smoking has been implicated in inducing oxidative stress which may be a risk factor for male infertility. This study aims to bridge this knowledge gap by evaluating the effect of hookah smoking on sperm quality and the expression of nuclear factor erythroid 2-related factor 2 (NRF2) and Cytochrome P450 family 24 subfamily A member 1 (CYP24A1) genes which are involved in antioxidant response. Young fertile men in reproductive age were categorized into hookah smokers (n = 32) and non-smokers (n=52). Sperm parameters (concentration, volume, motility, morphology) as well as gene expression of NRF2 and CYP24A1 were evaluated in each sperm sample. There was no meaningful change in sperm parameters in the case group compared with the controls. The mRNA levels of NRF2 was significantly higher in the case group compared with the controls (p = 0.01), while CYP24A1 expression did not significantly change between the study groups. A remarkable increase in NRF2 expression in hookah smokers might be a cellular defense mechanism, indicating activation of antioxidant response in sperm to counteract potential oxidative stress caused by hookah use. However, further research is needed to understand the functional consequences of this upregulation.
Introduction
Smoking tobacco is a risk factor for male infertility, with its adverse effects on sperm function attributed to oxidative stress (imbalance between the production of reactive oxygen species (ROS) and the body’s antioxidant) and DNA damage. The adverse impact is likely caused by the numerous toxic compounds found in tobacco, such as carbon monoxide and nicotine (Fesahat et al., 2023; Harlev et al., 2015; Kovac et al., 2015). These effects collectively impair sperm motility, morphology, and fertilization potential (Osadchuk et al., 2023). While the detrimental impact of cigarette smoking on male fertility is well-established (Qutub et al., 2022), the impact of hookah smoking remains relatively underexplored. Evidence suggests that hookah smoking can have similar effects as cigarette smoking (Fawzy et al., 2011; Firouzabadi, Henkel, et al., 2024). The underlying mechanisms by which hookah contributes to male infertility are not fully elucidated. One potential mechanism is that hookah smoke may damage sperm membranes, leading to impaired sperm function. Hookah smoke may disrupt hormonal regulation of spermatogenesis, further exacerbating to male infertility (Firouzabadi, 2024; Jandíková et al., 2017; Montjean et al., 2023).
Cellular stress responses, including the nuclear factor erythroid 2-related factor 2 (NRF2) pathway, play a crucial role in protecting cells from oxidative stress and maintaining cellular homeostasis (Ma, 2013). When cells experience oxidative stress, NRF2 translocate to the nucleus and binds to specific DNA sequences called antioxidant response elements (AREs) in the promoter regions of antioxidant genes (Baird & Yamamoto, 2020). This binding activates the transcription of these genes, leading to the production of various antioxidant enzymes and detoxifying molecules (Panieri et al., 2020). In the context of male infertility, NRF2 has been shown to play a crucial role in protecting sperm cells from oxidative stress. Studies in mice lacking the Nrf2 gene have shown significantly lower sperm counts, reduced sperm motility, and abnormal sperm morphology compared with their healthy mice (Liu & Ng, 2000; Nakamura et al., 2010). Conversely, activating Nrf2 has been demonstrated to improve sperm motility and morphology in mice (Ali et al., 2020). These findings suggest that NRF2 activation could be a potential therapeutic target for improving male fertility in humans.
Vitamin D possesses antioxidant and anti-inflammatory properties, potentially protecting sperm from oxidative stress (Kwiecinski et al., 1989). By enhancing these antioxidant defenses, vitamin D may neutralize ROS and mitigate their detrimental effects on sperm function (Maghsoumi-Norouzabad et al., 2021). Cytochrome P450 family 24 subfamily A member 1 (CYP24A1), an enzyme responsible for vitamin D metabolism, is expressed in the human sperm annulus (the outer membrane) and may serve as an indicator of vitamin D status and, consequently, sperm quality (Blomberg Jensen et al., 2010; Vahedi Raad et al., 2024). Studies have shown that lower CYP24A1 expression is associated with reduced sperm motility and morphology. This suggests that CYP24A1 activity may be crucial for maintaining optimal vitamin D levels within sperm cells and protecting them from oxidative damage (Bøllehuus Hansen et al., 2019; Helmi & Hameed, 2022).
Studies show infertile men who smoke have lower NRF2 gene expression, suggesting weaker antioxidant responses and increased vulnerability to sperm damage (Bidram et al., 2020; Elsamanoudy et al., 2017). Smoking may be associated with upregulated CYP24A1 expression in sperm, although the underlying mechanisms remain unclear (Kim et al., 2012). The impact of hookah smoking on NRF2 and CYP24A1 expression in sperm remains unclear. To address this knowledge gap, we designed this cross-sectional study to explore whether hookah smoking modulates NRF2 and CYP24A1 expression in sperm. Deciphering these pathways may hold the key to developing innovative therapeutic interventions to enhance fertility potential in male hookah smokers.
Material and Method
Participants of Study
A study was conducted at the Reproductive Science Institute in Yazd, between September 2023 and January 2024. Out of 100 infertile couples referred during this period, 84 infertile couples with female factor infertility were recruited. Following predefined inclusion criteria for male partners (normal sperm parameters and age eligibility) and obtaining informed consent, semen analysis was performed on all participants to assess sperm quality as the primary data collection method. Participants were then categorized into two groups: a case group of 32 male hookah smokers (minimum hookah use of 3 times per week for at least 30-45 min per session) and a control group of 52 non-smoker males. To ensure homogeneity within the groups and eliminate confounding factors, participants with chronic diseases (e.g., hypertension, diabetes, varicocele), urogenital infections, history of heavy cigarette smoking, alcohol consumption, specific medications, drug addiction, or abnormal sperm parameters were excluded (Figure 1).

Diagram of the Study’s Design
Semen Collection and Analysis
Adhering to standardized protocols, semen samples were collected via masturbation from all participants following a 2- to 7-day period of sexual abstinence. To ensure optimal sperm evaluation, each sample underwent liquefaction within a 37°C incubator for 20 min. Subsequent macroscopic analysis assessed semen characteristics including pH, visual appearance, liquefaction time, viscosity, and volume. Microscopic evaluation then focused on sperm morphology, motility, and concentration, employing the established criteria outlined by the World Health Organization (WHO) (2021) edition (WHO, 2021). Following these initial analyses, the remaining semen was designated for further investigations. This involved a centrifugation process at 1800 g for 10 min to achieve effective separation of spermatozoa from the seminal plasma. The resulting sperm pellets were then re-suspended in a phosphate-buffered saline (PBS) solution. This PBS wash eliminated residual seminal plasma constituents, thereby preparing the spermatozoa for subsequent molecular-based assessments.
Gene Expression Assessments
RNA Extraction
Following semen collection and analysis, sperm pellets were subjected to RNA extraction. The total RNA was extracted by applying the RNX-Plus kit (Sinaclon Bioscience, Cat. No.: RN7713C) according to our previous setup (Firouzabadi, Rezvani, et al., 2024; Tofighi Niaki et al., 2023). A NanoDrop spectrophotometer (Thermo Scientific, Waltham, MA, USA) was used to assess the concentration and purity of the extracted RNA at 260 nm and 260/280, respectively.
Complementary DNA Synthesis
To generate complementary DNA (cDNA) for subsequent Quantitative Real-Time PCR (qRT-PCR) analysis, 500 ng/µl of the purified RNA was used along with a cDNA synthesis kit (Parstous, Cat No: A101161; Tehran, Iran). The cDNA synthesis process involved adding specific volumes of buffer, water, RNA, and enzyme mix to each microtube, followed by thermocycler incubation according to the kit’s protocol.
Quantitative Real-Time PCR
QRT-PCR was employed to quantify the relative expression of target genes. The efficiency of each primer set was determined via the standard curve method, ensuring efficiency above 95% for subsequent qRT-PCR experiments. Each qRT-PCR reaction was run in duplicate using a Step One thermocycler (Applied Biosystems, USA) and included nuclease-free water, specific primers (Table 1), diluted cDNA (1:10), and Master Mix Green with high ROX™ (Amplicon). The thermal cycling program consisted of an initial holding stage at 95°C for 10 min, followed by 40 amplification cycles of 95°C for 15 s, 58°C for 30 s, and 72°C for 30 s. A final melt curve stage at 57°C verified primer specificity and reaction purity. Threshold cycle (Ct) values were obtained for each gene of interest and normalized to the housekeeping gene GAPDH for data analysis. The comparative Ct technique (2-ΔΔCt) was then applied to quantify relative gene expression levels.
The Sequence of Oligonucleotide Primers

Note. CYP24A1: Cytochrome P450 family 24 subfamily A member 1, GAPDH: Glyceraldehyde-3-Phosphate Dehydrogenase and NRF2: Nuclear factor erythroid 2-related factor 2.
Statistical Analysis
Data were presented as mean ± standard error of the mean (SEM). The normality of the data distribution in each group (case and control) was assessed using the Shapiro-Wilk test. Statistical comparisons between groups were performed using either the independent t-test (for normally distributed data) or the Mann–Whitney U test (for non-normally distributed data), depending on the results of the normality test. A p-value less than 0.05 was considered statistically significant. All statistical analyses were performed using GraphPad Prism 10 software (GraphPad Software, San Diego, CA, USA).
Results
Semen Analysis
Analysis of baseline characteristics revealed no significant difference in mean age between the two groups (case and control) Table 2. Semen volume in the control group (3.4 mL ± 1.78) tended to be higher compared with the case group (2.81 mL ± 1.11), although this difference wasn’t statistically significant. Similarly, sperm concentration in the control group (53.32 million/mL ± 33.45) showed a nonsignificant but higher average compared with the case group (40.5 million/mL ± 31.64). This translated to a trend of a higher total sperm number in the control group (182.4 ± 78.77 million) compared with the case group (132.4 ± 84.91 million). Evaluation of sperm motility revealed no clear differences between the groups. The percentages were as follows: progressive motility (control: 37.53% ± 1.30, case: 35.00 %± 17.20), non-progressive motility (control: 11.59 ± 0.12%, case: 11.97 ± 3.27%), and total motility (control: 49.12%± 1.44, case: 46.96% ± 17.73). Likewise, the percentage of sperm with normal morphology was comparable between the groups (control: 4.67% ± 0.89, case: 4.08% ± 1.21). Statistically significant and positive correlations were observed between age and total motility (p = 0.04, r = 0.32), although this correlation is weak. Hookah smoking duration exhibited a significant and negative correlation with sperm morphology (p = 0.006, r = −0.47). The results for NRF2 gene expression showed a significant and positive correlation with progressive motility (p = 0.01, r = 0.76). No statistically significant correlations were found between CYP24A1 gene expression and any of the sperm parameters (Figure 2).
Comparison of Sperm Parameters Between the Experimental Groups

Note. Values were expressed as mean ± SEM, Statistical significance was assessed using an independent student t-test for the normally distributed variables. p < 0.05 was regarded as a significant value.

Correlation Between Age, Hookah Smoking Duration and Gene Expression With Sperm Parameters
Gene Expression Profile
NRF2 expression showed a significant increase in the case group compared to the control group (p
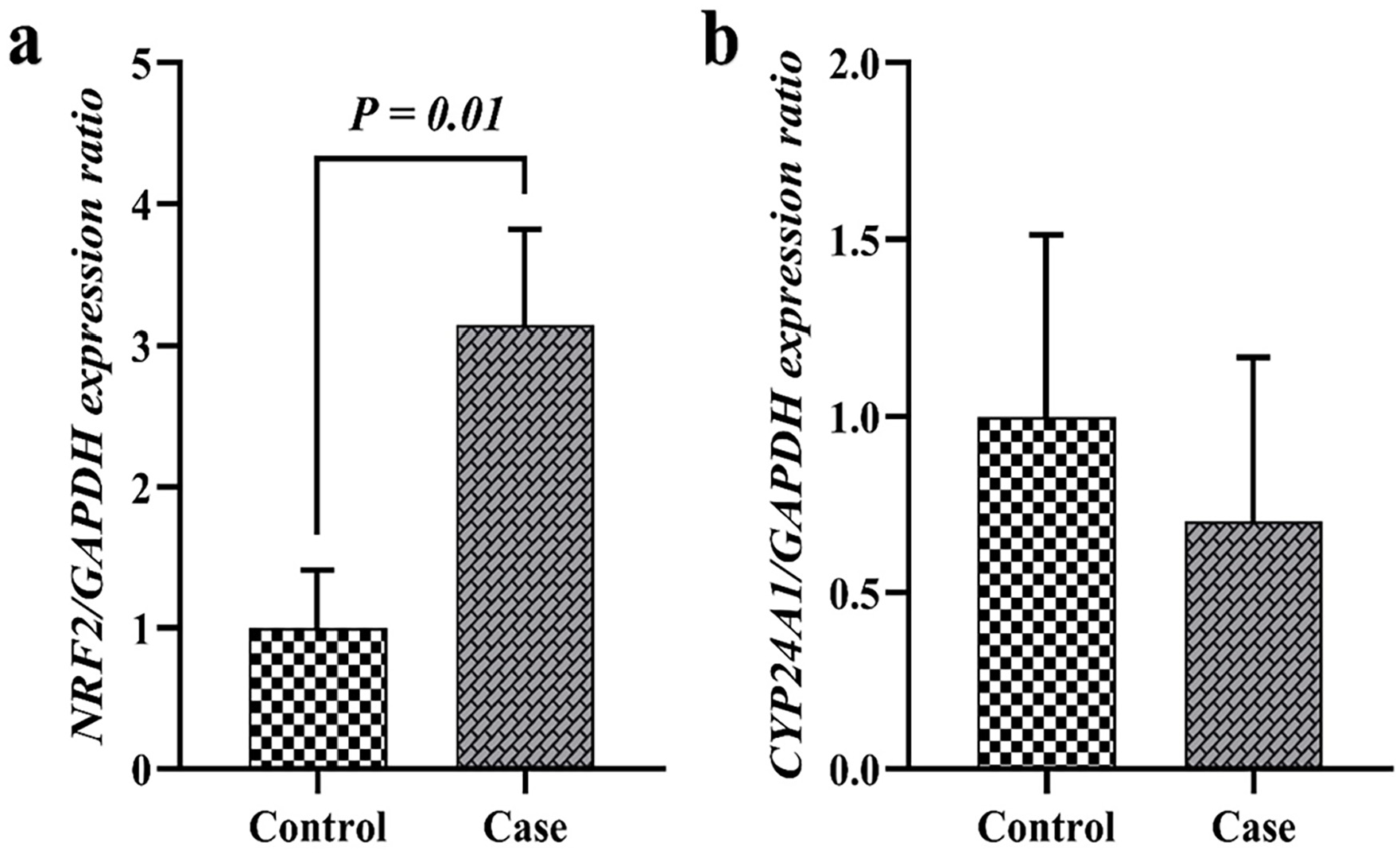
Comparison of Genes Expression Between the Experimental Groups
Discussion
Studies suggest a potential link between hookah smoking and decreased male fertility. Hookah tobacco contains toxins, like lead, that could disrupt sperm production and function in various ways. Isolating hookah’s specific impact on sperm quality is challenging due to external factors such as smoking habits, genetic, and environmental factors (Gandhi et al., 2017).
In line with our expectations, the study results demonstrated no detrimental effects of hookah smoking on sperm quality. This was anticipated as our case group comprised fertile males who consumed hookah. Conversely, the literature has indicated an adverse effect of cigarette smoking on sperm parameters (Osadchuk et al., 2023). This difference might be due to variations in smoke composition. Hookah smoke, compared with cigarettes, undergoes water filtration, potentially reducing the concentration of harmful chemicals, which could explain the varying effects on sperm (Qasim et al., 2019). Our findings revealed a statistically significant inverse association between the duration of hookah smoking and sperm morphology. This suggests that prolonged hookah use may be linked to a decline in the percentage of spermatozoa with normal morphology. This is consistent with Albeitawi’s findings, which indicated a significant association between both cigarette and hookah smoking and a reduced percentage of sperm with normal morphology (Albeitawi et al., 2024). Potential mechanisms include increased oxidative stress, lipid peroxidation of sperm membranes, DNA damage, and reduced activity of antioxidant enzymes, all of which are associated with tobacco smoke exposure (Elshal et al., 2009).
Our analysis revealed a significant increase in NRF2 expression in the sperm of hookah smokers compared with the control group. This finding aligns with other studies demonstrating NRF2 upregulation in response to oxidative stress, such as the work by Nemmar et al. (2018) who reported a slight increase in NRF2 expression in lung tissue and Ali et al. (2020) who observed a significant increase in Nrf2 expression in the testes of mice exposed to hookah smoke (Ali et al., 2020; Nemmar et al., 2018). In contrast, existing research on smoking and NRF2 expression shows inconsistencies. Studies by Elsamanoudy et al. (2017) and Bidram et al. (2020) reported decreased NRF2 expression in infertile smokers (Bidram et al., 2020; Elsamanoudy et al., 2017). This difference might be explained by the specific population studied. Our study involved fertile hookah smokers with normal sperm parameters, whereas the studies by Elsamanoudy et al. (2017) and Bidram et al. (2020) investigated infertile men. It is possible that decreased NRF2 levels are a consequence of infertility, potentially due to impaired antioxidant defense mechanisms. This hypothesis is supported by Nakamura et al. (2010) who demonstrated severe testicular dysfunction and reduced sperm production in Nrf2 knockout mice (Nakamura et al., 2010). The results for NRF2 gene expression, which plays a role in cellular antioxidant defense, showed a significant positive correlation with non-progressive motility (p = .01, r = .76). This suggests a potential link between antioxidant response and sperm motility. These results reinforce the existing evidence that NRF2 not only mitigates oxidative stress but also plays a pivotal role in preserving sperm functionality, particularly motility, under oxidative conditions (Signorini et al., 2024). To fully understand the link between hookah smoking, NRF2 expression, and individual susceptibility, further research is needed comparing NRF2 expression in fertile and infertile hookah smokers and investigating the functional consequences of NRF2 upregulation in our observed context (e.g., protein expression, enzymatic activity).
Our study found no significant change in CYP24A1 expression between the hookah smoking and control groups. This contrasts with the findings of Kim et al. (2012) who reported increased CYP24A1 expression in lung cancer patients exposed to cigarette smoke (Kim et al., 2012). The discrepancy might be due to several factors: the type of smoking (hookah vs. cigarettes), the target tissues (sperm vs. lung), and the presence/absence of preexisting health conditions (healthy men vs. lung cancer patients). Our results suggest that hookah smoking may not significantly influence CYP24A1 expression in healthy men. Further research with larger sample sizes and more sensitive techniques is needed to definitively draw this conclusion.
Conclusion
This study provides preliminary insights into the effects of hookah smoking on sperm quality, NRF2, and CYP24A1 expression in fertile men. While the findings suggest no statistically significant detrimental effects on traditional semen parameters like sperm count, motility, or morphology compared with non-smokers, the increased expression of NRF2 in sperm suggests a potential adaptive response to oxidative stress caused by hookah smoke. This upregulation may reflect the body’s attempt to counteract the harmful effects of reactive oxygen species, though its exact functional implications remain unclear. In contrast, the lack of significant changes in CYP24A1 expression calls for further exploration into the role of vitamin D metabolism in male fertility, especially in the context of hookah use. Despite the limitations, this study highlights the need for further research to better understand the mechanisms through which hookah smoking might impact male reproductive health. Larger, longitudinal studies incorporating advanced fertility assessments and diverse populations, particularly including infertile men, are essential to confirm these findings and explore potential therapeutic targets for male infertility in hookah smokers.
Footnotes
Acknowledgements
This study was done at Yazd Shahid Sadoughi University of Medical Sciences. The authors also thank the Reproductive Science Institute in Yazd for its research facilities.
Author Contributions
All authors contributed to the conception and design. Farzaneh Fesahat was responsible for overall supervision. Amir Masoud Firouzabadi and Maryam Imani Drafted the manuscript, which Amir Masoud Firouzabadi revised. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The Medical Ethics Committee of Shahid Sadoughi University of Medical Sciences, Yazd, approved this study with approval number IR.SSU.SPH.REC.1402.005.
Consent to Participate
All participants signed an informed consent form before sample collection.
Data Availability
The data supporting this study’s findings are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
