Abstract
The male factor is responsible for infertility in about 35–40% of all cases. Idiopathic oligo- and/or astheno- and/or therato-zoospermia is one of the most common male fertility disorders and remains a significant therapeutic challenge. The primary cause of idiopathic male infertility remains unknown but seems to be associated with oxidative stress. Objective: The use of antioxidative formulation to improve qualitative and quantitative deficiencies in the male gametes.
In total, 78 subjects were treated with a combination of 1,725 mg L-carnitine fumarate, 500 mg acetyl-L-carnitine, 90 mg vitamin C, 20 mg coenzyme Q10, 10 mg zinc, 200 µg folic acid, 50 µg selenium, and 1.5 µg vitamin B12 (Proxeed® Plus, Sigma-Tau, Italy) for 6 months; the preparation was taken twice daily from the time idiopathic infertility was diagnosed. Basic seminal parameters were evaluated by a European Society of Human Reproduction and Embryology (ESHRE) -certified embryologist following the fifth edition of the World Health Organisation (2010) guidelines at three time points: at baseline and 3 and 6 months of treatment.
Improvements in semen parameters (differing in terms of dynamics) were evident at 3 months and gradually improved over the 6 months of treatment. Each parameter: sperm concentration, total sperm count, sperm total and progressive motility improved significantly after treatment except for the percentage of sperm of abnormal morphology and ejaculate volume.
Proxeed Plus was effective for patients with idiopathic infertility; however, a long treatment period is needed to achieve optimal results.
In vitro fertilization (IVF) was approved in 1978 and rapidly became popular. However, until 1990, there was no available therapy that increased the pregnancy rate when male gametes were defective. A breakthrough came with the introduction of the intracytoplasmic sperm injection (ICSI) technique (Palermo et al., 1992). The subsequent development of molecular biological techniques has significantly expanded the opportunities afforded by ICSI. However, the basic problems remain; these are qualitative and quantitative gametic deficiencies.
The Pathogenesis of Male Infertility and Prospects for Treatment
According to the World Health Organisation (World Health Organization, 2010), inability to conceive after ≥1 year of regular unprotected intercourse may indicate infertility in one or both partners (Brugh & Lipshultz, 2004). Because the male factor is the cause of infertility in about 35–40% of cases, qualitative and quantitative sperm analyses must always be performed when investigating problems with conception (Comhaire et al., 2003).
In almost 90% of cases, male infertility is caused by a varicocele, an accessory gland infection, an immunological disorder, or so-called idiopathic infertility.
Varicoceles are the most common cause of male infertility. Most of its pathogenesis is related to oxidative stress, reflux of metabolites, increased temperature of the testicle. Varicoceles adversely affect the functioning of Sertoli and Leydig cells and therefore the quality of germ cells, triggering premature testicles failure, (Aitken, 2007; Comhaire, 1975). The standard treatment is surgery (Kunnen, 1981).
Male accessory gland infections are the second leading cause of infertility. Such infections usually involve the seminal vesicles, prostate, or epididymides. Disruption of the blood–testicle barrier caused by epididymal infection may be associated with secondary immunological abnormalities, including the production of antisperm antibodies (Comhaire, 2003). Antibiotic therapy is the basic treatment for accessory gland infections but does not ensure the return of normal gland functionality after the eradication of the infectious agent. Male accessory glans infections may also lead to blockage of epididymal tubules (obstructive azoospermia). (Depuydt et al., 2009).
Finally, so-called male idiopathic infertility is also common, occurring in almost 30% of all infertile males (even 60%) (Bonanomi et al., 2002; Cavallini et al., 2004; Hancke et al., 2006; Zhou, 2007). Idiopathic infertility is diagnosed when quantitative and qualitative semen data are not within the reference ranges and no other etiological cause is evident (Bonanomi et al., 2002) The most common abnormalities are idiopathic oligo- and/or astheno- and/or therato-zoospermia. Environmental factors have been suggested as possible causes, including alcohol and/or tobacco abuse, being overweight, an unhealthy diet (with an insufficient supply of omega-3 polyunsaturated fatty acids), and/or exposure to chemicals (including factors that accumulate in adipose tissue [xeno-estrogens] that disturb the hormonal homeostasis of the hypothalamic–pituitary–testicular axis) (Zalata et al., 1998).
Dietary supplements are commonly used to treat male idiopathic infertility. Such formulations should prevent abnormal sperm production at all stages of formation and maturation.
Oxidative Stress and the Use of Antioxidants to Treat Idiopathic Male Infertility
Oxidative stress is one of the major adverse factors affecting spermatozoal function and development (Aitken & Clarkson, 1987; Aitken & Krausz, 2001; Aitken et al., 2002; Agarwal & Said, 2005; Agarwal et al., 2003; Alvarez & Storey, 1982; Balercia et al., 2004; Khosrowbeygi & Zarghami, 2007; Lafuente et al., 2013; Makker et al., 2009; Rao et al., 1989; Saleh & Agarwal, 2002; Suleiman et al., 1996). Such stress affects cell membrane integrity, energy metabolism, and the mutation rates of genomic and mitochondrial DNA (Agarwal et al., 2004; Aitken et al., 1989, 1993; Comhaire et al., 2000, 2005; Eskenazi et al., 2005; Hosen et al., 2015; Rao et al., 1989; Sawyer et al., 2003; Sheweita et al., 2005; Showell et al., 2011).
Oxidative stress triggers disintegration of the cellular membrane and/or mitochondrial inactivation. Mitochondria are located in the midpieces of spermatozoa and synthesize adenosine triphosphate (ATP), which provides the energy required for motion. Oxidative stress partially impairs mitochondrial activity, reducing sperm motility and activity. In addition, mitochondrial energy production per se contributes to the generation of oxygen free radicals (Comhaire et al., 2000, 2005; Du Plessis et al., 2008). The frequency of genomic mutations (assessed by measuring oxidized guanine [8-OH-2dG] levels) is significantly higher in sperm exposed to oxidative stress (Comhaire et al., 2005; Hosen et al., 2015).
Molecules with antioxidative potential may protect sperm from damage induced by oxidative stress, significantly improving sperm quality and quantity and increasing the likelihood of pregnancy (Balercia et al., 2004; Lenzi et al., 1993; Showell et al., 2011). It is useful to include such materials in dietary supplements used to treat idiopathic male infertility.
Coenzyme Q10
Ubiquinone, or co-enzyme Q10, is a strong antioxidant that scavenges oxygen radicals generated by mitochondria (Comhaire et al., 2000; Du Plessis et al., 2008; James et al., 2004; Safarinejad, 2009); enhances the serum levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) in men (James et al., 2004; Safarinejad, 2009); and improves sperm morphology, concentration, and motility (Comhaire et al., 2000; Du Plessis et al., 2008; Hara et al., 2011; Lafuente et al., 2013; Safarinejad, 2009; Safarinejad et al., 2012).
Folic Acid
Folic acid acts as an antioxidant, limiting the number of round cells (one of the major sources of oxygen radicals) in semen (Aitken & De Iuliis, 2009; Bentivoglio et al., 1993) and increases the number of sperm (Aitken et al., 2009; Bentivoglio et al., 1993; Landau et al., 1978).
Selenium
Selenium occurs in the membranes of sperm in complexes with proteins (the selenoproteins mGPx4 and snGPx4) (Ahsan et al., 2014) and exhibits a remarkable antioxidative potential by stabilizing the membrane structure (Keskes-Ammar et al., 2003). Sperm concentration, morphology, and motility improve significantly after supplementation with selenium (Ahsan et al., 2014; Keskes-Ammar et al., 2003; Safarinejad & Safarinejad, 2009). In addition, selenium positively influences thyroid metabolism, which is important in terms of sperm production (Beckett & Arthur, 2005) and testosterone biosynthesis (Flohé, 2007).
Zinc
Testicular tissue contains high concentrations of the enzymes elongase and desaturase, and spermatogenesis at the time of sperm maturation may be inhibited if zinc (an essential cofactor for both enzymes) is deficient (Sæther et al., 2003; Stoffel et al., 2008). Adequate levels of vitamin B6 (also a cofactor for both enzymes) and zinc play important roles in spermatogenesis. Zinc has antioxidative effects during sperm production and maturation, increasing sperm motility and concentration (Ebisch et al., 2006; Hadwan et al., 2012, 2014) and even ejaculate volume (Hadwan et al., 2012). Zinc also protects against bacterial infection (Hadwan et al., 2012, 2014).
Vitamin C
Vitamin C, or ascorbic acid, is a strong water-soluble antioxidant required for various bodily processes such as biosynthesis of collagen and proteoglycan and certain hydroxylations and amidations (Linster & Van Schaftingen, 2006). Ascorbic acid in seminal plasma protects sperm from genetic material damage (Colagar & Marzony, 2009; Ko & Sabanegh, 2012).
L-carnitine/Acetyl-L-carnitine
Recent research (Aitken et al., 1993; Matalliotakis et al., 2000; Zhou et al., 2007), suggests that L-carnitine and acetyl-L-carnitine should be essential ingredients in dietary supplements used to treat male idiopathic infertility. Carnitines exhibit several useful properties. L-carnitine (3-hydroxy-4-N-trimethyl-aminobutyric acid) was first identified and isolated from bovine muscle by a Russian group in 1905, and its detailed structure was thoroughly investigated 22 years later in 1927 (Kerner & Hoppel, 1998; Peluso et al., 2000). Only the L-isomer is biologically active. L-carnitine is widely distributed in nature (Kerner & Hoppel, 1998). Only about 25% of bodily L-carnitine is synthesized endogenously from lysine and methionine; the remaining 75% must be supplied in the diet, principally by poultry, meat, and fish (Engel & Rebouche, 1984; Peluso et al., 2000). The first report on the beneficial effects of carnitines on male reproductive cell motility appeared in 1977. Tanphaichitr (Tanphaichitr, 1977) reported that acetylcarnitine significantly improved sperm motility when added to ejaculate.
L-carnitine plays an important role in energy production at the cellular level by transporting substrates of the energy generation pathway into mitochondria (Cavallini et al., 2004). L-carnitine is involved in the β-oxidation of long-chain fatty acids (Bremer, 1983; Kerner & Hoppel, 1998; Lenzi et al., 1992). Before being transported across the outer mitochondrial membrane, such fatty acids are activated by attaching to coenzyme A. These complexes alone cannot penetrate the inner mitochondrial membrane; L-carnitine mediates such translocation (Arduini et al., 2008; Bremer, 1983; Claudette et al., 1996; Jeulin et al., 1994). The fatty acids are coupled with L-carnitine, with transfer of an acetyl group to a CoA molecule located in the inner mitochondrial membrane; thus, β-oxidation commences and ATP is produced soon thereafter (Arduini, 1992).
L-carnitine is an antioxidant (Arduini, 1992; Juliet, et al., 2001; Kobayashi & Fujisawa, 1994). In addition to protecting genetic material from damage caused by oxygen free radicals, L-carnitine regulates the fluidity of phospholipid membranes (by adjusting phospholipid content and reducing the production levels of ceramides and insulin-like growth factor-1), aids in the restoration of mitochondrial membrane structure, and prevents apoptosis (Abad et al., 2013; Amendola et al., 1989; 1991; Arduini, 1992; Cavallini et al., 2004; Kanter et al., 2010; Lenzi et al., 1993, 2003; Mutomba et al., 2000; Okada et al., 2009). The highest L-carnitine concentration in the human body is that of epididymal fluid (2–100 mM). The blood level is almost 2,000-fold lower, not exceeding 50 µM under physiological conditions (Claudette, et al., 1996; Enomoto et al., 2002; Johansen & BØhmer, 1978; Lenzi et al., 1992; Radigue et al., 1996; Vicari et al., 2002). L-carnitine is secreted into the epididymal lumen by the epididymal epithelium (Arduini et al., 2008; Chiu et al., 2004). Sperm motility improves in direct proportion to the L-carnitine and acetyl-L-carnitine levels subsequently produced inside sperm (Chiu et al., 2004; Enomoto et al., 2002; Jeulin & Lewin, 1996; Johansen & BØhmer, 1978; Radigue et al., 1996; Yeste et al., 2010).
Acetyl-L-carnitine is a ready-to-use substrate of the Krebs cycle and, like L-carnitine, stabilizes cell membranes by facilitating phospholipid incorporation (Cavallini et al., 2004). It also exhibits antioxidative and antiapoptotic properties (preventing oxygen free radical–induced mitochondrial-dependent apoptosis) (Annadurai et al., 2011; Gunes et al., 2011; Tufekci et al., 2009).
The U.S. Food and Drug Administration (FDA) has approved carnitines for the pharmacological treatment of haemodialysis-induced deficits in patients with chronic renal failure (Zhou et al., 2007). The many positive effects and excellent safety profiles of carnitines render them an invaluable source of energy for male reproductive cells, significantly enhancing sperm parameters, particularly motility. Therefore, carnitines should be basic ingredients in dietary supplements used to treat male idiopathic infertility.
Materials and Methods
Materials
This retrospective study was based on a review of medical records collected at the Genesis fertility clinic (Bydgoszcz, Poland) between January 2015 and June 2017. The study was approved by the Bioethics Committee of the Nicolaus Copernicus University of Toruń; the Ludwik Rydygier Collegium Medicum in Bydgoszcz (approval no. KB564/2017). Patients signed an informed consent to participate in the study.
To obtain a group of males with idiopathic infertility, we excluded all patients with any detectable cause of infertility. We measured blood LH, FSH, and testosterone levels and performed genital examinations (including ultrasonography). We excluded patients with varicoceles, hormonal disorders, and/or infections. Of 163 patients who underwent scrupulous medical examinations to determine the causes of infertility, 78 (20- to 40-year-old Polish subjects) met our inclusion criteria. Each subject had a semen leucocyte concentration of less than 1 mln cells/mL and additionally met one or more of the following inclusion criteria: total sperm count in ejaculate <39 mln/mL (or a sperm concentration <15 mln/mL), progressive sperm motility <32%, and/or a morphologically pathological sperm level >96%.
All patients took a combination supplement twice daily for 6 months; each dose contained 1,725 mg L-carnitine fumarate, 500 mg acetyl-L-carnitine, 90 mg vitamin C, 20 mg coenzyme Q10, 10 mg zinc, 200 µg folic acid, 50 µg selenium, and 1.5 µg vitamin B12 (Proxeed® Plus, Sigma-Tau, Italy). Supplementation commenced at the time of diagnosis of idiopathic infertility.
The following semen parameters were assessed at baseline and after 3 and 6 months of treatment: ejaculate volume (mL), total sperm count (mln), sperm concentration per millilitre of semen (mln/mL), progressive (including both A and B forms) and total sperm motility (expressed as percentages and mln/mL), level of morphologically pathological sperm forms (%), and leukocyte concentration (mln/mL). Semen evaluations were performed by a single clinical embryologist certified by the European Society of Human Reproduction and Embryology (ESHRE) in accordance with WHO guidelines (fifth edition) (World Health Organization, 2010).
Statistical Methods
The statistical analysis was performed in Microsoft Excel 2010 and Statistica 13. We used the Shapiro–Wilk test to compare patient data with normal distributions. We compared sperm parameters at baseline and at 3 and 6 months of treatment using the Friedman test for dependent variables. p < .01 was considered to reflect statistical significance.
Results
General Semen Parameters
Except in the parameters sperm morphology and semen volume, significant improvements were observed after therapy. At least 45% of patients improved. The extent of abnormal sperm morphology did not improve greatly; in about 90% of patients no improvement occurred. Of all patients, 22–45% exhibited no change in semen volume or total or progressive sperm motility. Only a small proportion of patients evidenced deterioration Thus, the formulation was effective.
Sperm Motility in Ejaculate
Changes in the proportions of A-, B-, C-, and D-form sperm in semen after treatment are presented in graphical form in Figure 1. Semen quality improved after treatment; the median and quartile values of B-form sperm increased after treatment and those of D-form sperm decreased. The percentage of B-form sperm increased in 72% of patients and that of D-form sperm decreased in 35% of patients. The improvement was significant. Also, quartile values of A-form sperm increased, but the change was not statistically significant (p = .354). The percentage of C-form sperm did not change significantly, increasing in some patients but decreasing in others.
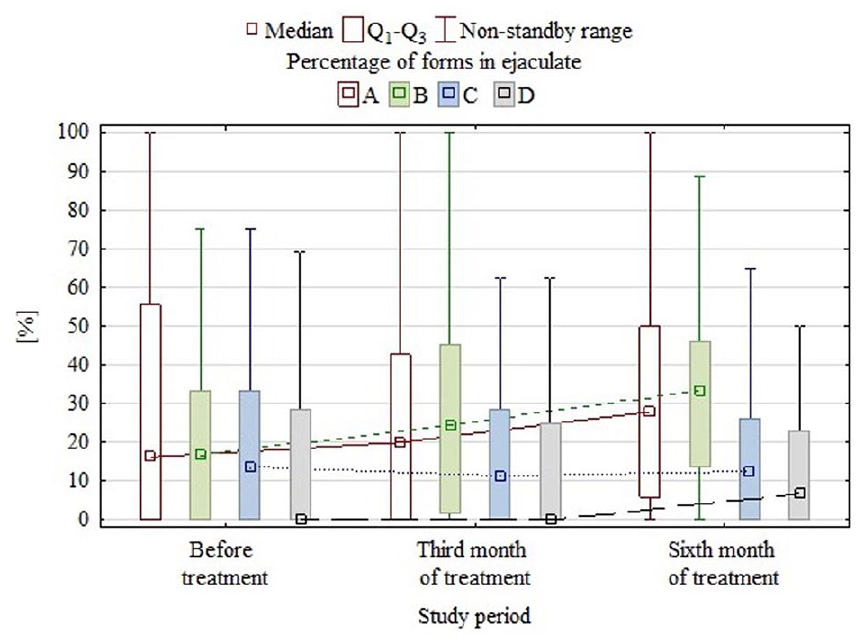
Graphical representation of the percentages of motile sperm forms at three time points.
Concentrations of Motile Sperm Forms in Ejaculate
Changes in the concentrations of A-, B-, C-, and D-form sperm in semen after treatment are presented graphically in Figure 2. Semen quality improved significantly after treatment, as evidenced by increases in the median and quartile values of the levels of A- and B-form sperm. A- and B-form sperm increased in 65% and 76% of patients, respectively Decreases in the concentrations of these forms were detected in 4% and 8% of patients, respectively, after treatment. Concentration of C-form sperm increased slightly in 54% of patients but decreased slightly in 21%. The concentration of D-form sperm increased slightly in 41% of patients but decreased slightly in 17%.

Graphical representation of concentrations of sperm of various motilities in ejaculate during treatment.
Total Counts of Sperm of Various Motilities in Ejaculate
Changes in the counts of A-, B-, C-, and D-form sperm in semen after treatment are presented in graphical form in Figure 3. Treatment improved the quality of semen, as evidenced by increases in the median and quartile values of the total counts of A- and B-form sperm. No significant changes in the total counts of D- or C-form sperm were apparent. Total counts of A- and B-form sperm increased in 71% and 79% of patients, respectively. Total counts of C-form sperm increased slightly in 56% of patients but decreased slightly in 22%. Total counts of D-form sperm increased slightly in 41% of patients but decreased slightly in 23%.

Graphical representation of total counts of sperm of various motilities in ejaculate during treatment.
Discussion and Conclusions
Idiopathic oligo- and/or astheno- and/or therato-zoospermia is one of the most common male fertility disorders and remains a considerable therapeutic challenge. The primary cause of such infertility remains unknown. It may be that oxidative stress plays an important role, triggering a variety of abnormalities. In particular, the progressive sperm motility essential for oocyte fertilization may be compromised by oxidative stress that extensively disturbs spermatozoal energy metabolism (Agarwal et al., 2003; El-Taieb et al., 2009; Iacono et al., 2013; Venkatesh et al., 2009; Zhou et al., 2007). We evaluated the utility of oral administration of a supplement containing L-carnitine fumarate, acetyl L-carnitine, vitamin C, coenzyme Q10, zinc, folic acid, selenium, and vitamin B12 (Proxeed Plus) in terms of improving general semen parameters, including ejaculate volume, sperm concentration, total sperm count, sperm motility, and sperm morphology evaluated at the time of diagnosis and at 3 and 6 months later. To the best of our knowledge, no prior report has examined the utility of Proxeed Plus in this context. Both we and others have confirmed the utility of oral antioxidant preparations in which acetyl-L- and/or L-carnitine is the principal active agent(s). The use of similar formulations has been associated with improvements in the semen parameters of patients with various conditions, including idiopathic infertility (Abad et al., 2013; Balercia et al., 2005; Cavallini et al., 2004; De Rosa et al., 2005; Lenzi et al., 2003; 2004; Lombardo et al., 2010; Suleiman et al., 1996; Vicari & Calogero, 2001; Vicari et al., 2002). We found that with the exception of sperm morphology and ejaculate volume, all quantitative and qualitative semen parameters (sperm concentration, total number of sperm, and motility), improved significantly after use of the dietary supplement. In terms of sperm motility, our results are consistent with those of others (Costa et al., 1994; Khademi et al., 2005; Lombardo et al., 2010) but, unlike us, Khademi et al. (Khademi et al., 2005) reported that L-carnitine significantly improved sperm morphology, and Sigman et al. (Sigman et al., 2006) found no clinically or statistically significant improvement in sperm motility in patients with idiopathic astheno-zoospermia who had previously been treated with L-carnitine. However, the effects of L-carnitine on semen parameters, including sperm morphology, are often inconsistent (Zhou et al., 2007).
We found significant increases in sperm concentration (1.9 mln sperm/mL ejaculate; from 6.9 ± 10.6 to 8.8 ± 10.4 mln/mL; mean values), total sperm count (7.3 mln; from 25.4 ± 47.3 to 32.7 ± 50.4 mln; mean values), sperm progressive motility (9%; from 53.8% ± 37.2% to 62.8% ± 34.3%; forms A and B; mean values), and total sperm motility (7.4%; from 72.6% ± 36.0% to 80% ± 28.7%; forms A, B, and C; mean values) after 6 months of treatment. The highest average increase in percentage of motile sperm levels in 7 of the 10 patients was that of the B form (average 6.9%). After 6 months of treatment, the concentration of the B form increased by about 1 mln/mL semen, and the total count increased by approximately 3.7 mln. In addition, the percentage, total count, and concentration of A-form sperm (fast progressive motility) increased by 2.2%, 4.1 mln and 1.1 mln/mL, respectively. Such changes were evident in more than half of the patients. Any change in the proportion of sperm of normal morphology was not significant.
Note that improvements were evident (although the dynamics differed) after only 3 months of supplementation but gradually improved further after 6 months. The treatment period should be long enough to obtain optimal results. Not all patients responded identically; beneficial effects were more pronounced in some than in others. Some patients did not exhibit any clinically significant change, and in a small number of patients semen parameters actually deteriorated (perhaps unrelated to supplementation, instead reflecting the progression of an existing idiopathic disorder). Similar observations were made by Abad et al. (Abad et al., 2013). Although we did not examine the stability of therapeutic effects after treatment was discontinued, data on similar formulations in the literature suggest that the beneficial effects are temporary: after discontinuation of treatment, semen parameters return to baseline values (Sofimajidpour et al., 2016).
It is presumed that the observed improvements reflect the combined activity of the individual components. L-carnitine and acetyl-L-carnitine positively affect semen parameters, including sperm motility (by serving as energy sources; (Ahmed et al., 2011; Lenzi et al., 2003, 2004; Li et al., 2007; Khademi et al., 2005; Zhou et al., 2007) and number of sperm (by promoting sperm maturation and protecting sperm from damage induced by oxidative stress). Zinc, coenzyme Q10, folic acid, selenium, and vitamin C perform certain functions important in spermatogenesis (e.g. coenzyme Q10 regulates FSH and LH serum levels) and facilitate sperm nuclear remodelling [(Abad et al., 2013)]. All of these materials reduce oxidative stress, substantially improving the qualitative and quantitative parameters of semen.
In summary, Proxeed Plus significantly improved sperm parameters in patients with idiopathic infertility, particularly the progressive sperm motility crucial for fertilization of the ovum. In addition, the sperm concentration per millilitre of semen and total sperm count increased, which may aid subjects with low sperm counts. It is advisable to take Proxeed Plus for at least 6 months to obtain optimal results. A lack of improvement or deterioration in semen parameters may be associated with idiopathic factors; such patients require careful analysis.
Supplemental Material
sj-doc-1-jmh-10.1177_15579883211036790 – Supplemental material for Comparison of 3- and 6-Month Outcomes of Combined Oral L-Carnitine Fumarate and Acetyl- L-Carnitine Therapy, Included in an Antioxidant Formulation, in Patients with Idiopathic Infertility
Supplemental material, sj-doc-1-jmh-10.1177_15579883211036790 for Comparison of 3- and 6-Month Outcomes of Combined Oral L-Carnitine Fumarate and Acetyl-L-Carnitine Therapy, Included in an Antioxidant Formulation, in Patients with Idiopathic Infertility by Marek Szymański, Tomasz Wandtke, Karolina Wasilow, Marek Andryszczyk, Radosław Janicki and Piotr Domaracki in American Journal of Men’s Health
Footnotes
Acknowledgements
MS designed research and had primary responsibility for final content; KW conducted patient’s medical examinations; MA analyzed data or performed statistical analysis; PD analyzed data or wrote the paper; RJ analyzed data or wrote the paper; TW conducted research and wrote the paper;
Authors’ Note
This retrospective study was based on a review of medical records collected at the Genesis fertility clinic (Bydgoszcz, Poland) between January 2015 and June 2017. The study was approved by the Bioethics Committee of the Nicolaus Copernicus University of Toruń; the Ludwik Rydygier Collegium Medicum in Bydgoszcz (approval no. KB564/2017).
The English in this document has been checked by at least two professional editors, both native speakers of English. For a certificate, please see:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
